Integration of Ozonation with Biodegradation for Dye Wastewater Treatment
Pranjal Tripathi and Singh RS*
Department of Chemical Engineering and Technology, IIT (BHU) Varanasi, Uttar Pradesh, India
Submission: November 04, 2022; Published: December 20, 2022
*Corresponding author: Singh RS, Department of Chemical Engineering and Technology, IIT (BHU) Varanasi, Uttar Pradesh, India
How to cite this article: Singh R, Pranjal T. Integration of Ozonation with Biodegradation for Dye Wastewater Treatment. Adv Biotech & Micro. 2022; 17(1): 555954. DOI:10.19080/AIBM.2022.17.555954
Abstract
Dye effluent contamination has recently reached unprecedented levels due to their widespread use in a variety of industries, including textile, leather, cosmetic, plastic, and paper. The abatement of these synthetic dyes is of a serious concern among the environmentalists. This review focus on integrated approach of ozonation and biodegradation for dye wastewater treatment. Ozonation has the potential to enhance the colour removal and biodegradability of wastewaters containing azo dyes as a pre-treatment for integrated chemical-biological treatment. This review is mainly focused on the mechanism of biodegradation and ozonation process and how their integration could be beneficial in reducing overall cost of the treatment. In order to stop dyestuffs from getting into drinking water sources, it is essential to comprehend all the phenomena and mechanisms that take place during the process of removing them from wastewater and their degradation products.
Keywords: Biodegradation; Ozonation; Textile wastewater; Hybrid process; Azo dye
Abbreviations: NADH: h nicotinamide adenine dinucleotide; FADH: Flavin Adenine Dinucleotide
Introduction
The dye and dyestuff industry is considered as one of the most polluting industries, releasing vast amounts of polluted effluents into the soil and water bodies, posing serious risks to aquatic life and the environment [1,2]. Over 10,000 commercially produced dyes are currently in use, with an estimated dye production of close to 100,000 tonnes, discharged into the environment globally [3,4]. In addition to making waterbodies unsightly, synthetic dyes reduce light penetration into the water, which has an impact on aquatic ecosystems ability to photosynthesize [5]. Therefore, it is of great concern to remove these toxic dyes from the environment, not only for aquatic life but for human life as well [6]. According to the chemical structure most commonly used dyes are azo dyes, which comprise one or more azo bonds (-N=N-). The conventional wastewater treatments fall short in deteriorating azo dyes due to their recalcitrant nature. Several physical and chemical processes comprising of adsorption, coagulation, and electrochemical oxidation have been utilized by various researchers for the degradation of the dyes. Due to their high cost and production of hazardous byproducts, these processes have a limited scope of application [7-9]. In recent years, bioremediation emerges as a suitable alternative compared to physical and chemical processes because of lesser sludge production and environmentally friendly nature [1,7,10]. However, because many of the organic substances are recalcitrant to biological treatment, the conventional biological processes alone will not provide beneficial results, particularly in case of industrial dye wastewater [11,12]. Therefore, the integration of biological processes with advanced oxidation processes (AOPs) are widely acknowledged as highly effective treatments for mutagenic dye wastewater [13,14].
The creation of hydroxyl radicals (OH•), a highly oxidizing potential radical (E0 (0) = 2.80 V) that can hasten the degradation of a variety of pollutants in a matter of minutes, is the fundamental basis of advanced oxidation processes (AOPs). The organic molecules are attacked by hydroxyl radicals produced in AOPs by removing a hydrogen atom from the molecule [15,16]. Ozonation is regarded as a highly efficacious method for dye degradation. Ozone has a strong attraction for chromophoric groups and reacts effectively with dyes resulted into simple degraded products [1,17]. The reaction of ozone with a various pollutant in aqueous solution is either directly through molecular ozone (in acidic condition) or indirectly through hydroxyl radical (OH•) (in alkaline condition) generated by ozone decomposition in water [18]. However, ozonation is best suited for total decolorization rather than mineralization. Furthermore, ozone production is also expensive because it consumes a lot of energy. This is the reason for this method to be used in conjunction with the others [11]. Biodegradation is the least expensive method of treating textile wastewater, and most frequently results in the detoxification of dyes [16]. The chromophoric groups of the dyes are recalcitrant to conventional biological treatment, which makes the integration of AOPs with biological processes a potential topic of research for dye effluent treatment [16,19].
Considering these factors, we decided to focus this review on integrated ozonation processes with biological techniques that have the best potential for use in the dye wastewater treatment. Particular emphasis was placed on comprehending the mechanisms of ozonation and biological process for dye degradation. We chose to give the readers a basic understanding of both ozonation, and biological processes used in the treatment of textile wastewater. Various ozonation reviews have been reported in the literature, but each covered this process independently. The primary goal of this review was to analyses research studies in which ozonation was used in conjunction with biological methods for dye wastewater treatment.
Biodegradation
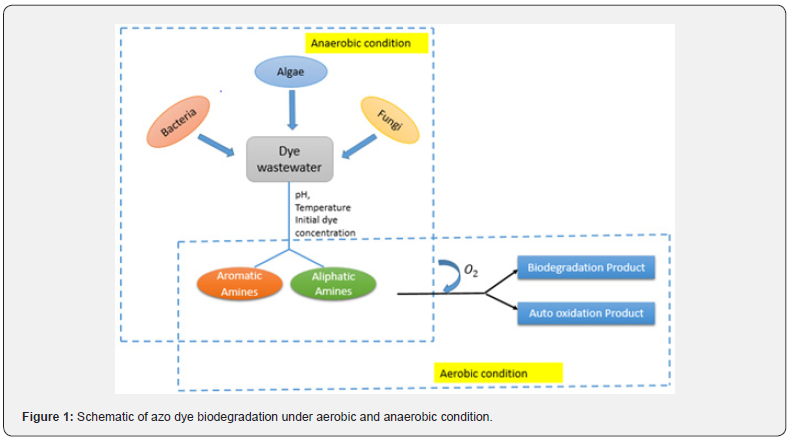
The fundamental of biodegradation is based on microbial metabolism, which degrades the pollutants in wastewater. Biodegradation is considered as the most prevalent and widely used technique for decomposing textile effluents [12]. Each microorganism has its unique degradation efficiency, and operating conditions. However, several process parameters such as pH, temperature, dye concentrations, and aerobic/anaerobic conditions have a remarkable impact on the biodegradation of various synthetic dyes [20]. The conjugate double (-N=N-) bonds in azo dye are broken down by bacteria under anaerobic conditions, producing a colourless solution that contains potentially dangerous aromatic amines. These aromatic amines are then broken down aerobically [21]. Biodegradation of dye wastewater is represented by Figure 1. Biodegradation can be classified as aerobic, anaerobic, or anoxic (both aerobic and anaerobic) depending on the nature of the microorganisms [22]. Enzymes (mainly azo reductase) primarily catalyze dye degradation under aerobic conditions. The azoreductase enzyme facilitates]. breakdown of chromophore azo bonds,].h nicotinamide adenine dinucleotide (NADH) and flavin adenine dinucleotide (FADH) acting as reducing agents [23]. This process involves transferring four electrons (reducing equivalents) to the azo linkage in two steps, with two electrons being forwarded to the azo dye (which functions as an electron acceptor) in every step, resulting in dye decolorization and formation of a colorless liquid [20,21]. It has been observed that in anaerobic environment, a relatively lower redox potential (50 mV) forms, causing the azo dyes to decolorize effectively [24]. For complete decolorization of azo dye, an amalgamation of anaerobic and aerobic process is typically preferred. The benefit of biodegradation comprises of production of less toxic sludge, low cost, and high removal efficiency. Compared to physical and chemical processes, biological processes produce sludge that is relatively less harmful to the environment [1,6]. The fact that these biological approaches can be used in situ (on site) or ex situ (off site) and microorganisms can be used as a consortium or as a pure strains, and even plants, demonstrates their adaptability [25,26].
Ozonation
Ozonation is an advanced oxidation process in which ozone is used as the oxidizing agent (O3). Because of the numerous benefits of this process, interest in using ozone in wastewater treatment has grown significantly in recent years. Because ozone is a strong oxidant, it reacts quickly with most organic pollutants, including aromatic rings and conjugated double bonds of azo dyes [27,28]. Ozonation mechanism: Ozone is a highly effective oxidizing agent that consists of a one-week single bond and one strong double bond. Ozone reacts with the substances in wastewater in two ways: in acidic environment it reacts directly as molecular ozone (O3) and in alkaline environment it reacts indirectly as secondary oxidants, such as hydroxyl radicals (OH•) [18,29-31]. Hoigne, Staehelin, and Bader (HSB) described the ozone decomposition process in aqueous solution [32,33]. The reaction sequence can be summed up as follows:

Now radical chain reaction takes place, during which H• are formed

OH⋅ that have formed, react with additional ozone via following mechanism
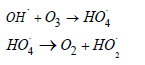
In termination step radical scavengers which consume OH• without generation of O-2 superoxide radicals. Thus, the decay of ozone may be remarkably accelerated in an alkaline solution but less effective in an acidic solution. In alkaline pH, ozone can form a more powerful hydroxyl radical oxidizing agent (E0 = 2.8 V) than its own redox potential (E0 = 2.08 V), which can break down the complex structure of dyestuffs and result in decolorization [34]. Some researchers prefer direct ozonation for dye degradation because molecular ozone have relatively higher selectivity for dye when compared to the hydroxyl (OH•) radicals. Ozone has the ability to quickly break down the double bond of azo dye, resulting in colour removal in short time intervals and subsequently it increases the biodegradability of certain dyes [35]. The ozonation process is known as one that produces relatively less sludge compared to other AOPs. The remaining ozone degrades into water and oxygen. However, ozonation is best suited for total decolorization rather than mineralization. Furthermore, ozone production is costly due to high energy consumption. That is the reason why this method must be used in conjunction with the others [31,36].
Integration of Ozonation and biodegradation: Industrial dye wastewater is generally resistant to biological treatment, an integration of chemical and biological processes for this effluent treatment is of interest. The ozonation process produces toxic byproducts and consumes a lot of energy. Because biological methods are environmentally beneficial and cost-effective, combining ozonation and biodegradation can provide a costeffective dye wastewater treatment method shown in Table 1 [12,16,37,38]. Most ozonation research is currently focused on its use as a pre-treatment in conjunction with biological processes. This is usually due to the fact that industrial effluents may contain compounds which are harmful to the microorganisms used in biological treatment.
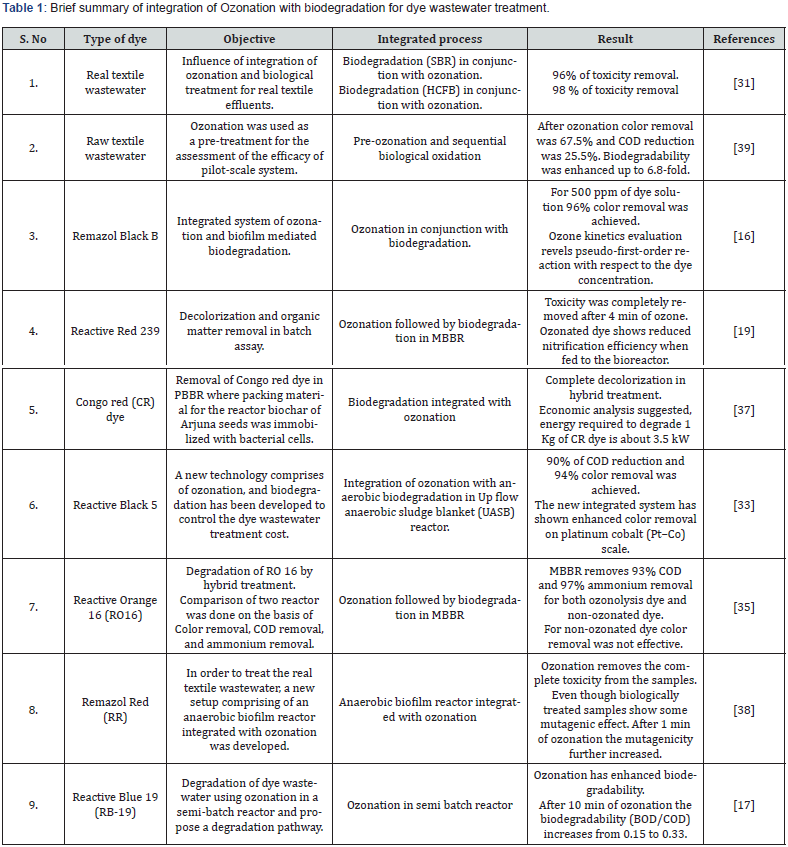
As a result, the pre-treatment aims to improve the biodegradability of that effluent samples. Paździor et al. have found 96% toxicity removal for biodegradation followed by Ozonation in SBR and 98% toxicity removal for Horizontal Continuous Flow Bioreactor (HCFB). Dias et al. have found ozonation removed colour very quickly, but the mineralization achieved was quite low. Venkatesh et al. has used ozonation in conjunction with anaerobic biodegradation using up flow anaerobic sludge blanket (UASB) and found 90% COD reduction and 94% color removal. Castro et al. have used Ozonation followed by biodegradation in moving bed bioreactor (MBBR) and found that after Ozonation, color removal was more than 97% but COD removal was only 48%. After the treatment of ozonolysis product in MBBR the COD removal was 93%. Punzi et al. found that the integrated anaerobic-ozonation process has successfully removed up to 99% of the colour and COD removal was up to 85-90%, and toxicity was also reduced for 100-1000 mg/l of the Remazol Red dye.
Conclusion
In the case of non-biodegradable dye sample, it is advised to use chemical processes as pre-treatment in order to improve its biodegradability. Despite the fact that chemical pretreatment is used to increase biodegradability, it is also possible that intermediate formed after chemical treatment may be toxic for the microorganisms and inhibit their metabolic pathways. Ozonation as a pre-treatment for integrated ozonation-biodegradation has the potential to remove the colour and enhance the biodegradability of dye-containing wastewaters. Once the process parameters are optimized for the ozonation process, it becomes economically advantageous because it requires less ozone for compound mineralization, and after that complete mineralization will be achieved by biological process at lower operational costs than chemical processes. It has been demonstrated that ozonation and biodegradation combined have the potential to completely decolorize and significantly detoxify dyeing wastewater. Because the process uses a lot of electricity, solely ozonation-based technology is typically not a cost-effective way to process textile wastewater on a large scale.
References
- Kumar Sonwani R (2021) Construction of integrated system for the treatment of Acid orange 7 dye from wastewater: Optimization and growth kinetic study. Bioresour Technol 33: 125478.
- Bhatia D, Sharma NR, Singh J, Kanwar RS (2017) Biological methods for textile dye removal from wastewater: A review. Crit. Rev. Environ Sci Technol 47: 1836-1876.
- Maurya KL, Swain G, Sonwani RK, Verma A, Singh RS (2021) Bioremediation of Congo red in an anaerobic moving bed bioreactor: Process optimization and kinetic modeling. Bioresour Technol Reports 16: 100843.
- Singh R, Singh P, Singh R (2014) Bacterial decolorization of textile azo dye acid orange by staphylococcus hominis RMLRT03. Toxicol Int 21(2): 160-166.
- Sonwani RK, Swain G, Giri BS, Singh RS, Rai BN, et al. (2020) Biodegradation of Congo red dye in a moving bed biofilm reactor: Performance evaluation and kinetic modeling. Bioresour Technol pp. 302.
- Bharti V (2019) Biodegradation of methylene blue dye in a batch and continuous mode using biochar as packing media. Environ Res 171: 356–364.
- Abu Talha M (2018) Bioremediation of Congo red dye in immobilized batch and continuous packed bed bioreactor by Brevibacillus parabrevis using coconut shell bio-char. Bioresour Technol 252: 37-43.
- Padmanaban VC, Geed SRR, Achary A, Singh RS (2016) Kinetic studies on degradation of Reactive Red 120 dye in immobilized packed bed reactor by Bacillus cohnii RAPT1. Bioresour Technol 213: 39-43.
- Mohan S, Oke N (2022) Application of the Optimized Pre-ozonation Treatment for Potential Resource Recovery from Industrial Textile Effluent. Ozone Sci Eng 44: 236-249.
- Ledakowicz S, Żyłła R, Paździor K, Wrębiak J, Sójka-Ledakowicz (2017) J. Integration of Ozonation and Biological Treatment of Industrial Wastewater from Dyehouse. Ozone Sci Eng 39: 357-365.
- Ledakowicz S, Pázdzior K (2021) Recent achievements in dyes removal focused on advanced oxidation processes integrated with biological methods. Molecules 26(4): 870.
- Chaturvedi A, Rai BN, Singh RS, Jaiswal RP (2022) A comprehensive review on the integration of advanced oxidation processes with biodegradation for the treatment of textile wastewater containing azo dyes. Rev Chem Eng 38: 617-639.
- Irani R, Khoshfetra AB, Forouzesh M (2021) Real municipal wastewater treatment using simultaneous pre- and post-ozonation combined biological attached growth reactor: Energy consumption assessment J Environ Chem Eng 9: 104595.
- Asghar A, Raman AAA, Daud WMAW (2015) Advanced oxidation processes for in-situ production of hydrogen peroxide/hydroxyl radical for textile wastewater treatment: A review J Clean Prod 87: 826-838.
- Malik SN, Ghosh PC, Vaidya AN, Mudliar SN (2020) Hybrid ozonation process for industrial wastewater treatment: Principles and applications: A review J Water Process Eng 35: 101193.
- De Souza SM de AGU, Bonilla KAS de Souza, AAU (2010) Removal of COD and color from hydrolyzed textile azo dye by combined ozonation and biological treatment. J Hazard Mater 179 (1-3): 35-42.
- Fanchiang JM, Tseng DH (2009) Degradation of anthraquinone dye C.I. Reactive Blue 19 in aqueous solution by ozonation. Chemosphere 77: 214-221.
- Tapalad T, Neramittagapong A, Neramittagapong S, Boonmee M (2008) Degradation of congo red dye by ozonation. Chiang Mai J Sci 35: 63-68.
- Dias NC, Bassin JP, Sant’Anna GL, Dezotti M (2019) Ozonation of the dye Reactive Red 239 and biodegradation of ozonation products in a moving-bed biofilm reactor: Revealing reaction products and degradation pathways. Int Biodeterior Biodegrad 144: 104742.
- Srivastava A, Rani RM, Patle DS, Kumar S (2022) Emerging bioremediation technologies for the treatment of textile wastewater containing synthetic dyes: a comprehensive review. J Chem Technol Biotechnol 97: 26-41.
- Kishor R (2021) Efficient degradation and detoxification of methylene blue dye by a newly isolated ligninolytic enzyme producing bacterium Bacillus albus MW407057. Colloids Surfaces B Biointerfaces 206: 111947.
- Geed SR, Prasad S, Kureel MK, Singh RS, Rai BN (2018) Biodegradation of wastewater in alternating aerobic-anoxic lab scale pilot plant by Alcaligenes sp. S3 isolated from agricultural field. J Environ Manage 214: 408-415.
- Jamee R, Siddique R (2019) Biodegradation of synthetic dyes of textile effluent by microorganisms: an environmentally and economically sustainable approach. Eur. J Microbiol Immunol 9: 114-118.
- Bromley-Challenor KCA (2000) Decolorization of an azo dye by unacclimated activated sludge under anaerobic conditions. Water Res 34: 4410-4418.
- Vikrant K (2018) Recent advancements in bioremediation of dye: Current status and challenges. Bioresour Technol 253: 355-367.
- Ghosh A, Dastidar MG, Sreekrishnan TR (2017) Bioremediation of chromium complex dyes and treatment of sludge generated during the process. Int Biodeterior Biodegrad 119: 448–460.
- Zhang H, Duan L, Zhang D (2006) Decolorization of methyl orange by ozonation in combination with ultrasonic irradiation. J Hazard Mater 138: 53-59.
- Gökçen F, Özbelge TA (2006) Pre-ozonation of aqueous azo dye (Acid Red-151) followed by activated sludge process. Chem Eng J 123: 109-115.
- Bilińska L, Gmurek M (2021) Novel trends in AOPs for textile wastewater treatment. Enhanced dye by-products removal by catalytic and synergistic actions. Water Resour Ind 26: 100160.
- Arslan-Alaton I, Alaton I (2007) Degradation of xenobiotics originating from the textile preparation, dyeing, and finishing industry using ozonation and advanced oxidation. Ecotoxicol Environ Saf 68: 98-107.
- Paździor K (2017) Influence of ozonation and biodegradation on toxicity of industrial textile wastewater. J Environ Manage 195: 166-173.
- Staehelln J, Hoigne J (1985) Decomposition of Ozone in Water in the Presence of Organic Solutes Acting as Promoters and Inhibitors of Radical Chain Reactions. Environ Sci Technol 19: 1206-1213.
- Venkatesh S, Venkatesh K, Quaff AR (2017) Dye decomposition by combined ozonation and anaerobic treatment: Cost effective technology. J Appl Res Technol 15: 340-345.
- Gharbani P, Tabatabaii SM, Mehrizad A (2008) Removal of Congo red from textile wastewater by ozonation. Int J Environ Sci Technol 5: 495-500.
- Castro FD, Bassin JP, Dezotti M (2017) Treatment of a simulated textile wastewater containing the Reactive Orange 16 azo dye by a combination of ozonation and moving-bed biofilm reactor: evaluating the performance, toxicity, and oxidation by-products. Environ Sci Pollut Res 24: 6307-6316.
- van Leeuwen J (2009) Ozonation within an activated sludge system for azo dye removal by partial oxidation and biodegradation. Ozone Sci Eng 31: 279-286.
- Goswami M (2020) Application of Arjuna (Terminalia arjuna) seed biochar in hybrid treatment system for the bioremediation of Congo red dye. Bioresour Technol 307: 123203.
- Punzi M (2015) Combined anaerobic-ozonation process for treatment of textile wastewater: Removal of acute toxicity and mutagenicity. J Hazard Mater 292: 52-60.
- Somensi CA, Simionatto EL, Bertoli SL, Wisniewski A, Radetski CM (2010) Use of ozone in a pilot-scale plant for textile wastewater pre-treatment: Physico-chemical efficiency, degradation by-products identification and environmental toxicity of treated wastewater. J Hazard Mater 175(1-3): 235-240.






























