Screening of Anti-aflatoxigenic Potential of Neem Seed Kernel Extracts in Solid Culture Media
Venkataiah Bhootham, Mir Zahoor Gul, Suresh Velpula, Sashidhar Rao Beedu and Karuna Rupula*
Department of Biochemistry, University College of Science, Osmania University, India
Submission:October 01, 2020;Published: November 19, 2020
*Corresponding author:Karuna Rupula, Department of Biochemistry, University College of Science, Osmania University, Hyderabad 500 007, Telangana, India
How to cite this article: Venkataiah B, Mir Zahoor G Suresh V, Sashidhar R B, Karuna R. Screening of Anti-aflatoxigenic Potential of Neem Seed Kernel Extracts in Solid Culture Media. Adv Biotech & Micro. 2020; 16(1): 555928.DOI:10.19080/AIBM.2020.15.555928
Abstract
Introduction: This study was aimed to screen the antifungal activity of various neem seed kernel extracts with reference to the inhibition of Aspergillus parasiticus (NRRL 2999) fungal growth and production of aflatoxins in solid culture media.
Methods: Aqueous extract (0.05, 0.25, 0.5 and 1.0% w/v), n-hexane, methanolic, ethyl acetate and acid-methanolic extracts (0.01, 0.1, 0.5 and 1.0% w/v) were prepared from the neem seed kernels and tested on the YES agar solid medium inoculated with the above fungal strain.
Results: A gradual increase in inhibition of fungal growth and toxin production was observed with increasing concentration of the aqueous, methanolic, ethyl acetate extracts and found to be statistically significant (p<0.05 and p<0.01 respectively). Among all the extracts, the methanolic extract was more profound in inhibiting the fungal growth and toxin elaboration, while hexane extract was less effective.
Conclusion: Neem seed kernel extracts showed anti-aflatoxigenic potentials and may find applications in formulating anti-fungal preparations for crop protection in field and in human and animal health care.
Keywords:Aflatoxins; Azadirachta indica; Aspergillus parasiticus; Neem seed kernel extracts
Introduction
Aflatoxins, are toxic secondary metabolites produced predominantly by Aspergillus group of fungi and have been categorized as class I carcinogens by the International agency for research on cancer [1]. Ingestion of food/feed contaminated with aflatoxins causes chronic and acute toxicity including hepatotoxicity, teratogenicity, immunotoxicity and even death in humans and animals [2, 3]. These fungi are cosmopolitan organisms that grow naturally on a variety of natural substrates including cereal grains, oil seeds, cotton seeds etc., [4,5]. With respect to their air-borne propagules, they can be easily distributed from their natural ecological niches to susceptible plants and crops all over the world [6]. Several strategies have been employed to minimize aflatoxin contamination, which include synthetic and natural antifungal compounds. The natural compounds offer relatively lower incidence of adverse reactions and reduced cost over modern synthetic pharmaceuticals [7]. In this context introduction of eco-friendly and safe alternative control strategies using natural products as a source of antimicrobial agents was felt. Plants are known to produce an enormous variety of phytoalexins which constitute heterogeneous group of compounds including terpenoids, phenolic compounds and glycosteroids. These phytoalexins protect the plants against pathogens and pests and are also considered as molecular markers in plant disease diagnosis [8]. Neem (Azadirachta indica A. Juss.) is well known in the Indian sub-continent with wide spectrum of biological activities in ecological, medicinal, and agricultural sectors. In Telangana, south of central India, neem is known as ‘Vepa’ or the purifier of air. Mere presence of the neem tree near human dwellings is believed to materially improve human health and even act as a prophylactic against malarial fever and even cholera and also people lay a patient suffering from smallpox, chickenpox, or even syphilis on a bed of neem leaves and fanned with a neem branch, while leaves and fruits are also used in treating tuberculosis and heart diseases [9,10]. Because of such diverse curative properties, neem is appropriately known as The Village Pharmacy in rural India and has secured a place in the Indian Pharmacopoeia.
Neem’s medicinal properties are listed in the ancient documents Carak- Samhita and Susruta-Samhita, the books at the foundation of the Indian system of natural treatment, Ayurveda. It is described in the Ayurvedic texts as sarva roga nivarini (a universal reliever of all illness). Neem has been used in Ayurveda and Unani as anti-ulcer, antioxidant, anticancer, spermicidal, antidiabetic, immunomodulating and immunocantraceptive in humans and animals [11-13]. Use of neem extract for the treatment of variety of diseases including malaria, rheumatism, gastric ulcer, cardiovascular diseases, osteoporosis, and vaginal infections, as well as mosquito repellent have been reported. The agrochemical uses of neem have also been established with respect to restraining insect feeding and impeding the development of other pests and nematodes [14]. Furthermore, the role of neem extracts is not only restricted in the treatment of many diseases, but also in combating the microbial infections. Several studies reported anti-bacterial, antiviral, insecticidal and fungicidal activities [15,16] by the crude extracts of different parts of neem. Among its various parts, neem seed kernel has maximum number of bioactive terpenoids such as azadirachtin, nimbidin and gedunin with proven antifungal activity [17,18]. Earlier studies reported the inhibitory effect of neem leaf extracts, oil and cake on various group of fungi [19,20] . The effect of various neem seed kernel extracts on A. parasiticus (NRRL 2999) a toxigenic strain known to produce copious amounts of aflatoxins, has not been studied so far using solid media. Hence, the present study was aimed to screen and evaluate the anti-fungal and antiaflatoxigenic potential of various neem seed kernel extracts (aqueous and solvents) in yeast extract sucrose (YES) agar solid medium, inoculated with A. parasiticus (NRRL 2999).
Materials and Methods
Chemicals
Fresh neem seeds were procured from the local market vending Ayurvedic and Herbal materials (Hyderabad, Telangana, India.). The voucher specimen for neem seeds. (OU-Biochem/ VS/BSR/KR-01) has been preserved in the laboratory for future reference. Reference standards for aflatoxins (AFB1, AFB2, AFG1 and AFG2) were obtained from Sigma Aldrich Chemicals Co., St. Louis, USA. Silica gel coated TLC 60 (20 x 20cm; make: Merck KGaA, Darmstadt, Germany) plates were procured from Merck Life Science Pvt. Ltd, Mumbai. All the other chemicals, reagents and solvents used were of analytical grade.
Strain
A highly toxigenic fungal strain, Aspergillus parasiticus (NRRL 2999) known to produce Aflatoxin B1, B2, G1 and G2 was obtained from United State Department of Agriculture (USDA), Peoria, Illinois, USA. Cultures were maintained on potato dextrose agar (PDA) slants for 8 days at 28.0±1.0 0C in a cooling incubator (Remi, Laboratory Instruments, Mumbai, India)
Yeast extract sucrose (YES) agar solid medium
Yeast extract sucrose (YES) agar solid medium with 2% (w/v) yeast extract, 15% (w/v) sucrose was prepared, and the pH was adjusted to 6.0. Later 1.5% (w/v) agar was added to the media and was sterilized by autoclaving at 1.05 kg cm-1 (103 kPa) pressure at 121 0C for 15 min.
Preparation and Incorporation of different neem seed kernel extracts in YES solid agar medium
Neem seed kernels were taken and powdered in a laboratory mechanical grinder (Sumeet, Mumbai, India) to a fine particle. The preparation of the various extracts of the neem seed kernel powder, containing various types of bioactive compounds was based on the method described by [21].
Aqueous extract
The neem kernel powder (50 g, moisture adjusted) was mixed with 1 L of sterile potassium phosphate buffer (10 mM, pH 7.0) and was left overnight at 4 0C. Later it was filtered by using sintered funnel and then the pH of the aqueous solution was adjusted to 6.0. The solution was further filter sterilized by using 0.45 μm membrane filter unit (Millipore, USA) under aseptic conditions. Aqueous neem kernel extracts of volume 1.0 mL, 5.0 mL, 10.0 mL and 20 mL were taken and added separately to a volume of 99.0 mL, 95.0 mL, 90.0 mL and 80.0 mL of YES media respectively to prepare 1.0%, 5.0%, 10.0% and 20.0% (v/v) concentrations of aqueous extracts. These extracts prepared are equal to the respective weights of 0.05%, 0.25%, 0.5% and 1.0% (w/v) neem kernel powder. The aqueous extract of neem seed kernel is a rich source of carbohydrate, amino acids, pigments and soluble bitters [21].
Hexane extract
The neem kernel powder (250g) was defatted with n-hexane by using Soxhlet apparatus [22]. Hexane was evaporated using flash evaporator (Superfit, Mumbai, India) at 45-50 °C that resulted in brown viscous oil which was used as n-hexane extract. To 4.0 mL (equivalent to 4 g) of oil, 2 mL of petroleum ether (boiling point, 52-55⁰C) and was thoroughly mixed. To this total volume (6.0 mL) of the mixture, equal volume of sterile distilled water was added and was shaken vigorously, and the pH of the emulsion was adjusted to 6.0. This solution was filter sterilized by using 0.45 μm membrane filter unit under aseptic conditions. A volume of 0.03 mL, 0.3 mL, 1.5 mL and 3.0 mL of the above emulsion was added to 99.97 mL, 99.7 mL, 98.5 mL and 97.0 mL of YES media respectively under aseptic conditions to get the 0.01%, 0.1%, 0.5% and 1.0% concentrations of the n-hexane extract. Appropriate blanks were also prepared by using petroleum ether without n hexane extract. The n-hexane extract of neem seed kernel have been reported to also contain active bioactive constituents such as salannin, nimbin, azadiradione and epinimbin [21]. The defatted neem seed kernel powder was then re extracted with the respective solvents as described below.
Methanolic extract
Hundred grams (100.0g) of defatted powder was extracted three times with 50 mL portion of the 95% ethanol. The ethanol layer was separated by using sintered funnel, and later subjected to flash evaporation to remove ethanol. The resultant thick residue (46.0g) obtained by this process was subjected to two quick, efficient partitioning between petroleum ether and 95% aqueous methanol. The petroleum ether layer was discarded and the methanolic fraction was dried by using flash evaporator. The resultant methanolic extract was dried under nitrogen and 4.0 g was dissolved in 8.0 mL of sterile distilled water and the pH was adjusted to 6.0. It was filtered sterilized by 0.45 μm membrane filter unit. Methanol extract of volume 0.02 mL, 0.2 mL, 1.0 mL and 2.0 mL was added to 99.98 mL, 99.8 mL, 99.0 mL and 98.0 mL respective volumes of YES media to get 0.01%, 0.1%, 0.5% and 1.0% (w/v) concentrations respectively. The methanol extract is found to be rich in proteins, sugars and azadirachtin [21].
Ethyl acetate extract
The above methanolic extract (10. 0 g) was dissolved in ethyl acetate (50 mL) and was partitioned with equal volume of double distilled water. The ethyl acetate portion was separated and was dried by using flash evaporator. The resultant thick brown residue (4.3 g) was again dried under nitrogen. Four grams of this dried material was again dissolved in 2 mL of ethyl acetate and 2 mL of methanol. This mixture was shaken vigorously and was mixed with 4 mL sterile distilled water. The pH of the emulsion was adjusted to 6.0 and was filter sterilized by 0.45 μm membrane filter unit. This emulsion of volume 0.02 mL, 0.2 mL, 1.0 mL and 2.0 mL was added to 99.98 mL, 99.8 mL, 99.0 mL and 98.0 mL respectively of YES media to get 0.01%, 0.1%, 0.5% and 1.0% (w/v) concentrations, respectively. Azadirachtin, a terpenoid is a main constituent of ethyl acetate extract of neem seed kernel [21].
Acid-methanolic extract
Defatted neem kernel powder (100 g) was extracted with 60 mL of 1.0% (v/v) HCl in methanol for 1 hr in a mechanical shaker. The methanolic portion was flash evaporated at 45-50 0C to remove methanol and HCl. The residue (4.0 g equivalent) was then re-dissolved in 20 mL of sterile water and pH was adjusted to 5.8- 6.0 with 0.1 N NaOH, before addition to the liquid medium. A volume 0.05 mL, 0.5 mL, 2.5 mL, 3.75 mL and 5.0 mL of this acid methanol extract was added separately to 99.95 mL, 99.5 mL, 97.5 mL, 96.25 mL and 95.0 mL respective volumes to prepare 0.01%, 0.1%, 0.5%, 0.75% and 1.0% respective concentrations of the extracts. The acid methanolic extracts of neem seed kernel contain many biologically active compounds, mainly phenols and polyphenols [21].
Incorporation of different neem kernel extracts into YES agar solid medium
All the extracts were filtered by using sintered glass funnel and then pH was adjusted to 6.0 with sodium hydroxide (0.1M). Later, filter sterilized by using 0.45 μm membrane filter unit (Millipore products divisions, MA, USA). Appropriate respective volumes of each extract were incorporated in to YES agar media just before the solidification (45 0C) to get 0.05, 0.25, 0.5 and 1.0% (w/v) of aqueous and 0.01, 0.1, 0.5 and 1.0% (w/v) concentrations for hexane, methanolic, ethyl acetate and acid-methanolic extracts under aseptic conditions. In the present study, SDS concentration at 0.001% in YES media was maintained for solubility of hydrophobic compounds of neem seed extracts, as our earlier studies revealed no effect on growth of A. parasiticus (NRRL 2999) aflatoxin elaboration at this concentration [25]. Immediately 4.0 mL of the medium was poured into polycarbonate petri plates (6.0 cm diameter) and allowed to solidify. At the centre of the petri plates, a well was punched by using a gel puncher (well size, 3 mm diameter).
Inoculation of the YES solid agar medium
Spore suspension of A. parasiticus (NRRL 2999) was prepared and 25 μL of it (spores 5 x 105/mL) was dispensed into the well, under aseptic conditions in the laminar air flow chamber (Microfilt, Pune, India). Appropriate controls were also set up in a same manner except that the extract replaced with sterile distilled water and the experiments were conducted in triplicates. After inoculation, the Petri plates were incubated for 8 days, at 28±1 °C in a cooling incubator. Eight-day old cultures were monitored for the fungal growth and aflatoxin elaboration. The mean colony diameter (mm) was measured for monitoring fungal growth. The solid media along with the mycelial mat was processed for the extraction of aflatoxins.
Extraction of aflatoxins
The YES solid agar medium containing mycelial mat was processed using extraction solvent (methanol: water, 55:45), and homogenized at 15,000 rpm using a high speed mechanical blender (Boss, Mumbai, India). The homogenate was later filtered through siliconized glass wool and was washed twice with 10 mL distilled water. Aflatoxin was extracted from the filtrate by solvent extraction procedure. The filtrate containing aflatoxins was initially defatted with 25 mL n-hexane in the presence of sodium chloride (80 mg/mL), followed by extraction with 10 mL chloroform, twice. The pooled chloroform extracts containing aflatoxin was passed through anhydrous sodium sulfate and flash evaporated to dryness at 45 °C. The residue was reconstituted in benzene: acetonitrile (98:2).
Estimation of aflatoxins
Aflatoxins were estimated by digital image-based TLC-flourodensitometric method [23,24]. Sample extracts in benzene: acetonitrile (98:2) were spotted (10 μL) on the activated TLC plates. The TLC plates were developed in toluene: ethylacetate: formicacid (6:3:1) and visualized under long wave UV light (λ 365 nm). Annotated digital images of the spots were recorded by the CCD camera (UV tech, Cambridge, UK) and saved in PC-compatible file format (tif file) in a CD. Later, the digital density images of different toxins were analysed by the software supplied by the manufacturer and used for determining the net density, which was measured as peak volume. Reference standards AFB1, AFB2, AFG1 and AFG2 were used for calculating the aflatoxin content in the samples. The aflatoxin content in the samples was expressed as μg mL-1 of YES used to prepare the YES solid agar culture media.
Results and Discussions
In view of the eco-friendly properties associated with natural plant products for application in agriculture and medicine, various extracts of potent medicinal plant parts have been used extensively as natural antimicrobials and antioxidants. Hence, in the present study, the neem seed kernel extracts (aqueous, hexane, methanolic, ethyl acetate and acid-methanolic) were evaluated for their antifungal activity in YES solid agar media inoculated with A. parasiticus (NRRL 2999). In controls, the mean colony diameter (fungal growth) was found to be 40.0±3.2 mm and total toxin production was found to be 1.87±0.07 μg mL-1 on 8th day of incubation period.
Aqueous extract
Presence of aqueous extract of the neem seed kernel in YES agar medium showed an inhibitory effect on growth of A. parasiticus (NRRL 2999) as well as aflatoxin production. A gradual increase in inhibition of fungal growth from 22.5 to 80% was observed with increasing concentration of the extract from 0.05 to 1.0% (w/v) and found to be statistically significant (p<0.01) when compared to controls (Figure 1a). The total toxin production was inhibited by 21, 62, 62 and 80% at 0.05, 0.25, 0.5 and 1.0% concentrations, respectively on 8th day of incubation and was statistically significant (p<0.01) (Figure 2a). Previously, [27], Reported. < 90% inhibitory effects of aqueous extracts of neem leaf (50 % v/v) on aflatoxin production by A. parasiticus on Sucrose Low Salts (SLS) a semi synthetic medium at the end of 4 days of the incubation period. Their studies also concluded that the activity of glutathione S transferase was reduced upto 80 % in the toxigenic culture of A. parasiticus exposed to neem extract at a concentration of 1.56% (v/v).
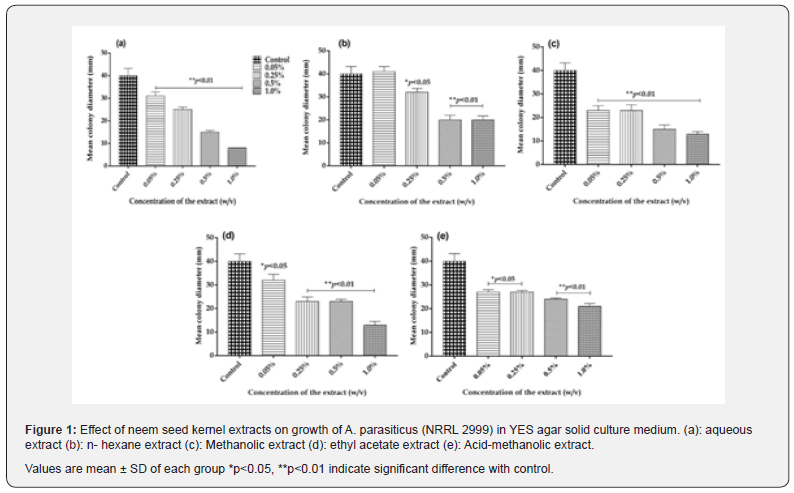
Hexane extract
Hexane extract of the neem seed kernels at 0.1, 0.5 and 1.0% concentrations showed a significant inhibition (p<0.05, p<0.01) on fungal growth (Figure1b) and the total toxin production was inhibited by 24, 54 and 53% on 8th day of the incubation respectively, and the inhibition was found to be statistically significant (p<0.05; p<0.01)(Figure 2b). However, at the lowest concentration of 0.01%, the extract was not effective against fungal growth and toxin production. Earlier [24-27]. Reported complete inhibition of the growth of Fusarium moniliforme, Aspergillus niger, Drechslera rostratum and Macrophomia phaseolina when treated with (10%) of neem seed oil extracts
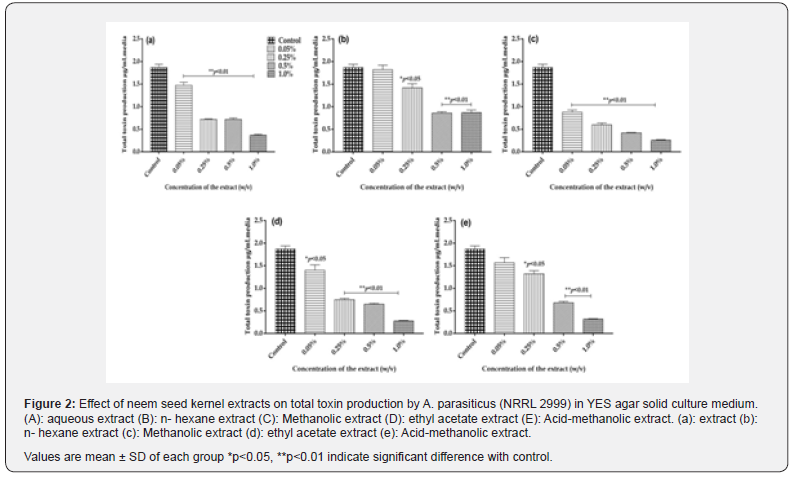
Methanolic extract
Various concentrations (0.01 to 1%) of methanolic extract of the neem seed kernels showed a significant inhibition (p<0.01) on fungal growth and toxin production. At 0.01 and 0.1% concentrations, the fungal growth was inhibited by 45%, whereas, at the higher levels of 0.5 and 1.0% the inhibition was found to be 63 and 68% respectively (Figure 1c). The toxin production was inhibited by 53, 68, 78 and 86% at 0.01, 0.1, 0.5 and 1% levels, respectively and it was statistically significant (p<0.01). The inhibition in toxin production was found to be dose dependent (Figure 2c). Earlier [28] reported that the methanolic extracts of leaves of Azadirachta indica exhibited higher inhibitory activity against Aspergillus niger as compared to its aqueous extracts. Studies by [29] also have demonstrated that triterpenoids mixture derived from the methanolic extract of neem oil was effective against phytopathogenic fungi Drechslera oryzae, Fusarium oxysporum, and Alternaria tenuis. However very limited information on the effects of methanolic extracts of neem seed kernels on Aspergillus fungal growth and aflatoxin production are available. In this regard the present study revealed potent inhibitory effects of methanolic extracts of need seed kernels on the Aspergillus parasiticus (NRRL 2999) fungal growth and aflatoxin production using YES agar (solid) media. Further the effect of methanolic extract of the neem seed kernels (0.5%) was found to be 3 folds reduced in the solid media in the present study when compared to the liquid media culture conditions in our earlier studies [30].
Ethyl acetate extract
Ethyl acetate extract of the neem seed kernels incorporated in YES agar medium had inhibited fungal growth and toxin production. Inhibition of fungal growth was found to be 20, 43, 43 and 68% at 0.01, 0.1, 0.5 and 1.0% levels respectively (Figure 1d). In the presence of the extract, toxin production was less when compared to control group and was found to be 25, 60, 65 and 69% at 0.01, 0.1, 0.5 and 1.0% concentrations, respectively. The inhibition in fungal growth and toxin production was dose dependent and statistically significant (p<0.05, p<0.01) (Figure 2d). Previously [31] also reported that organic extracts (ethanol and ethyl acetate) of neem leaves at (20% w/v; concentration) caused 100 % inhibition of A. flavus and 88.77% of A. niger fungal growth.
Acid methanol extract
At a higher concentration of 1 % the acid methanol extract of the neem seed kernels inhibited fungal growth by 47.5% (Figure 1e) and was statistically significant (p<0.01). Inhibition of total toxin production in the presence of this extract was found to be 16, 24.5, 63.2 and 83% at 0.01, 0.1, 0.5 and 1% concentrations in a dose dependent manner respectively (p<0.01) (Figure 2e) However the fungal growth and toxin production at various concentrations of the neem seed acid methanol extract was less as compared to other extracts. In the present study, all the five types of the neem seed kernel extracts (aqueous, hexane, methanol, ethyl acetate and acid methanol) were found to inhibit fungal growth and aflatoxin production. Among the various extracts, methanolic extract at 0.01, 0.1 and 0.5% was more effective in inhibiting the fungal growth and toxin elaboration. These results indicated that the inhibition of fungal growth by neem seed kernel extracts was associated with aflatoxin production. The difference in the fungicidal activity of various neem seed kernel extracts, which was evident from the present experimental investigations, is important in evaluating new antifungal compounds with different mode(s) of action. In our earlier study, various neem seed kernel extracts were found to inhibit fungal growth and aflatoxin production by A. parasiticus (NRRL 2999) in YES liquid culture medium. In the present investigation, the study was carried out on solid agar medium. Further, in the solid culture media exposed to methanolic extracts the total aflatoxin production was significantly reduced at all concentrations tested (p <0.01) while in cultures exposed to ethyl acetate it was significantly reduced at 0.1 ,0.5 and 1% (p < 0.01) as compared to their effects in liquid culture media. The production of total toxin in controls on solid agar media was found to be nearly 2 folds less (1.87 μg/mL) when compared to the liquid culture medium (3.9 μg/mL), due to enhanced uptake of nutrients by the fungus in the liquid culture media. In the present studies, using the agar diffusion principle, the spores inoculated were monitored for their germination into fungal biomass during which they interact with neem seed kernel extracts present in the YES solid agar medium. Consequently, the variations in the fungal biomass observed when compared to the controls indicated the direct effect of these extracts on the fungal growth. In this perspective, the use of solid culture media for screening the inhibitory effects of new anti-fungal/ anti-aflatoxigenic agents provides as an alternative and suitable method.
Study Limitations
The present study in solid media indicated a two - three fold reduction in the total aflatoxin production between controls and methanolic extracts when compared with the liquid culture media. Natural infestation of agricultural commodities by toxigenic strains of fungi simulates solid media. Hence the present study was aimed at screening of anti-aflatoxigenic potential of neem seed kernel extracts in solid culture media.
Two major cytosolic enzymes Fatty acid synthase and Glutathione synthase of the toxigenic strains of Aspergillus parasiticus are reported to be higher in the toxigenic strains as compared to the non-toxigenic stains. Further toxigenic strains of Aspergillus parasiticus exposed to neem seed extracts in liquid culture media resulted in inhibition of Glutathione S transferase activity. However, there are no reports on the influence of neem seed extracts on these enzymes in toxigenic strains of Aspergillus parasiticus on solid culture media. Hence the present study can be extended for further investigation in this regard.
Conclusion
The neem seed kernel extracts were found to possess inhibitory effects on A. parasiticus (NRRL 2999) fungal growth and aflatoxin production in the solid culture media tested. Further, the extracts might have practical applications as novel antimicrobial agents, especially as seed dressing agents to contain fungal infestation during storage. Additionally, formulations based on neem seed kernel extracts along with detergents can be successfully employed as fungicidal or fungi-static agents for field crops as well as in antimicrobial medicines for human applications.
Author’s contribution
VB and MZG carried out all the experimentation, acquisition and analysis of data and drafting of the manuscript. SV assisted with the analysis of data. BSR provided the toxigenic strains and co-supervised the study. KR conceived, designed, and supervised the study and revised manuscript. All authors have read and approved the final manuscript.
Declaration of competing interest
The authors declare that there are no conflicts of interest.
Ethical Approval
N/A
Acknowledgment
All the authors acknowledge the, DST-PURSE - II programme (C-DST-PURSE-II/6/17) Government of India New Delhi, implemented at Osmania University, Hyderabad for the infrastructural facilities. The author MZG Acknowledge the CSIR, New Delhi for Research Associate fellowship vide sanction order No. 09/132(0883)/2019-EMR-1. Author SV also acknowledges the University Grants Commission, New Delhi for the BSR-SRF fellowship.
References
- IARC, IAfRoC (2002) Summaries& Evaluations. In: Aflatoxins 82: 171.
- Peraica M, Radić B, Lucić A, Pavlović M (1999) Toxic effects of mycotoxins in humans. Bulletin of the World Health Organization 77(9): 754-766.
- Wagacha JM, Muthomi JW (2008) Mycotoxin problem in Africa: current status, implications to food safety and health and possible management strategies. Int J Food Microbiol124(1):1-12.
- Diener UL, Cole RJ, Sanders TH, Payne GA, Lee LS, Klich MA (1987) Epidemiology of Aflatoxin Formation by Aspergillus Flavus. Annu Rev Phytopathol 25(1):249-270.
- Cotty PJ, Bayman DS, Egel DSa, Elias KS (1994) Agriculture, aflatoxins and Aspergillus. In: Powell KA, Renwick A, Peberdy JF (Eds.) The Genus Aspergillus: From Taxonomy and Genetics to Industrial Applications, Plenum Press. New York, NY, USA. pp. 1-27.
- Payne GA (1998) Process of contamination by aflatoxin-producing fungi and their impact on crops. In: Sinha KK, Bhatnagar D (Eds.) Mycotoxins in Agriculture and Food Safety, Marcel Dekker Inc, New York, USA. pp. 279-306.
- Anastacio PM, Lutzhoft HC, Halling-Sorensen B, Marques JC (2000) Surfactant (Genapol OX-80) toxicity to Selenastrumcapricornutum. Chemosphere 40(8): 835-838.
- Ahuja I, Kissen R, Bones AM (2012) Phytoalexins in defense against pathogens. Trends Plant Sci, 17(2):73-90.
- Mitra CR (1963) Neem Indian Central Oil Seeds Committee. Hyderabad, India.
- Susana OM, Tonny AA, Mary AA, Peter AAJ, Daniel B (2019) Medicinal Plants for Treatment of Prevalent Diseases.
- Akula C, Akula A, Drew R (2003) Somatic embryogenesis in clonal neem, Azadirachta indica Juss. and analysis for invitroazadirachtin production. In VitroCell Dev Biol Plant 39:304-310.
- Biswas K, Chattopadhyay I, Banerjee R, Bandyopadhyay U (2002) Biological activities and medicinal properties of neem (Azadirachta indica). Curr Sci 82(11): 1336-1345.
- Subapriya R, Nagini S (2005) Medicinal properties of neem leaves: a review. Curr Med Chem Anticancer Agents 5(2): 149-146.
- Bhambhani S, Lakhwani D, Gupta P, Pandey A, Dhar YV, et al. (2017) Transcriptome and metabolite analyses in Azadirachta indica: identification of genes involved in biosynthesis of bioactive triterpenoids. Scientific Reports 7: 5043(2017).
- Atawodi SE, Atawodi JC (2009) Azadirachta indica (neem): a plant of multiple biological and pharmacological activities. Phytochemistry Reviews 8(3):601-620.
- Jacobson M (1986) The Neem Tree: Natural Resistance Par ExcellenceNatural Resistance of Plants to Pests, American Chemical Society. Washington, DC, United States pp. 220-232.
- Rawat NS (1994) Neem products and their pesticidal characteristics. Everyman's Science, Indian Science Congress Association, Calcutta pp. 194-199.
- Srivastava P, Chaturvedi R (2011) Increased production of azadirachtin from an improved method of androgenic cultures of a medicinal tree Azadirachta indica Juss. Plant Signal Behav6(7): 974-981.
- Bansal RK, Sobti AK (1990) An economic remedy for the control of two species of Aspergilluson groundnut. Indian Phytopath 43(3): 451-452.
- Eswaramoorthy S, Muthusamy S, Mariappan V (1989) Inhibitory of Neem, Acacia, Prosopis and Ipomoea extracts on Sarocladium oryza and Fusarium oxysporium. Neem NewsLetter 6(1): 4 -5.
- Schroeder DR, Nakanishi K (1987) A Simplified Isolation Procedure for Azadirachtin. J Nat Prod 50(2): 241-244.
- Govindachari TR, Sandhya G, Ganeshraj SP (1990) Simple method for the isolation of azadirachtin by preparative high-performance liquid chromatography. Journal of Chromatography513: 389-391.
- Wacoo AP, Wendiro D, Vuzi PC, Hawumba JF (2014) Methods for Detection of Aflatoxins in Agricultural Food Crops. J Appl Chem 15.
- Tanuja K, Kavitha G, Karuna R, Sashidhar RB (2012) Substrate suitability of neem seed kernel for the growth and elaboration of aflatoxins by Aspergillus parasiticus (NRRL 2999). Indian J Nat Prod Res 3(3): 395-406.
- Tanuja K, Hemalatha K, Karuna R, Sashidhar Rao B (2010) Effect of various surfactants (cationic, anionic and non-ionic) on the growth of Aspergillus parasiticus (NRRL 2999) in relation to aflatoxin production. Mycotoxin Res 26(3): 155-170.
- Allameh A, Razzaghi Abyane M, Shams M, Rezaee MB, Jaimand K (2002) Effects of neem leaf extract on production of aflatoxins and activities of fatty acid synthetase, isocitrate dehydrogenase and glutathione S-transferase in Aspergillus parasiticus. Mycopathologia 154(2):79-84.
- Vir D, Sharma A (1985) Studies on the fungicidal properties of neem oil. Indian J Plant pathol 3(2): 241-242.
- Sharma D, Lavania AA, Sharma A (2009) An In vitro comparative screening of antibacterial and antifungal activities of some common plants and weed extracts. AsianJ Exp Biol Sci 33(1):169-172.
- Govindachari TR, Suresh G, Gopalakrishnan G, Banumathy B, Masilamani S (1998) Identification of Antifungal Compounds from the Seed Oil of Azadirachta indica.Phytoparasitica 26(2):109 -116.
- Godugu K, Rupula K, Sashidhar RB (2014) Inhibitory role of neem seed kernel extracts and terpenoids on growth and aflatoxin production by Aspergillusparasiticus (NRRL 2999). Indian J Nat Prod Resour 5(1):20-33.
- Mahmoud DA, Hassanein NM, Youssef KA, Abou Zeid MA (2011) Antifungal activity of different neem leaf extracts and the nimonol against some important human pathogens. Braz J Microbiol 42(3): 1007-1016.






























