Identifying Potential Inhalation Risks Associated with Exposure to Different Forms of Silica at the Nanomolecular Level as it Relates to Antimicrobial Agents
Hamid A Khan1* and Benjamin O Bienia2
1American Microsurgical Orthoplastic Society, Foot and Ankle Deformity Correction Center, USA
2Department of Neuro Radiology, AMITA Health Hinsdale, USA
Submission:June 06, 2021; Published: June 17, 2021
*Corresponding author: Hamid A Khan, American Microsurgical Orthoplastic Society, Foot and Ankle Deformity Correction Center, Chicago, IL. USA
How to cite this article: Hamid A K, Benjamin O B. Identifying Potential Inhalation Risks Associated with Exposure to Different Forms of Silica at the Nanomolecular Level as it Relates to Antimicrobial Agents. Adv Biotech & Micro. 2021; 16(3): 555937 DOI:10.19080/AIBM.2021.16.555937
Abstract
Silica is a naturally occurring element found in abundance throughout the earth’s crust and its use dates back centuries. There are different variations of silica and each one contains specific physio- chemical characteristics which help differentiate one another, these characteristics also aid in understanding whether or not there are increased risks for health hazards that may result from long term or occupational exposure to various silica types. Although Silica is most known for its presence in construction, mining, and sandblasting, there are numerous other applications for its existence, use and potential. Recently, there has been interest raised around the utilization of nanomolecular silica formulated in such a manner that there is proven efficacy in terms of disinfecting surfaces and protecting surfaces from harmful pathogens. Several studies have demonstrated astonishing log reductions when it comes to distinct microbes, however the safety of this silica at a nanomolecular level is not well understood as this advanced biocidal process is still a relatively new one. The purpose of this literature is to review the facts behind the different types of common silica and determine if they have the potential for increasing the risk for hazards to human health.
Keywords: Silica, Antimicrobial, Lungs, Colloidal, Amorphous, Nanomolecular
Abbreviation: SiO2: Silicon dioxide; COPD: Chronic obstructive lung disease (COPD); TB: Tuberculosis
Introduction
Silica is the mineral compound of silicon dioxide (SiO2) and this element is one of the most abundant naturally occurring materials that is present on Earth. As technology and science continue to evolve, so do the applications for silica. Historically, silica has been understood as being present in plants, certain vegetables, water, and rocks [1,2]. Today the use of silica has gone far beyond that which we naturally encounter on a daily basis, silica is now being utilized as diagnostic and therapeutic applications in medicine, as well as in cosmetics, drugs, varnishes and even in household and industrial anti-microbial cleaning agents and pesticide products [1-3]. Although the biocidal effects of nano silica are not completely understood due to the fact that it is a relatively new concept, independent studies and the research and literature currently available prove that there is tremendous antimicrobial versatility when silica is used at the nanomolecular level to combat harmful pathogens. Depending on the type of silica being used within various antimicrobial agents, there are many benefits of utilizing nanomolecular SiO2 as the base material for modifying a surface and creating an antimicrobial environment that allows for a less-toxic, and more environmentally friendly substance as a safer alternative to the more toxic volatile chemical disinfectants and antimicrobial products commonly used today.
In the case of silica being used for antimicrobial processes, there must be a clear understanding of the type of silica being used and a concise understanding that the phrase ‘crystalline-like’ silica or ‘microcrystalline-like’ silica is not the same as ‘crystalline’ silica. The main impression posed with the use of nanomolecular silica for antimicrobial purposes lays in the foundation of a technique referred to as a ‘mechanical kill’, where in which elemental silicon dioxide is fractionated and formulated with other inert or nonchemical ingredients to create microcrystalline-like structures on the surface it is applied to, after it has been given time to dry. These crystalline-like structures that form have been found to be [4-6] nanometers in diameter and are not visible to the naked eye but work in harmony to attach to the targeted surface via covalent bonding. The strong bonds that occur at this nanomolecular level create a physical barrier/surface that coats the underlying surface and protects it from future adherence of harmful microbes. Studies have shown that these newly formed crystalline-like structures contain microscopic spikes at their tips, that physically work to penetrate the outer membranes or bodies of whatever it comes into contact with ( i.e. bacteria, viruses, fungi, mold, algae), essentially deflating and eliminating that particular pathogen.
One of the most important factors related to this newly created surface, aside from its ability to combat microbes, is the associated longevity. The research completed on these surfaces has indicated that the surface barrier created by the nano molecular silica is one that can be viewed as being permanent, unless abraded off or soiled over. It is for this very reason why the use of silica within disinfectants and pesticide products is so promising. By avoiding the constant need for applying harmful and volatile chemicalbased products, nano molecular silicon dioxide (SiO2)-based agents offer a safer and more long-term alternative that are just as effective and, in some cases, even more efficacious than traditional chemical-based solutions [4].
Literature Review
For years, the use of silica and its impact on human health has been investigated and well- documented. Aside from silica being a natural occurring element of the earth’s crust, it is also naturally found within the human body and serves to help strengthen the connective tissue of the brain, spinal cord, and nerve cells. Studies have suggested that silica aids in the stabilization of insulin release from the pancreas, there is also literature describing how silica is necessary for strong and healthy skin, hair and nails.5 In terms of adverse effects relating to silica, most of this testing and research has been completed on animal test subjects and many of these studies focused primarily on crystalline micron- sized silica. There is a limited amount of literature that places emphasis on amorphous and nano- molecular sized silica as it relates to health and safety, but the available information does offer sufficient scientific evidence to support the claim that when it comes to the different forms and characteristics of silicon dioxide (SiO2) there are major dissimilarities associated with toxicity [6]. The notion that silica has the potential of being a toxicological hazard to human health, is completely dependent on the physio-chemical characteristics of the silica material being discussed. In General, there are two forms that silica can be classified as, crystalline and amorphous [7]. There are several differences related to each form of silica and these variations are what help to separate and distinguish the safety of silicon dioxide based on those characteristics.
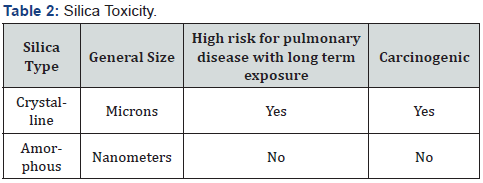
Historically, the guidelines set forth by large health related entities such as the World Health Organization primarily described negative and harmful health effects related to silicon dioxide. However, as stated previously, through the advancements made in science, this is no longer the case. Since silicon dioxide is found in plants and drinking water, the general understanding is that it is considered as being safe and this holds truth to a great extent. It is almost universally accepted that the silica which human beings consume through the diet does not accumulate within the body. In fact, research has proved that the silica consumed through the diet is actually completely removed by the kidneys in what is understood to be an unchanged manner. Silicon dioxide is becoming a quite common ingredient throughout numerous consumer products, and it is important that these consumers are aware of the different ways each form of silica can affect the human body. Silica is primarily synthesized from an aqueous solution with dissociated monomeric acid or from a vapor of a silicon compound [7,8]. Occupational exposure to crystalline silica has been linked with several pulmonary diseases and current literature suggests that there is a direct correlation between an increased amount of crystalline silica inhalation and the increased risk for silicosis, Chronic Obstructive Lung Disease (COPD), Tuberculosis (TB), and lung cancer [9-11]. There is a clear pulmonary hazard that has been determined with chronic exposure to crystalline silica and this becomes extremely important in those individuals who are already suffering from some sort of an underlying pulmonary dysfunction or disease [11]. By understanding the key differences between the different types of silica, a better understanding of the related cause and affect can be easily achieved.
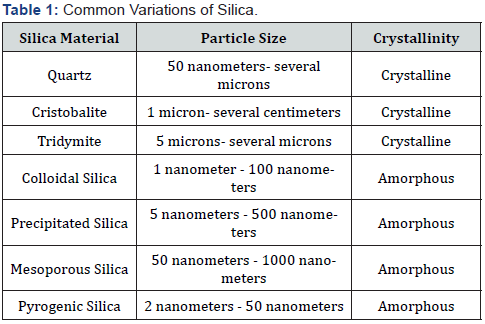
Crystalline silica can be broken down into three major typesquartz, cristobalite and tridymite [12]. Of these, the quartz form of silica is the most common and widely recognized of its kind. It is often utilized to manufacture surfacing materials such as granite flooring and counter tops. Crystalline silica is well known for its role in construction, mining, and sandblasting. The imposed health risks associated with prolonged and excessive exposure to crystalline silica dust particles from these occupational related materials continues to be examined as there have been obvious respiratory related diseases that have occurred secondary to exposure of this crystalline silica. The theory behind this is due to the fact that as individuals are constantly in direct contact with these crystalline dust particles, millions upon millions of these structures are able to breach into the respiratory tract through the nares as a person normally inhales to breathe. Overtime, the crystalline silica begins to build up within the pulmonary system and causes an array of symptoms, eventually leading to pulmonary disease and or possibly lung cancer [10,11,13]. Amorphous Silica on the other hand, is a form of silica which is most often synthetically manufactured and does not contain crystalline silica. There are three major classifications associated with amorphous silica- wet process silica, chemically or physically modified silica, and pyrogenic silica [14]. Amorphous silica is commonly used as silicon dioxide within food additives and is generally regarded as safe by different governmental agencies. The table provided below helps to further distinguish between crystalline and amorphous silica.
It is clear from the Table 1 above, that particle size and crystallinity are very dependent on one another. The potential hazards associated with inhalation of crystalline silica exposure tend to be associated with those structures which are primarily in the micron-size range, while the safer amorphous silica is always visualized in nanometers. With that said, it is important to note that due to the nanomolecular size of these particles, the only practical way of examining and visualizing these structures is through the use of a high-powered electron microscope and due to the high costs and limited quantity of these microscopes, it becomes very difficult to conduct mass amounts of studies related to nanomolecular silica. When using a high-powered electron microscope to assess the phenotypic characteristics of the different types of silica, crystalline structures such as quartz, resemble an arrangement of molecules which are ordered in an array of proportions with structures pointing in every direction.
Generally, Amorphous silica can present some selective order but lacks the sharp pattern. However, in certain manufactured goods where the silica is fractionated and altered for a specific use it is possible to create a ‘crystalline-like’ structure from an amorphous silica and this is particularly true for certain antimicrobial cleaning agents and pesticide products. In this instance, a colloidal or food grade silica is altered and used as a base material in order to modify solid surfaces at the nanomolecular level and become inherently resistant to microbial contamination and in fact biocidal to viral, bacterial and fungal species. Since many of these antimicrobial products do in fact use non-crystalline silica and instead alter colloidal silica to form a crystalline-like structure, the concerns relative to inhalation toxicity and pulmonary disease can be reduced as there have not been any studies that allow for the classification of amorphous silica as a silica that causes direct damage from inhalation nor have there been any findings which link amorphous silica as being carcinogenic in humans [15,16]. With that said, it is understandable that in certain individuals who have a history of pulmonary disease or respiratory issues there must be extra precaution taken when being exposed to amorphous silica as this and numerous other factors including breathing polluted air, can possibly create further difficulty Table 2. In terms of animal inhalation studies with synthetic amorphous silica, some of the results indicated that at higher concentrations, inflammation and granuloma formation did occur but these were both reversible and there were no signs of progressive fibrosis of the lungs that were evident [16-19].
Summary
In conclusion, there is no doubt that silica continues to find itself as a useful component to numerous industries around the world. As an ingredient used within antimicrobial agents, there have been extraordinary efficacy results demonstrated for many bacteria, viruses, and other harmful pathogens. Most consumers who understand the science and ‘mechanical kill’ mechanism of action, associated with nanomolecular silica and microbes are convinced that this process is effective and longer lasting than traditional ‘chemical kill’ agents. What is not well understood is the safety related to using nanomolecular silica within these antimicrobial agents, and the answer to this is entirely dependent on the type of silica being utilized as an ingredient. Although the concept of applying nanomolecular silica for antimicrobial purposes is relatively new, the use of silica has been practiced for centuries and with a clear understanding of the phrases used to define or describe silica, such as crystalline, amorphous, noncrystalline, and crystalline-like, an overall comprehension of the safety and toxicity related to silica can be determined. There are direct pulmonary hazards that have the potential to occur secondary to long-term or occupational exposure with crystalline silica. These include the increased risks for silicosis, Chronic Obstructive Lung Disease (COPD), Tuberculosis (TB), and even lung cancer. In contrast, non-crystalline or amorphous silica has not been proven to increase the risk for any of the previously mentioned pulmonary diseases. With the exception of limited case reports in conjunction with inadequately described exposure quality, there is no proof that intentionally manufactured synthetic amorphous silica, even that which has been fractionated and altered to be viewed as ‘crystalline-like’ pose any serious harm or increased risk for disease or irreversible damage to the human lung. As with all human exposure to various elements, continuous studies are highly recommended, and this is particularly important for nanomolecular silica since its use is expanding throughout several industries.
Conflict of Interest
The authors mentioned above have no conflict of interest to declare and did not receive any specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
- Moghimi SM, Hunter AC, Murray JC (2005) Nanomedicine: current status and future prospects. FASEB J 19(11): 311-330.
- Hirsch LR, Stafford RJ, Bankson JA, Sershen SR, Rivera B, et al. (2003) Nanoshell-mediated near-infrared thermal therapy of tumors under magnetic resonance guidance. Proc Natl Acad Sci USA 100(23): 13549-13554.
- Barik TK, Sahu B, Swain V (2008) Nanosilica-from medicine to pest control. Parasitol Res 103(2): 253-258.
- Mubshar Hussain, Khawar Jabran (2018) Mechanical Weed Control in Non-Chemical Weed Control.
- Araújo LA, Addor F, Campos PM (2016) Use of silicon for skin and hair care: an approach of chemical forms available and efficacy. An Bras Dermatol 91(3):331-335.
- Nel A, Xia T, Madler L, Li N (2006) Toxic potential of materials at the nanolevel. Science 311(5761): 622- 627.
- Peter V Sushko et al. (2005) J. Phys.: Condens. Matter 17 S2115.
- Lopatin SI, Stolyarova VL, Sevastyanov VG, et al. (2012) Determination of the saturation vapor pressure of silicon by Knudsen cell mass spectrometry. Russ. J. Inorg. Chem. 57: 219-225.
- Hessel PA, Sluis-Cremer GK (1987) Silica, silicosis, and lung cancer among ceramic workers: a case- referent study. Am J Ind Med 12: 219-222.
- Hnizdo E, Vallyathan V (2003) Chronic obstructive pulmonary disease due to occupational exposure to silica dust: a review of epidemiological and pathological evidence. Occup Environ Med 60(4): 237-243.
- Checkoway H, Franzblau A (2000) Is silicosis required for silica-associated lung cancer? Am J Ind Med 37(3): 252-259.
- Federico Musso, Piero Ugliengo, Xavier Solans-Monfort, Mariona Sodupe (2010) Periodic DFT Study of Radical Species on Crystalline Silica Surface. The Journal of Physical Chemistry C 114 (39): 16430-16438.
- Calvert GM, Rice FL, Boiano JM, Sheehy JW, Sanderson WT (2003) Occupational silica exposure and risk of various diseases: an analysis using death certificates from 27 states of the United States. Occup Environ Med 60(2): 122-129.
- Merget R, Bauer T, Küpper HU, Philippou S, Bauer HD, et al. (2002) Health hazards due to the inhalation of amorphous silica. Arch Toxicol 75(11-12): 625-634.
- Checkoway H (2000) Epidemiological evidence on the carcinogenicity of silica: factors in scientific judgement. Ann Occup Hyg 44: 483-484.
- Warheit DB (2001) Inhaled amorphous silica particulates: what do we know about their toxicological profiles? J Environ Pathol Toxicol Oncol 20(Suppl 1): 133-141.
- Lee KP, Kelly DP (1992) The pulmonary response and clearance of Ludox colloidal silica after a 4-week inhalation exposure in rats. Fundam Appl Toxicol 19(3): 399-410.
- Wilson RK, Stevens PM, Lovejoy HB, Bell ZG, Richie RC (1981) Respiratory effects of inhaled amorphous silica. In Health effects of synthetic silica particulates. Edited by: Dunnom DD. American Society for Testing and Materials pp. 185-198.
- Hamilton RF, Thakur SA, Holian A (2008) Silica binding and toxicity in alveolar macrophages. Free Radic Biol Med 44(7): 1246-1258.






























