Evaluation of Robustness and Effectiveness of the BV-STOP, A Novel Antiseptic Compound with Specific and Anti-Microbial Activity
Elena Toniato1, Daniele Savio2, Gian Luca Falleti3, Riccardo Martinotti4, Riccardo Pulcini1,5, Jessica Elisabetta Esposito1, Francesco Avolio1 and Stefano Martinotti1*
1University of Chieti, Center of Advanced Studies, and Technology (CAST), Chieti, Italy
2R & D Solutions, srl, Greggio (VC), Italy
3Nanoprom Chemicals srl, Research and Development Group, Veggia-Villalunga, (RE), Italy
4University of Rome “La Sapienza”, Residency Program in Medical Oncology, Policlinico Umberto I, Rome, Italy
5PhD program in Science and Technology for Sustainable Development, Scuola Superiore “G. d’Annunzio”, University of Chieti, Italy
Submission:February 12, 2023; Published: March 02, 2023
*Corresponding author: Stefano Martinotti, Director of the Unit of Predictive Medicine and Clinical Pathology, CAST- Department of Innovative Technology in Medicine and Dentistry, University of Chieti, Chieti, Italy
How to cite this article: Elena Toniato, Daniele Savio, Gian Luca Falleti, Riccardo Martinotti, Riccardo Pulcini, et al. Evaluation Of Robustness and Effectiveness of the BV-STOP, A Novel Antiseptic Compound with Specific and Anti-Microbial Activity. Adv Biotech & Micro. 2023; 17(2): 555956 DOI:10.19080/AIBM.2023.17.555956
Abstract
We have studied the effects of a new compound defined as a surface antimicrobial product, commercially named BV-STOP, whose antimicrobial activity has been evaluated in vitro according to specific guidelines from the UNI-EN. The BV-STOP has been assembled as a mixture of substances which confer potent anti-microbial activity with potential 99.9% bacterial growth inhibition. Such antimicrobial activity affects several pathogenic and saprophytic species in particular Pseudomonas , Staphylococcus, Enterococcus and Escherichia coli. Moreover thanks to the formulation conditions of the product, once distributed over flat surfaces in any type of temperature conditions, the BV-STOP can retains its antimicrobial activity even though the target surfaces are treated with conventional detergent product for routine cleaning programs. Finally, due to its particular formulation characteristics, the BV-STOP is sensitive to UV irradiation. Therefore, its adherence to surfaces can be monitored over time in order to guarantee a proper antiseptic activity. Thus, the BV-STOP may represent an ideal device for correct and perfect monitoring of antimicrobial conditions.
Introduction
There is a sense of unease in modern societies regarding infections and infectious diseases as a continuous threat to human health. According to reports from most of the major health agencies in the US and in western countries, more than four million people are estimated to acquire a healthcare-associated infection (HCAI) every year in Europe and almost 2 milions in the USA) [1]. The number of deaths occurring as a direct consequence of these infections is estimated to be at least 37,000, and these infections are thought to contribute indirectly to an additional 110,000 deaths each year (in the USA, a total of 99,000 deaths is estimated).
It is known that most sources of microbial contagions occurred on solid–air interfaces in healthcare units, such as on tables, flat surfaces of different types, computer keyboards, textiles as well as on work surfaces in factories and in private houses [2,3]. Moreover, solid–liquid surfaces are also of great concern in hospitals such as taps, showers and drains, where biofilms appear frequently. New methods, in addition or as an alternative to appropriate use of disinfectants and antibiotics, are required to reduce microbial activity [4,5]. Such new products should be associated with longer lasting effects with particular regards to the capacity of neutralizing associated infections and to reverse the increase in antimicrobial resistance. A potential and promising method to fight bacterial growth is the possibility to develop new products with intrinsic antimicrobial (nano)coatings activity. (AMC) [6]. It could be plausible to project new generation antiseptic products that activate their self- disinfecting surface through the application of coatings with bactericidal properties [7-12].
And the Bactericidal coatings are interesting in healthcare because of the capability of these coatings to kill pathogens upon contact. Antibacterial coatings may contain active eluting agents (e.g., ions or nanoparticles of silver, copper, zinc, or antibiotics, chloride, iodine), immobilized molecules that become active upon contact or active due to light exposure (e.g., TiO2 or photosensitizAbstract ers) [4]. According to this view, we present in this report the BVSTOP compound which may represent a new product capable of increasing the efficiency of bactericidal activity on hard surfaces [13-15]. It consists of a patented formulation that links properties of nanotechnology to the antimicrobial activity capable of lasting for long period and to resist the action of many passages of liquid detergents during cleaning cycles. We will show how the application of the BV-STOP in bacteria cultures can stop bacteria growth up to 99,9% of efficiency in in-vitro studies. Furthermore, the BVSTOP can be monitored through the application of UV light exposure which would detect the amount and the correct spreading of the product on target surfaces giving a high degree of confidence in the guarantee of a bacteria free item. The capability of the BVSTOP to give a long staying of antiseptic conditions is also related to the resistance up to 300 consecutive washing cycles using different sets of detergents. This of course offers a prerequisite condition for using in a large variety of surfaces either in sanitary as well in industry or home applications [16].
Test Results
All the experiments were set according to the defined Guidelines as in UNI EN 13727.
Each bacteria species was first grown and prepared as a stock culture. A sample of the product (BV-STOP) as delivered and/or diluted with hard water is added to a test suspension of bacteria in a solution of interfering substance. The mixture is maintained at 37°C for a contact time of 30 minutes. At the end of the contact time an aliquot was taken and the bactericidal and/pr bacteriostatic action in this portion is immediately neutralized or suppressed by a validated method. The method used was the neutralization/ dilution method. For each test organism two different suspensions of bacteria were prepared: the test suspension and the test validation suspension to perform the control and method validation. Preservation and stock cultures of test organism. The test organism and their stock cultures were prepared and kept in accordance with EN 12353. Working cultures. To prepare the working cultures of the test organism, we prepared a subculture from a stock culture by streaking on TSA plates and incubating at 37°C. After 18 hours we prepared a second subculture plate in the same way, and we incubated for 18 hours. We considered that a working culture.
Then we prepared a test suspension culture (N) as follows
We took 10 ml of diluent broth and placed it into a 100 ml flask with 5 grams of glass beads. We took loopfuls of bacteria from the working culture and vigorously shaken to dissolve loopfuls into the diluent solution. After that, we aspirated the 10 ml culture, and we measured bacteria concentration with a spectrophotometer at 620 nm with plastic cuvette. We adjusted the number of bacteria between 1.5 x 108 and 5 x 108 cfu/ml. We considered this a Test Suspension Culture (N). At this point we diluted the test suspension culture at 10-6 and 10-7. We plated 1 ml from these dilutions spreading onto TSA plates and incubate at 37°C for 24 hours. We kept the Vc values counting cells from plates in duplicate.
For validation method we prepared other suspension. Briefly we prepared a validation suspension culture.
We prepared a Validation Suspension (NV ) and a Validation Suspension for the neutralizing controls (NVB).
For Validation Suspension (NV ) we diluted the Test Suspension Culture with diluent to have a concentration of bacteria between 3 X 102 and 1.6 X 103 cfu/ml. For Validation Suspension for Neutralizing Control (NVB) we diluted the Test Suspension to have a concentration between 3 X 104 and 1.6 X 105 cfu/ml. For counting preparation, we diluted each Suspension (NV and NVB) 10-1 and 10-3, respectively. We plated in duplicate using TSA plates. Set of controls for validation of the dilution-neutralization system and verification of the absence of toxicity of the neutralizer.
To set the proper conditions for the method validation before calculating the bactericidal activity of the BV-STOP, we operated as follows.
i. For the validation of experimental conditions, we mixed 1 ml of interfering substance plus 1 ml of the bacteria validation suspension (range concentration: 3 X 102 - 1.6 X 103 cfu/ml.). Then we let it stand for 2 minutes. At the end of this time, we added 8 ml of sterile water and incubated for the contact time at 37°C. Finally, we took 1ml and spread onto TSA plates. This experiment was applied to test validity of the interfering substance.
ii. For the validation and absence of toxicity of the neutralizer solution, we took 1ml of bacteria suspension from the NVB (range concentration: 3 X 104 - 1.6 X 105 cfu/ml) and 9 ml of neutralizing solution. We let it stand for a few minutes at 37°C. Then we took 0.5 ml from this mixture and made a 10-1 dilution, repeating the procedure in order to have a 10-2 dilution. Finally, we plated 1 ml from the 10-2 dilution onto TSA plates. This experiment was used for testing absence of toxicity from the neutralizing solution.
iii. For the validation of the dilution-neutralizing method, we took 1 ml of the interfering solution and added 1ml of the diluent. Then we put 8ml of the BV-STOP and allowed to strand for the proper contact time. Then we took 1 ml from this mixture and 8 ml of Neutralizing solution. We let it stand for 5 minutes before adding 1 ml of the Validation Suspension. Then we incubated for 30 minutes at the target temperature. Finally, we took 1 ml and plated onto TSA plates in duplicate.
Determination of the bactericidal concentration of the BVSTOP.
The procedure for determining bactericidal concentrations was performed as follows.
We pipetted 1ml of interfering substance, and we added 1ml of the test suspension. We mixed and we let it stand for 2 min utes. Then we added 8 ml of the target BV-STOP test solution. We mixed and we let it stand for the proper contact time. After that we took 1 ml of this mixture (Na solution), we mixed with 8ml of neutralizing and 1ml of sterile water. Let it stand for 2-5 minutes. Finally, we took 1ml, and we plated duplicate onto TSA plates. This final preparation includes the neutralizing, the test product (BVSTOP), the interfering substance and the test bacteria solution. In this case any type of bactericidal activity can be tested and will determine the clear efficacy of the product. We applied these procedures to all the organisms we used for checking the bactericidal property of the BV-STOP. As you can see from data obtained from our experiments, in all organisms tested so far, we obtained a clear decrease of the bactericidal activity of BV-STOP that was accounted to be more 99% (99,9%). Such calculation was made considering the log decimal reduction of growth when we compared the TSA plating and bacteria colony formation from Na (Incubation of the BV-STOP test product with bacteria, suing interfering and neutralizing solutions) with respect to plating from N, or the plating from the validation method that tested the procedures for this experimental assay.
Caption of (Table 1). The test product considered as a potential bactericidal compound has been analysed according to uni en 13727. The test product in different concentration analysed for inhibiting a bacterial growth of stafilococcus aureus has given a value greater than 5 decimal log reduction when used undiluted and a 3,37 decimal log reduction when diluted at 50%. This means that we reached a percentage of bacterial growth reduction of more than 99%. Caption of Table 2. The test product considered as a potential bactericidal compound has been analysed according to uni en 13727. The test product in different concentration detected for inhibiting bacterial growth of pseudomonas aeruginosa has given a value greater than 5 decimal log reduction when used undiluted and a 3,34 decimal log reduction when diluted at 50%. This means that we reached a percentage of bacterial growth reduction of more than 99%.
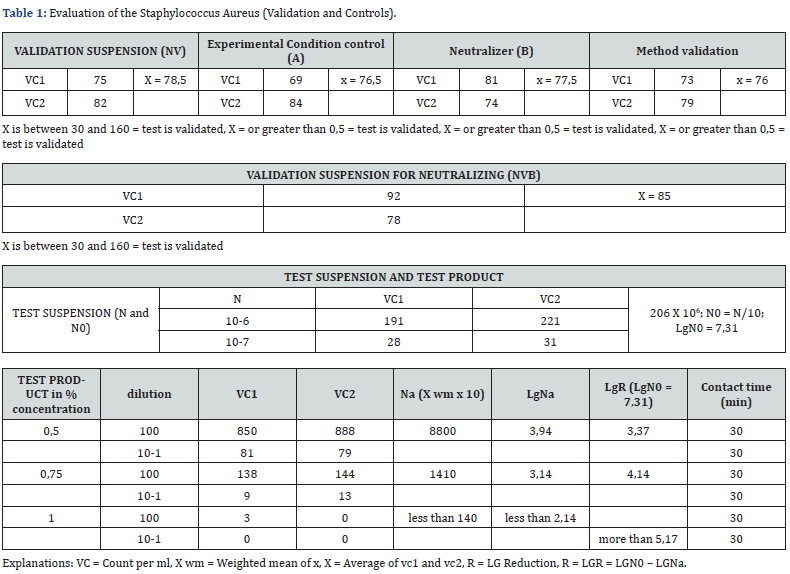

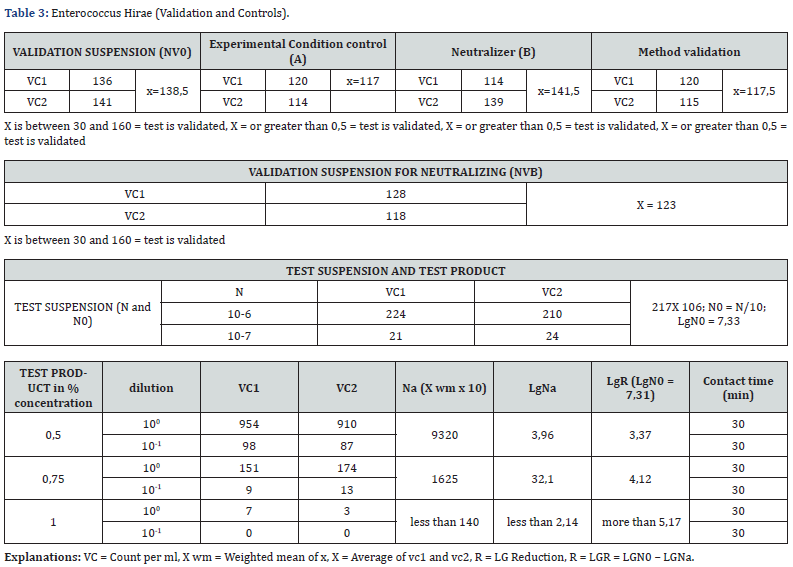
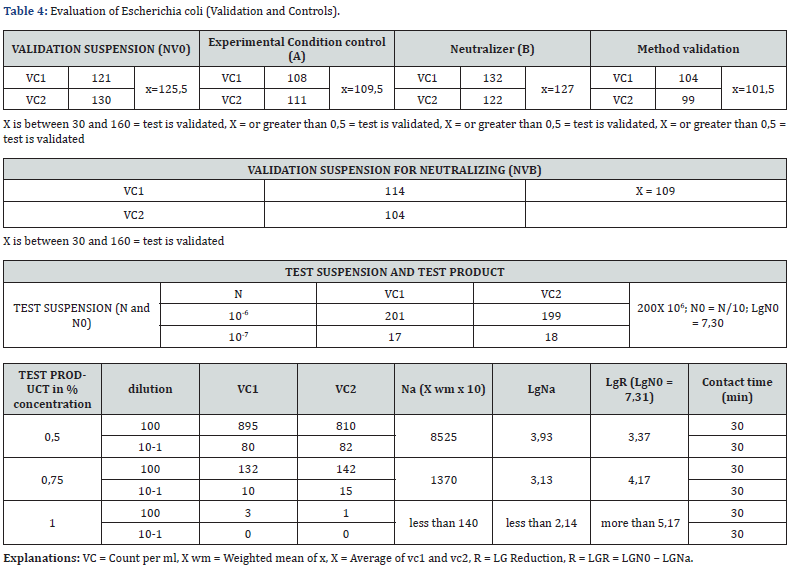
Caption of Table 3. The test product considered as a potential bactericidal compound has been analysed according to uni en 13727. the test product in different concentration detected for inhibiting a bacterial growth of enterovirus hirae has given a value greater than 5 decimal log reduction when used undiluted and a 3,37 decimal log reduction when diluted at 50%. this means that we reached a percentage of bacterial growth reduction of more than 99%. Caption of Table 4. The test product considered as a potential bactericidal compound has been analysed according to uni en 13727. The test product in different concentration detecetd for inhibiting a bacterial growth of escherichia coli has given a value greater than 5 decimal log reduction when used undiluted and a 3,37 decimal log reduction when diluted at 50%. This means that we reached a percentage of bacterial growth reduction of more than 99%.
Special Remarks Regarding the Results
A. All controls and validation were within the basic limits.
B. At least one concentration of the product demonstrated a log reduction of more than 5 lg.
C. No precipitation during the test procedure (so test mixture were homogeneous)
Conclusion
As we stated in the Results, we could conclude that the BVSTOP has a wide capacity to inhibit bacteria growth despite the virulence and aggressiveness of bacterial infection and independent of its genetic and biological property. As it is possible to check by analyzing all data, the Lg reduction value is very similar across the different bacteria species tested so far.
a) For the staphylococcus aureus the BV-STOP product at 1% concentration in the Test Product assay the R (Lg reduction) value was greater than 5,17.
b) For the staphylococcus aureus the BV-STOP product at 0,75% concentration in the Test Product assay the R (Lg reduction) value was greater 4,14.
c) For the staphylococcus aureus the BV-STOP product at 0,5% concentration in the Test Product assay the R (Lg reduction) value was greater 3,94.
d) For the Enterococcus Hirae the BV-STOP product at 1% concentration in the Test Product assay the R (Lg reduction) value was greater than 5,17.
e) For the Enterococcus Hirae the BV-STOP product at 0,75% concentration in the Test Product assay the R (Lg reduction) value was 4,12.
f) For the Enterococcus Hirae the BV-STOP product at 0,5% concentration in the Test Product assay the R (Lg reduction) value was 3,37.
g) For the Pseudomonas aeruginosa the BV-STOP product at 1% concentration in the Test Product assay the R (Lg reduction) value was greater than 5,17.
h) For the Pseudomonas aeruginosa the BV-STOP product at 0,75% concentration in the Test Product assay the R (Lg reduction) value was 4,12.
i) For the Pseudomonas aeruginosa the BV-STOP product at 0,5% concentration in the Test Product assay the R (Lg reduction) value was 3,34.
j) For the Escherichia coli the BV-STOP product at 1% concentration in the Test Product assay the R (Lg reduction) value was greater than 5,17.
k) For the Escherichia coli the BV-STOP product at 0,75% concentration in the Test Product assay the R (Lg reduction) value was 4,17.
l) For the Escherichia coli the BV-STOP product at 0,5% concentration in the Test Product assay the R (Lg reduction) value was 3,37.
Materials and Methods
The BV-STOP Product.
The BV-STOP is manufactured by Nanoprom Chemicals (Veggia-Villalunga, (RE), Italy.
a. Storage conditions (temp and other) of the product were at room temperature (RT), and in darkness.
b. The appearance of the product can be described as liquid, clear, yellowish.
c. Active substance(s) and their concentration(s): were not indicated unless present in the formulation (see patent….). Light sensitive additives capable to entrap UV light and to release in the visible spectrum were present and determine the capability to check adhesiveness of the product during the time.
d. Product diluent recommended by the manufacturer for use: used as such. If applicable, it could be diluted into potable water.
Experimental conditions and test organisms
Product diluent: hard water; concentrations of the product tested.
Test-organisms: Pseudomonas aeruginosa ATCC 15442, Staphylococcus aureus ATCC 6538, Enterococcus hirae: ATCC 10541, Escherichia Coli ATCC 10536
Test temperature: test temperature 25°c; contact time: 30 min.
Interfering substance: 0,3 g/ml bovine albumin.
Incubation temperature: 37°C.
All bacteria tests were performed according to UNI EN 13727.
General Description of The Procedure
Principle
A sample of the product as delivered and/or diluted with sterile water is added to a test suspension of bacteria in a solution of interfering substance. The mixture is maintained at one temperature and the contact time applied. At the end of this contact time an aliquot is taken; the bactericidal or bacteriostatic action in this portion is immediately neutralized by a validated method. The method of choice we used is the dilution/neutralization method. The numbers of surviving bacteria in each sample are determined and the reduction is calculated as LgR value that represents the Lg reduction comparable to the controls.
Culture media and reagents
i. Double distilled water autoclaved and keeps sterile.
ii. Tryptone Soya Agar Plates
a. Tryptone (15g)
b. Soya peptone (5g)
c. Sodium chloride (5g)
d. Agar (15g)
e. Water up to 1000ml
f. Autoclave and then make plates for bacteria growth
iii. Diluent for bacteria suspension
a. Tryptone (1g)
b. Sodium chloride (8.5 g)
c. Water up to 1000ml
d. Autoclave, check pH adjusting at pH 7.2.
iv. Neutralizer
a. Sodium thiosulfate (3g/L)
b. Polysorbate 80 (30g/L)
c. Lecithin (3g /L)
d. Catalase (0.25g/L)
v. Hard water
a. Make solution A (19.84 g MgCl2, 46.24g CaCl2 dilute into 1000 ml water, autocave)
b. Make solution B (35.02g NaHCO3, dilute to 1000 ml, sterilize by filtration [0.22 nm].
c. To make Hard water take 6 ml of solution A and 8 ml solution B dilute to 1000ml. Check pH (7.2).
vi. Clean Conditions
Prepare 0.3 bovine serum albumin (BSA) into 100 ml water. Sterilize by filtration. This is a 10X solution.
Procedure
Preparing Test Organism Suspensions and Product Test Solutions
Test organism suspension (N)
Prepare a working culture of the test organism by streaking from the stock.
a. Incubate at 37°C and repeat the procedure at least two times.
b. Then make a Test Suspension (N) of bacteria growth. Proceed adding 10 ml of broth diluent into a 100 ml sterile flask. Then take all bacterial cells from the TSA plate and dislodge into the diluent. Add glass beads and shake the flask for 3 minutes. Then aspirate 10 ml and transfer it into a bacterial tube. Using a spectrophometer read the bacteria density at 620 nm using a cuvette. Adjust to have between 1-5 and 5 x 108 bacteria cells/ml.
c. To carefully calculate the number of bacteria in the suspension, count plating from a 10-6 and 10-7 dilution of the N bacterial suspension. Prepare TSA plates in duplicate, apply 1 ml of the diluted bacterial, spread with a speed and incubate at 37°C as usual. Count in duplicate. Take a record. The good number for the N suspension should be around 3 x 108/ml.
Validation Suspension (make NV and NVB)
a. To prepare the Validation Suspension (NV) just take the test suspension (N) and dilute with the broth diluent to obtain between 3 x 102 cfu/ml – 1,6 x 103 cfu/ml. This is about one fourth of the 10-5 dilution of the N suspension.
b. To prepare the validation suspension for the neutralizer control (NVB) just take the test suspension (N) and dilute to obtain between 3 x 104 and 1,6 x 105 cfu/ml.
c. Then plate in duplicate using the TSA plates and record the number of cfu obtained.
d. It is important to count with precision that you make a 10-1 dilution from NV and a 10-3 dilution from NVB.
Product test solutions
a. The product test solution should start from a product that has 1.25 times the desired test concentration because any test for evaluating the bactericidal property of the product itself has a higher test concentration of 80%. However, we use the final product and then we arrange a 0.75% and a 0.5% concentration diluting the test solution with double distilled water. For ready-to-use products for BV-STOP it is recommended to use double distilled water instead of hard water. However, a test with hard water as a dilution solution could be made, but a further control for hard water could be considered since we must exclude that hard water may interfere with the bactericidal activity.
b. Experimental conditions should be as follows: contact time 30 minutes, temperature of contact time, 25°C.
c. The deviation for every contact time should be +/- 10 seconds.
d. Choice of method to proceed should be the dilution-neutralization method.
Test Na that means determination of bactericidal concentrations
a. Pipette 1 ml of interfering substance (10X BSA solution) and 1 ml of test suspension (N)
b. Contact time at 25°C for 2 minutes.
c. Add 8 ml of test solution and apply contact time for 30 minutes at 25°C.
d. Then take 1 ml of this suspension, then add 1 ml water, 8 ml of neutralizer.
e. Plate using TSA plates.
f. Consider that in this case the test mixture Na contains neutralizer, product test solution, interfering substance, and test suspension (N).
Validation method and performing control A, B and C
i. For control A validation we want to verify any lethal effect in the test conditions, so we must proceed as follows.
ii. Pipette 1 ml of interfering substance. Add 1 ml of the validation suspension (NV)
iii. Contact time 2 minutes at 25°C.
iv. Then add 8 ml of water.
v. Contact time 30 minutes.
vi. Then take 1ml and plate in duplicate using TSA plates. Incubate at 37°C.
vii. For Control B Validation we do want to verify the absence of toxicity of the neutralizer.
viii. Pipette 9ml of neutralizer then add 1 ml of validation suspension (NVB ).
ix. Then take 0.5 ml of this mix and transfer in tube containing 4.5 ml of neutralizer (10-1 dilution).
x. Repeat taking 0.5 ml from this mix and put into a tube with 4.5 ml neutralizer (10-2 dilution)
xi. Contact time for 5 minutes.
xii. Then plate in duplicate from the 10-2 dilution.
xiii. For Control C validation. We do want to validate the dilution neutralization method
xiv. Pipette 1ml of interfering substance, 1 ml of broth diluent and 8 ml of product test solution. Make this experiment only from ready to use product. Do not make the same for the 0.75% and 0.5% dilutions.
xv. Contact time for 30 minutes at 25°C.
xvi. Take 1 ml of this mixture and add 8 ml neutralizer.
xvii. Contact time for 5 minutes at 25°C.
xviii. Then add 1 ml of validation suspension (NV)
xix. Contact time for 30 minutes.
xx. Then take 1ml and plate in duplicate using the TSA plates.
xxi. Incubate
Calculation And Applying General Criteria for Bactericidal Activity of The Product.
Determining the Vc value
i. The Vc value is the number of colonies once we have plated 1 ml into TSA plates. For good counting colonies from TSA plates should be between 15 and 300. In this UNI EN a 10% deviation is accepted. So, you should count between 14 and 330.
ii. For calculation of N and N0 consider that N is the test suspension and bacteria concentration is determined plating from a 10-6 and 10-7 dilution. N0 is the counting of test suspension after adding the product and the interfering substance at time 0 that is before the contact time. Bacteria concentrations should not be affected, and it is usually one tenth of the N as test suspension.
iii. Formula for calculating N. N is number of cell bacteria per ml in the test suspension.
So it is:

Where
c = sum of the Vc values
n1= is the number of Vc values, usually two for the 10-6 dilution
n2= is the number of Vc values, usually two for the 10-7 dilution
10-6 is the factor corresponding to the lower dilution.
Calculation of Na
a. Na is the number of bacteria survivors per ml in the test mixture. at the end of contact time and before neutralization. It is tenfold higher than the Vc values due to the adding of neutralizer and water.
b. So, if you calculate the meaning of dilution spit, we will have Na and Na-1.
c. Formula is:
Na= 10c/n
Where
c= sum of the Vc values considered
n= number of Vc values
Calculation of NV, NV0, NVB
NV is the number of cells per ml in the validation suspension and it is tenfold higher than the counts in terms of Vc values due to the 10-1 dilution step.
a) NV0 is the number of bacteria cells in mixture A, B or C at the beginning of contact time (time 0). In the case of neutralizer control B – dilution/neutralization method it is the number of cells per ml after 100-fold dilution. NV0 is one-tenth of NV, in case of NVB is one-thousandth.
b) So according to the we must consider the formula:
c) NV = 10c/n
d) NVB = 1000c/n
e) NV0 = c/n
Calculation of A, B, and C
1. A, B and C are the bacteria survivors in the experimental conditions control A, Neutralizer control B, and method validation C.
2. Let us consider the formula:
3. A, B, and C = c/n
4. Where
5. c is the sum of Vc values considered
n is the number of Vc values
References
- European Centre for Disease Prevention and Control (ECDC) (2013) Annual epidemiological report on communicable diseases in Europe
- Bhattacharyya A, Duraiswamy S (2018) Antimicrobial (nano) coatings for fighting hospital-acquired infections: Prospects, challenges, and opportunities. Journal of Materials Chemistry B 6(22): 3573-3590.
- Hashem A, Abd-El-Haleem D (2017) Antimicrobial coatings for textiles. Journal of Textile Science and Technology 3(2): 1-7.
- Huang X, Li Y, Wang Z, Li Z (2019) A review: Photocatalytic disinfection on indoor air and surface with TiO2-based materials. Journal of Environmental Sciences 78: 51-64.
- Jaiswal S, Duffy B, Jaiswal A K (2019) Antimicrobial coatings for the prevention of bacterial and fungal biofilms. Applied Microbiology and Biotechnology 103(3): 1049-1062.
- Xiu Z M, Zhang Q B, Puppala H L, Colvin V L, Alvarez P J (2012) Negligible particle-specific antibacterial activity of silver nanoparticles. Nano Letters 12(8): 4271-4275.
- aruso F, Donath E (1999) Mössbauer spectroscopy: a novel method to study the self-assembly of thin films. Accounts of Chemical Research 32(5): 417-424.
- Chou S, Ka J A, Kim M (2018) Fabrication of antimicrobial film using clay and silver nanoparticles. Materials Today Communications 17: 481-486.
- Haldar J, An D, de Cienfuegos L Á (2019) Nanoparticles for antimicrobial purposes. In Nanoparticles for Biomedical Applications (pp 401-429). Elsevier.
- Hedayati MK, Taheri S (2019) Nanotechnology applications for the detection and disinfection of pathogens. Applied Microbiology and Biotechnology 103(21-22): 8565-8579.
- Huang K, Wu J (2019) Nanotechnology in food science: Functionality, applicability, and safety assessment. Journal of Food and Drug Analysis 27(3): 637-648.
- Lemire J A, Harrison J J, Turner R J (2013) Antimicrobial activity of metals: mechanisms, molecular targets, and applications. Nature Reviews Microbiology 11(6): 371-384.
- Nogueira G M, Santos V M, Sabino M A, Alves C J (2020) Advances in materials with antimicrobial properties. Trends in Biotechnology 38(9): 916-930.
- Sileika T S, Barrett D G, Zhang R, Lau K H (2017) Antibacterial and antifouling polymer coatings for marine applications. Marine Drugs 15(9): 268.
- Su Y, Guan Y, Lai K, Wang L, Huang H (2021) Coatings with bioactive agents: towards multifunctional surfaces for biomedical applications. Journal of Materials Chemistry B 9(13): 3002-3022.
- Rodrigues L, Oliveira A, Sousa S, Henriques M (2018) Candida sp. infections in patients with diabetes mellitus. Journal of Clinical Medicine 7(12): 528.






























