The Histology of Background Pathological Processes in Cynomolgus Monkeys (Macaca fascicularis) from Control Groups in Toxicology Studies
Aurika G Anikina1*, Mariia A Varavko2, Aleksei A Aleksandrov3 and Yakow Yu Ustiugov4
1Senior Research Associate, Laboratory of Pathology, JSC BIOCAD, Russia
2Head of Laboratory of Pathology, JSC BIOCAD, Russia
3Head of Experimental Biology Division, Preclinical Studies Department, JSC BIOCAD, Russia
4Head of the Preclinical Studies Department, JSC BIOCAD, Russia
Submission:September 12, 2022; Published: October 14, 2022
*Corresponding author: Aurika G Anikina, Senior Research Associate, Laboratory of Pathology, JSC BIOCAD, Russia
How to cite this article: Anikina A G, Varavko M A, Aleksandrov A A, Ustiugov Ya Yu. The Histology of Background Pathological Processes in Cynomolgus Monkeys (Macaca fascicularis) from Control Groups in Toxicology Studies. Adv Biotech & Micro. 2022; 17(1): 555953 DOI:10.19080/AIBM.2022.17.555953
Abstract
Histological samples of organs obtained from 72 (35 male and 37 female) cynomolgus monkeys (M. fascicularis) from negative control groups in 12 toxicological studies were analyzed retrospectively. The most common processes where focal accumulations of exogenous pigment were found in 76.4% of animals in the lungs, non-atrophic chronic gastritis, which was observed in 37.4% of animals, focal chronic quiescent colitis was noted in 36.1% of cases, infiltrate in the liver were observed in 33.3% of animals, focal chronic quiescent gastritis was identified in 30.5% of animals, lymphoplasmacytic infiltrates in the kidneys were found in 26.4% of animals. Identification and accumulation of information on background pathology findings in primates from control groups are essential since these data allow differentiating spontaneous pathology from abnormalities associated with administration of a drug test, to correctly interpret the results of toxicology studies.
Keywords: Preclinical studies; Cynomolgus monkeys; Background pathology; Histopathology
Introduction
Nonclinical safety studies with biopharmaceuticals usually use nonhuman primates, primarily cynomolgus monkeys (M. fascicularis), as a test system, since, due to their close phylogenetic relationship with humans, primates are often the only relevant animal species for assessing the safety of targeted drugs [1,2]. However, in some cases, clinically healthy monkeys have some background pathology changes [3,4] which develop either before administration of the drug or during the experiment and, in turn, when identified in a histological examination, may affect the accuracy of the results of an evaluation of toxicity studies. The correct interpretation of pathomorphological abnormalities is of utmost importance, since, in some cases, it can affect the general conclusion on a safety study of a drug and the characteristics of its toxicological profile.
The aim of this work was to describe the background histopathological findings in tissues from control cynomolgus monkeys received from the breeding facilities of the Sochi Research Institute of Medical Primatology, Russia, as well as primates imported from Vietnam. Since previous toxicology studies most often evaluated histological abnormalities in the internal organs of primates primarily from Mauritius, China and Vietnam, [5,6] it seems relevant to analyze the morphology of background lesions in monkeys used in preclinical studies conducted in the Russian Federation, followed by a comparative assessment between animals grown in the breeding facilities of Sochi and primates obtained from Vietnam.
Materials and Methods
Histological specimens of organs obtained from 72 (35 males and 37 females) cynomolgus monkeys (M. fascicularis) from negative control groups in 12 toxicology studies conducted at the Sochi Research Institute of Medical Primatology were analyzed retrospectively. All research on non-human primates was conducted in accordance with the guidelines for conducting animal experiments at the Institute of Medical Primatology in Sochi and was approved by the ethical commission of JSC “BIOCAD”, which confirms the use of animals for scientific purposes in accordance with generally recognized scientific, ethical and legal standards and imposes responsibility on researchers whose activities are subject to control by the commission on bioethics.
The study animals were aged 3 to 8 years old (the data is presented in Table 1), with body weight ranging from 2.2 to 6.1 kg. Of these, 42 monkeys (21 males and 21 females) were imported from Vietnam with subsequent quarantine, 30 animals (14 males and 16 females) were born and raised in the enclosures of the breeding facilities in Sochi. The animals were kept in individual metal cages equipped with feed bins and drinkers. The food ration consisted of complete feed, fruits, and vegetables according to the average standards of feed consumption. The animals received water from the central water supply. During the study, the following environmental conditions were maintained in the animal holding room: ambient temperature (24.5±3.5°C), relative humidity (65±5%), natural daylight duration.
Euthanasia was performed by intravenous injection of 5.0 ml of Lysthenon® (Nycomed Austria GmbH, Austria) with preliminary general anesthesia by intravenous injection of 0.10ml/kg of 2% Xylazine (Interchemie Werken “de Adelaar” BV, the Netherlands) and 0.05ml/kg of Zoletil® (Virbac Sante Animale, France). The pathological evaluation was carried out according to the regulatory requirements for preclinical studies [7]. This article provides an analysis of morphological findings in the organs of the bronchopulmonary (trachea, bronchi, lungs), cardiovascular (heart, aorta), urinary (kidney, bladder), immune (thymus, spleen), and endocrine (thyroid gland, adrenal glands, endocrine pancreas) systems, as well as the gastrointestinal organs (tongue, salivary glands, esophagus, stomach, small and large intestine, exocrine pancreas) and the organs of the hepatobiliary system (liver, gallbladder).
Samples of the studied organs were fixed in a 10% neutral buffered formalin, dehydrated through a series of alcohol solutions with increasing concentrations and cytosols, and embedded in paraffin HISTAMIX (BioVitrum, Russia). [3-4] μm sections prepared on a Rotary Microtome Microm HM355S (Thermo Scientific) were stained with eosin and hematoxylin using a standard technique, as well as Perls’s staining was used to detect hemosiderin. We also used polarizing microscopy to confirm the presence of exogenous pigment in the lungs. For statistical analysis, the Pearson’s Chisquare Test was used for values of expected frequencies greater than 5, otherwise, the exact Fisher’s test was used (Statistical data processing was performed using SAS JMP 11 packages). A P-value less than 0.05 indicated statistical significance.
Results
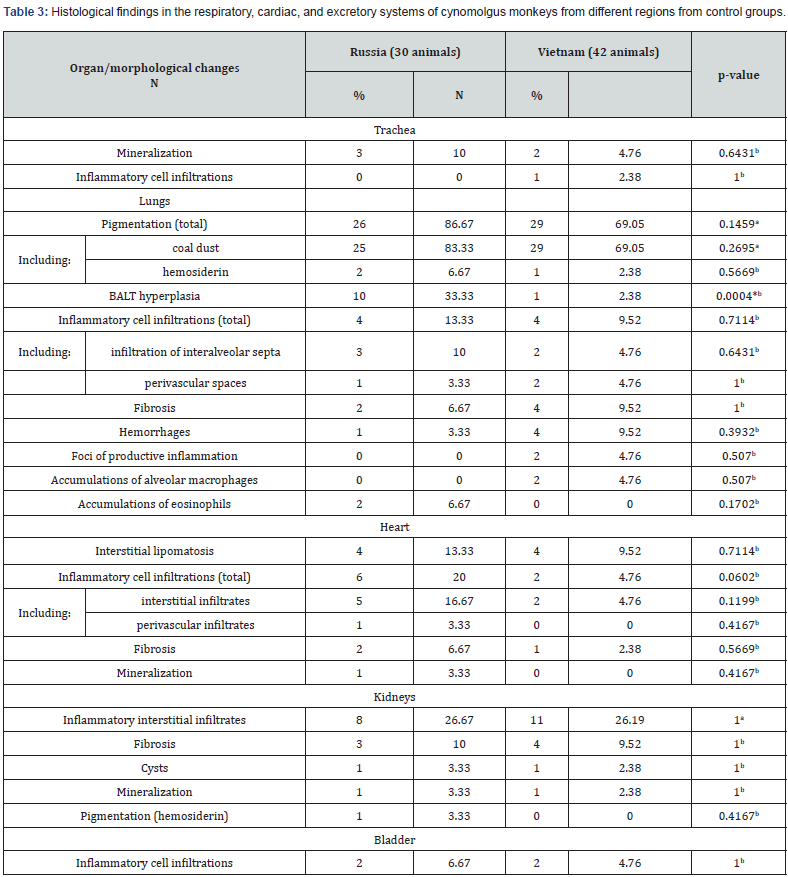
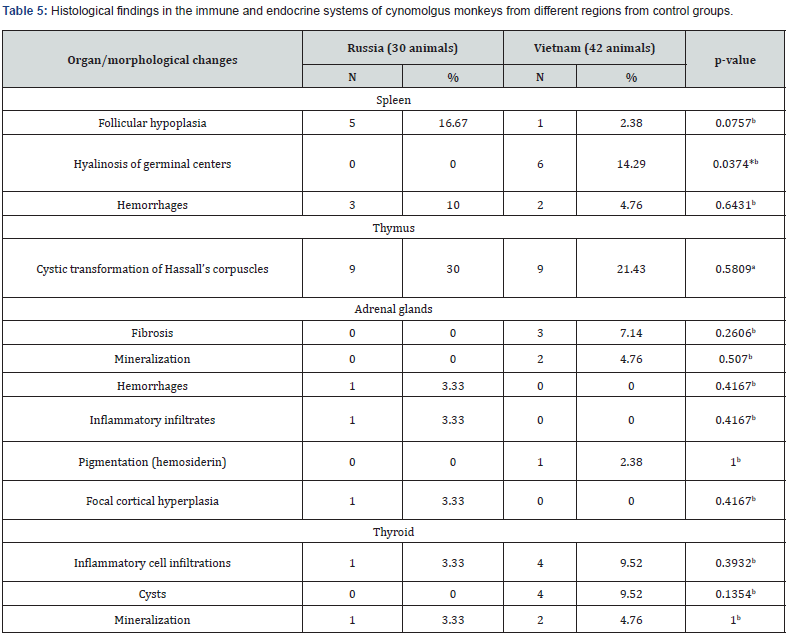
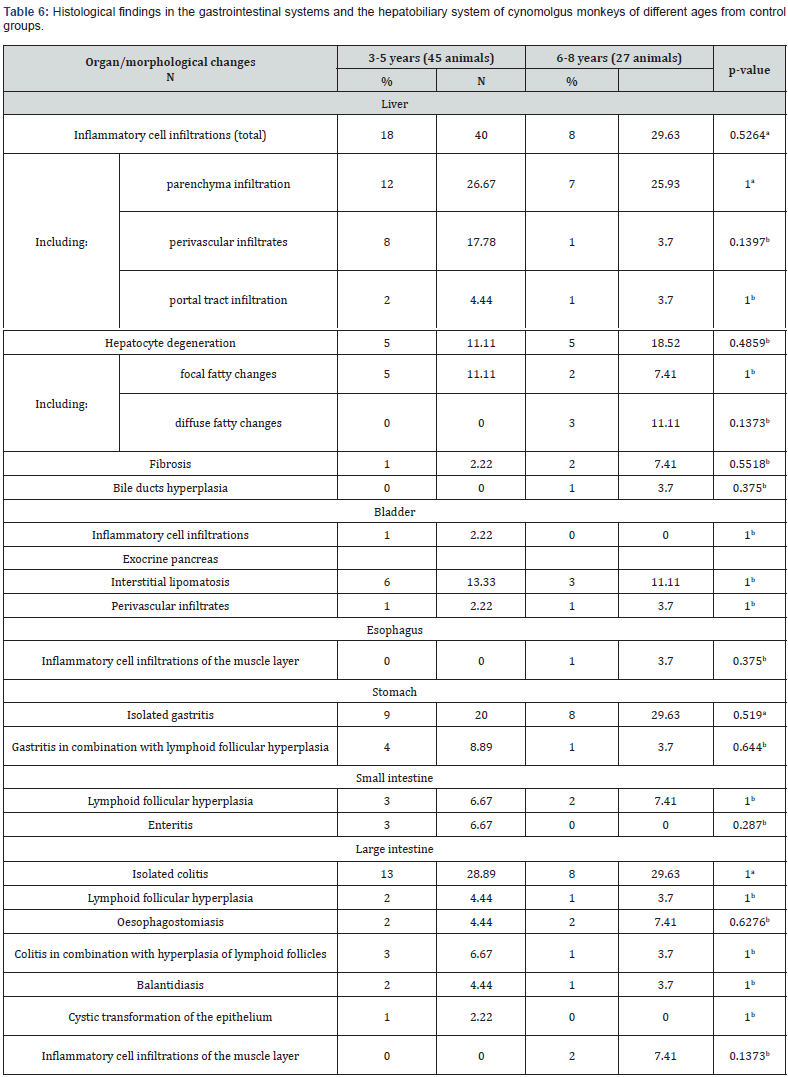
The most common lesions found in the studied organs were inflammatory infiltrates. They were found in the trachea, lungs, heart, kidneys, bladder, adrenal glands, thyroid glands, liver, gallbladder, pancreas, esophagus, stomach, and intestines. Infiltrates were usually mild, were not accompanied by destructive changes in the adjacent tissues and were mainly represented by lymphocytes and macrophages. Less common were small areas of mineralization in the tracheal cartilages, lungs, heart, kidneys, adrenal glands, and thyroid gland. Focal fibrosis of the lungs, heart, kidneys, adrenal glands, and liver was detected in some fields of view in individual animals. Other pathological processes, such as hemorrhages, cysts, hemosiderosis, interstitial lipomatosis, were found in rare cases. The following text shows the percentage of occurrence of a particular trait in all studied animals, regardless of age and birthplace. Tables 2-7 show the frequency of background findings based on age and birthplace.
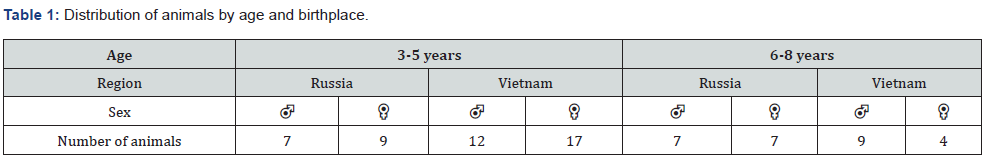
When examining the bronchopulmonary system, small foci of mineralization were found in the tracheal cartilages (Figure 1a, b) in 6.9% of the animals, while they were found only in monkeys in the age group of 6-8 years (Table 2). Mineralization of the tracheal cartilages is a common aging change, which, according to the literature, is found in some laboratory animals [8]. Isolated lymphoplasmacytic infiltrates were found in the submucosal layer of the trachea in 1.4% of primates. In the lung tissue, background changes were represented by focal accumulations of pigment, hyperplasia of bronchial-associated lymphoid tissue (BALT), focal inflammatory infiltration, focal fibrosis, and minor hemorrhages, which correlates with the literature data. [9,10] Pigment accumulations were observed in 76.4% of cases, which is 75.0% of animals was represented by small, dark gray or black particles of exogenous origin (Figure 1c, 1d), located in the cytoplasm of macrophages, in perivascular or peribronchial spaces, less often in the interalveolar septa. Granules of exogenous pigment can be explained by the proximity of the breeding facility to a large city.
Note: aPearson’s Chi-Square & Yates’s Continuity Correction, bFisher’s exact test; *P˂ 0.05.
In 4.2% of cases, the pigment was represented by golden brown hemosiderin grains, which were found predominantly in the interalveolar septa and sometimes in the alveolar lumina in siderophages. BALT hyperplasia was observed in 15.3% of monkeys. In the most majority of cases, BALT hyperplasia was found in primates from Russia (Table 3). Focal fibrosis, mainly subpleural, was observed in 6.9% of monkeys, minor hemorrhages were observed in the same percentage of cases. In addition, proliferative infiltration in the form of accumulations of lymphocytes, macrophages, plasmacytes, and fibroblasts, as well as isolated infiltration with macrophages or eosinophils (Figure 1b) were found in individual primates (2.8% of cases). Small infiltrates and BALT hyperplasia may be the result of a nonspecific response to an infectious agent or allergen; in some cases, such inflammatory foci resolve by the formation of small foci of fibrosis.
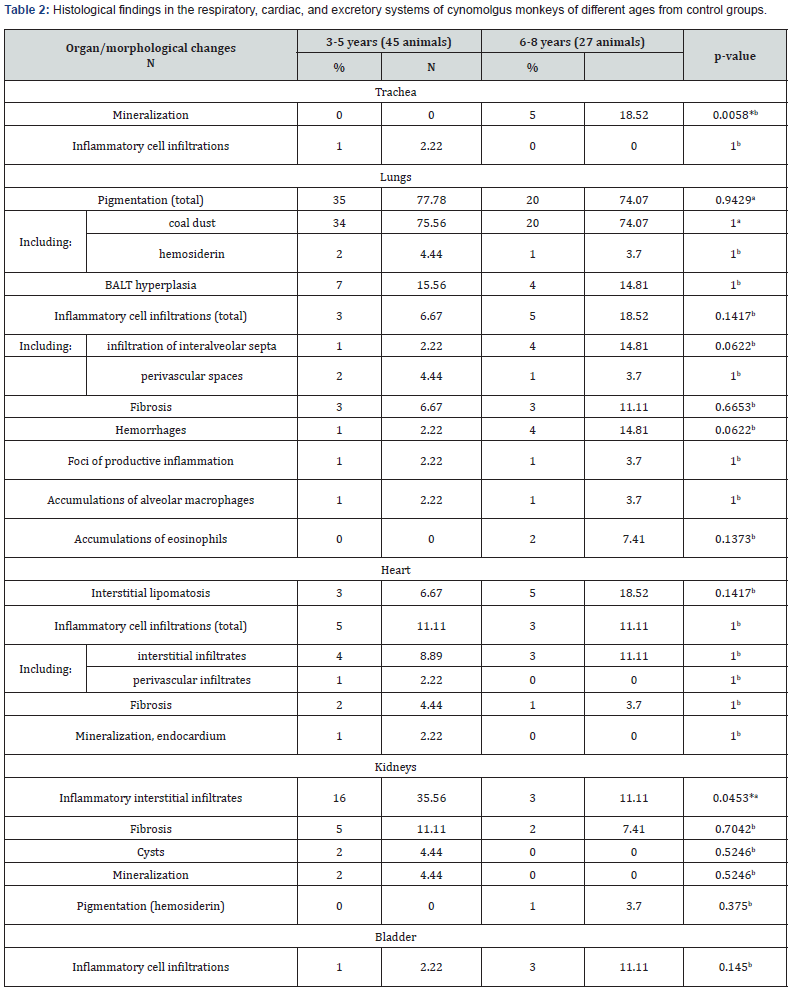
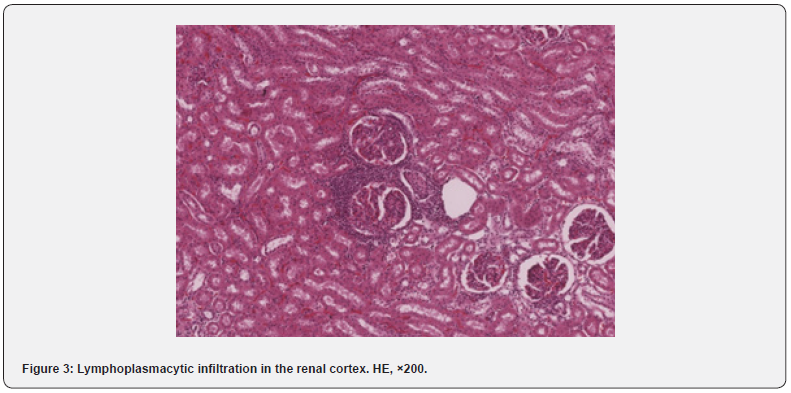
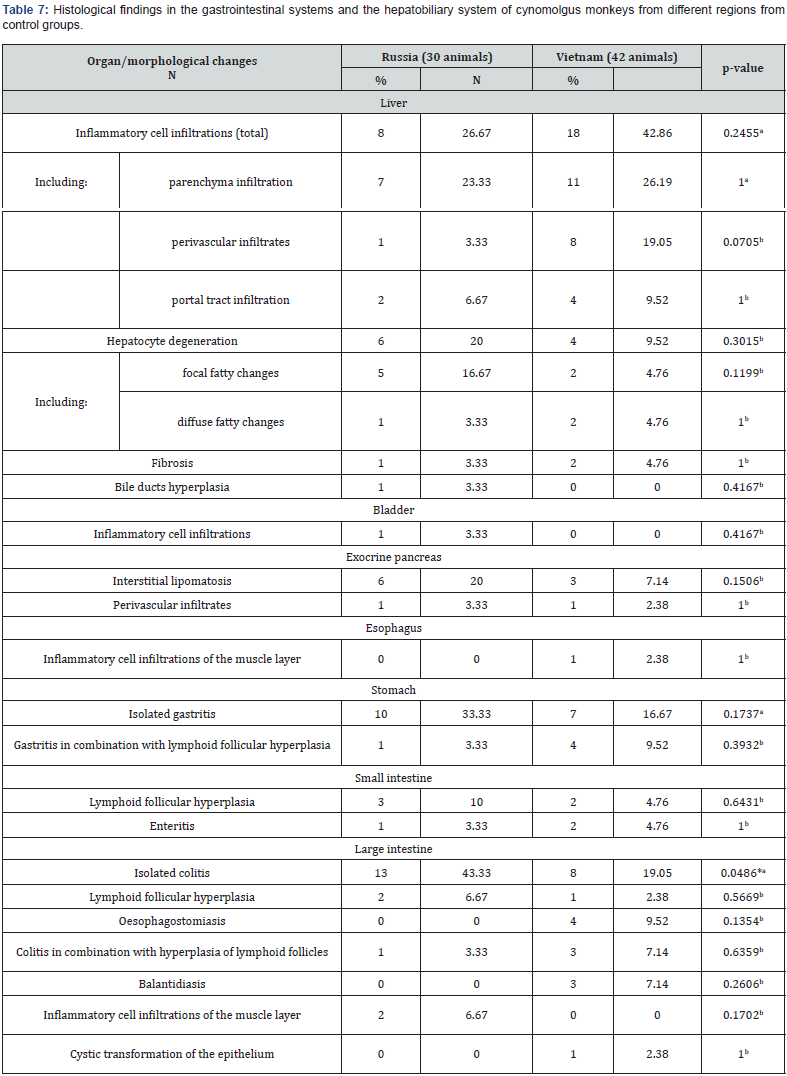
In the heart, minimal interstitial lymphoplasmacytic cell infiltration was found most often (11.1% of cases) (Figure 2a) which also occurred in the perivascular spaces of both the left and right ventricles. Areas of focal interstitial lipomatosis were observed with a similar frequency (Figure 2b), mainly in the right ventricular wall. Small foci of interstitial fibrosis were detected in 4.2% of monkeys. In one animal (1.4% of cases), a large focus of mineralization was found in the endocardium of the right ventricle (Figure 2c). No pathological changes were found in the aorta of all the animals studied. The detected changes in the myocardium were not related to the age and place of origin of primates. The detected small infiltrates and small foci of fibrosis are also described by some authors [4]. Predominant background lesions in the kidneys were minimal interstitial lymphoplasmacytic infiltrate, located mainly in the cortex (Figure 3), less often in the medulla, in 26.4% of the study animals. Infiltrates in the kidneys were more common in younger animals (Table 2). Small foci of fibrosis were found in 9.7% of cases. Cysts were detected in 2.8% of animals; foci of mineralization were found with a similar frequency, located mainly in the interstitium or in the lumina of the distal tubules of the medulla.
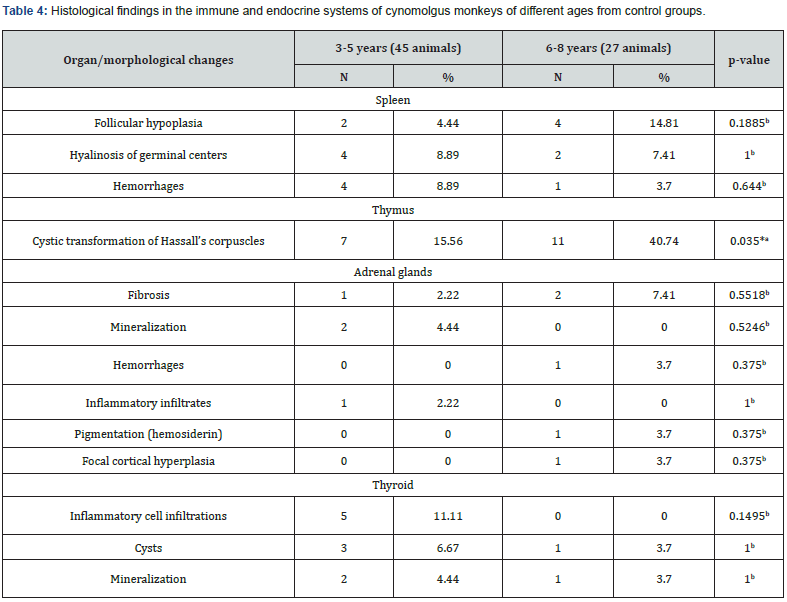
Accumulations of isolated siderophages were found in the cortical stroma in 1.4% of animals. Small lymphoplasmacytic infiltrates were most often found in the kidneys, which corresponded to the available literature data [5]. The prevalence and number of detected infiltrates do not allow suggesting interstitial nephritis; their appearance is likely to indicate compensation and adaptation as a non-specific response to adverse environmental factors. The appearance of fibrotic lesions and cysts, presumably of retention nature, as well as mineralization foci, may result from the resolution of inflammatory infiltrates. In a small number of animals (5.6%), small infiltrates represented by lymphocytes and macrophages were identified in the submucosa of the bladder. In the spleen, follicular hypoplasia and hyalinosis of germinal centers of lymphoid follicles were observed as background findings in 8.3% of animals. The appearance of hyalinosis of germinal centers may reflect the process of recent involution [11]. Follicular hyperplasia was only found in monkeys imported from Vietnam (Table 5). Slightly less often, in 6.9% of cases, there were small areas of hemorrhage in the red pulp. We have not been able to establish the obvious reasons for follicular hypoplasia that has occurred in a small number of animals and is probably a transient condition.
In 25.0% of animals, a cystic transformation of Hassall’s corpuscles was detected in the thymus, cysts were more often detected in primates in the age group of 6-8 years (Table 4). The formation of cysts at the site of necrotic Hassall’s corpuscles are physiological and is a process of thymus involution [8].
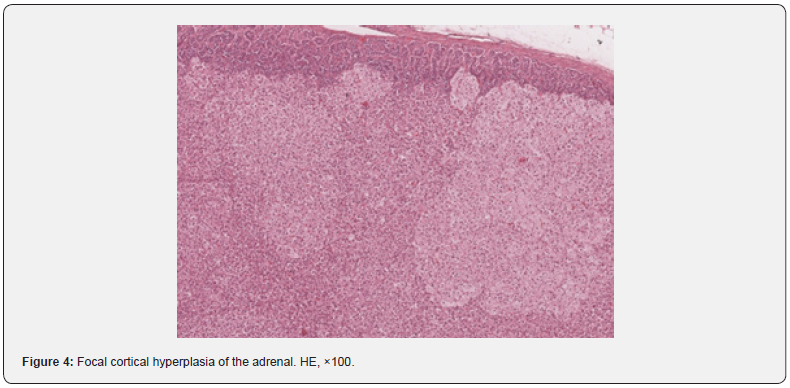
Background changes in the adrenal glands were represented by focal fibrosis, with a frequency of 4.2%. The proportion of small hemorrhages and isolated foci of encrustation with calcium salts accounted for 2.8%. One animal (1.4%) had small deposits of hemosiderin grains and in one case (1.4%) focal cortical hyperplasia occurred (Figure 4), which also has been described in cynomolgus monkeys [4]. Thyroid gland pathologic changes were found in a small number of animals. They were represented by small interstitial lymphoplasmacytic infiltrate (6.9% of cases) and single cystically dilated follicles at 5.6% of animals. Also, small foci of mineralization were found in the thyroid gland in 4.2% of cases, which were located either in the stroma or in the lumen of cystically dilated follicles. The reasons for the development of thyroid cysts are not entirely clear,4 but it is suggested that they originate from embryonic duct remnants [12].
Note: aPearson’s Chi-Square & Yates’s Continuity Correction, bFisher’s exact test; *P˂ 0.05.
Note: aPearson’s Chi-Square & Yates’s Continuity Correction, bFisher’s exact test; *P˂ 0.05.
Note: aPearson’s Chi-Square & Yates’s Continuity Correction, bFisher’s exact test; *P˂ 0.05.
Note: aPearson’s Chi-Square & Yates’s Continuity Correction, bFisher’s exact test; *P˂ 0.05.
Note: aPearson’s Chi-Square & Yates’s Continuity Correction, bFisher’s exact test; *P˂ 0.05.
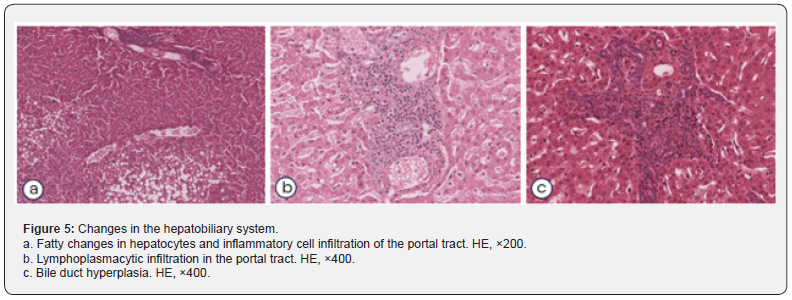
The examination of the gastrointestinal tract most often revealed background lesions in the liver. In 33.3% of cases, focal lymphoplasmacytic infiltrates were found in the liver: in the parenchyma, in the portal tracts (Figure 5a), or perivascular spaces. Also, focal fibrosis was detected in individual animals, in 4.2% of cases, mainly in portal tracts. Dystrophic changes of varying severity in hepatocytes were detected in 13.8% of animals. In 9.7% there was a focal fatty change (Figure 5b), 4.2% of animals had diffuse fatty changes of hepatocytes. Bile ducts hyperplasia (Figure 5c) was found in 1.4% of animals. Focal lymphoplasmacytic infiltration of the submucosa of the gallbladder was detected in 1.4% of animals. In the pancreas, small foci interstitial lipomatosis was detected in 12.5% of cases, focal perivascular lymphoplasmacytic infiltrates were found in 2.8% of animals.
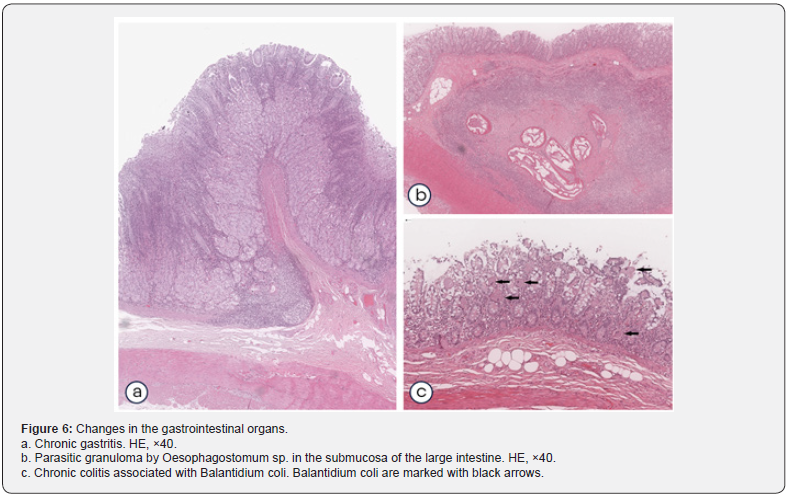
Background findings in the esophagus were represented by single small perivascular lymphoplasmacytic infiltrates in the muscle layer, which was observed in 1.4% of cases. Signs of superficial non-atrophic chronic gastritis were detected (Figure 6a) in 37.4% of monkeys, while, in 30.5% of cases, no response of the gastric lymphoid tissue was observed. In the remaining 6.9% of cases, gastritis was associated with lymphoid follicular hyperplasia. Histological changes in the small intestine were represented by isolated hyperplasia of lymphoid follicles, which was observed in 6.9% of animals, or by focal chronic enteritis detected in 4.2% of cases. The histology picture of chronic colitis was observed in 36.1% of the animals, in monkeys from Russia colitis was more common. Isolated hyperplasia of the lymphoid follicles of the large intestine was detected in 9.7% of the animals.
In 6.9% of cases, helminth infestation (Oesophagostomum sp.) occurred in the larval stage (Figure 6b), which was found only in animals exported from Vietnam (Table 4). Colitis in combination with hyperplasia of lymphoid follicles was found in 5.6% of monkeys. In 4.2% of cases, Balantidium coli infestation of the colon mucosa was detected (Figure 6c). Lymphoplasmacytic infiltration of the muscle layer of the colon was detected in 2.8% of monkeys, and cystic transformation of the epithelium was observed in 1.4% of cases. Chronic gastritis and enterocolitis often are asymptomatic and quite common in cynomolgus monkeys [13]. The development of focal chronic gastritis is mainly associated with Helicobacter pylori infection [13], which should be taken into consideration when conducting preclinical studies of drugs with an oral route of administration. Enteritis was extremely rare, and we did not establish any obvious reasons for its development. Various infectious agents, such as some species of Campylobacter, S. flexneri, Y. enterocolitica, enteropathogenic E. coli,[14] can serve as the etiological factor of chronic enteritis. In addition, physical or stressful factors, such as a change in the diet, relocation of the animal from the enclosure to the isolator during the formation of experimental groups, may also contribute to the development of both chronic gastritis and enteritis. The above may also be causes for focal colitis, in addition, colitis could be associated with parasitic diseases that included oesophagostomiasis and balantidiasis [15] in the study animals. The histological structure of the tongue and salivary glands was normal, with no signs of inflammatory infiltration, destruction, or other morphological changes.
Discussion
Most of the changes we found in the internal organs of primates both the breeding facilities in Sochi and imported from the breeding facilities in Vietnam. These findings were consistent with the descriptions by other authors. Most of the revealed histological changes in the organs from the study were found to the same extent both in animals raised in the territory of the breeding facilities in Sochi and monkeys imported from Vietnam. The frequency of detected finds also in most cases did not depend on the age of the animals. The number of animals with BALT hyperplasia and isolated colitis were statistically different, which were more common in primates raised in the breeding facilities in Sochi. Hyalinosis of the germinal centers of the spleen was more common in monkeys imported from Vietnam.
Age differences were represented by more frequent detection of mineralization foci in the tracheal cartilage and cysts in the thymus in the animals from the group of 6-8 years old. Whereas in the group of animals 3-5 years old, inflammatory infiltrates in the kidneys were more common. The number of monkeys with parasitic diseases (esophagostomosis, balantidiasis) did not differ statistically between the study regions, but there was a tendency to the predominance of parasitic infestations in imported animals. Thus, the abnormalities identified in primates used in our preclinical are generally similar to the findings described by other authors. It is advisable to further monitor and accumulate histological data on background lesions in primates, the description, and publication of which will allow better differentiating spontaneous changes from abnormalities associated with administration of a drug tested, which is essential since an incorrect interpretation of the data can lead to an incorrect assessment of safety profiles of innovative drugs.
References
- Han L, Wei X, Liu C, Miguel A Esteban, Xun Xu, et al. (2022) Cell transcriptomic atlas of the non-human primate Macaca fascicularis. Nature 604(7907): 723-731.
- Iwasaki K, Uno Y, Utoh M, Yamazaki H (2019) Importance of cynomolgus monkeys in development of monoclonal antibody drugs. Drug Metab Pharmacokinet 34(1): 55-63.
- Saravanan C, Flandre T, Hodo CL, Lewis AD, Turner, OCD et al. (2020) Research Relevant Conditions and Pathology in Nonhuman Primates. ILAR J 61(2-3): 139-166.
- Sato J, Doi T, Kanno T, Wako Y, Tuschitani M, Narama Isao et al. (2012) Histopathology of incidental findings in cynomolgus monkeys (Macaca fascicularis) used in toxicity studies. J Toxicol Pathol 25(1): 63–101.
- Chamanza R, Naylor SW, Gregori M, Boyle M, Romeike A, et al. (2022) The Influence of Geographical Origin, Age, Sex, and Animal Husbandry on the Spontaneous Histopathology of Laboratory Cynomolgus Macaques (Macaca Fascicularis): A Contemporary Global and Multisite Review of Historical Control Data. Toxicol Pathol 50(5): 607-627.
- Weinbauer G, Mecklenburg L (2022) Does Geographical Origin of Long-Tailed Macaques (Macaca fascicularis) Matter in Drug Safety Assessment? A Literature Review and Proposed Conclusion. Toxicol Pathol 50(5): 552-559.
- (2014) OECD Series on Principles of Good Laboratory Practice and Compliance Monitoring. No 16: Advisory Document of the Working Group on Good Laboratory Practice, Guidance on the GLP Requirements for Peer Review of Histopathology.
- McInnes E Background Lesions in Laboratory Animals. A Color Atlas (2012) Saunders Elsevier.
- Yamakawa Y, Ide T, Mitori H, Oishi Y, Matsumoto M, et al. (2016) Accumulation of brown pigment-laden macrophages associated with vascular lesions in the lungs of cynomolgus monkeys (Macaca fascicularis). J Toxicol Pathol 29(3): 181-184.
- Gregori M, Naylor SW, Freke MC, Chamanza R, Piaia A, Hall AP, et al. (2021) Multisite Analysis of Lesions in the Respiratory Tract of the Rat and Nonhuman Primate (Cynomolgus Monkey) Exposed to Air, Vehicle, and Inhaled Small Molecule Compounds. Toxicol Pathol 49(2): 349-369.
- Everds NE, Reindel J, Werner J (2019) Variability of Spleen and Mesenteric Lymph Node in Control Cynomolgus Monkeys (Macaca fascicularis) from Nonclinical Safety Studies: A Retrospective Assessment. Toxicol Pathol 47(1): 53-72.
- Nakamura T, Ichii O, Irie T, Mizoguchi T, Shinohara A et al. (2018) Cotton rats (Sigmodon hispidus) possess pharyngeal pouch remnants originating from different primordia. Histol Histopathol 33(6): 555-565.
- McKeag S, McInnes EF (2012) The Incidence of Lymphoplasmacytic Gastritis in the Fundus and Antrum of Cynomolgus Monkey (Macaca fascicularis) Stomachs. J Toxicol Pathol 25(4): 249-256.
- Johnson AL, Keesler RI, Lewis AD, Reader AD, Laing ST, et al. (2016) Common and Not-So-Common Pathologic Findings of the Gastrointestinal Tract of Rhesus and Cynomolgus Macaques. Toxicol Pathol 50(5): 638-659.
- Zanzani SA, Gazzonis AL, Epis S, Manfredi MT (2016) Study of the gastrointestinal parasitic fauna of captive non-human primates (Macaca fascicularis). Parasitol Res 115(1): 307-312.






























