Histological Analysis in the Process of Somatic Embryogenesis of Cycas Revoluta Thunb., from Callus Cultivated in vitro
Rodríguez de la O JL1* Lázaro J Flores VA2, Robledo Monterrubio MS3 and Contreras Cruz F3
1Department of Plant Science, Academy of genetics, Autonomous University Chapingo, Mexico
2Department of Plant Science, Thesis Master of Science in Biotechnology, Mexico
3Autonomous Chapingo University Carr Fed Mexico
Submission: May 05, 2020;; Published: September 09, 2020
*Corresponding author:Rodríguez de la O JL, Department of Plant Science, Academy of genetics, Autonomous University Chapingo, Mexico
How to cite this article: Rodríguez d l O J Lázaro J F V, Robledo M M, Contreras C F. Histological Analysis in the Process of Somatic Embryogenesis of Cycas Revoluta Thunb., from Callus Cultivated in vitro. Adv Biotech & Micro. 2020; 15(5): 555925. 10.19080/AIBM.2020.15.555925
Abstract
C. revolutacycical callus obtained from in vitro cultured megagametophyte tissues, were subculture to promote somatic embryogenesis ES, evaluating four variants of the Murashige and Skoog 1962 (MS) medium. Combining three growth regulators benciladenine (BA), 2.4-Dichlorophenoxyacetic (2.4-D), and kinetin (K), as well as picloram. The histology of tissues showed the organization and differentiation as previously to the formation of somatic embryos. The effect of the Ac. (ABA), at 0, 0.38, 1.13, 3.78 and 5.67 μM influenced the obtaining and development of embryogenic responses; and the combinations of BA, kin, 2.4-D, as well as giberélic acid (AG3)) and naphthalenacetic acid (ANA), influenced the maturity of embryos and their subsequent germination. Calluses were fixed in Formaldehyde Acetic Alcohol (FAA) and dehydrated in a histokinette brand leica TP 1020. Histological cuts were made in a rotational microtome American optical 820 with a thickness of 12-16 m, and dyed with safranin and fast green, placed in synthetic resin and dried in an oven at 45 °C for 15 days. The cuts were observed and photographed through a microscope carl zeiss, analyzed with the software zin lite 2012. The embryonic responses in the cells were evident by combining 4.44 + 1.36 μM BA + 2.4-D respectively, as well as 4.65 + 4.14 μM kin + picloram, at 170 and 345days. The histology performed, allowed to observe the cell differentiation and formation of somatic embryos from cells with dense cytoplasm and prominent nuclei, differentiating pro embryos globular type, and epidermal cells with divisions of pericline type. The combination of 4.44 + 1.35 μM BA + 2.4-D promoted the maturity and germination of embryos. Histology identified areas with a cellular symbiotic relationship and other regions with a wide cell division, associated with a meristematic activity (bipolar) that precedes the formation of somatic embryos.
Keywords: Histology; C. revoluta; Embryogenic responses; Growth regulators; Cellular processes
Introduction
Cycads are a group of dioecious and ancestral plants that appeared in the permian, flourished during the Mesozoic and reached their maximum distribution during the Jurassic [1]. They evolved from the progymnosperms of free spores and preceded the Gingkoales and Gnetales, [1-3] has dubbed them living fossils, and some authors point out that the permanence of such plants would be like having living dinosaurs. Cycas revoluta Thunb., commonly known as the palm of Sagú, is the oldest of the living cycads [4], the species symbolizes the economic importance of the order [5]; however, by its nature, it presents some permanence contradictions such as slow growth, dioiscism, asynchrony in sexual maturity, short periods of receptivity on the part of the female and long periods between pollination and fertilization; and in the development of THE ZE, among other things [6-8]. Propagation techniques are therefore limited to the use of branches and cuttings Robinson, (2010). Tissue culture techniques in the propagation of such a species, and cycads in general, are used at a relatively low frequency. The development of ES-based biotechnological protocols as an alternative to conventional propagation methods, through the use of non-gametical fusion tissues is essential for the conservation of fossil species such as cycads; in vitro research with this type of plants has been few in recent years and regeneration has been sought from ZE [9]; leaves, central vein and epicotyls [10,11]; endosperm and megagametophytes By [11,12] however, the use of megagametophytes as an explant has generated organogenetic responses (buds and roots), which ash more according to have limited use for the conservation of genetic resources due to the genetic instability inherent in this type of plants [10]. claimed the presence of embryogenic masses in ZE-derived calluses; however, no evidence of such structures was presented. Cabrera, Chávez, Sandoval, [13] pointed out the presence of similar driving elements traded in three-month-old calluses, derived from Dioon merolae’s megagametophytes; Dioon merolae however, the morphogenic differentiation after six months of cultivation was unsuccessful [14]. reported unorganized structures in ZEderived calluses and Zamia skinneri megagametophytes; but stated that it was not possible to determine whether these structures responded to a process of somatic organogenesis or embryogenesis. In many cases, when the formation of callus is induced for further differentiation to seedlings, this does not happen and it is not known whether the process started and could not continue to either Valverde, [15]. Therefore, it is very important to develop histological analysis to help assist the study and understanding of implicit cellular processes within the ES, in order to establish the best strategies for the conservation and in vitro propagation of cycads. In this main objective research was to analyze histologically the process of induction of somatic embryogenesis from C. revolutacalluses obtained in vitro from the tissues of megagametophytes.
Materials and Methods
The callogenesis obtained at 3 months of tissue culture of C. revoluta megagametophytes, were sub cultivated in vitro to promote ES, using a basic MS medium (1962), considering different stages, in which the combination of types and concentrations of growth regulators were used (Table 1). Table 1 Tipo y concentraciones de reguladores de crecimiento empleados en el proceso de ES de Cycas revoluta encada una de sus etapas. To analyze the processes involved at each stage of embryogenic development, they were selected calluses with physical characteristics primarily associated with cell differentiation before forming somatic embryos ES. Calluses were fixed in Formaldehyde Acetic Alcohol (FAA) and dehydrated combining gradual alcohols by a Histokiner Leica TP 1020 of 12 seasons, to be subsequently included in solid paraffin. The cuts (12-16 m) were cut using an American Optical 820 rotational microtome, then rum is selected and placed on a slide. The obtained sections were dyed with safranin and fast green. The staining process lasted 84 minutes, then they were permanently assembled with synthetic resin and allowed to dry in an oven at 45 °C for 15 days. The samples were observed through a Carl Zeis Microscope, model Axio Scope A1, with digital camera Axio Cam ICc 5 and analyzed with Zen lite 2012 software.
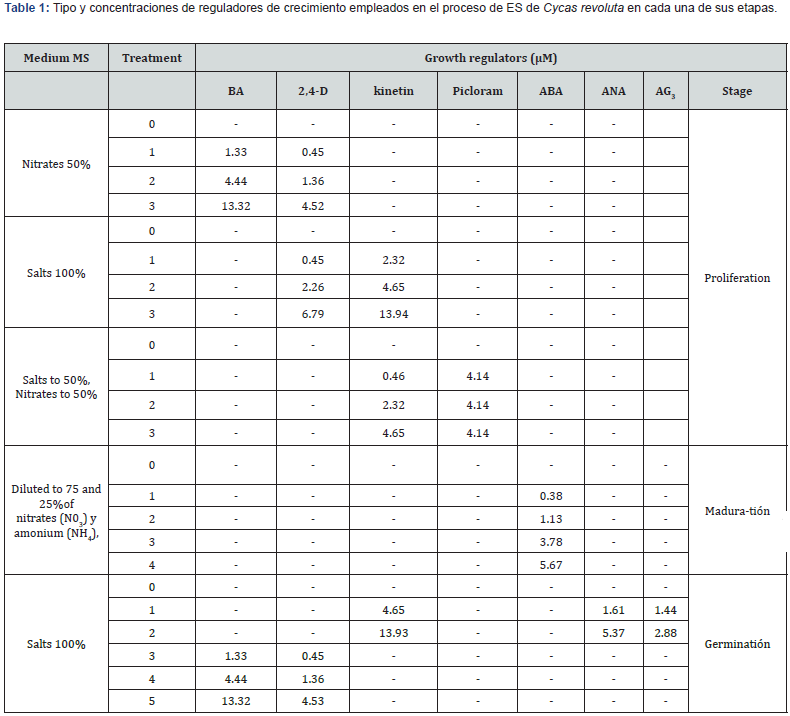
Results and Discussions
The results of the histological cuts made, allowed to identify the associated processes in the cellular differentiation promoted by the addition of means and growth regulators involved to promote somatic embryogenesis in vitro ES, from the calluses obtained from tissues taken from the mega gametofytes de C. revoluta. Histology performed, allowed to identify the conditions in vitro that stimulated somatic cells, to dedifferentiated them and subsequently promote their de novo cell differentiation to obtain somatic embryos, reach their mature and their subsequent germination in vitro.
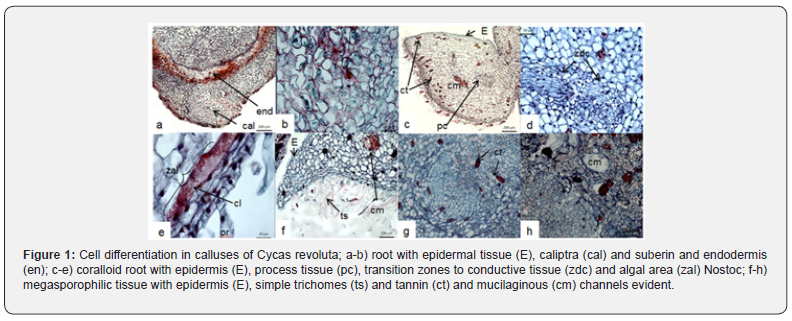
The histological cuts realized, showed that calluses cultivated in vitro may have associated responses towards the differentiation of structures of pro embryogenic type, included each stages after of obtain embryos. Some initials responses were generated at from the combination of 4.44 + 1.36 μM (BA + 2.4-D), and importantly by combining 2.32 + 4.14 μM (kin + picloram).In the first case the regulators employed promoted the development of coralloid roots types at 170 dds, this response can be observed from cells with extensive meristematic activity in the apicals. regions Figure 1. Coralloid roots, originate from tissue epidermal cells with thin cuticle, and from parenchymatous us-type peripheral cells that, by their radial arrangement, differentiated tissues associated with the region of caliptre (cal); and endodermis (en), basically consisting of a belt of compact cells and a discontinuous distribution of suberine; which allows you to define as a normal root structure (non-coraloid). ). The coralloid roots develop from the apical area of the normal root already described; and showed the development of process cells, epidermal cells and tannin channels. The differentiation of cells in this region and the histology carried out allowed to observe the mucilaginous channels s (cm) in some structures, uninucleated radical hairs (pr) and even concentric areas of difference to conductive tissues (zdc); other important histological responses, allowed to identify elongated cells with intercellular spaces near the periphery of the tissue, with homogen single-celled arrangements ors in linear chains (cl) which in presumably shows of fossil bacteria dt he cyanobacterial (Nostoc), within the tissues, so it involves the evolutionary association between plant cells and bacteria this type in cells, algal area (zal), in this type of roots. Similarly, tissues like megasporophyles were observed, which were singularized by the development of single-celled simple trichomes (ts), epidermis and parenchymatic cells with large deposits of amyloplasts and starch (Figure1). The promotion of coralloids roots in cultured in vitro tissue is a sample and see that both the participation of media and regulators allow both does differentiation and cell differentiation from cultivated megagametophytes, highlighting the presence of Nostoc-cyanobacteria, who are evolutionary able to keeped themselves intracellular form, as mentions, so in this research, it is confirmed that in cells cultivated under in vitro conditions cell differentiation that promotes the appearance of coral roots, has been a mechanism of survival in plants in relation to their habitat. The combinations of 2.32 + 4.14 μM (kin + picloram) stimulated at 40 days, an excellent development of helical tracheids of up to 290,245 mm of length; the development of funnel-like structures, which were characterized by the presence of parenchymatic cells in the basal part and apical globular-looking (mag) meristematic growth where there was a high correlation between cell and nuclear size (Figure 2) [13]. reported the presence of similar driving elements in Dioon merolae calluses; however, the differentiation of the tissues is not clear; also, the structures are relatively small compared to those identified in this work.
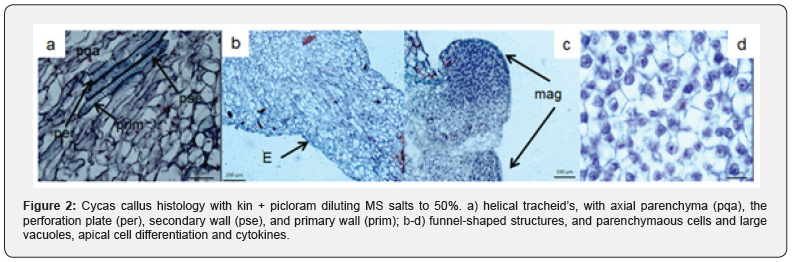
Embryogenic responses
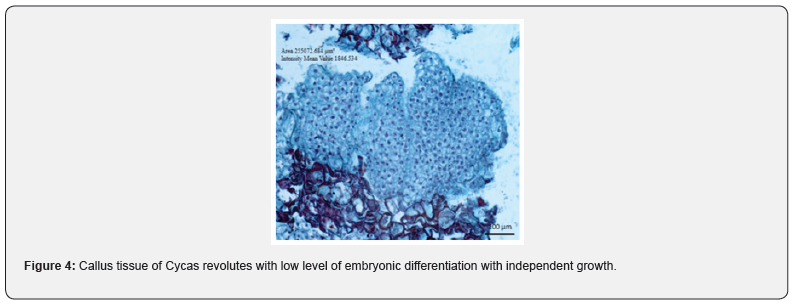
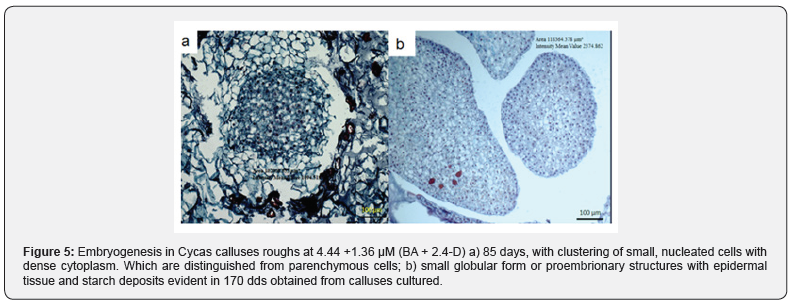
The cellular differentiation promoted towards formation of pro-embryonic structures or globular like, were observed through a microscopy from the calluses that samples were fixed according to the protocol described above and whose provenance resulted from the combination of both medium and regulators used. The results observed in the different tissue samples showed that both cell organization and maturation is strongly stimulated by the influence by the types and dosages of the growth regulators employed. The combination of 6.79 + 13.94 μM (2.4-D + kin respectively, differentiated pro-embryonic or globular type, from the 10 dds of culture, in calluses whose characteristic was marked by extreme friability, and according to their histology, they showed a lack of epidermal tissue in the EPEs Figure (3- a). In other results found the potentially embryogenic calluses the regulators employed did not promote cell differentiation, according to the combination, and concentration, nor were the incubation time enough for the development of potentially embryogenic tissues. On the other hand, the combination of 4.65 + 4.14 μM (kin+picloram) respectively and diluting the inorganic salts of MS to 50%, stimulated the formation of small globular structures of approximately (305. 200 m2) at 40 days, with the definition of epidermal tissue, and cells with dense cytoplasm defining interphase nuclear and other non-nucleated vacuolate cells that appeared to form a cell suspensory zone of approximately (76. 000 m2) (Figure 3-b). The 80-day-long cell samples of growth differentiated areas with larger downtowns (245. 600 m2) whose cells showed profasic nucleic stage and a more obvious development in epidermal tissue (Figure 3-c); without embarking on microscopic observations other cell growths in the calluses was limited embryogenic response since only superficially differentiated nodular tissue. At 354 days the cells constituted in the callus, differentiated other cell clusters (255. 000 μm2) without coding embryonic structures with more developed epidermal tissue, histological analysis in the samples, demonstrated independent growth (Figure 4). The combination of 4.44 + 1.36 μM (BA + 2.4-D) respectively, promoted the development of calluses of compact texture, showing an acceptable level of embryonic differentiation and whose progressive process was defined in later stages. Embryogenic calluses at 85 days differentiated concentric areas (-102,000 m2) consisting of small cells with broad starch content, dense cytoplasm, and thin wall, as well as prominent nuclei characteristics that define their mitotic activity, located on the periphery of established calluses, these results match or reported by [5]. In embryonic responses, non-nucleated parenchymátic cells were identified, with large, vacuoles linked to pro-embryonic structures, or embrionary globular form, probably fungi as suspensor cells whose function could be anchoring (Figure 5-a). Other samples with 170 days of cultivation, presented larger concentric areas (-780. 812 μm 22) and some relatively small EPEs (-118. 300 mm2) characterized by nucleated cells of dense citoplasm (ci), thin wall, starch, and epidermal cells (Figure 5-b). Although there are few histological studies reported to understand or explain in vitro regeneration processes in cycads via somatic embryogenesis, the results obtained, allowed to identify the important effect by combining BA and 2.4-D to promote cellular differentiation until somatic embryogenesis was obtained, coinciding with what was reported by [14], in addition histologically the embryogenic responses could be specified. It was also possible to determine that cells cultured in vitro C. revoluta, have a small size and with relatively large nucleus, their cytoplasm is dense and possess large concentrations of starch, as characteristics that define potentially embryogenic cells, these characteristics were also defined by [15] in pejibaye calluses.
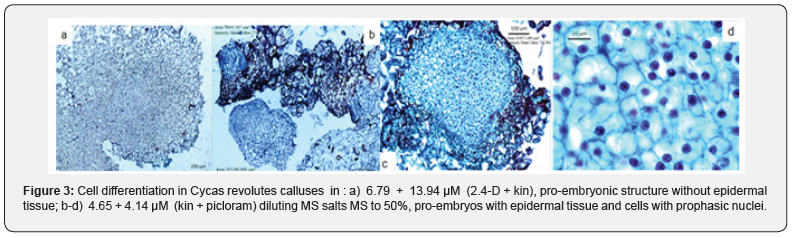
Maturation of Embryogenic Structures
According to the results observed in the histological samples, the differentiation and maturity of embryonic structures from subcultured calluses was successfully presented in the media mainly if the presence of growth regulators and with the influence of light (Figure 6). Calluses grown in the culture medium with the inorganic salts of MS (1962), with the presence of 4.44 + 1.36 μM BA + 2.4-D respectively with 170 days of culture, differentiated somatic embryos, with a rosacea coloration, derived independently of the formed callus tissue. The concentric zones in the calluses surface consisting of elongated cells, with poorly defined nuclei and large vacuoles (Figure 6a-c). Somatic embryos derived from the embryogenic cellular processes, had an area of 51,911 μm2 responses located in the first 20 days after the subculture, and unlike the development of pro-embryonic structures (EPEs), their cell differentiation was characterized by a proliferation of cells lacking starch. Initially formed embryos start from large, non-nucleated vacuolate cells; other responses associated with these originate within the periphery of calluses from relatively smaller nucleated cells, with meristematic activity or cell division (periclinal division), which could be identified as the onset of embryonic bipolarity (Figure 6-d).
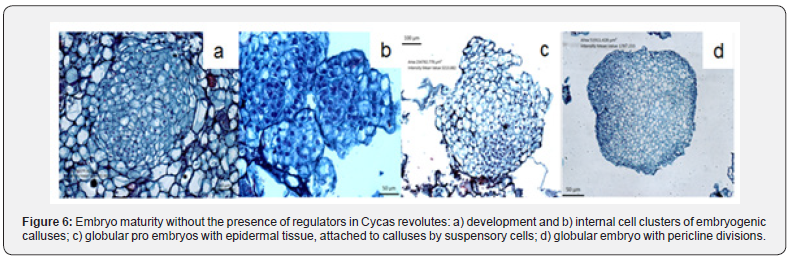
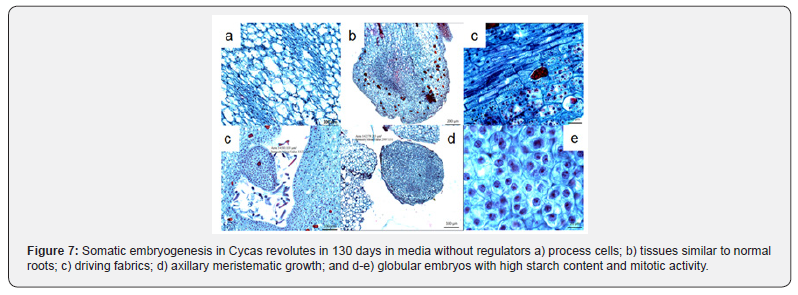
The answers that precede the formation of somatic embryos in each of the cellular stages have to be well defined since this process will allow them to reach their maturity [16,17] they emphasized that the globular or proembrionary phases are considered in order to reach the maturity of somatic embryos of Quercus suber and olive, respectively, and highlight the participation of vacuolate cells, while on the outside it was possible to specify a high mitotic or meristematic activity associated with their bipolar embryogenic development. The embryos with a length greater than 5 mm (>140 days), in the culture media used to induce maturity, acquired the ability to elongate, observing meristematic bipolar zones located in the apical zones. process that observe célls associated with its process activity, which will define the primary conduction tissues, (xylem and floem). The histological samples of 150 days of culture, showed the primary differentiation of tissues los those located in the area of the root, as epidemis, caliptra and endodermis even with the absence of radical hairs; in addition, the development of trichomes was identified in axillary meristematic areas of 54. 500 μm2, the latter generated small globular structures of 142. 000 μm2 with célls consisting of a dense cytoplasm, prominent nucleus and with a wide major mitotic activity (Figure 7).
Germination
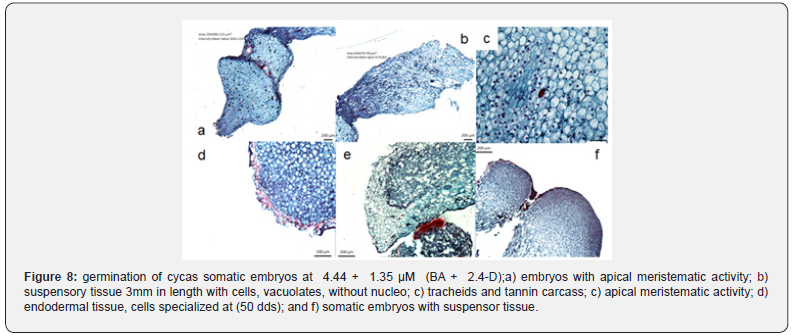
The best responses associated with the development and germination of somatic embryos were presented by combining the regulators 4.44 +1.35 μM BA and 2.4-D respectively, levels that promoted un elongation of the embryonic structure with 5 mm (40 dds), the levels of cell differentiation achieved gave way to the formation of tracheid’s whose function is associated with the phenomenon of absorption and conduction. The meristemátic activity located in the apical regions, also promoted the formation of primary strategies towards the formation of cotyledons. The cellular differentiation observed from meristematic activity, in apical regions, consequently derived an obvious suspensory tissue characteristic at this stage (Figure 8). The responses described above coincide with what was reported by [14], who noted, that a long side embryogenic cells there are other elongated, vacuolate and embryogenic cells that activate the embryogenic process very importantly later. Starch deficiency in cells involved in the development of apical and radical meristematic growth areas; as well as its extensive meristematic activity before the embryonic stages, also coincide with what [2] where reported in embryogenic culture of C. mexican. Moreover, the development of vascular tissue in some somatic embryos have also been reported by [16-20] in embryogenic culture of ulmus minor crops. The development of somatic embryos of C. revoluta, according to the histological results performed, coincide with the studies of embryonic development. According to the histological cuts made for each stage of somatic embryogenesis (ES), it was identified that the growth regulators used and their doses allowed to observe each of the cellular processes associated with the production, maturation and germination of somatic embryos in cycas, besides can be considered in addition, a new contribution to the results also achieved by Rinaldy and [10,11].
Conclusion
According to the histology work carried out in the callus obtained in vitro of C. revolutashowed different levels of organization and cell differentiation according to the inorganic salts used, constituted in the culture media, as well as the growth regulators used. The cell differentiation observed from embryogenic calluses in each of the cellular stages within embryogenic processes were initially located in small cells with dense cytoplasm and prominent nucleus, characteristics that define the formation of somatic embryos. The pro-embryonic or globular stages, until the formation of embryos, were initially located on the periphery of calluses with relatively small nucleated cells with divisions of periclinal type. The histology work carried out allowed to observe and specify the development of areas of apical meristematic growth, as well as the conduction tissues and the role of the cells associated with the stages of germination. The cell differentiation observed in histology, allowed to identify the embryonic stages until the maturation of the embryos, highlighting in a very significant way discovering at the histological level of the structures and the symbiotic relationship between bacterial cells and plant cells of cycas, considered fossil plants studied within the scientific contribution of this research.
Acknowledgment
The authors thanks to the National Council of Science and Technology for their support for the project (736230/595557) and the program Master of Science in Agricultural Biotechnology, as well as the Ing. Mario Iván Venegas Ayala, technician of the Laboratory of Histology and General Cytology of Agricultural Preparatory belonging to the UACh for the technical orientation and significant contribution to this research.
References
- Gilbert S(1984) Cycadsstatuas, trade, exploration and protection 1977-1982. Washington, USA: TRAFFIC (USA).
- Chávez VM, Litz RE,MarquezJ (1995) Histology of somatic embryogenesis of the cycad Ceratozamiamexicana var. Robusta (Miq.) Dyer. Plant Science108(2): 191-200.
- Crane PR, Upchurch GRM (1987) Drewriapotomacensis gen. et sp. nov., an early Cretaceous member of the Gnetales from the Potomac Group of Virginia. American Journal of Botany 74(11): 1722-1736.
- Jones DL (1993) Cycads of the World. London, Inglaterra: Reed Book.
- Teixeira da Silva JA, Woondenberg WR,Songjun Z (2014)Cycads in vitro. Plant Tissue Culture and Biotechnology 24(2): 287‐301.
- Kono M,Tobe H (2007)Is Cycas revoluta (Cycadaceae) wind-or insect-pollinated? American Journal of Botany 94(5): 847-855.
- Litz RE, Chávez VM, Moon PA (1995) Somatic embryogenesis in the Cycadales. Somatic Embryogenesis in Woody Plants 1-15.
- Motohashi T, Toda M,Kondo K (2008) Adventitious embryo formation derived from zygotic embryos in Cycas revoluta. Plant Biotechnology 25(6): 589-591.
- Rinaldi LMR,Leva AR (1995) in vitroorganogénesis from diploid tissues of Cycas revoluta Plant Cell, Tissue and Organ Culture 43(1): 37-41.
- LingPKA, Thing YS, Gansau JA,Hussein S (2008) Induction and multiplication of callus from endosperm of Cycas revoluta. African Journal Biotechnology 7(23): 4279-4284.
- De Luca P,Sabato S (1980) Regeneration of coralloid roots on cycad megagametophytes. Plant Science Letters 18(1): 27-31.
- Webb DT, Nevarez M,De Jesús S (1984) Further in vitro studies of light‐induced root nodulation in the Cycadales. Enviromental and Experimental Botany24(1): 37‐44.
- CabreraHSL, Chávez AVM, Sandoval ZE,Litz RE, y Cruz SF (2008) Morfogénesisin vitro de Dioonmerolae De Luca, Sabato & Vázquez-Torres (Zamiaceae, Cycadales) a partir de megagametofitos y embrionescigó Interciencia 33(12): 929-933.
- Zúñiga PT (2007) Estudiomorfogénico de ZamiiaskinneriWarszewicz, empleandomegagametófitos y embrioneszigó TecnologìaenMarcha 20(2): 58-70.
- Valverde R, Arias Oy, Thorpe T (1992) Estudiohistológicoencallos de pejibaye (Guilielmagasipaes). AgronomíaCostarriciense 16(2): 225-229.
- Pinto G, Valentim H, Costa A, Castro S,Santos C (2002) Somatic embryogenesis in leaf callus from a mature Quercus suber L. tree. in vitro Cellular and Developmental Biology Plant 38(6): 359-572.
- Capelo AM, Silva S, Brito G,Santos C (2010) Somatic embryogenesis induction in leaves an petioles of a mature wild olive. Plant Cell Tissue Organ Culture 103: 237-242.
- Ashmore SE (1997) Statue report on the development and application of in vitro techniques for the conservation and use of plant genetics resources. International Plant Genetics Resources Institute: Rome.
- Murashige T, SkoogF (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiologia Plantarum15: 473-497.
- Robinson ML (2010) Cycads in Southern Nevada.






























