QMicrobial and Biochemical Changes Occurring During Production of Njemani, a Traditional Zimbabwean Beverage
Sadza Beverly1 and Parawira Wilson1,2*
1 Department of Science and Mathematics Education, Faculty of Science Education, Bindura University of Science Education, Zimbabwe
2Department of Biological Sciences, Bindura University of Science Education, Zimbabwe
Submission: February 29, 2020; Published: May 11, 2020
*Corresponding author:Parawira Wilson, Department of Science and Mathematics Education, Department of Biological Sciences, Faculty of Science Education, Bindura University of Science Education, Zimbabwe
How to cite this article: Sadza B, Parawira W. Microbial and Biochemical Changes Occurring During Production of Njemani, a Traditional Zimbabwean Beverage. Adv Biotech & Micro. 2020; 15(5): 555921.DOI:10.19080/AIBM.2020.15.555921
Abstract
Njemani is a type of traditional palm wine produced in the Shangaan tribe of Zimbabwe and plays an important role in many traditional functions and ceremonies. The wine is consumed in a variety of flavors from sweet to sweet-sour and vinegary. The main aim of the research work was to identify, analyze the biochemical and microbial changes that occur during production and documenting the whole procedure of Njemani production in an attempt to upgrade the traditional processing to a commercial scale. During fermentation microbial population increased with fermentation time. The most dominant Lactic acid bacteria were Bacillus, Lactobacillus spp. and Leuconostic species that were responsible for production of inorganic acids and lactic acid during lactic fermentation. Glucobacter and Acetobacter were the dominant Acetic acid bacteria identified and were responsible for acetic acid fermentation using alcohol as a substrate. The yeast species identified were Saccharomyces cerevisiae, Saccharomyces pombe, Saccharomyces uvarum and Candida spp, however the most dominant was Saccharomyces cerevisiae and were responsible for the conversion of sweet palm sap to an alcoholic beverage. The biochemical parameters varied as fermentation progressed Ethanol 0.8%-16.3%, acetic acid 2.8%-7.1%, lactic acid 3.8%-6.9% resulting in Total Titratable Acidity ranging from 0.05% to 0.8% and pH from 6.36 to 3.09. The initial sugar content was 15.7% and dropped to 0.7% after fermentation. If biotechnological techniques are applied to the production of Njemani a number of benefits will be harnessed. The palm sap fermentation involves alcoholic-lactic-acetic acid fermentation, due to the presence mainly of yeasts and lactic acid bacteria. Further research will be needed to isolate and identify all the microbial isolates in Njemani.
Keywords: Njemani; Biochemical changes; Microbial changes; Palm wine; Lactic acid; Alcoholic fermentation; Yeasts
Introduction
Palm wine is an alcoholic drink obtained by the natural fermentation of the sap of various types of palm trees; this beverage is produced and consumed in several tropical regions of the world, [1]. It plays an important role in traditional practices as an alcoholic beverage, so it is important to determine the physicochemical characteristics and microbiological aspects of its fermentation. Though research in this field is still at its infancy stage in Zimbabwe, a number of researches have been documented so far both locally and abroad. The following are examples of traditional foods which have been studied, documented and some commercialized sour milk, Amarula wine, maheu, masvusvu, mangisi, ngome doro/hwahwa in Zimbabwe; gari, dawa- dawa, ogi, urwagwa beer among others in East and West Africa. In addition, fermented plant sap juices such as shoto sake (Japan), muratina (Kenya), Basi (Philippines), pulque (Mexico), ulanzi (Nigeria) have also been studied and documented. Palm wine in Zimbabwe is made from murara tree (Hyphaene petersiana Mart). It is also known as Llala in Ndebele, muzira /murara in Shona. Other English variations are palm, Real palm and vegetable ivory palm. The plant is a native tree found along the Limpopo flood plains. The plant is single stemmed, very tall and stands erect. The leaves are usually crowded on top of the stem. They are fan shaped in short spikes. The female flowers form large sprays which develop at the end of the branch next to the male flowers. The species is very similar and closely related to H. coriacea. Palm wine has been produced and consumed in tropical regions of the world for centuries. Palm wine has a pivotal role in the diet, income and traditional practices of these regions [1, 2, 3].
Palm wine in Zimbabwe is consumed for its nutritional and medicinal benefits. Earlier studies by [4] reported that palm wine was consumed for its nutritional effect because of its probiotic contents. also reported in their study that palm wine is a good source of single cell protein and various vitamins since the micro biota responsible for fermentation were taken in together with the wine. That is the reason why pregnant women and breastfeeding women were consuming the palm wine, it’s nutritious for the foetus and promotes milk production respectively. Fresh palm wine is also consumed as an energy drink. At this stage palm wine is very sweet because of high sugar content, [5]. This research contribute to this field of research of fermented foods and beverages and biotechnology in the sense that it focused on the unreported microbial and biochemical changes during the production of Njemani, a traditional beverage in Zimbabwe. Further research needs to be done on this beverage so as to increase the potential benefits economically, medically and beyond.
Materials and Methods
The study was carried out in the Eastern part of Masvingo Province specifically in the Shangaan lands of Mahere, Gonarezhou strip, Malipati-Gezani area, Mwenezi and Chilonga. The data was collected using interviews, participative and experimental methods. Interviews were done to obtain information on tapping of palm sap and on the procedures in Njemani production from the tappers and Njemani brewers. Interview questions or interview schedules used were similar to those used by researchers in similar studies and were modified to suit this study. The interview method was ideal since some of the tappers and brewers could not read and write. This allowed room for probing and clarification. The interview augmented the observation method giving valid and reliable information. The researcher had the opportunity to participate and see exactly how the beverage was made. The researcher observed and participated in the process from tapping, collection of sap and fermentation until the finished product was obtained. Furthermore, the researcher had the opportunity to note physical characteristics such as the colour of collected sap, the taste and odor etc. The researcher also took note of the time frames in the Njemani production procedures. The data was collected when the process was actual happening. This method helped the researcher to fill in the information gaps left during interviews. The observation method together with the interview method made the data collected very valid and reliable.
Tapping
The palm sap was extracted at an incision made between the kernels. A sharp machete or a knife known as gwese was used to make an incision between the kernels. A lot of effort was needed to make the incision since the kernels are closely packed forming a thick protective layer on the palm stem. The incision was covered by a wet warm cloth (40 °C) for 10 minutes, this was done to stimulate flow of the sap. To collect the sap a light container was used and was fastened just below the incision. Gourds were used as containers for tapping/collection of the palm sap. The sap oozed slowly into the container. About 2ml of sap was tapped per minute giving a total of 1,440 ml per day. Cut pieces of palm tree leaves were put in the collecting containers. This was done to slow the rate of fermentation during tapping and collection.
Plant Sap collection and transport from the plantation
The sap was left to collect during the day and during the night. Collection was done early mornings at 5:00 am and evenings at 6:00 pm. Collection is more efficient during the day and during the summer season since the temperatures will be high enough to promote tapping and collection process. In the rainy season sap collected ranges from 2-4 liters per adult tree. In the dry winter season, on average 1-2 liters were collected per tree. The collected sap from each tree was poured in 2-liter plastic gourds. The samples of the palm wine were taken to the laboratory and were carried in a medical cooler bag with temperatures of 15 °C to reduce the rate of fermentation before reaching the laboratory.
Fermentation
One thousand and five hundred milliliters (1500 ml) of collected palm sap was left to ferment in the gourds with a capacity volume of 2 liters in triplicates. The gourds used were borrowed from the palm wine brewers. The palm sap was left to ferment for 48 hours in the laboratory at room temperature (32 °C - 35 °C).
Sample analyses
Samples (10 ml) for analyses were collected during tapping and collection of the sap and subsequently after every 6 hours during fermentation for 48 hours. The following physicochemical parameters were determined: pH, total titratable acid, sugar content, volume of alcohol and the microbiological changes.
Biochemical assays
All assays where done in triplicate. Titratable acidity was measured immediately. The supernatant (5ml) collected during fermentation was transferred into a conical flask and diluted twice with distilled water. This solution was titrated against 0.1M NaOH using phenolphthalein as the indicator. The amount of acid produced was calculated as percent lactic acid according to the formula [6].

A Hanna Acidity Titrator (MT682) was also used to verify the results, this is a machine that takes 30 seconds to give accurate TA reading. The Hanna Acidity Titrator is used by many commercial wine makers and it is suitable for large batches of samples. The pH was measured using a pH meter (Hanna, USA).
Measurement of Sugar Content during Njemani Production
A refractometer and a hydrometer were used to measure sugar content during the fermentation process. A drop of the sample was placed on the lens and left for 30 seconds for the sample to adjust to the temperature of the refractometer prism. A line that splits the blue area and white area was viewed and the Brix reading was obtained. The refractometer have Brix reading on one side and specific gravity at the other side. The formula that was used to calculate the total sugar was [6]:

A hydrometer was placed in 20 ml sample of Njemani and was given a spin stir until it stabilized at 20 °C. A reading was taken on the top of liquid where it meets the scale on the hydrometer. Determination of alcohol content during the fermentation was measured by using an Alcometer.
Microbiological Analysis
Palm wine subsamples collected for microbiological analysis were serially diluted using peptone water and spread in triplicate on different media to assess microbiological changes during the production of Njemani. Total aerobic mesophilic bacterial counts were made on plate count agar (DIFCO) after incubation at 37 °C for 24 hours. Lactic acid bacterial counts were made on plate on De Man, Rogosa and Sharpe (MRS) (Oxoid) medium and incubated at 37 °C for 48 hours. Yeast counts were made on plates of Glucose Yeast Agar (GYA containing 0.05 mg/ml of chloramphenicol to inhibit bacterial growth) incubated at 28 °C for 48 hours. Determination of Acetic Acid Bacteria were on plates Glucose Yeast extract Carbonate (GYC) incubated for 24 hours at 28 °C.
Results and Discussion
At collection the palm sap was very sugary and contained very low volumes of alcohol. The initial concentration of sugar was 15.7%, this value dropped to 4.6% within 24 hours of fermentation. The total sugar percentage further dropped to 0.7% after 48 hours. At collection of palms sap the alcohol content was 0.8, the sap was sweet and non-intoxicating, however the percentage volume of alcohol rose to 4.4 in 24 hours. The percentage of lactic acid and acetic acid resulted in an initial TTA of 0.05 and increased to 0.08 in 44 hours. Since lactic acid bacteria are essential for fermentation the percentage of lactic acid in the first 12 hours of fermentation was 0.3% whereas the percentage of acetic acid was 0.4% at the end of fermentation. This is because acetic acid is responsible for the aroma of mature palm wine.
Njemani wine production
According to the results fermentation in palm wine production can be described as a 3-stage process, with the first stage occurring at the incision cut in the palm tree itself. Fermentation at this point occurs as continuous culture fermentation. This is because palm sap continually oozes out and is exposed to microbiota from the air and the cut/incision area. in their study also reported that fermentation on cut area was described as a continuous culture fermentation. The second stage occurs during tapping in the tapping container. Here the alcohol build up is faster than on the tree trunk, though it is being constantly diluted by new sap trickling in. High alcohol volumes in the containers could be attributed to the presence of high population of microbiota found in cleaves of the collection containers. The collection containers are not washed in between collections there by making the containers a suitable habitat for the microbiota responsible for fermentation. The highest build-up of alcohol occurred during fermentation in the fermentation/collection containers, the third stage. During the first 24 hours the collection containers were closed which favour alcoholic fermentation by yeasts. Fermentation of palm wine is a triple sequential process. It begins with lactic acid fermentation, followed by alcoholic fermentation and ending with acetic acid fermentation. The products of each stage serve as metabolites for the next stage [2,7] reported that palm wine is produced by natural lactic-alcoholic-acetic fermentation of the sugary sap of palm wine. Production of palm wine is popular in summer than in the winter season. The summers are characterized by wet conditions and high temperatures, which favour efficient palm sap tapping, collection and fermentation. While the winter season is characterized by dry spells and low temperatures. Tapping is very minimal in this season since the trees will be trying to serve the little water available and fermentation which normally take 48 hours will take 72 hours or more. If biotechnological techniques are applied, it would be good to tap in summer and preserve some palm sap and wine for the winter season.
Biochemical Changes
Changes in alcohol content and total sugars during njemani wine production
The initial alcohol content at collection was 0.8%, with time the level of alcohol increased significantly to 16.3% within the 48 hours of fermentation. The initial concentration of sugar was 15, 7%, this value dropped to 4.6% in the 24 hours of fermentation. The total sugars percentage further dropped to 0, 7% after 48 hours. The graph shows a drastic decrease in total sugars while a significant increase in alcohol is noted as fermentation progressed, Figure 1.
pH changes during Njemani wine production
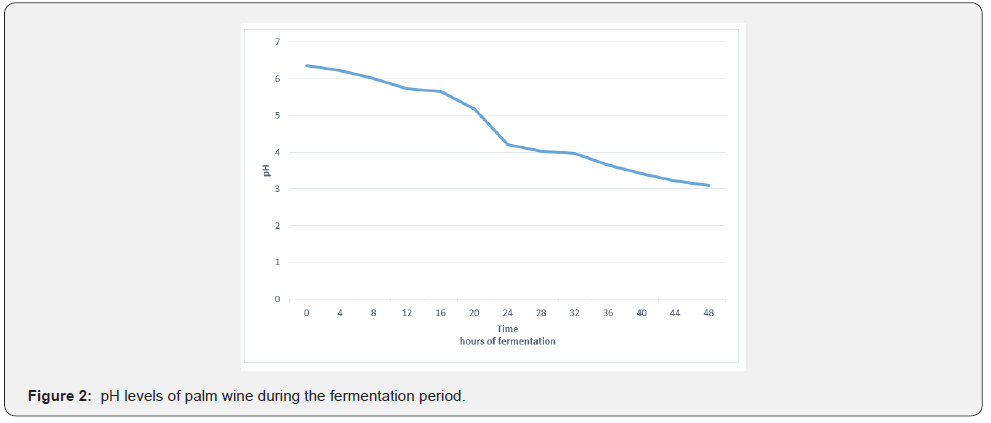
The pH of fresh palm sap at collection was 6.36. However, the pH dropped drastically to 4.2 within 24 hours, during fermentation. The drop in pH continued as fermentation progressed until it was 3.09 in the 48th hour. The trend shown by the results was a generous decrease in the pH as shown by the trend line in Figure 2. The decrease in pH was attributed to lactic fermentation and acetic fermentation which produced lactic acid and acetic acid respectively. This was evidenced by an increase in titratable acidity. Similarly, in also reported that pH value in their research decreased from pH 6.8 to pH 3.8 as fermentation progressed, thus acidity increased. The lactic acid bacteria responsible for lactic acid production in palm wine production are the Leuconostic strains. They ferment foods and increase their nutritive characteristics by producing a distinctive flavor, in the process carbon dioxide is produced. The accumulation of the alcohol rapidly lowers the pH, coupled with inhibition of the development of undesirable microbes. Similarly, in their assessment of microbial growth and survival in fresh raphia palm wine also mentioned that production of lactic acid lowered the pH [7]. also reported that during fermentation by yeasts ethanol, carbon dioxide and other secondary products are produced as a result pH is lowered and good flavor is enhanced.
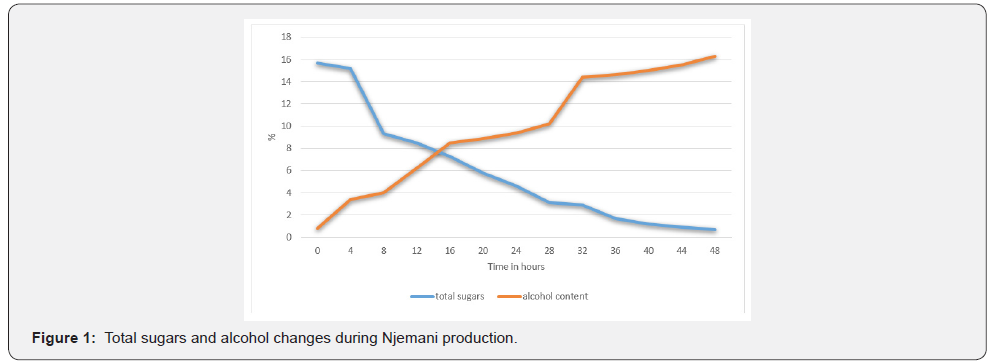
Total titratable acidity changes during Njemani production
The percentage of lactic acid and acetic acid resulted in an initial TTA of 0.05%. The percentage of TTA did not change from the time of collection to the 8th hour of fermentation. There was a slight rise from 0.05 to 0.06%, from the 8th hour to the 12th hour of fermentation. The percentage of TTA was constant from the 12th hour to the 16th hour of fermentation. A slight rise was also observed from the 16th hour to the 24th hour, the percentage of TTA rose from 0.06 to 0.08%. The percentage of TTA from the 24th hour to the 44th hour of fermentation was constant at 0.08%. The TTA rose to 0.09% in the last 4 hours of fermentation, Figure 3.
Microbial changes
After tapping the collected sap was milky and very sugary. Palm sap is a rich substrate for various microbial growth because of its high sugar content. The following microorganisms were isolated Lactic acid bacteria, acetic acid bacteria, aerobic mesophiles and yeasts.
Lactic acid bacteria changes during production of palm wine
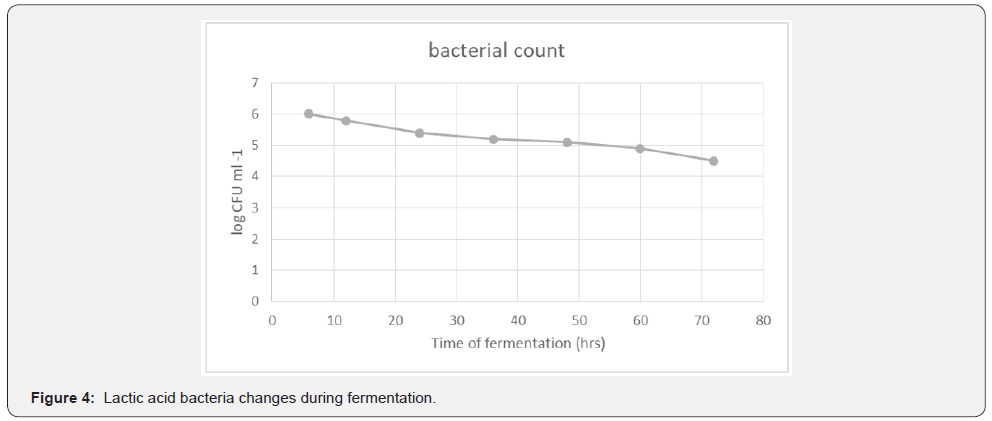
The lactic acid bacteria showed an initial bacterial count of 6.0 x 10³ CFUml⁻¹ at 4 hours after collection of palm sap. The population declined progressively as fermentation occurred to a count of 4.5 x 10³ CFUml⁻¹ in the 72nd hour, Figure 4. This decline in population of LAB showed that LAB were active at the onset of fermentation, since fermentation started soon after tapping. Lactic acid bacteria are responsible for the onset of lactic fermentation. The lactic acid bacteria are found on the surface of the palm leaves, though the population is very low on the trees. These bacteria can also be found on tapping and collecting equipment, gourds and clay containers have cleaves that hibernate the bacteria [8]. Lactic acid bacteria are responsible for producing lactic acid and rapid acidification of the product. That is why the population of lactic acid is high at the onset of fermentation and decreases as fermentation progresses. Presence of lactic acid bacteria influences the composition of the product [2]. Previous research by [9] reported that Bacillus, Lactobacillus spp. and Leuconostic species dominated in palm wine [1]. in their research also reported that Lactobacillus plantarum and Leuconostic mesenteroides have were reported as the predominant LAB. In this research no colour change of Njemani was observed from tapping to the finished product. Contrary to this current research reported that LAB causes change in colour of palm wine, during tapping colour changes from transparent to whitish. This is also supported by [10] who reported that heavy suspension of yeast and bacteria are responsible for the milky-white appearance of palm wine. The LAB have been identified in previous work by [8] as responsible for the sour taste in palm wine. Similarly, reported that LAB are responsible for the taste and the characteristic flavour. Bassir 1968 cited in also reported that L. plantarum and L mesenteroides were responsible for the souring of palm wine tapped from live upright palm trees. Presence of Lactobacilli in palm wine samples have also been reported (Figures 5&6).

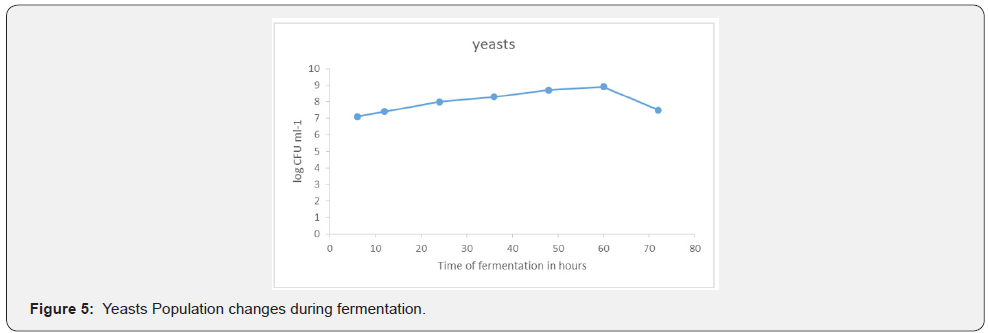
Yeast population changes during fermentation
There was a gradual increase of the yeast population as fermentation progressed between 4 hours and 60 hours. The initial population count at 4 hours was 7.1 x 10⁻³ CFUml⁻¹ and increased to 8.9 x 10⁻³ CFUml⁻¹. After 60 hours the population of yeast dropped significantly to 7.5 x 10⁻³ CFUmI⁻¹ in the 72nd hour of fermentation. The other observations recorded of yeasts was the unique earthly smell, the cream color and the oval, round shape. The morphological feature dominant in the yeast corresponded to that of Saccharomyces cerevisiae. This result showed that the nutritional status of fresh wine and the products of lactic acid fermentation were favorable conditions for the growth of yeasts [2] This was supported by the findings of who revealed that fresh sap contains several sugars (sucrose, fructose, raffinose) and various growth factors such as vitamin C and B12. At 48 hours of fermentation a decline in the yeast population was observed, that is from 8.9 x 10³ml⁻¹ at 48 hours to 6.2 x 10³ml⁻¹. The decline was caused by a number of factors namely depletion of metabolites, increase in volume of alcohol and increase in the population of acetic acid bacteria. At this point there was enough alcohol to serve as substrate for acetic acid fermentation [2,7]. Similarly, reported that the decline in the population of yeast after 60 hours of fermentation was due to competition for nutrients, oxygen, and space among microorganisms as a result of fermentation. Yeast cells were the most abundant organisms in the palm wine sample. The species of yeast which were present in the palm wine were Saccharomyces cerevisiae, Saccharomyces pombe, Saccharomyces uvarum and Candida spp. However, the most abundant species was S cerevisiae. Contrary to this current study Faparusi and in [2] identified a total of 188 yeast isolates from palm wine production. However, in this study only four were identified. These yeasts are responsible for the conversion of the sweet substance (palm sap) into an alcoholic beverage. Saccharomyces cerevisiae was partially confirmed in this present research as the dominant yeast species responsible for the fermentation of palm wine tapped from erect and live palm trees. This is in agreement with other previous studies carried out by the following. The studies reported presence of S. cerevisiae through restriction fragment length polymorphism to identify the yeast isolates from samples of palm wine from several towns in Southern Ghana.
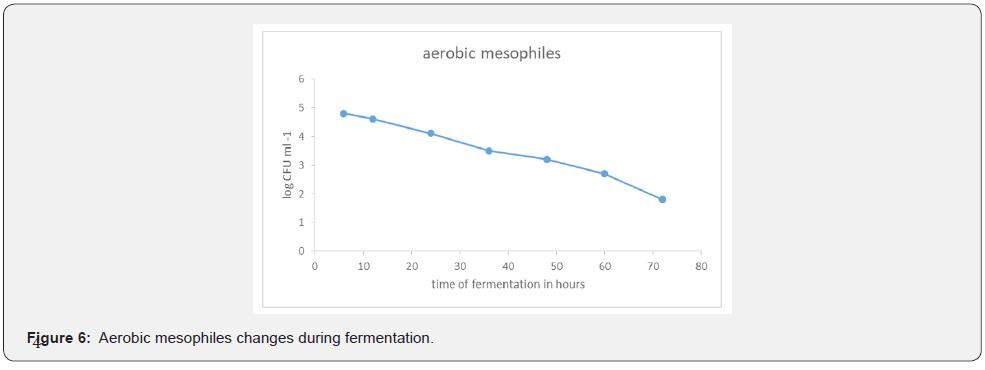
Aerobic mesophiles population changes during fermentation
The initial population of aerobic mesophiles 4.8 x 10³ CFU ml⁻¹ during the first 4 hours of fermentation. The population then gradually dropped to 1.8 x 10³ CFU ml⁻¹ after 72 hours of fermentation.
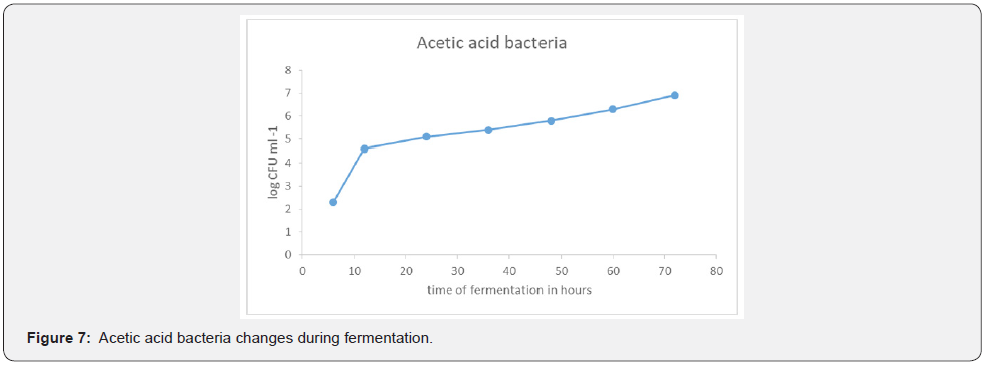
Acetic acid bacteria changes during fermentation
After 6 hours of fermentation the concentration of Acetic acid bacteria was 3.8 x 10³ml⁻¹. A gradual increase in population was noticed as fermentation progressed. At the 72nd hour the population recorded was 6.9 x 10³mI⁻¹, Figure 7. Acetic acid bacteria were responsible for acetic acid fermentation. The predominant Acetic acid bacteria which were partially identified in this research were Glucobacter and Acetobacter genera. Similarly, Santiago-Urbina et al., (2014) reported the predominant AAB as Glucobacter and Acetobacter in their research. The gradual increase in alcohol content triggered the increase in acetic acid bacteria to a detectable concentration in the 48th hour. Similarly, [2,3,7,11] also reported that palm sap constituents also favour growth of AAB. Ref. Furthermore, [12,13] also reported that ethanol is the main substrate used by AAB for the acetic fermentation in palm wine production. This was so because the AAB were now using the alcohol produced as a carbon source for their growth. The AAB present in the wine were Gram negative, Catalase positive and Oxidase negative. This was identified through pigments they produced. The pigments were dissolved in water and were able to dissolve the calcium carbonate at the bottom of the GYC standard medium. These bacteria produced acetic acid which gave the wine a distinctive aroma. The AAB were also believed to be responsible for the vinegary taste of the Njemani wine. Similarly, [9] reported that AAB was also responsible for the acidic, bitter and/or vinegary taste of palm wine reducing the palm wine shelf life.
Conclusion
This current study recorded the existence of palm wine (Njemani) in the Shangaan state of Zimbabwe. Palm wine (Njemani) is a beverage obtained from the fermentation of palm sap of Murara/LIala tree (Hyphaene petersiana Mart) by indigenous microbes found in tapping and collecting equipment and from the atmosphere. There is natural fermentation process involving lactic acid, alcohol and acetic acid fermentation and is termed the lactic-alcohol-acetic fermentation. The following microbes were partially identified in this research: acetic acid bacteria (predominantly Glucobacter and Acetobacter); yeasts (S. cerevisiae, S. pombe, S. uvarum and Candida spp.) and the most abundant being S. cerevisiae and lactic acid bacteria (Bacillus, Lactobacillus spp., Leuconostic species). Yeasts, lactic and acetic acid bacteria are all important in the fermentation of palm wine and influence the composition of the product. The results obtained could serve as the starting point to understand the microbial and biochemical processes in Njemani production, with the aim of improving the efficiency of the process.
Acknowledgement
The authors would like to thank the members of staff of Tongaat Hullets Laboratory in Triangle for their help and for allowing us to use their laboratory. We also acknowledge the assistance of Mr P. J. Chauke and Mrs Chipirikisa from Chikanga for their knowledge on the art of Njemani beer production.
Recommendations
Further research will be needed to isolate and identify all the microbial isolates in Njemani. Further study is recommended to evaluate bacteria diversity in palm wine, effects of traditional plant preservatives on the microbial load and shelf life of Njemani. Once fermentation process is known and the microbes involved are known it is possible to begin to refine and improve the process focusing on manipulating environmental conditions: temp, pH, moisture content, aeration, substrate pretreatment in the laboratory. Laboratory simulations enables a better scientific understanding of the process critical steps, conditions, Study effect of varying substrate concentrations and varieties, and production time and detection of potential problems. The pilot stage is the next step which is the first clear departure from smallscale production and should be based on the result of laboratory experiments.
References
- Mueller I, Zimmerman PA, Reeder JC (2007) Plasmodium malariae and Plasmodium ovale the ‘bashful’ malaria parasites, 23(6): 278 - 283.
- Beare NA, Taylor TE, Hardin SP, Lawallen S, Molyneux ME (2006) Malarial Retinopathy: A newly established diagnostic sign in severe malaria. American Journal of Tropical Medicine and Hygiene 75(5): 790-797.
- Behera BC, Verma N, Sonone A, Makhija U (2006) Determination of antioxidant potential of Lichen Usnea ghattensis in vitro LWT 39: 80- 85.
- Pabon A, Carmona J, Burgos LC, Blair S (2003) Oxidative stress in patients with non - complicated malaria. Clinical Biochemistry 36: 71 -78.
- Isah MB, Ibrahim MA (2014) The role of antioxidants treatment on the pathogenesis. Parasitology Research 113(3): 801-809.
- Peter W, Portus H, Robinson L (1995) The four-day suppressive in vivo antimalarial test. Ann Trop Med Parasitol 69: 155-171.
- Rocha eSilva LF, Pinto ACS, Pohlit AM, Quignard ELJ, Vieira PPR, et al. (2011) In vivo and in vitro antimalarial activity of 4-nerolidylcatechol. Phytother Res 25:1181-1188.
- Reitman S, Frankel S (1957) A colorimetric method for the determination of serum oxaloacetatic and glutamic pyruvic transaminases. American Journal of Clinical Pathology 28(1): 56–63.
- Englehardt A (1970) Measurement of alkaline phosphatase. Aerztl Labor 16: 42.
- Pearlman FC, Lee RT (1974) Detection and measurement of total bilirubin in serum, with use of surfactants as solubilizing agents. Clinical chemistry 20: 447-453.
- Zoppi FPA, Felini D, Marcovina S, Ramella C (1976) Metodo per la determinazione della bilirubin totale e conlugata. Uso di un tensioaltivo cationico come agentte solubilizzantte. Giorn ital chim clinica 1: 343-359.
- Jendrassik L, Grof P (1938) Estimation of total serum bilirubin level by spectrophotometrically in serum and plasma. Biochemische Zeitschrift 297: 81-89.
- Nanyunja M, Orem JN, Kato F, Kaggwa M, Katureebe C, Saweka J (2011) Malaria treatment policy change and implementations: The case of Uganda. Malaria Research and Treatment.
- Cui L, Su XZ (2009) Discovery, mechanisms of action and combination therapy of artemisinin. National Institute of Health: Expert Rev Anti Infect Ther 7(8): 999-1013.
- Wintergerst ES, Maggini S, Hornig DH (2006) Immune enhancing role of vitamin C and zinc and effect on clinical conditions. Ann Nutr Met 50: 85-94.
- Marva E, Golenser J, Cohen A, Kitrossky N, Harel R (1992) The effects of ascorbate induced free radicals on Plasmodium falciparum. Tropical Medical Parasitology 43: 17-23
- Mohammad A (2012) Effect of serum antioxidant ascorbic acid concentration by malarial infection Man. The Experiment 3(4): 214-215.
- D’Souza V, D’Souza B (2006) Comparative study on lipid peroxidation and antioxidant vitamins E and C in falciparum and vivax malaria. Indian Journal Clinical Biochemistry 21(2): 103-106.
- Gaw A, Cowman RA, O'Reilly DS, Shepherd J (1995) Clinical Biochemistry: An Illustration color Text. Clinical Biochemistry, New York.






























