Ovicidal Activity of Chemical Disinfectants and the Nematophagous Fungus Duddingtonia flagrans and Toxocara canis
Carolina Perin Motta1, Carolina Magri Ferraz1, Filippe Elias de Freitas Soares2, Thaís Schmidt Ferreira1, Rafael de Souza Stabenow1, Jackson Victor de Araújo3*, Fernando Luiz Tobias1, Gabriel Augusto Marques Rossi1, Thais Ferreira Feitosa4, Gabriela Sorrentino Vieira1, Vinícius Longo Ribeiro Vilela4*, and Fabio Ribeiro Braga1*
1Laboratório de Parasitologia Experimental e Controle Biológico, Universidade Vila Velha, Brasil
2Departamento de Química, Universidade Federal de Lavras, Brasil
3Laboratório de Parasitologia Veterinária, Universidade Federal de Viçosa, Brasil
4Instituo de Ciências Biológicas, Universidade Federal de Minas Gerais, Brasil
Submission:August 19, 2022; Published: September 07, 2022
*Corresponding author: Fabio Ribeiro Braga, Laboratório de Parasitologia Experimental e Controle Biológico, Universidade Vila Velha, Brasil
How to cite this article: Carolina P M, Carolina M F, Filippe E d F S, Thaís St F, Rafael d S S, et al. Ovicidal Activity of Chemical Disinfectants and the Nematophagous Fungus Duddingtonia flagrans and Toxocara canis. Adv Biotech & Micro. 2022; 17(1): 555952 DOI:10.19080/AIBM.2022.17.555952
Abstract
Toxocara canis is a zoonotic canine geohelminth that contaminate the environment with eggs which later hatch the larvae that can infect human beings. Its environmental control is a challenge and require studies focused on establishing efficient strategies for reducing the risks for public health. Thus, this study aimed to evaluate the in vitro ovicidal efficacy of two commercially disinfectants used singly, combined, or in association with the nematophagous fungus Duddingtonia flagrans (AC001) against T. canis eggs. For this purpose, seven groups were created as follow: G1 (AC001), G2 (sodium hypochlorite 2%), G3 (benzalkonium chloride 15%), G4 (AC001 + sodium hypochlorite 2%), G5 (AC001 + benzalkonium chloride 15%), G6 (sodium hypochlorite 2% + benzalkonium chloride 15%), and G7 (water - control group). There were statistically significant differences (p < 0.05) between the treated and control groups, and the egg reductions were G1 (48.2%), G2 (100%), G3 (60.9%), G4 (100%), G5 (26.9%), and G6 (100%). The use of disinfectants alone or combined among them was more effective in destroying eggs than D. flagrans. In conclusion, sodium hypochlorite 2% and benzalkonium chloride 15% were effective for destructing T. canis eggs in vitro, while D. flagrans had a moderate ovicidal action. However, the application of this fungus is promising to be used as a biological and safety strategy for decontaminating the environments where leisure activities occur and exist risks for public health.
Keywords: Toxocariasis; Biological Control; Sodium Hypochlorite 2%; Benzalkonium Chloride 15%
Introduction
The ascarid nematode Toxocara canis parasite dogs and is a zoonosis distributed worldwide. T. canis larvae can cause a severe disease in humans: the visceral larva migrans (VLM) syndrome OJHA et al. [1,2]. T. canis life cycle occurs also in the environment (soil) and consequently has been considered as a “geohelminth” [3]. Generally, helminth infections in domestic animals are controlled through administration of anthelmintic drugs. However, cases of therapeutic failure and drug resistance among canine nematodes have been described [4]. Thus, other strategies focused on controlling this parasite are required. In the case of geohelminths, although disinfectants such as sodium hypochlorite 2% and benzalkonium chloride be considered as effective, their use is limited on impermeable surfaces such as concrete or ceramic floors [5]. Thus, the application of nematophagous fungi may be as a viable control strategie [6], mainly in sandy and permeable soils. In the environment, Duddingtonia flagrans produces traps that capture and fixate these nematodes and later destroy their internal structures [7]. In Brazil, the presence of zoonotic parasites in the soil of public squares is an environmental and public health problem, requiring a One Health approach for the control. There is a risk for the infection among children and adults who use these leisure environments [8,9] because the soils are areas where these parasites are difficult to control, and their occurrence are frequently reported. Thus, the aim of this study was to evaluate the in vitro ovicidal efficacy of two commercial disinfectants and the nematophagous fungus D. flagrans against T. canis eggs, through singly or combined treatments.
Materials and Methods
This study was submitted to and approved by the Ethics Committee on the Use of Animals (CEUA- Vila Velha University, Process nº 406). The T. canis eggs were obtained through dissecting the uterus of adult females that were obtained from two parasitized dogs. The process of recovering and preparing the parasite eggs was performed according to the methodology described by Okul et al. (2010). The Sodium hypochlorite 2% (Água Sanitária Q’Boa®, Anhembi, Brazil) and benzalkonium chloride 15% (Herbalvet T.A.®, Ourofino, Brazil) were used and prepared according to manufactures instructions. The nematophagous fungus D. flagrans (AC001) was donated by the Veterinary Parasitology Laboratory of the Federal University of Viçosa. This fungus is maintained with permission at the Experimental Parasitology and Biological Control Laboratory of Vila Velha University.A solution of conidia of AC001 was used in this study. Initially, the fungus was cultured in a Petri dish of 9 cm in diameter containing potato-dextrose-agar 2% (PDA 2%) where the mycelial growth occurred after seven days. Afterwards, 5 mL of distilled water were placed in the Petri dish and the conidia were scraped from the agar surface using a glass slide. Subsequently, the suspension of conidia in water was poured into a 15 mL Falcon tube (Araújo & Maia, 1993).
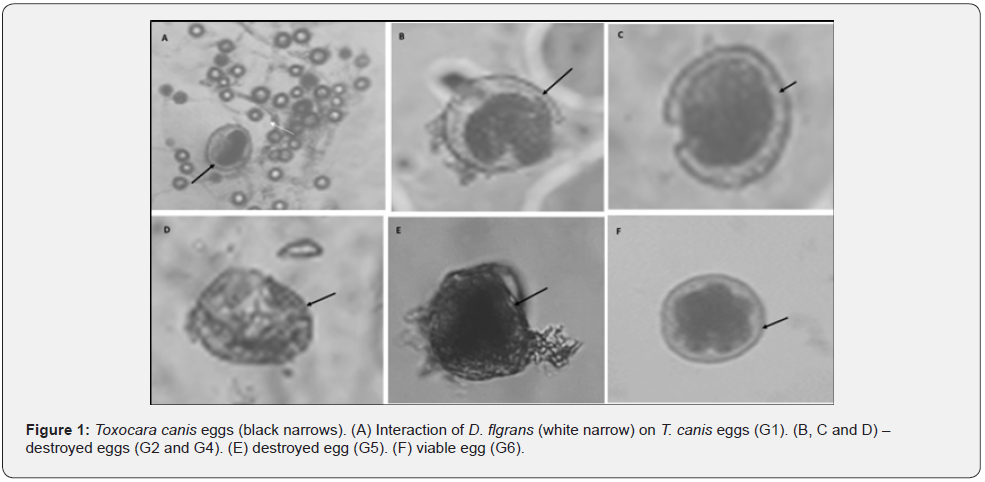
Seven experimental groups were created in 1.5 mL microtubes, and six replicates were performed for each group (Table 1). The readings were performed on the following days after assembling the tests: day one (24 hours), day seven (168 hours), day 14 (336 hours) and day 21 (504 hours). The quantities of eggs and conidia added to the microtubes was standardized by means of aliquots, such that the concentrations were approximately 120 eggs/18 μl and 120 conidia/10 μl. The volumes of sodium hypochlorite 2%, benzalkonium chloride 15% and distilled water were calculated to obtain a final volume of 100 μl per microtube.
The aliquots were homogenized, and then the content of the tubes was pipetted and deposited on glass slides under coverslips. The readings occurred under an optical microscope at 40x and 100x magnification. The remaining eggs were counted with the using a statistical manual counter and were assessed visually considering their general appearance, color, shell integrity, thickness, content appearance and other conditions that differed from the initial state. Only the eggs with visual characteristics like the initial ones were counted as surviving eggs. The results from the experiment were evaluated by means of analysis of variance (ANOVA) and Tukey’s test at 5% probability, using the BioEstat 5.0 software [10]. The reduction percentages were calculated through the equation (MENDOZA-DE-GIVES & VASQUEZ-PRATES, 1994): % Reduction= (Mean number of eggs in the control group -Mean number of eggs in the treated group)/ (Mean number of eggs in the control group) x 100
Results and Discussion
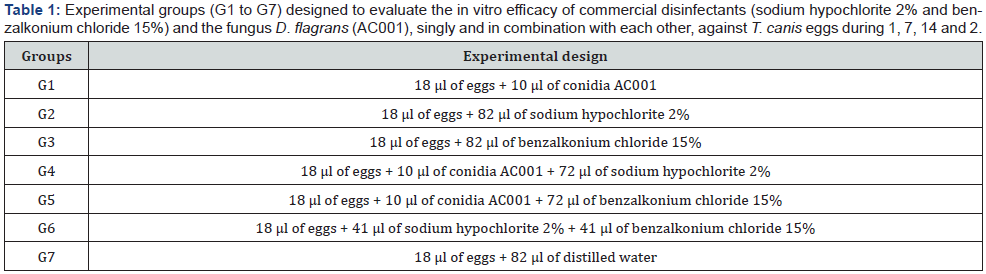
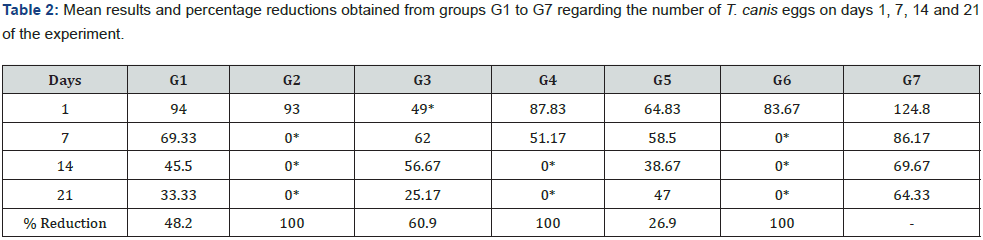
Table 2 presents the results of the efficacy of both disinfectants and D. flagrans against T. canis eggs. The egg reductions were G1 (48.2%), G2 (100%), G3 (60.9%), G4 (100%), G5 (26.9%), and G6 (100%) after 21 days. The highest ovicidal reductions were observed for groups G2 and G6, containing sodium hypochlorite 2-2.5% and sodium hypochlorite 2% + benzalkonium chloride 15% without combination to D. flagrans. The ovicidal activity of D. flagrans (G1), even lower, is important. Previously, the action of this fungus on helminth eggs had already been suggested by [11]. The authors evaluated the ovicidal capacity of AC001 on Ascaris lumbricoides and demonstrated that this fungus adhered to the eggs, suggesting an ovicidal capacity. In the present study, AC001 was able to promote a lytic effect without morphological damage to the eggshell, where hyphae and chlamydospores were adhered (Figure 1). Considering the same parasite used in this study, [12] established a reduction of 63% on T. canis larvae (L2), after 24 hours of applying AC001. This approach is very interesting due the risks that this parasite can cause for public and environmental health.
Additionally, this fungus can adhere to Schistosoma mansoni eggs and may have affect its development [13]. These finds have provided evidence of an ovicidal effect due the production of proteolytic enzymes from AC001. Morgan Jones et al. (1993) stated that fungi’s adhesion to parasite eggs can result on nematodes be unfeasible. Geohelminth eggshell are composed of protein and lipidic structures (Wharton, 1980) and the application of fungus can support on destructing them due enzymatic action [7]. Thus, our data contribute for a better understanding of an integrated and strategic control because the groups G4 and G5 (those containing D. flagrans) had an ovicidal effect, however, evidence of antagonism effects of chemical disinfectants can be observed for some groups.
The groups G2, G4 and G6, which contained 2-25% sodium hypochlorite, had morphological changes in the eggs, mainly referring to the shell, and they progressed during the time. At day one, the eggshells had thinner thicknesses and some broken eggs were observed, exposing their content, while at days 7, 14 and 21 an irregularity in the shell was observed, with smaller diameter and deterioration of the content. Additionally, it was not possible to fully visualize them, leaving only fragments of eggs and dispersed contents at days 14 and 21. [14] evaluated in vitro the hypochlorite solution of 2-2.5%, benzalkonium chloride 15% (Herbalvet T.A.®) and others of commercial genesis on the evolution of embryogenic of T. canis during 36 days. These authors observed that the sodium hypochlorite promotes the degradation of the out layer of eggs. Additionally, [5] established that this same solution was able to degenerate 50% of T. canis eggs after 24 days. In this study, eggs degeneration occurred after 21 days and agree with these studies.
The groups G3 and G5, which contained 15% benzalkonium chloride, showed lower ovicidal activity, after G1. There are reports in the literature of 15% benzalkonium chloride presenting ovicidal and larvicidal potential on geohelminths [15]. In the present work, the ovicidal efficacy of this disinfectant was also proven throughout the experiment, and after 21 days the reduction was 44.2%. The remaining eggs at 21 days showed slightly altered morphology, but many had their contents intact. On the other hand, there are studies demonstrating that the ovicidal potential of 15% benzalkonium chloride is significantly lower compared to the effect of 2-2.5% sodium hypochlorite and 70% alcohol [5,14,16]. Therefore, further comparative studies are needed, as well as associating chemical disinfectants and biological agents, to optimize the ovicidal and larvicidal potential of these agents. Thus, more updated studies are required for a better comprehension of their effects and compare chemical disinfectants and biological strategies to optimize their effects against eggs and larvae.
The G6 group had the highest percentage of ovicidal reduction. The activity of both disinfectants used (sodium hypochlorite + benzalkonium chloride) was potentiated and presented a 75.7% of ovicidal activity. This is an interesting practical result considering that many persons use two or more associated chemical products as household disinfectants, mainly in dog’s kennels and cat shelters. Ribeiro (2004) reported the importance of kennel and soil hygiene to avoid maintaining eggs and larvae in a direct transmission cycle and to prevent the infection of intermediate and paratenic hosts. However, Prats et al. (2005) highlighted that the resistance of gastrointestinal parasitic nematode eggs makes any disinfectant-based prophylaxis in soil impossible. Thus, studies performed using a soil matrice must be done for a better practical comprehension of their effects, mainly associated with nematophagous fungus D. flagrans.
Regarding the number of days used in this study, we aimed to mimic the life cycle time and the pre-patent period (21 days) of T. canis (Urqhuart et al. 1998). Since the first day of the experiment (day 1) a reduction in the number of eggs was clearly noted, mainly by the action of the chemical compounds (disinfectants) used. Ascarids have eggs with thick shells capable of resisting desiccation and temperature variations, remaining for a long time in the environment, making their elimination difficult (Tavares, 2011). According to [17-31], it is important that prevention and control of parasites be implemented to reduce environmental contamination by the infective eggs and larvae. These authors highlighted the importance of using methods to prevent eggs in the environment through disinfectants routinely used at the environments frequented by dogs and cats and our results may be extrapolated to them, contributing for reducing the possible risks for public health. However, further studies are required to improve the ovicidal percentage of these products, mainly trying to potentiate the activity when associated or with a biological control method, such as the nematophagous fungi.
Conclusion
Sodium hypochlorite 2% and benzalkonium chloride 15% were effective for destructing T. canis eggs in vitro, while D.flagrans had a moderate ovicidal action. The application of this fungus is promising to be used as a biological and safety strategy for decontaminating the environments where leisure activities occur and exist risks for public health. However, further studies using soil matrices are required to prove this practical application.
Acknowledgement
Cooperação CAPES/FAPES (Programa de Desenvolvimento da Pós-Graduação – PDPG - Edital 018) and Scolarship CNPq (Fábio Ribeiro Braga).
References
1. Selek MB, Karagoz E, Baylan O (2016) Toxocariasis: a review. Medicine Science International Medical Journal 5 (4): 1063-1067.
- Chen J, Liu Q, Liu GH, Zheng WB, Hong SJ et al. (2018) Toxocariasis: a silent threatwith a progressive public health impact. Infectious Diseases of Poverty 7(59).
- Lim-Leroy A, Chua TH (2020) Prevalence and risk factors of geohelminthiasis among the rural village children in Kota Marudu, Sabah, Malaysia. PLoS One 15.
- Castro PDJ, Howell SB, Schaefer JJ, Avramenko RW, Gilleard JS et al. (2019) Multiple drug resistance in the canine hookworm Ancylostoma caninum: an emerging threat? Parasites & Vectors 12: 576.
- Morrondo P, Diez MC, Pedreira J, Diez BN, Sánchez AR, et al. (2006) Toxocara canis larvae viability after disinfectant-exposition. Parasitology Research 99: 558-561.
- Lima JAC, Ferraz CM, Sobral SA, Geniêr HLA, Soares FEF, et al. (2020) Combined use of chemical and biological compounds to control hookworm. Journal of Helminthology 94: 1-4.
- Braga FR, Araújo JV (2014) Nematophagous fungi for biological control of gastrointestinal nematodes in domestic animals. Applied Microbiology and Biotechnology 98: 71-82.
- Batista SP, Silva FF, Valêncio BA, Carvalho GMM, Santos A, et al. (2019) Parasitos zoonóticos em solos de praças públicas no município de Sousa, Paraí Revista Brasileira de Ciência Veterinárias 26(3): 82-86.
- Gorgônio SA, Sousa DLC, Bezerra CS, Monteiro GDF, Paulo FS, et al. (2021) Agentes parasitários de importância em Saúde Única em solos de praças públicas em condições semiá Research, Society & Development 10(1).
- Ayres M, Ayres JR, Ayres DL, Santos AS (2003) BioEstat 3.0: aplicações estatísticas nas áreas das ciências biológicas e mé Sociedade Civil Mamirauá, Belém, Brasília, pp. 290.
- Braga FR, Araújo JV, Campos AK, Carvalho RO, Silva AR, et al. (2007) Observação in vitro da ação dos isolados fúngicos Duddingtonia flagrans, Monacrosporium thaumasium e Verticillium chlamydosporium sobre ovos de Ascaris lumbricoides (Lineu, 1758). Revista da Sociedade Brasileira de Medicina Tropical 40: 356-358.
- Braga FR, Ferraz CM, Silva EN, Araújo JV (2020) Efficiency of the Bioverm® (Duddingtonia flagrans) fungal formulation to control in vivo and in vitro of Haemonchus contortus and Strongyloides papillosus in sheep. 3 Biotech 10(62).
- Braga FR, Araújo JV, Campos AK, Siva AR, Araújo JM, et al. (2008) In vitro evaluation of the effect of the nematophagous fungi Duddingtonia flagrans, Monacrosporium sinense and Pochonia chlamydosporia on Schistosoma mansoni eggs. World Journal of Microbiology and Biotechnology 24: 2713-2716.
- Verocai GG, Tavares PV, Ribeiro FA, Correia TR, Scott FB (2010) Effects of Disinfectants on Toxocara canis Embryogenesis and Larval Establishment in Mice Tissues. Zoonoses and Public Health 57(7-8): e213-216.
- Suzuki T, Coelho FAS, Marson FG, Coelho MDG, Araújo AJUS (2013) Eficácia de desinfetantes comerciais na inibição da evolução de ovos de Ancylostoma spp. obtidos de cães naturalmente infectados. Revista Biociência 19: 86-92.
- Ursache AL, Mirean V, Dumitrache M, Andrei L, Stefanut L, et al. (2019) Is routine disinfection effincient in preventing contamination with Toxocara canis eggs? Journal of Helmintology 94: e60.
- Santos ECF, Carneiro MB, Tavares PV, Batista LCSO, Melo RMPS et al. (2012) Ação de diferentes desinfetantes sobre viabilidade e mortalidade de larvas de terceiro estágio de Ancylostoma spp. Revista Brasileira de Medicina Veterinária 34: 55-59.
- Braga FR, Soares FEF, Giuberti TZ, Lopes ADCGL, Lacerda T, et al. (2015) Nematocidal activity of extracellular enzymes produced by the nematophagous fungus Duddingtonia flagrans on cyathostomin infective larvae. Veterinary Parasitololgy 212: 214-128.
- Braga FR, Araújo JV, Soares FEF, Araújo JM, Genier HLA, et al. (2011) Optimizing protease production from an isolate of the nematophagous fungus Duddingtonia flagrans using response surface methodology and its larvicidal activity on horse cyathostomins. Journal of Helminthology 85: 164-170.
- Braga FR, Ara jo JV, Silva AR, Araújo JM, Carvalho RO, et al. (2009) Biological control of horse cyathostomin (Nematoda: Cyathostominae) using the nematophagous fungus Duddingtonia flagrans in tropical southeastern Brazilian Journal of Veterinary Parasitology 163: 335-340.
- Chen J, Liu Q, Liu GH, Zheng WB, Hong SJ, et al. (2018) Toxocariasis: a silent threatwith a progressive public health impact. Infectious Diseases of Poverty 7(1): 59.
- Ferraz CM, Sobral SA, Senna CC, Fidelis Junior O, Moreira TF, et al. (2019) Combined use of ivermectin, dimethyl sulfoxide, mineral oil and nematophagous fungi to control Rhabditis spp. Veterinary Parasitology 275: 108924.
- Frassy LN, Braga FR, Silva AR, Araújo JV, Ferreira SR, et al. (2010) Destruição de ovos de Toxocara canis pelo fungo nematófago Pochonia chlamydosporia. Revista da Soceiedade Brasileira de Medicina Tropicala 43: 102-104.
- Gorgônio SA, Sousa DLC, Bezerra CS, Monteiro GDF, Paulo FS, et al. (2021) Agentes parasitários de importância em Saúde Única em solos de praças públicas em condições semiá Research, Society & Development 10(1).
- Mendoza GP, Vazquez PVM (1994) Reduction of Haemonchus contortus infective larvae by three nematophagous fungi in sheep faecal cultures. Veterinary Parasitology 5: 197-203.
- Santos ECF, Carneiro MB, Tavares PV, Batista LCSO, Melo RMPS, et al. (2012) Ação de diferentes desinfetantes sobre viabilidade e mortalidade de larvas de terceiro estágio de Ancylostoma spp. Revista Brasileira de Medicina Veterinária 34: 55-59.
- Suzuki T, Coelho FAS, Marson FG, Coelho MDG, Araújo AJUS (2013) Eficácia de desinfetantes comerciais na inibição da evolução de ovos de Ancylostoma spp. obtidos de cães naturalmente infectados. Revista Biociência 19: 86-92.
- Taylor M, Coop R, Wall R (2017) Parasitologia Veteriná (3), Guanabara Koogan, Rio De Janeiro, Brazil.
- Ursache AL, Mirean V, Dumitrache M, Andrei L, Stefanut L, et al. (2019) Is routine disinfection efficient in preventing contamination with Toxocara canis eggs? Journal of Helmintology 94: e60.
- Verocai GG, Tavares PV, Ribeiro FA, Correia TR, Scott FB (2010) Effects of Disinfectants on Toxocara canis Embryogenesis and Larval Establishment in Mice Tissues. Zoonoses and Public Health 57(7-8): e213-216.
- Vilela VLR, Feitosa TF, Braga FR, Araújo JV, Souto DVO, et al. (2022) Biological control of goat gastrointestinal helminthiasis by Duddingtonia flagrans in a semi-arid region of the northeastern Brazilian. Veterinary Parasitol.ogy 188: 127-133.






























