Prevalence of Bovine Trypanosomosis and Apparent Density of Tsetse Flies in Dale Sadi District, West Oromia, Ethiopia
Darge Lulu Hordofa*, Firaol Bekele Bayecha and Desta Goshu Nurfeta
Dale Sadi Woreda Livestock Development and Fishery Office, Ethiopia
Submission: May 22, 2019; Published: July 17, 2019
*Corresponding author: Darge Lulu Hordofa, Dale Sadi Woreda Livestock Development and Fishery Office, Kelem Wollega, Oromia, Ethiopia
How to cite this article:Darge Lulu Hordofa, Firaol Bekele Bayecha, Desta Goshu Nurfeta. Prevalence of Bovine Trypanosomosis and Apparent Density of Tsetse Flies in Dale Sadi District, West Oromia, Ethiopia. Adv Biotechnol Microbiol. 2019; 14(3): 555887. DOI: 10.19080/AIBM.2019.14.555887
Abstract
Across sectional study was conducted from October to November 2016 to determine the prevalence of bovine trypanosomosis and the apparent density of tsetse flies in six peasant association of dale sadi district. For the prevalence study, dark phase contrast Buffy coat examination and giemsa stained thin blood smears were used. Whereas, for entomological survey, to determine apparent density of tsetse flies, traps were deployed. The entomological survey indicated that total mean catches per trap per day of tsetse flies in the study area was 5.34. G. moristance submoristance, G.pallidipes, G.furcipes, G.tachinoides was the four species of glossina recorded in the study area and the highest FTD was recorded chemo village. For the prevalence study, out of total of 532 cattle examined, 19(3.57%) were found infected with trypanosomes. out of infected animals highest prevalence was recorded in cattle with poor body condition (52.63%) followed by medium (31.58%) and good (15.79%). The overall mean PCV value of examined cattle is 29.29 and the overall mean PCV value of non infected cattle was higher (29.70%) than that of infected animals (24.25%). Among the species of trypanosomes isolated, Trypanosoma vivax was identified as the major causes of trypanosomosis in the study area accounting for 63.16% of total infections observed followed by trpanosoma congolense (36.84%) respectively. Taken as a whole, the present work evidenced that tsetse and trypanosomosis has continued to pose a considerable threat to cattle of the study area warranting an integrated control to safeguard cattle production and productivity.
Keywords: Bovine trypanosomosis; Prevalence; Tsetse fly; Dale said
Introduction
Trypanosomosis is a complex disease caused by unicellular parasites found in the blood and other tissues of vertebrates including livestock, wild life and people. The most important trypanosome species affecting livestock in Ethiopia are Trypanosoma congolense, Trypanosoma vivax, and Trypanosoma brucei in cattle, sheep and goats, Trypanosoma evansi in camels and Trypanosoma equiperdium in horses [1].
The influence of tsetse on African agriculture through the transmission of trypanosomosis continues to be a major constraint to the development of national economies and their achievement of self sufficiency in basic food production. The general distribution of tsetse flies is determined principally by climate and influenced by altitude, vegetation, and presence of suitable host animals [2]. Tsetse flies in Ethiopia are confined to southern and western regions between longitude 330 and 380 East and latitude 50 and 120 North which amounts to about 220,000Km2 [1]. Tsetse infested areas lied in the low lands and also in the river valleys of Blue Nile, Baro Akobo, Didessa, Ghibe and Omo. Out of the nine regions of Ethiopia five (Amhara, Beninshangul Gumuz, Gambella, Oromia and Southern Nation Nationalities and peoples) are infested with more than one species of tsetse flies. To date five species of Glossina (Glossina morsitans submorsitans, G. Pallidipes, G. tachnoides, G. f. fuscipes and G. longipennis) have been recorded from Ethiopia. Apart from the cyclical transmission of trypanosomosis by the Glossina species, it is highly considered that mechanical transmission is a potential threat to livestock productivity in some parts of Ethiopia [1,3].
In dale sadi district trypanosomosis was found to be one of the factors that hampered livestock rearing in most peasant associations. Therefore, a study on the status of the disease and investigating the vectors and their relative abundance is crucial for a successful control in the area. Therefore, the present work aimed at determining the prevalence of bovine trypanosomosis and apparent density of tsetse flies described in the transmission of trypanosomosis.
Material and Methods
Descriptions of study area
The study was conducted from October to November 2016 in Dale sadi district located in kelem Wollega zone of Oromia regional sate, situated at 562 Kilometers West of Addis Ababa. The mean annual rain fall in Dale sadi district ranges from 1150-1300mm. The annual temperature ranges from 26-34 °C. The district has altitudes ranging from 1200-2000 m. a. s.1. The district has large rivers conducive for tsetse fly habitat like Birbir, Mardafo and Kile river. The areas have got a number of wild animals such as African buffaloes, Bush pigs, warthog, bush buck, kudu, hippopotamus, crocodiles, hyena, antelopes and snakes which are claimed to serve as sources of food for the fly as reservoir for trypanosomes. Study population the cattle in the district are local breeds that are kept under traditional extensive husbandry systems with communal herding. Agriculture is the main stay of the livelihood of the society with mixed farming system and livestock play an integral role for agriculture [4]. The animal population of the district is estimated to be 85,189 cattle, 28,600 sheep, 19,699 goats, 181 horses, 470 mules, and 3839 donkeys [5].
Sample size and study animals
The study was conducted on local breed cattle from six Peasant Associations (PAs). The body condition score were classified as poor, medium and good by observing the body condition of the animals in the field [6]. The ages of animals were also estimated by the dentition method [7] and from owner information. Sample size was determined using 95% confidence level, 50% expected prevalence and 0.05 desired absolute precision using the formula described by Thrust field [8]. Therefore, a total of 384 cattle were needed for the study. However, sample was collected from 532 animals to increase precision.
Study design and protocol
The PA’s were selected based on their accessibility to transport and information from the district’s administrative body. Multistage sampling was used to sample animals, where, herds were selected from each PA’s by simple random sampling as primary sampling units. From the selected herds, individual animals to be sampled were selected by simple random sampling techniques as secondary sampling units.
A cross-sectional study design was employed. For the trypanosomosis prevalence study, dark phase contrast buffy coat examination and Giemsa stained thin blood smears were used. Blood sample collection was performed by piercing the marginal ear vein with a sterile lancet and blood was drawn by a heparinized capillary tube. Then one end (the heparanized end) of capillary tubes were sealed with crystal sealant and centrifuged at 12,000 rpm for five minutes to separate the blood cells and to concentrate trypanosomes using centrifugal forces, as buffy coat. Then the PCV was determined and recorded. The capillary tubes were then broken just below buffy coat using diamond pencil and expressed on microscopic slide and covered with a cover slip. Then it was examined under 40× objective of microscope to detect the presence of the parasites. Trypanosome species were identified using Giemsa stained thin blood films. For the entomological survey, a total of 66 baited traps were deployed along suitable tsetse habitats to assess the apparent densities, distributions and species of tsetse flies and other biting files involving in transmission of trypanosomosis. All traps were baited with acetone, Octenol (1-3-Octane) and cow urine filled in separated bottles and labeled and deployed at an interval of 100-150 meters. The coordination and altitude of each trap were recorded using GPS. The vegetation type, the prominent feature within 100 m radius and the canopy of each trap were recorded. After 48 hours of trap deployment, the cages were collected, and captured flies were identified and sexed according to morphological characteristics and counted. The tsetse flies were identified to species level. The apparent density of the tsetse fly was calculated as the number of tsetse catch/trap/ day [9].
Data management and analysis
Raw data were entered into a Microsoft Excel spreadsheet and descriptive statistics were used to summarize the data. The prevalence was calculated for all data as the number of infected individuals divided by the number of individuals examined and multiplied by 100. The density of fly population was calculated by dividing the number of files caught by the number of traps deployed and the number of days of deployment and expressed as fly /trap/ day (FTD). The PCV of sampled animals was measured using PCV reader and the result between infected and non infected animals was compared.
Results
Entomological survey result
A total of 66 mono-pyramidal, bi-conical, mono-conical and NGU traps were deployed in the district. The apparent density of tsetse flies is 5.34 fly/trap/day. Savana and riverine species of Glossina particularly pallidepes 17.28%, G.morsistance submorsistance 29.75%, G.furcipes 28.19% and G.tachinoides 24.79% were caught from the district (Table 1). Out of total caught tsetse flies, 25.35% were male and 74.65% were female (Table 2). The highest FTD was recorded in Chemo villages from the study areas (Table 2).
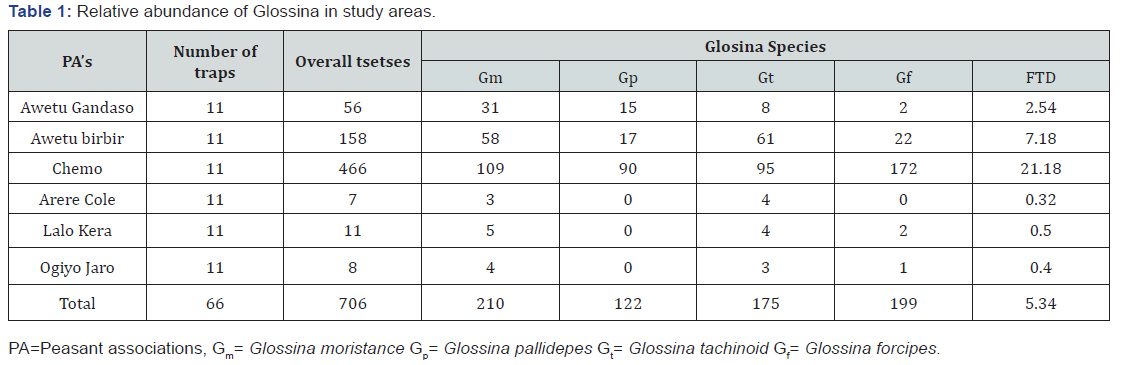
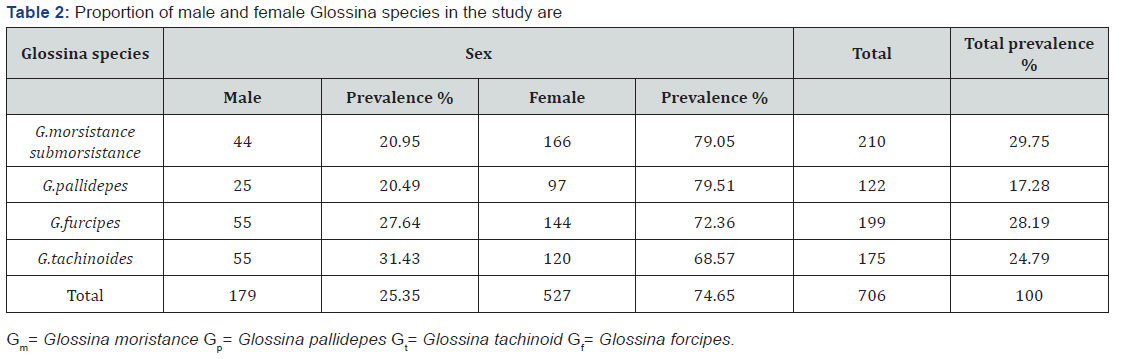
Trypanosomes survey results
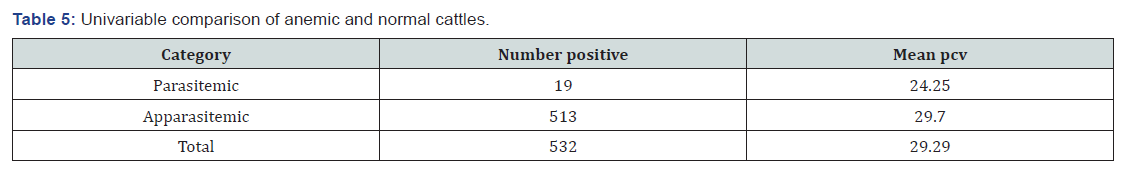
A total of 532 cattle were examined. The overall prevalence of trypanosomosis was 3.57%. The prevalence of trypanosomosis was determined to be 3.48% (3/86), 4% (4/100), 6.75% (5/74), 2% (2/100), 4.17% (3/72) and 2% (2/100) in Awetu Gandaso, Awetu Birbir, Chamo, Arere Chole, Lalo kera and Ogiyo Jaro villages respectively (Table 3). Out of the parasitaemic cattle 36.84% were infected with T. congolense and 63.16% were infected with T. vivax. Therefore, T. vivax is considered as the dominant species responsible for infection of cattle residing in the district. the Highest prevalence was recorded in cattle with poor body condition (52.63 %) followed by medium (31.58%) and good (15.79%) out of the parasitaemic cattle. Highest infection rate was recorded in Adult (52.63%) cattle followed by old (26.32%) and young (21.05%). The prevalence in female animals (57.89%) was higher than in males (Table 4).
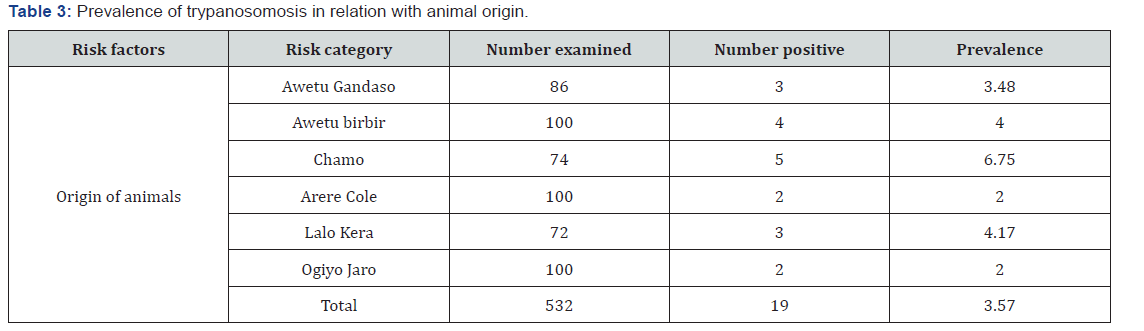
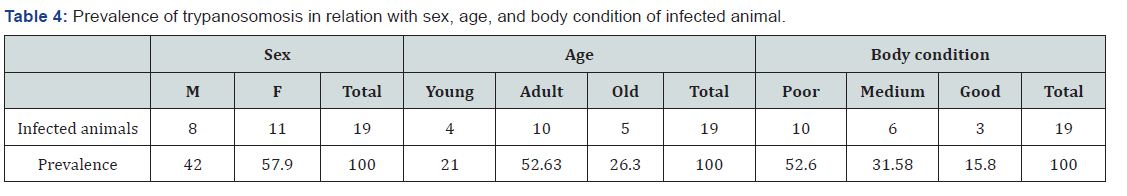
Hematological survey result
The overall mean PCV value of examined cattle was 29.29. The mean PCV of non-infected cattle was higher (29.70%) than that of infected animals (24.25%) (Table 5&6). In fact, the difference in mean PCV between parasitaemic animals and aparasitaemic animals indicated that trypanosomosis may be involved in adversely lowering the PCV values of infected animals.
Discussion
In the present study, the apparent density of tsetse flies caught was 5.34 fly/trap/day in dale sadi district. G.morsistance submorsistance, G.pallidipes, G.furcipes and G.tachinoides were the species recorded. The current result is lower than what was reported from the same study area (9.8%) and Dale Wabera (9.1%) [10,11] 6.7% from Hawa Gelan district. The number of traps deployed, site of trap installation, the months of study etc. could contribute for the variation [12]. The fly species identified also agrees with the reports of [11,13]. The low apparent density of tsetse fly by this study may be attributed to application of tsetse control measures such as target impregnated insecticides and insecticide treatment of cattle. The survey revealed that the overall trypanosome prevalence was 3.57%. This result is consistent with the work reported in Esitsa (3.45%) [14]. The current prevalence of trypanosomosis in the woreda was lower than preveous result reported by Bedaso 6.7% in Hawa Gelan district [11]. The lower prevalence of trpanosomosis in this study may be attributed to application of control measures such as target impregnated insecticides, insecticides treatment of cattle and regular treatment of sick animals. The highest prevalence was reported in Chamo village (6.75%) and the lowest was in Ogiyo Jaro and Arere Cole (2%). The Prevalence among Peasant associations in this study could be attributed to the tsetse fly type present in each peasant association which is dependent on micro climate, animal heard density, distance between herds and other various factors [11]. Finding of this study is lower than that of Adale & Yasine [15] which reported 6.3% in Wolaita zone of Kindo Koish district, Southern Ehiopia; Degneh [16] wich reported 6.86% in Lalo Kile district of Oromia regional state; Alamayehu [17] which reported 6.9% in Chena district, south west Ethiopia and Denbarga [18], Zecharias A & Zeryehun T [19] reported 27% in Arbaminch; Begna [20] reported 14.2% in Humbo district, southern Ethiopia; Melaku & Abebe [21] reported 10.67% in Debre Elias district, North west Ethiopia; Dagnachew [22] reported 11.33% in Jawi district of Amhara region, and Achukwi & Musongong [23] reported 14.3% in Faro division, northern Cameroon. .
However, findings of this work is higher than the result reported by Hunde [24] 1.02% in West Gojjam, Ethiopia; and Tadesse [25] 2.66% in Tselemti Woreda, Western Tigray, Ethiopia. The lower prevalence of trypanosomosis in this study may be attributed to application of control measures such as target impregnated insecticides, insecticide treatment of cattle and regular treatment of cattle. The proportion of Trypanosoma vivax in all sites of this study was higher (63.16%) followed by T. congolense (36.84%). This result is consistent with the report by Cherenet [10] & Cherenet [11] in tsetse free areas of Amhara region. This result disagreed with the report in which they reported 58% of the total trypanosomes detected were T. congolense. Zecharias & Zeryehun [19] Mulaw [13] also reported higher proportion of T. congolense than T. vivax. Such difference could possibly be attributed to the presence of major mechanical vectors and more efficient transmitters of T. vivax. In this study, out of infected animals, age-wise comparison revealed that a highest infection rate was recorded in Adult ones (52.63%). These results agree with that of Tasew & Duguma [26] and Dagnachew [22]. out of trypanosomosis infected animals higher prevalence is recorded in poor body conditioned animals (52.63%) than other groups. This finding is similar to the report by Bacha [27]. The prevalence of infection between sex categories was 42.1% for male and 57.89% for females. Similarly, Daya & Abebe [28] and Teka [29] also reported the same trend in susceptibility between the two sexes. This shows that both male and female cattle were more or less equally susceptible to trypanosomosis infection [30].
The overall mean PCV value of examined cattle was 29.29. The mean PCV of non infected cattle was higher (29.70%) than that of infected animals (24.25%). In fact, the difference in mean PCV between parasitemic animals and aparasitemic animals indicated that trypanosomosis may be involved in adversely lowering the PCV values of infected animals. Parasitemic animals had generally lower mean PCV than the corresponding aparasitemic ones, though there is appearing of parasitological negative animals within the PCV values of less than the threshold value. This may be due to in adequacy of detection method [30] or delayed recovery of anemic situation after current treatment with trypanocidal drugs and may be other blood parasites infection, malnutrition associated with long draught in the areas.
Conclusion
The present work showed a relatively low prevalence of trypanosomosis and apparent density of tsetse flies in Dale sadi district. However, this is an evidence not to be neglected that tsetse and trypanosomosis has yet continued to pose a considerable threat to cattle of the study area warranting an integrated parasite and vector control to safeguard cattle production and productivity
During this study a number of challenges were observed in this woreda. Some of them are:-scarcity of transportation, laboratory materials, lack of budget for this work, poor farmers participation on the work in some kebeles.
Acknowledgement
The authors are grateful to the National Tsetse and Trypanosomosis Investigation, Control and eradication Center and Dale Sadi Livestock development and Fishery office for facilitating the fieldwork.
Recommendations
Creating continues Community awareness regarding the purpose of trypasomes and tsetse fly survey in the district to improve their participations. The budget should be allocated to solve the problem of infrastructure, transportation and to fulfill all necessary materials used for surveying purpose. Effective and well targeted control measures of tsetse flies should be developed with special concern to minimize the prevalence of trypanosomosis.
References
- Abebe G (2005) Current situation of Trypanosomosis. In: review article on: Trypanosomosis in Ethiopia. Ethiop J Biol Sci 4: 75-121.
- Leak SG (1999) The role in the epidemiology and control of trypanosomosis Tsetse biology and ecology, Wallingford (UK), CAB International, pp. 152-210.
- Regassa F, Abebe G (2004) Current epidemiological situation of bovine trypano- somosis in Limu Shay tsetse controlled area of upper Didessa Valley. Ethiop Vet J 13 (2): pp. 19-33, 2004.
- Dale said (2006) Agricultural development office, Harosebu, Ethiopia.
- Dale said (2016) Livestock development and Fishery office, Harosebu, Ethiopia.
- Nicholson M, Butterworth T (1996) A guide to body condition score in zebu cattle international livestock center for Africa, Addis Ababa, Ethiopia.
- Gatenby R (1991) The Tropical Agriculture, London and Beging Stock Mc Millan Edu- cation Ltd. ACCT, pp. 6-10.
- Thrusfield M (2005) Veterinary Epidemiology (3rd Ed), Oxford, England: Blackwell Science, Ltd, pp.332.
- STEP (2012) Field operation manual of tsetse and trypanosomosis control and monitoring, Ministry of Science and Technology, Southern tsetse eradication project (step), Addis Ababa, Ethiopia, pp. 5-63.
- Cherenet TA (2004) Epidemiology of bovine and its economic impact in tsetse-infested and tsetse-free areas of Amhara region, North-west Ethiopia, PhD thesis, University of Putra, Malaysia.
- Cherenet T, Sani RA, Panandam JM, Nadzr S, Speybroeck N, et al. (2004) Seasonal prevalence of bovine trypanosomosis in a tsetse-infested zone and a tsetse-free zone of the Amhara Region, north-west Ethiopia. Onderstepoort J Vet Research 71(4): 307-312.
- Abebe G, Jobre Y (1996) Trypanosomosis: A threat to cattle production in Ethiopia. Rev Vet Med 147: 897-902.
- Mulaw S, Addis M, Fromsa (2011) A Study on the Prevalence of Major Trypanosomes Affecting Bovine in Tsetse Infested Asosa District of Benishangul Gumuz Regional State, Western Ethiopia Global Veterinaria 7: 330-336.
- Tafese W, Melaku A, Fentahun T (2012) The prevalence of bovine trypanosomosis and in two districts of East Wollega Zone ,Ethiopia Onderstepoort journal of veterinary research 79(1): 34-43.
- Adale E, Yasine A (2013) Prevalence of bovine trypanosomosis in Wolaita Zone Kindo Koish District of Ethiopia,’’ African Journal of Agricultural Research 8: 6383-6387.
- Degnah E, Fereda B, Bekuma B, Hunde A, Duguma M (2013) Prevalence of Bovine Trypanosomosis in Lalo Kile District, Kelem Wollega Zone, Oromia Regional State, Western Ethiopia. Acta Parasitological Globalis 4: 34-40.
- Alemayehu B, Bogale B, Fentahun T, Chanie M (2012) Bovine trypanosomosis: A threat to cattle production in Chena district, Southwest Ethiopia. Open J of Ani Sci 2: 287-291.
- Denbarga Y, Ando O, Abebe R (2012) Trypanosoma Species Causing Bovine Trypanosomosis in South Achefer District, Northern Ethiopia. J Vet Adv 2: 108- 113.
- Zecharias A, Zeryehun T (2012) Prevalence of Bovine Trypanosomosis in Selected District of Arba Minch, SNNPR, Southern Ethiopia. Global Veterinaria 8: 168-173.
- Begna F, Abebe S, Bekele M Bovine Trypanosomosis in Selected Villages of Humbo District, Southern Ethiopia. Global Veterinaria 7: 192-198.
- Melaku A, Abebe A (2012) Bovine Trypanosomosis and Its Vector Type and Density at Debre Elias District, North-western, Ethiopia. Journal of Advanced Veterinary Research 2: 247-251.
- Dagnachew S, Girma H, Abebe G (2011) A cross-sectional study on bovine trypanosomosis in Jawi district of Amhara Region, Northwest Ethiopia. Ethiop Vet J 15: 69-78.
- Achukwi MD, Musongong GA (2009) Trypanosomosis in the Doayo/Namchi (Bos taurus) and zebu White Fulani (Bos indicus) cattle in Faro Division, North Cameroon. Journal of Applied Biosciences 15: 807-814.
- Hunde AB, Chanie TA, Ferede BW, Tesfaye AM (2012) Prevalence of Bovine Trypanosomosis in Addisamba and Amarit District of West Gojjam Zone, Amhara Regional State. American-Eurasian Journal of Scientific Research 7: 112-117.
- Tadesse A, Hadgu E, Mekbi B, Abebe R, Mekuria S (2012) Mechanically transmitted bovine trypanosomosis in Tselemty Woreda, Western Tigray, Northern Ethiopia. Agricultural Journal 6: 10-13.
- Tasew S, Duguma R (2012) Cattle anaemia and trypanosomiasis in Western Oromia State, Ethiopia. Revue Méd Vét 163: 581-588.
- Bacha B, Beyene Z, Woyessa M, Hunde A (2013) Prevalence of Small Ruminants Trypanosomosis in Assosa District of Benishangul Gumuz Regional State, Western Part of Ethiopia. Acta Parasitologica Globalis 4: 99-104.
- Daya T, Abebe G (2008) Seasonal dynamics of tsetse and trypanosomosis in selected sites of Ethiopia. Ethiop Vet J 12: 77-98.
- Teka W, Terefe D, Wondimu A (2012) Prevalence study of bovine trypanosomosis and tsetse density in selected villages of Arbaminch, Ethiopia. Journal of Veterinary Medicine and Animal Health 4: 36-41.
- Murray M, Murray PK, McIntyre WIM (1997) An improved parasitological technique for the diagnosis of African trypanosomosis. Transactions of the Royal Society of Tropical Medicine and Hygiene 71: 325-326.






























