Bioactivities of Liuwei Dihuang Extracts and Its Role in the Treatment of Diabetes
Ting Shien Teng1,2 and William Wei Ning Chen1,2*
1School of Chemical and Biomedical Engineering, Nanyang Technological University, Singapore
2Food Science and Technology Programme, Nanyang Technological University, Singapore
Submission:February 01, 2022; Published: February 28, 2022
*Corresponding author:W N Chen, school of Chemical and Biomedical Engineering, Nanyang Technological University, Food Science and Technology Programme, Nanyang Technological University, 62 Nanyang Drive, Block N1.2-01-12, 637459, Singapore
How to cite this article: Ting Shien T, William Wei Ning C. Bioactivities of Liuwei Dihuang Extracts and Its Role in the Treatment of Diabetes. Adv Biotech & Micro. 2022; 16(5): 555949 DOI:10.19080/AIBM.2022.16.555949
Abstract
Diabetes is a chronic metabolic disease that plagues millions of people in the world. Current methods of diabetes treatment are greatly hindered by the complications which would ensue due to increased level of oxidative stress. Liuwei Dihuang, a patented Traditional Chinese Medicine (TCM) formula, is a combination of herbal materials commonly prescribed for diabetic patients. TCM prescriptions generally fall short of the definition for medicine and be considered as nutraceuticals instead. While the effectiveness of the formula as treatment is widely proven in clinical and mouse studies, little is known of its mechanism of action. Herein, the study aims to characterise and evaluate the extracts of Liuwei Dihuang using antioxidant and anti-diabetic assays, coupled with results from GC-MS metabolite profiling. In vitro antioxidant and antidiabetic activity assays, total phenolic content and total flavonoid content were determined from extracts of Liuwei Dihuang pills and herbal formula, using different solvents. Methanol and aqueous extracts of Liuwei Dihuang from pills and herbal formula exhibited strong antioxidant activity in ABTS and DPPH assays, achieving near 100% inhibition of assay radicals at concentrations lower than 10 mg/ml. Results from anti-diabetic assays also demonstrated good inhibition of α-amylase at approximately 60% for methanol extracts. Majority of metabolites identified by GC-MS were sugar and organic acids found in methanol and aqueous extracts. Sorbose, fructose glucopyranose, malic acid and quinic acid were among the metabolites found in high abundance. Overall, our study suggested that Liuwei Dihuang could possibly improve diabetic conditions by reducing oxidative stress as the dominating factor, while also partially inhibiting enzymatic activities to reduce circulating glucose.
Keywords: Liuwei Dihuang; Antioxidant; Anti-Diabetic; Metabolomics; Diabetes
Introduction
The global occurrence of diabetes is on the rise. A total of 422 million adults were identified as diabetic in 2014 which represented an increase from 4.7% to 8.5% within the adult population [1]. Diabetes mellitus (DM) is a chronic metabolic disease of the endocrine system characterised by hyperglycaemia (ASSOCIATION & DEFINITION, 2009). Patients affected by diabetes face serious complications which include heart disease, nephropathy, retinopathy, cerebrovascular disease, and nerve damage [2]. Treatment of DM generally targets the immediate lowering of blood glucose level of the patients to prevent further complications brought about by hyperglycaemia. The pathogenesis of diabetes has also been commonly known to involve an elevated level of oxidative stress in the body caused by the presence of radical oxygen species (ROS) participating in free radical reactions [3]. ROS is comprised of a variety of chemical entities, ranging from an unstable group of superoxide or hydroxylradicals to longer lasting and diffusible hydrogen peroxide [4]. They play a critical role in activating important signalling pathways within the cells that alter gene expression or initiate cell deaths [5]. However, at high concentrations, free radicals can result in biomolecular damages to the cells [6]. The level of ROS is maintained at equilibrium by the presence of endogenous and exogenous antioxidant under physiological conditions [7]. Endogenous antioxidants refer to the body’s inherent defence antioxidant such as superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPx) [8] whereas exogenous antioxidants are natural nutrients consumed through daily diets which includes ascorbate, tocopherols, carotenoids, polyphenols and flavonoids [9,10]. These natural nutrients are readily found in fruits, vegetables and medicinal plants [10,11].
Traditional Chinese medicine (TCM) has a different view on the development of diabetes as a chronic disease. Termed asxiaokezheng in TCM, the disease is attributed to yin-deficiency of the body caused by improper diet, emotional distress and excessive sexual activities [12]. Patients of xiaokezheng are usually prescribed with Liuwei Dihuang (LWDH), a classical herbal formula known to restore the functional balance of the body by replenishing the kidney yin and invigorating the spleen qi. The formula is comprised of 6 different herbal plants, namely radix rehmanniae preparata, fructus corni, rhizome dioscoreae, poria, rhizome alismatis and cortex moutan. Studies of LWDH treatments on mouse models shown improvement in diabetic condition through the regulation of PI3K/Akt signalling pathway [13] while clinical studies of the formula demonstrated beneficial effects to diabetic microvascular complications and lowers two forms of oxidative stress biomarkers CML and 8-IsoP [14,15]. Comprising of plant materials entirely, LWDH can be positioned as nutraceuticals that provides as pharmaceutical alternative to conventional western medicinal treatment for diabetes.
Studies have shown that LWDH is effective in managing and preventing deterioration of diabetic symptoms and complications [16,17]. However, despite the proven effectiveness of LWDH treatment for diabetic cases, the mechanism of actions for the treatment has yet to be shown. The medicinal values of herbal plants may lie in its abilities to reduce oxidative stress to the body by increasing its antioxidant activity. It is hypothesised that LWDH alleviates diabetic symptoms via the reduction of oxidative stress and lowering of blood glucose in the body through the actions of the compounds synthesized by the herbal plants. Herein, the present work aims to characterise the antioxidant and antidiabetic capacities of LWDH and its phytochemical presence with the use of different solvents as an extraction medium.
Materials and Methods
Chemicals and Materials
Chemicals used in the extraction process, namely methanol, ethyl acetate, chloroform, and hexane, were purchased from Sigma-Aldrich (St. Louis MO, USA). Derivatisation reagents methoxyamine hydrochloride, pyridine, and N-methyl-N- (trimethylsilyl)-trifluoroacetamide (MSTFA) were purchased from Sigma-Aldrich (St. Louis MO, USA). Folin-Ciocalteau’s reagent, aluminium chloride, sodium bicarbonate, 2,2’-azinobis (3-ethylben-zothiazoline-6-sulfonic acid) diammonium salt (ABTS), 2,2’-diphenyl-1-picrylhydrazyl (DPPH), gallic acid and quercetin used in the assays were purchased from Sigma-Aldrich (St. Louis MO, USA). Concentrated pills of LWDH used in the present study were obtained commercially. The pills were manufactured by Hefei Shenlu Double-Crane Jiuhua Pharmaceutical Co. Ltd, Anhui, China. Raw herb materials of the LWDH formula, namely radix rehmanniae preparata, fructus corni, rhizome dioscoreae, poria, rhizome alismatis and cortex moutan, were purchased from Sinchong TCM Clinic, Singapore.
Pills Extraction Process
LWDH pills were reduced to powder form with pestle and mortar. To prepare the extracts in different solvents, the powdered LWDH pills were vortexed at a concentration of 0.1g/ml with methanol, ethyl acetate, chloroform and hexane. For aqueous extract, LWDH pills powder were suspended in distilled water at a concentration of 0.1g/ml before decocting at 105°C for 30 mins. All extracts were centrifuged at top speed for 10 mins before collecting the supernatant in aliquots. Each aliquot was aired to dryness before storing in -80°C for further processing.
Herbal Formula Extraction Process
The herbal formula is prepared with radix rehmanniae preparata, fructus corni, rhizome dioscoreae, poria, rhizome alismatis and cortex moutan in the ratio of 8:4:4:3:3:3, combined to a total weight of 2.5 g. 25 ml of distilled water was added before heating the mixture to 105°C for 30 mins. For methanol extract, 25 ml of methanol was added to the mixture and sonicated for 30 mins. Both extracts were collected and centrifuged at top speed before collecting the supernatant in aliquots. Each aliquot was aired to dryness before storing in -80°C for further processing.
Sample Preparation for Assays
Methanol, ethyl acetate, chloroform and hexane extracts were reconstituted in 95% methanol while aqueous extracts were reconstituted in distilled water. All extracts were added solvents equivalent to its original volume prior to drying. Methanol and distilled water served as blank reference for all assays. Considering some of the extracts were colored, control references were prepared by mixing the samples with only the solvents of the assay solution in the same volume.
Quantitative Determination of Antioxidant Activities
ABTS radical scavenging activity
The 2,2’-azino-bis (3-ethylben-zothiazoline-6-sulfonic acid diammonium salt) radical cation scavenging activity assay, more commonly known as ABTS assay, was used to quantitatively determine the antioxidant activities of each extract. The assay was performed in accordance to the method described by Re et al. with modifications [18]. Briefly, ABTS+ ions were produced by mixing 7 mM ABTS stock solution with 2.45 mM potassium persulfate in 1:1 ratio and allowing the reaction to reach completion by leaving the mixture in the dark for 16 hours. A working stock is prepared by diluting the ABTS+ solution with ethanol until a stabilised absorbance reading of 0.700 ± 0.05 when measured at 734 nm was achieved. Antioxidant activities of the extracts were assessed by adding 180 μl of ABTS working stock to 20 μl sample in each well of the microplate. The absorbance reading is taken after allowing the mixture to incubate for 15 minutes. The level of antioxidant activity was expressed as percentage inhibition of the ABTS+ ions and can be determined by the following equation:
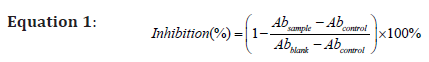
The higher the percentage, the higher the level of antioxidant activity of the extract. Samples were later serially diluted (1 mg/ ml to 100 mg/ml) for further investigation of antioxidant activity at different concentrations.
DPPH Radical Scavenging Activity
The free radical scavenging activities of the various extracts were assessed by using the 2,2’-diphenyl-1-picrylhydrazyl (DPPH) assay, performed in accordance to the method described by Takao et al. [19], applied with suitable modifications from the procedures of [20]. Briefly, 100 μl of sample was mixed with 100 μl DPPH in methanol (0.2 mM). The mixture was incubated in darkness for 30 mins before absorbance measurement was taken at 517 nm. The level of antioxidant activity was expressed as percentage inhibition of the DPPH free radicals and can be determined by the following equation:
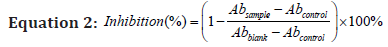
The higher the percentage, the higher the level of antioxidant activity of the extract. Samples were later serially diluted (1 mg/ ml to 100 mg/ml) for further investigation of antioxidant activity at different concentrations.
Quantitative Analysis of Phytochemicals
Total Phenolic Content
The total phenolic content of each extract was measured by the Folin-Ciocalteau’s reagent method as described by McDonald et al [21]. Briefly, 200 μl of 0.2 N Folin-Ciocalteau’s reagent was added to 100 μl of sample, followed by 800 μl of 700 mM Na2CO3. The mixture was incubated in the dark for 2 hrs before absorbance measurements were taken at 765 nm. A standard calibration curve was plotted with the absorbance readings of gallic acid (0 μg/ml to 120 μg/ml) dissolved in distilled water. The total phenolic content of each extract was expressed as gallic acid equivalents. Samples were also serially diluted (1 mg/ml to 100 mg/ml) for further investigation of total phenolic content at different concentration
Total Flavonoid Content
The total flavonoid content of each extract was measured using method described by Chang et al., performed with modifications [22]. Briefly, 1 ml of AlCl3 (2%) dissolved in methanol was added to 1 ml of sample. The mixture was incubated for 1 hour in the dark before the absorbance measurements were taken at 415 nm. A standard calibration curve was plotted with the absorbance readings of quercetin (0 μg/ml to 20 μg/ml) dissolved in distilled water. The total flavonoid content of each extract was expressed as quercetin equivalents. Samples were also serially diluted (1mg/ml to 100 mg/ml) for further investigation of total flavonoid content at different concentrations.
Quantitative Determination of Anti-diabetic Activities
The α-amylase assay was carried out in accordance with the procedure described by [23] with modifications. Briefly, 15 μl Afolayan, of dried extract reconstituted in PBS is added with 5 μl of enzyme porcine pancreatic solution (2U/ml) and incubated for 10 mins at 37℃. 20 μl of starch solution is added at concentration 10 mg/ml and the mixture is further incubated for 30 mins at 37℃. 10 μl of 0.1M HCl was added to stop the reaction after incubation and 75 μl of iodine was added to the mixture. Absorbance reading was taken at 580 nm. Samples were measured in triplicates.
GC-MS Metabolites Profiling
GC-MS analysis was conducted according to procedure described by Chen et al. [24]. For sample preparation, the extracts were dried and derivatized by first dissolving in 50 μl of 20 mg/ml solution of methoxyamine hydrochloride in pyridine, incubated for 60 mins at 37℃. Following which, 100 μl of N-methyl-N-(trimethylsilyl)-trifluoroacetamide (MSTFA) with 1% trimethylchlorosilane (TMCS) was further added to each extract, and further incubated at 70 °C for 30 min. Once derivatization is complete, the mixtures were centrifuged at 14,000 rpm for 10 mins and the supernatant transferred to glass vials for GC-MS analysis.
Samples were injected at 1 μl into a HP5-MS column by an autosampler using a spitless mode. Helium gas was used as a carrier gas with flow rate of 1.1 mL/min. The inlet and MS source temperature were kept constant at 250℃ and 230℃ respectively. The oven temperatures were initialized at 80℃ for 2 mins, and subsequently increased to 165℃ at 10℃/min for 2 mins, 180℃ at 1℃/min for 3 mins, 275℃ at 20℃/min with no pause and 280℃ at 2℃/min for 5 min. post-temperature is set at 80℃ for 3 mins. Total run time is 42.75 mins. Chromatogram acquisition and mass spectra identification were obtained with Agilent MSD Chemstation Data Analysis software and the compounds were identified with NIST02 mass spectral database and Golm database. The mass spectra were deconvoluted with AMDIS. The compounds were normalised to the peak intensity of the internal standard ribitol. The order of sampling was randomised and adjusted for consistency by batch analysis.
Statistical Analysis
All samples were measured in triplicates and the results were expressed in terms of mean value (n = 3) ± standard deviation of the mean value. Linear regression was used to analyses the data obtained from the measurements.
Result
Methanol, aqueous, ethyl acetate, chloroform and hexane extracts were prepared for LWDH pills while methanol andaqueous extracts were prepared for LWDH herbal formula. These extracts were assessed for their antioxidant activities and anti-diabetic activities and quantified for their total phenolic content and total flavonoid content. Pills and herbal formula were selected as representing samples for Liuwei Dihuang to observe for any changes to extraction efficacy of the compounds due to matrix differences of the sample materials. While LWDH could be grounded to fine powder, it was not possible to reduce the plant materials of the herbal formula to similar fineness prior to extraction. However, results from the study would later show that both types of samples were able to produce similar antioxidant effects despite differences in the sample matrix.
Antioxidant Activities
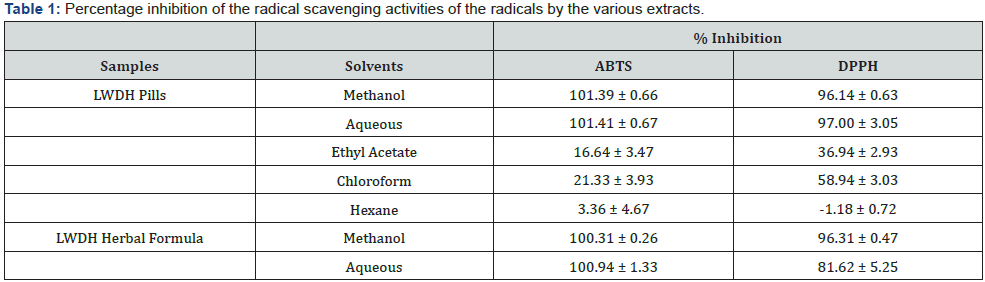
Table 1 showed the result of the antioxidant activities of the various extracts as assessed by the ABTS and DPPH assays. The highest antioxidant activities were consistently observed in the methanol and aqueous extracts of both LWDH pills and herbal formula across both assays, reaching near 100% inhibition. Chloroform extract of LWDH pills also exhibited notable antioxidant activities, with more than 50% inhibition observed in DPPH assay. However, it should be noted that the measurements were corrected for their initial absorbance readings due to some of the extracts being colored, resulting in values exceeding 100% and lower than 0%. Furthermore, presence of white emulsion could be seen when reagents of ABTS and DPPH assays were added to the aqueous extracts of LWDH pills and herbal formula due to the use of methanol and ethanol as diluting solvents. Considering that methanol and aqueous extracts of LWDH pills and herbal formula achieved significantly higher level of antioxidant activities compared to the other extracts, the four extracts were investigated further for their dose dependent effects and the influence on antioxidant activities. Figures 1 & 2 illustrated the percentage inhibition of the radicals for ABTS and DPPH assays at different concentrations of the extracts respectively.
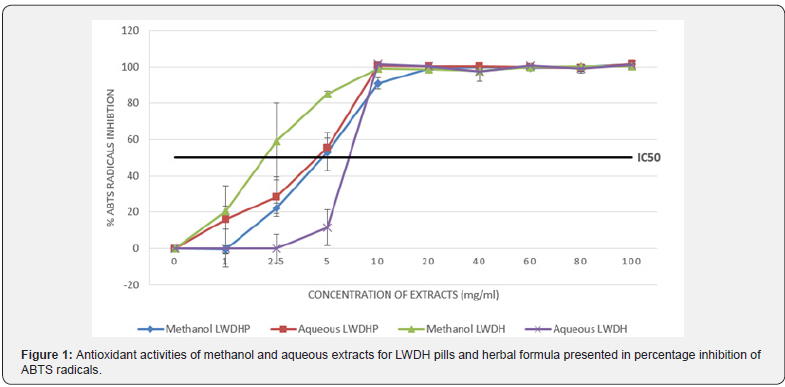
The antioxidant activities were determined by spectrophotometric measurements at 734 nm and 517 nm respectively for ABTS and DPPH assays.


TPC is expressed in terms of gallic acid equivalent (y = 0.0064x + 0.0663R² = 0.9514) and TFC is expressed in terms of quercetin equivalent (y = 0.0132x + 0.0022R² = 0.9956).
From Figure 1, it can be observed that all extracts eventually reached 100% inhibition of ABTS radicals at 10 mg/ml. By comparing the IC50 concentrations of the extracts, methanol extract of LWDH herbal formula demonstrated the strongest antioxidant activity, with a low IC50 concentration between 1 mg/ml and 2.5 mg/ml whereas aqueous extract of the herbal formula yielded the weakest antioxidant activity level, with IC50 concentration between 5 mg/ml and 10 mg/ml. On Figure 2, all extracts approached 100% inhibition of DPPH radicals at concentration between 2.5 mg/ml and 5.0 mg/ml. Estimation of IC50 concentrations showed that methanol extract of LWDH herbal extract had again exhibited the strongest antioxidant effect, together with aqueous extract of LWDH pills. Methanol extract of LWDH pills had the least potent antioxidant effect, requiring concentrations between 1 mg/ml to 2.5 mg/ml to reach 50% inhibition. While this is inconsistent with the data obtainedfrom ABTS assay, the variation is marginal. The discrepancy may be attributed to the affinity of the compounds extracted from both solvents with the radicals of the antioxidant assays [25], with some compounds inhibiting ABTS radicals more effectively than DPPH radicals, and vice versa.
Phytochemicals Quantification
Phenolic compounds are a common class of secondary metabolites found in plant materials [26]. The hydroxyl groups present in these phytochemicals confer scavenging ability for the plants [27] which is believed to be responsible for their antioxidant activity [28]. Reports of qualitative analysis conducted on plant metabolites revealed large presence of phenols from extracts using polar solvents [29]. Of the different categories of phenolic compounds found in plants, flavonoids form a major component. Flavonoids are thought to possess significant antioxidant activities [30] derived from their abilities to scavenge most oxidizing molecules and radicals [31]. The quantification of phenolic content was carried out to determine its presence. A further quantification was carried out to determine the flavonoid content of each extract. Table 2 showed the results of the quantification tests for phenolic and flavonoid contents expressed as gallic acid and quercetin equivalent respectively.
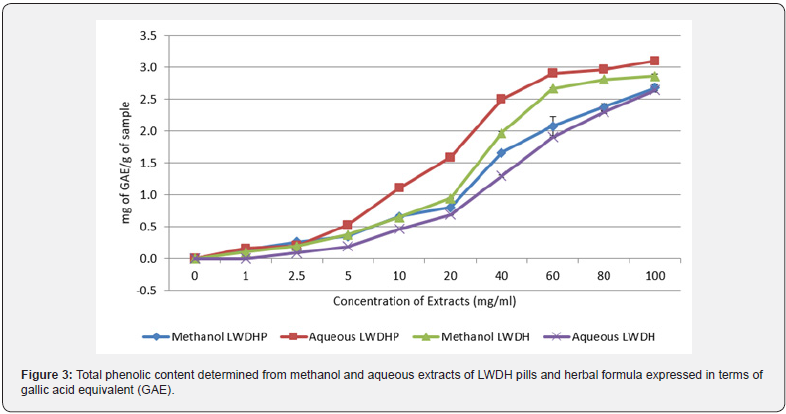
Consistent with the results of antioxidant activities, methanol and aqueous extracts showed the highest amount of phenolic content with more than 2mg of gallic acid equivalent per gram of pills or herbs sample. Chloroform also exhibited a significant level of phenolic presence, amounting to 0.692 mg of gallic acid, which may be the explanation for the slight antioxidant activity detected in the assays. The results are supported by other studies which have also established a positive correlation between phenol concentration and antioxidant activity [32,33]. However, high phenolic contents did not correspond to high level of flavonoid contents. Methanol and aqueous extracts of LWDH herbal formula were determined to only contain 0.092 mg and 0.035 mg equivalent of quercetin, almost half the value detected in methanol extract of LWDH pills. This suggests that the classes of antioxidant compounds extracted from LWDH pills and herbal formula may be different, with LWDH pills extracts having higher amount of flavonoid than LWDH herbal formula. There is no observable correlation between flavonoid content and antioxidant activity. The phenolic and flavonoid contents in the methanol and aqueous extracts were further investigated at different concentrations. Figure 3 compared the phenolic contents of the extracts at each concentration level. Despite having the lowest IC50 values in both antioxidant assays, methanol extract of LWDH herbal formula did not report to have the highest phenolic content. Methanol and aqueous of both types of LWDH samples were able to achieve 50% inhibition in the antioxidant assays with phenol content lesser than 1 mg of gallic acid equivalent. Figure 4 illustrated the changes in the flavonoid content of the extracts at different concentration levels, showing a much content when compared to the phenolic contents.
Anti-diabetic Activities
As summarized in Table 3, methanol extracts of LWDHP and LWDH showed the highest positive inhibition of the hydrolytic function of α-amylase. Interestingly, aqueous extracts of both samples exhibited inhibition, suggesting that starch content was more than the amount initially added. To understand the situation, methanol and aqueous extracts of LWDH pills and herbal formula were further investigated for starch presence by replacing α-amylase solution and starch solution with PBS. Table 4 showed the amount of starch detected in each extract at various concentrations. It should be mentioned that the intensity of absorbance for aqueous LWDH pills was beyond the limit of the spectrometer. Given the linearity of the relationship between the concentration of the extract and the starch content, the amount of starch present was extrapolated from the linear curve fitted to the graph, with a correlation of 0.9635.
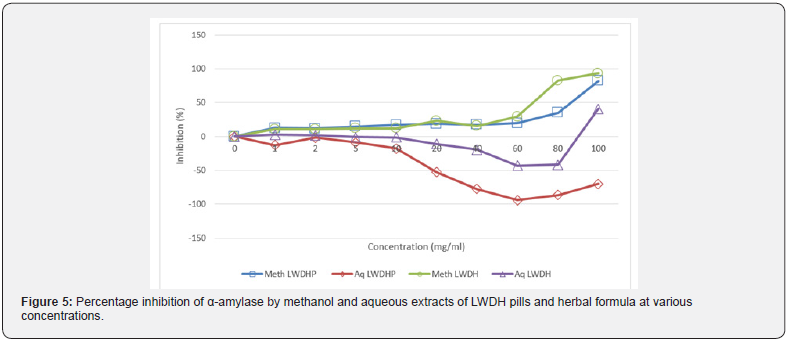
It was observed that aqueous LWDH pills contained high content of inherent starch when compared to the aqueous counterpart of LWDH herbal formula. However, the same discrepancy in starch content could not be seen between the twomethanol extracts. Ironically, methanol extract of LWDH herbal formula reported a higher amount of starch content than aqueous extract of the same sample at low concentrations. Considering the starch content recorded in each extract, the percentage inhibition of α-amylase digestive action was calculated by subtracting theinherent starch content from the final value. Percentage inhibition in Table 3 was hence calculated after accounting for the starch present in the extract prior to performing the assay. Figure 5 summarized the level of inhibition for each extract at various concentrations.



Readings that were beyond the limit of the spectrometer was extrapolated from the linear curve fitted to the curve. Equation used: y = 0.0587x +0.4431 with r2 = 0.9635.
There is a gradual increase in level of inhibition of the enzyme for methanol extracts of both LWDH pills and herbal formula, eventually approaching 100% inhibition, relative to inhibition level observed with acarbose, at 100 mg/ml with the sharpest increment observed between 60 mg/ml to 80 mg/ml. On the contrary, there was almost no inhibition recorded with the aqueous extracts of either LWDH pills or herbal formula except for an anomaly seen in aqueous LWDH herbal formula where inhibition reached approximately 50%. By negative percentage inhibition, the change in starch content determined from the assay was found to be more than the amount added.
Metabolomics Profiling
Figure 6 showed a heatmap summarizing the distribution and relative abundance of detected compounds across the seven extracts of LWDH pills and herbal formula. Of which, the methanol and aqueous extracts of both LWDH pills and herbal formula contained most of the detected compounds. The compounds were identified with minimum 75% similarity match to the mass spectra found in the database. The abundance of each compound is determined with reference to the peak area of 20 ul of ribitol (20 mg/ml) added to each sample prior to drying. There were generally two main groups of compounds: sugars and organic acids. Sugar compounds were of the highest abundance among the compounds, specifically fructose, sorbose and glucopyranose. There were many identified organic acids found in the list of detected compounds as well, with malic acid detected in higher concentration in the aqueous extracts relative to methanol and chloroform extracts (Table 5).

It was surprising to observe that large amount of glucose and sucrose was found only in the aqueous extract of LWDH herbal formula while their presences were almost negligible in the aqueous extract of LWDH pills and in both methanol extracts. These sugars may have originated from an even greater quantity of polysaccharides which are undetected by GC-MS due to their large molecular weight, but nonetheless exist in the extracts which could hence relate to the results determined by the previous two assays. There were also 18 detected compounds that were unable to be accurately identified, that is the similarity match to the database mass spectra fell below 75% and were hence unannotated. However, visual analysis of the chromatogram highlighted these compounds as having distinct peaks with reproducible retention time across majority of the samples and were therefore recorded.
AMDIS was used to deconvolute the GC-MS data where the overlapping components of the spectra were removed prior to identifying with the database. Two databases were used in the identification process: NIST02 and Golm Metabolome Database. Golm Metabolome Database was used as primary database for identification due to its collection of mass spectra of phytochemical compounds which were relevant to our study. NIST02 was used as a supportive database in cases where AMDIS was able to identify features in the chromatogram but was unable to match it to any spectra in Golm database.
Discussion
The formation of emulsion caused by addition of alcoholic solvents indicated the presence of proteins, enzymes, polysaccharides or other natural products [34]. Emulsion is believed to have resulted in aqueous extracts of LWDH pills and herbal formula reporting an inhibition level above 100%. This is technically impossible as 100% inhibition should indicate absolute clearance of the assay radicals. Centrifuging the samples at top speed and longer duration can help to reduce the erroneous readings, suggesting that compounds causing the emulsion can be further precipitated and removed through centrifuging. Nevertheless, an observable trend can be seen from both Figures 1 & 2 where all extracts showed rise in antioxidant activity levels as concentrations increase, eventually reaching maximum inhibition levels of the assays.
Given the high levels of antioxidant activity detected in the methanol and aqueous extracts of both types of LWDH samples, phenolic compounds were speculated to be responsible on the inhibition on the radicals in the assays [35]. However, while having the lowest IC50 for both assays, methanol extract of LWDH herbal formula did not reflect the highest phenolic content. This further suggest that other classes of antioxidative compounds were present in the extracts. Across the different concentrations of extracts, flavonoids represent only a small portion of the phenolic contents that were detected, and the data do not correspond to the antioxidant activity levels of the extracts – high flavonoid content did not correlate to high antioxidant level. While flavonoid has been reported to have significant antioxidant effects [36-38], the results from the study may suggest that flavonoid did not play a significant role as antioxidants. Other compounds may possess stronger antioxidant activity.
The aqueous extract of LWDH pills was found to exhibit α-amylase inhibition over 100%. In fact, the intensity of the absorption was over the limit of the spectrometer and was thus unable to provide a meaningful reading. The upper reading limit was therefore used as the value for aqueous extract of LWDH pills. Nevertheless, this suggested that aqueous LWDH pills extract was either able to inhibit the enzymes more than acarbose, or there could inherently be starch present in the aqueous extract. Observing the starch amount present in the extracts however, revealed that the amount of starch detected in aqueous extract of LWDH pills exceeded the original amount of starch added to the extract at the start of the assay. While approximately 0.400 mg of starch was added to the extracts, the amount detected in the aqueous extract was 0.457 mg. This concluded that there were already starch-related compounds present in the extract prior to adding starch. Hence, it is necessarily to isolate the starch equivalent compound extracted from LWDH pills and herbal formula to accurately determine the level of inhibition. Serial dilution was made for aqueous and methanol extracts of both LWDH pills and LWDH herbal formula as described previously in the antioxidant assay. While it was observed that aqueous extract of LWDH pills had high starch content relative to LWDH herbal formula, the methanol extract of both samples did not exhibit the same pattern. The result was inconclusive in terms of the starchcontent difference between the two samples nor the efficiency of the solvents with extracting starch-related compounds. Nevertheless, it showed the importance of accounting for the inherent starch content when calculating the inhibition level of each extract content difference between the two samples nor the efficiency of the solvents with extracting starch-related compounds. Nevertheless, it showed the importance of accounting for the inherent starch content when calculating the inhibition level of each extract.
It is worth mentioning the discrepancy observed between the starch content amount observed in the antidiabetic assay and interference of precipitate seen in antioxidant assay. While both aqueous extracts of LWDH pills and herbal formula resulted in precipitation following the addition of ethanol in antioxidant assay, suggesting the presence of polysaccharides, proteins, enzymes and natural products, high starch content was only observed in the aqueous extract of LWDH pills in the antidiabetic assay. This may suggest that the interference recorded in antioxidant assay may ultimately be caused by compounds besides polysaccharides, which encourages the theory that LWDH pills and herbal formula have relatively different chemical profile constituents despite similar extraction solvent and herbal ingredients.
The GC-MS analysis of the samples provided an insight into the chemical compositions of LWDH in both pills and herbal formulas. The compounds identified will be useful for understanding the effects of LWDH, possibly through metabolic pathway analysis when administered as treatment for diabetic patients. Fundamentally, it will help to explain the results of the bioactivities observed in the previous assays by focusing on the possible compounds responsible for producing the antioxidant or antidiabetic activities. It will also be insightful to compare the compounds found in methanol and aqueous extracts of LWDH, given that the two extracts exhibited the most significant bioactivities. By pinpointing the difference between the aqueous and methanol extracts of LWDH pills and herbal formula in terms of the chemical compounds present and the relative abundance of the compounds, it may be possible to explain the difference in bioactivities found in the four extracts. For example, a different in the compositions of the compounds in the aqueous and methanol extracts of LWDH may reveal the compounds that were responsible for the diverging effects in the antidiabetic assay.
It is undeniable that GC-MS is a useful technique to elucidate the chemical compounds present in LWDH. However, it should be noted that analysis by GC-MS is limiting, and it will not be able to provide a complete global profile of the samples. Compounds which can be detected have to be sufficiently volatile and of relatively small mass, approximately 800 Dalton. Also, the high temperature involved in vaporizing the compounds may cause degradation, especially for natural products such as phytochemicals, which reduces the accuracy of the detections. The long processing time needed for sample preparations may also indirectly cause degradation of the compounds. As such, it is necessary to complement the results obtained by GC-MS analysis with that of LC-MS analysis. LC-MS analysis detects larger molecules and does not involve vaporizing the compoundsprior to detection. This reduces the uncertainty of compound degradations. However, LC-MS has lower reproducibility when compared to GC-MS. Coupling LC-MS analysis with GC-MS analysis will provide a more complete picture of the content in LWDH, both pills and herbal formula. Eventually, this allows further results to be attributed and explained by comparing it to the identified compounds with the associated pathways and reactions.
Conclusion
To our knowledge, the study is the first to investigate and compare the efficacy of different solvents on the metabolite extraction of LWDH before further characterizing the extracts based on their antioxidant activity, anti-diabetic activity, and GCMS metabolite profiling. Methanol and aqueous were determined to be the most effective solvent for extraction, given by its strong antioxidative and anti-diabetic effect, as well as by the high number of detected compounds from the metabolomic data. The results of the present study have suggested that LWDH, as a TCM prescribed treatment for kidney-yin deficient forms of diabetes, is antioxidative and anti-diabetic and could hence potentially alleviate symptoms of diabetes by lowering enzymatic activities that releases glucose and reducing the oxidative stress in the body.
Acknowledgement
We thank Dr. Jaslyn Lee, Dr. Kuan Rei Ng, and Dr. Cui Xi for their technical assistance and support.
Authors’ Contribution
TST performed the experiment, analysed the data, and wrote the paper. WCN supervised the study. All authors have read and approved the final manuscript.
Funding
We thank Nanyang Technological University for awarding the scholarship to TS Teng.
References
- WHO (2016) Global Report on Diabetes.
- Cade WT (2008) Diabetes-Related Microvascular and Macrovascular Diseases in the Physical Therapy Setting. Physical Ther 88(11): 1322-1335.
- Asmat U, Abad K, Ismail K (2016) Diabetes mellitus and oxidative stress—A concise review. Saudi Pharm J 24(5): 547-553.
- Finkel T, Holbrook NJ (2000) Oxidants, oxidative stress and the biology of ageing. Nature 408(6809): 239-247.
- Cho K-H, Wolkenhauer O (2003) Analysis and modelling of signal transduction pathways in systems biology. Biochem Soc Trans 31(Pt 6): 1503-1509.
- Halliwell B (2007) Biochemistry of Oxidative Stress. Angewandte Chemie International Edition in English. 25(12): 1058-1071.
- R O, L N, OO (2014) Oxidative Stress and Diabetic Complications: The Role of Antioxidant Vitamins and Flavonoids. In Antioxidant-Antidiabetic Agents and Human Health.
- Ighodaro OM, Akinloye O A (2018) First line defence antioxidants-superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPX): Their fundamental role in the entire antioxidant defence grid. Alexandria Journal of Medicine 54(5): 287-293.
- Giugliano D (2000) Dietary antioxidants for cardiovascular prevention. Nutr Metab Cardiovasc Dis 10(1): 38-44.
- Pandey KB, Rizvi SI (2009) Plant polyphenols as dietary antioxidants in human health and disease. Oxid Med Cell Longev 2(5): 270-278.
- Dong-Ping, X Ya, L Xiao, M Tong, Z Yue, et al. (2017) Natural Antioxidants in Foods and Medicinal Plants: Extraction, Assessment and Resources. Int J Mol Sci 18(1): 96.
- Li W L, Zheng HC, Bukuru J, De Kimpe N (2004) Natural medicines used in the traditional Chinese medical system for therapy of diabetes mellitus. J Ethnopharmacol 92(1): 1-21.
- Dai B, Wu Q, Zeng C, Zhang J, Cao L, et al. (2016) The effect of Liuwei Dihuang decoction on PI3K/Akt signaling pathway in liver of type 2 diabetes mellitus (T2DM) rats with insulin resistance. J Ethnopharmacol 192: 382-389.
- Zhao Y, An X, Liu J, Liu S, Xu W, et al. (2018) The improvement of oxidative stress by two proprietary herbal medicines in type 2 diabetes. Complement Ther Med 40:120-125.
- Zhao Y, Yu J, Liu J, An X (2016) The role of liuwei dihuang pills and ginkgo leaf tablets in treating diabetic complications. Evid Based Complement Alternat Med 2016: 7931314.
- Liao T, Zhao K, Huang Q, Tang S, Chen K, et al. (2020) A randomized controlled clinical trial study protocol of Liuwei Dihuang pills in the adjuvant treatment of diabetic kidney disease. Medicine 99(31): e21137.
- RS, YW, XA, JM, TW, et al. (2019) Efficacy of Co-administration of Liuwei Dihuang Pills and Ginkgo Biloba Tablets on Albuminuria in Type 2 Diabetes: A 24-Month, Multicenter, Double-Blind, Placebo-Controlled, Randomized Clinical Trial. Frontiers in Endocrinology, 10(FEB).
- Re R, Pellegrini N, Proteggente A, Pannala A, Yang M, et al. (1999) Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radical Biology and Medicine 26(9-10): 1231-1237.
- Takao T, Kitatani F, Watanabe N, Yagi A, Sakata K (1994) A Simple Screening Method for Antioxidants and Isolation of Several Antioxidants Produced by Marine Bacteria from Fish and Shellfish. Bioscience, Biotechnology, and Biochemistry 58(10): 1780-1783.
- Mo ZZ, Liu YH, Li CL, Xu LQ, Wen LL, et al. (2017) Protective Effect of SFE-CO2 of Ligusticum chuanxiong Hort Against d -Galactose-Induced Injury in the Mouse Liver and Kidney. Rejuvenation Research.
- McDonald S, Prenzler PD, Antolovich M, Robards K (2001) Phenolic content and antioxidant activity of olive extracts. Food Chemistry 73(1): 73-84.
- Chang CC, Yang MH, Wen HM, Chern JC (2002) Estimation of total flavonoid content in propolis by two complementary colometric methods. Journal of Food and Drug Analysis.
- Odeyemi SW, Afolayan A (2018) Identification of Antidiabetic Compounds from Polyphenolic-rich Fractions of Bulbine abyssinica A. Rich Leaves. Pharmacognosy Res 10(1):72-80.
- Chen L, Chen WN (2014) Metabolite and Fatty Acid Analysis of Yeast Cells and Culture Supernatants. Bio-Protocol 4(17): e1219.
- Shalaby EA, Shanab SMM (2013) Comparison of DPPH and ABTS assays for determining antioxidant potential of water and methanol extracts of Spirulina platensis. Indian Journal of Marine Sciences.
- Lin D, Xiao M, Zhao J, Li Z, Xing B, et al. (2016) An overview of plant phenolic compounds and their importance in human nutrition and management of type 2 diabetes. Molecules 21(10): 1374.
- Saeed N, Khan MR, Shabbir M (2012) Antioxidant activity, total phenolic and total flavonoid contents of whole plant extracts Torilis leptophylla L. BMC Complement Altern Med 12:221.
- Petridis A, Therios I, Samouris G, Tananaki C (2012) Salinity-induced changes in phenolic compounds in leaves and roots of four olive cultivars (Olea europaea L.) and their relationship to antioxidant activity. Environmental and Experimental Botany.
- Čanadanović-Brunet J, Ćetković G, Djilas S, Tumbas V, Bogdanović G, et al. (2008) Radical Scavenging, Antibacterial, and Antiproliferative Activities of Melissa officinalis L. Extracts J Med Food 11(1): 133-143.
- Nunes XP, Silva FS, Almeida JR G da S, Lima JT de, Ribeiro, LA de A, et al. (2006) Biological oxidations and antioxidant activity of natural products. In Phytochemicals as nutraceuticals - Global approaches to their role in nutrition and health.
- Arun KB, Chandran J, Dhanya R, Krishna P, Jayamurthy P, et al. (2015) A comparative evaluation of antioxidant and antidiabetic potential of peel from young and matured potato. Food Bioscience 9: 36-45.
- Borneo R, León A E, Aguirre A, Ribotta P, Cantero JJ (2009) Antioxidant capacity of medicinal plants from the Province of Córdoba (Argentina) and their in vitro testing in a model food system. Food Chemistry 112(3): 664-670.
- Piluzza G, Bullitta S (2011) Correlations between phenolic content and antioxidant properties in twenty-four plant species of traditional ethnoveterinary use in the Mediterranean area. Pharm Biol 49(3): 240-247.
- Gong X, Wang S, Li Y, Qu H (2013) Separation characteristics of ethanol precipitation for the purification of the water extract of medicinal plants. Separation and Purification Technology.
- Tosun M, Ercisli S, Sengul M, Ozer H, Polat T, et al. (2009) Antioxidant properties and total phenolic content of eight Salvia species from Turkey. Biological Research.
- Pietta PG (2000) Flavonoids as antioxidants. Journal of Natural Products 63(7): 1035–1042.
- ASSOCIATION, AD DEFINITION (2009) Diagnosis and classification of diabetes mellitus. Diabetes Care.
- Bravo L (2009) Polyphenols: Chemistry, Dietary Sources, Metabolism, and Nutritional Significance. Nutr Rev 56(11): 317-333.






























