Comparison of Five Methods for Lipid Extraction from the Phaeodactylum tricornutum Microalga and Determination of Fucoxanthin and Fatty Acids Profiles
Caroline Schmitz1*, Anabela Pizzatto dos Passos1, Claudia Marlene Bauer1, Juliana Cunha1, Bruna Mattioni2 and Marcelo Maraschin1
1 Laboratório de Morfogênese e Bioquímica Vegetal, Centro de Ciências Agrárias, Universidade Federal de Santa Catarina, Brazil
2Laboratório de Nutrição de Espécies Aquícolas, Centro de Ciências Agrárias Universidade Federal de Santa Catarina, Brazil
Submission: June 03, 2019; Published:June 26, 2019
*Corresponding author: Caroline Schmitz, Laboratório de Morfogênese e Bioquímica Vegetal, Centro de Ciências Agrárias, Universidade Federal de Santa Catarina, CEP 88040-970, Florianópolis, Brazil
How to cite this article:Caroline S, Anabela P d P, Claudia M B, Juliana C, Bruna M, et al. Comparison of Five Methods for Lipid Extraction from the Phaeodactylum tricornutum Microalga and Determination of Fucoxanthin and Fatty Acids Profiles. Adv Biotechnol Microbiol. 2019; 14(3): 555886. DOI: 10.19080/AIBM.2019.14.555886
Abstract
In this study the effectiveness of five protocols for lipid (FA) and carotenoid extraction from the Phaeodactylum tricornutum microalga was assayed. The protocols of Folch, Lee and Stanley, Bligh and Dyer, and Bligh and Dyer + UAE (ultrasound-assisted extraction) showed the higher yields of total lipids determined through gravimetric analysis. Folch, Lee and Stanley protocol was more effective in extracting fucoxanthin in the methanolic phase. It is noteworthy that there was no significant difference in fucoxanthin extraction in chloroform phases among the methods evaluated. On the other hand, the Folch, Lee and Stanley method showed to be more selective for MUFAs recovery from the microalgal biomass. However, following the market demands for efficient extraction of oils rich in omega-3, Bligh and Dyer and Bligh and Dyer + UAE protocols are recommended. The protocol established by Bligh and Dyer + UAE can be recommended as an effective methodology in biomedicinal oil extraction.
Keywords: Organic solvent extraction protocols; Microalgae; Fatty acids; Fucoxanthin; Lipid extraction; Phaeodactylum tricornutum
Abbrevations: MUFAs: Monounsaturated Fatty Acids; PUFAs: Polyunsaturated Fatty Acids; EPA: Eicosapentaenoic acid; DHA: Docosahexaenoic Acid; FA: Fatty Acids; UAE: Ultrasound-Assisted Extraction; BD: Bligh and Dyer method; FLS: Folch Lee and Stanley method; SÖ: Selstam and Öquist
Introduction
Microalgae produce a large amount of bioactive compounds that can be used by society as natural supplements. These bioproducts have been gaining ground in the market due to their broad panel of bioactivities. Marine bioactive compounds in a broad-sense, can be polysaccharides, pigments, proteins, vitamins, and lipids with several activities, among them, anti-tumor, antioxidants, antibiotics and photoprotectors [1-3]. These bioproducts have been marketed as encapsulated fatty acids, amino acids, and pigments for human consumption and pharmaceutical and cosmetic industries. Despite the increasing market of microalgae bioproducts, the raw material for the extraction of bioactive molecules of aquatic organisms has in fish stock biomass its main source yet [4].
Due to sustainability concerns of fish farming systems, microalga cultivation systems are claimed as a viable alternative to obtain oils rich in omega-3 fatty acids (ω-3). Currently the slow development at industrial scale of cultivation systems for microalgae has been related to the limited growth of these organisms in open ponds and the high costs of these cultures in photobioreactors [5]. Other problems are associated with the processing of biomass, which, when not improved, has a direct relationship with the yield of this crop. One of the obstacles to the extraction of microalgae’s oil fraction is the nonexistence in the target species of an optimized method to recover that fraction, despite several reports in the literature. Therefore, more studies are necessary to fit a standard method specifically directed to the species of interest in this study (i.e., a tailored protocol), considering their structural differences (i.e., chemical composition of the cell wall) and metabolic differences between microalgal biomasses traditionally used as raw materials [5].
Furthermore, the works related to the evaluation of extraction methods are commonly directed to green microalgae and cyanobacteria, as they are species already cultivated for Abstract In this study the effectiveness of five protocols for lipid (FA) and carotenoid extraction from the Phaeodactylum tricornutum microalga was assayed. The protocols of Folch, Lee and Stanley, Bligh and Dyer, and Bligh and Dyer + UAE (ultrasound-assisted extraction) showed the higher yields of total lipids determined through gravimetric analysis. Folch, Lee and Stanley protocol was more effective in extracting fucoxanthin in the methanolic phase. It is noteworthy that there was no significant difference in fucoxanthin extraction in chloroform phases among the methods evaluated. On the other hand, the Folch, Lee and Stanley method showed to be more selective for MUFAs recovery from the microalgal biomass. However, following the market demands for efficient extraction of oils rich in omega-3, Bligh and Dyer and Bligh and Dyer + UAE protocols are recommended. The protocol established by Bligh and Dyer + UAE can be recommended as an effective methodology in biomedicinal oil extraction. Keywords: Organic solvent extraction protocols; Microalgae; Fatty acids; Fucoxanthin; Lipid extraction; Phaeodactylum tricornutum Abbrevations: MUFAs: Monounsaturated Fatty Acids; PUFAs: Polyunsaturated Fatty Acids; EPA: Eicosapentaenoic acid; DHA: Docosahexaenoic Acid; FA: Fatty Acids; UAE: Ultrasound-Assisted Extraction; BD: Bligh and Dyer method; FLS: Folch Lee and Stanley method; SÖ: Selstam and Öquist this purpose. Among microalgae of biotechnological interest, Phaeodactylum tricornutum has been extensively exploited for its interesting traits such as:
a. to store large amounts of neutral lipids, i.e., about 30% of its dry weight [6],
b. a short cropping cycle [7],
c. to synthesize nutraceutical pigments [8],
d. adaptability, modifying its shape according to the environment conditions [9],
e. not demanding an organic carbon source for growth in culture medium, surpassing the productivity of any agricultural crop [10], and
f. its genome has been sequenced, being a relevant model for metabolic engineering studies involving the biosynthesis and accumulation of molecules of biotechnological importance as fucoxanthin, for instance [11].
One attractive bioproduct in P. tricornutum is its high added value lipid fraction, due to its content in ω-3 fatty acids, named long chain Polyunsaturated Fatty Acids (PUFAs) such as Eicosapentaenoic Acid (EPA) and Docosahexaenoic Acid (DHA), [12]. These metabolites play important biochemical roles in the brain and retina, augment the absorption of fat-soluble vitamins, and still exhibit anti-inflammatory action [13-15]. Besides, in the lipid fraction of microalgal biomasses the carotenoid compounds are also highlighted.
In this context, the diatom P. tricornutum has fucoxanthin as its major carotenoid and, due to its nonpolar trait, such secondary metabolite is usually found associated to the lipid fraction in that species. This pigment has a potent antioxidant action, preventing diseases associated with the damage effects of free radicals in cells such as cancer [16]. The aforementioned bioproducts, i.e., fatty acids and carotenoids, are already commercially available from the ingestion of the microalgal matrix of species such as Chorella sp. and Spirulina sp., being used in human food. Additionally, they can also be extracted from microalgae to be sold separately, or in formulations containing the microalgal lipid extract, the last one seeking the synergism of its benefits to the human health. Importantly, studies have suggested that a diet rich in fatty acids may increase the absorption of fucoxanthin in human beings, for instance [17]. This occurs because esterified fucoxanthin is probably incorporated into the lipid core and transported to a variety of tissues, facilitating their absorption and enhancing the bioavailability, given its highly apolar nature [18]. This occurs due to a connection to an esterification process of those metabolites in cells of the human and mice intestine, facilitating their absorption and enhancing the bioavailability, given the highly apolar nature described in models [19].
The choice of the extraction methodology can directly impact the design of microalgae production bioprocesses, with positive repercussions on the possibility of expanding the market of bioproducts containing fatty acids and carotenoids to human food, i.e., nutraceuticals. Based on the above, it is assumed as a working hypothesis that a standardized protocol for the extraction of the oil fraction eventually containing carotenoids of P. tricornutum biomass is relevant to improve the process for the recovery of those high added value metabolites. In this sense, this study aimed to determine and to compare the extraction efficiency of five protocols in recovering the lipid fraction, fatty acids, and carotenoids from P. tricornutum biomass. For that, gravimetric and chromatographic analytical techniques were applied to determine the yields of the extraction protocols and the fatty acids and fucoxanthin profiles in the organosolvent samples thereof.
Material and Methods
Culture and biomass preparation
The marine microalgae Phaeodactylum tricornutum, strain CCAP 1052/A, was cultured in 500 ml Erlenmeyer flasks for 7 days in f/2 culture medium, following the protocol of [20]. Initial concentration of cells per flask was 22k cells/mL/L, receiving 65μmol m-2s-1 of irradiance on average. At the beginning, the culture medium presented pH values ~ 8 and 31% salinity. No relevant changes occurred in these variables over the cultivation period, i.e., seven days. At the end of the experiment (day 7th), the cell cultures were collected, centrifuged (2276g, 9 min) and the supernatant rejected. The collected biomass was desalinated by quickly washing it with 0.5M ammonium format solution, following freeze-drying (36 h, 0.001 mbar) and storage at -20 °C until extraction. This biomass will serve as a pool for lipid extraction of three independent experiments, according to three protocols [21-23]. Hereafter, such protocols will be assigned in the text to the acronyms BD, FLS, and SÖ, respectively. Additionally, the BD protocol was performed three times by adding a pre-treatment of cell disruption with the aid of microwaves and Ultrasound- Assisted Extraction (UAE).
Extraction of total lipids
Using the BD method, 100mg of dry microalgal biomass were added of 3ml methanol (MeOH), 3 ml chloroform (CHCl3) and stirred (vortex) for 30s. In the sequence, 1ml CHCl3 1ml MeOH and 2ml water were added, following incubation for 10 min, with eventual shaking (vortex, 30s, every 2 min). Subsequently the extract was filtered. The filter was rinsed with 4 ml CHCl3 to allow a better recovery of the lipid fraction. This process resulted in a three-phase extract of chloroform, methanol and water, roughly at ratio 2: 2: 1.8 (v/v/v). This procedure was repeated three times, two of them by performing a pre-treatment for cell disruption using microwaves (557 W) for 1 min and ultrasound using a sonicator for 30 minutes (47 160kHz, 60 W, Branson B-2200) before adding solvents.
In the FLS method, a sample of P. tricornutum biomass (100mg dry weight) was homogenized in a CHCl3 MeOH (2: 1, v/v) solution, following shaking (vortex) for 1minute. The extract was filtered, and the biomass resuspended with the extract solution above described. Subsequently, a 0.7% aqueous NaCl solution was added, affording a final solvent system of CHCl3: MeOH: water (2: 1: 0.8, v/v/v). As this protocol requires small amounts of biomass and solvent, it was necessary to adapt it in order to be able to collect the extract after filtering. Thus, the extract was withdrawn and a new resuspension with 3 ml CHCl3 was carried out in the vacuum flask in order to recover a larger amount of extract.
The method of SÖ was followed keeping in mind the adaptations proposed by [24]. For that, a sample of 100mg microalgal biomass was homogenized with a mixture of CHCl3 and a solution of methanol: aqueous 0.7% NaCl (4: 1, v/v), resulting in extract of chloroform: methanol: water (1: 2: 0.5, v / v / v). The solvent solution was added, resulting in a solution of chloroform: methanol: water (2: 3.6: 0.9, v / v / v) that was centrifuged. Finally, a solution of chloroform: methanol and aqueous solution (0.73% NaCl) was added, resulting a system of chloroform: methanol: water (1:1:0.8, v / v / v). i.e., After the extraction, the CHCl3 phase was concentrated under reduced pressure (rotaevaporator), at 40 °C, until dryness. After this procedure, the flasks were weighed in order to determine the lipid fraction content by gravimetry.
Fatty acid analysis
For the analysis of fatty acids, the lipids were extracted and quantified following the methods specified above. After measuring the lipid fraction of samples by gravimetry, an aliquot (0.3g) was collected and the fatty acid methyl esters were obtained according to O’Fallon, [25]. Methyl esters were injected (1.0 μL) into a gas-liquid chromatograph (Agilent 7890B series GC system) equipped with an automatic injector (240 °C), a fused silica RTx 2330 column (90% biscyanopropyl/10% cyanopropylphenyl polysiloxane - 105 m x 0.25 mm internal diameter x 0.20 μm) at 120 ºC, and a FID detector (300 °C). For the chromatography of the methyl derivatives of fatty acids, the column was initially thermostitized at 120 °C, increasing to 186°C at a rate of 3 °C/min following injection and held for 5 min; then ramped at 3 °C/min to a final temperature of 231°C. Helium as carrier gas was used at 2.5 ml/min flow rate and samples were injected in a 1: 40 split ratio. The composition of fatty acids was determined by comparing the retention times of the peaks with those of the respective fatty acid standard (C23: 0 - Sigma Aldrich, MO USA) and normalization area, being expressed as percentage for quantitation purpose.
Fucoxanthin analysis
High-performance liquid chromatography (HPLC) was used for fucoxanthin quantification in both the methanolic fraction and chloroformic one (lipid phase) of the P. tricornutum extract. The later fraction was previously dried in a speed-vac apparatus and resuspended in MeOH. An aliquot of each sample (10 μL, n = 3) was injected into a liquid chromatograph (Shimadzu LC-10 A), equipped with a C18 reverse phase column (Vydac 218TP54, 250 mm x 4.6 mm, ∅ 5 μm, 30 °C), protected by a C18 reverse-phase guard column (Vydac 218GK54, ∅ 5 μm), and a UV-vis detector (450 nm). Elution was performed with MeOH/ CH3CN (90: 10, v/v) at a flow rate of 1 ml min-1. Fucoxanthin identification was performed using its retention time and co-chromatography of analytical standard (Sigma-Aldrich, St. Louis, MO, EUA). Metabolite quantification was based on standard curve of fucoxanthin (1 - 25 μg ml-1; y= 0.0513x; r2 = 0.999. Readings were taken in triplicate and results were expressed in mg/g.
Statistical analyzes
Data were collected, summarized, and submitted to the analysis of variance ANOVA, followed by the post hoc Tukey test, when appropriate. The results were considered significant at p <0.05. Statistical analyzes were conducted using scripts written in R language (v. 3.1).
Results and Discussion
The total lipid content of Phaeodactylum tricornutum extracts is shown in Figure 1. The most effective protocols for the extraction of total lipids were BDyer and FLS. The lipid yield increased when ultrasound was added to the process, though the results did not differ statistically. The phenomenon of cell disruption is observed with the aid of ultrasound and occurs through cavitation bubbles formed into the organosolvent extraction solution [26]. In liquids exposed to sonication at high intensities, the sound waves that propagate into the liquid media result in alternating high- and low-pressure cycles. During the low pressure cycle, high-intensity ultrasonic waves create small vacuum bubbles or voids in the liquid. When the bubbles attain a volume at which they can no longer absorb energy, they collapse violently during a highpressure cycle giving rise to the cavitation phenomenon [27]. In our study, the protocol associating ultrasound with chemical extraction (i.e., UAE) increased the yield and reproducibility of the method since considerably lower standard deviation value has been found.
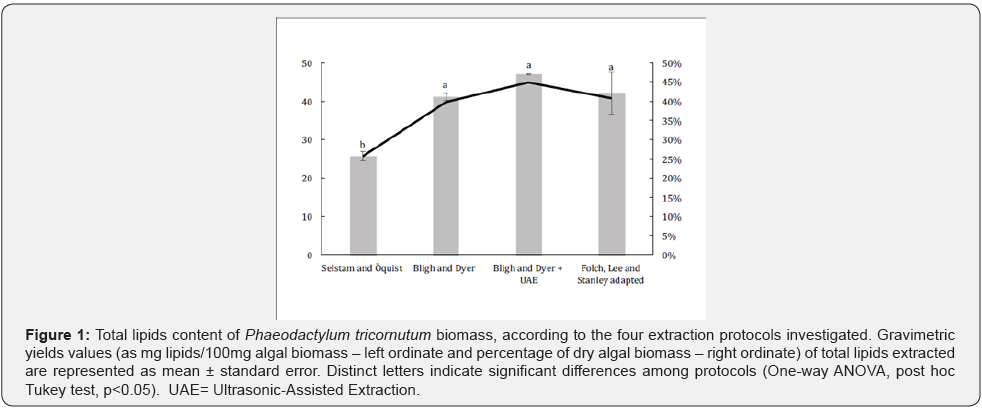
The FLS’s protocol when performed as originally described presented the lowest yield of extraction, affording only 18% of lipids on average of the dry biomass. The work of [28], that also investigated the yield of lipidic extraction in Chlorella vulgaris microalga found similar values to the ones presented here. Chlorella vulgaris presented yields around 40% and 15% in Bligh and Dyer method and Folch, Lee and Stanley, respectively, as in this study. However, the low yield of Folch, Lee and Stanley method could be related to the fact of being the only method that uses the recovered mixture for washing the paper filter in the protocol’s second step. In order to reduce the steps and increase the recovery content, the Folch protocol has been adapted. For this, the recovery step of the extract in the filtration was performed with additional 3 ml of chloroform. With this protocol adaptation the lipid content was increased from 18% to 41%. In the other methods the paper filter is washed with pure solvent, what reduces the number of filtration steps. This additional stage of recovery and washing influenced extractions in small quantities considering the volatility of the solvents used. When the protocol was adapted with the addition of 3 ml chloroform for washing the paper filter, the yield increased, resembling Bligh and Dyer extraction. This difference in yield may imply that when using a small amount of solvents, it is necessary to add a washing step of the filtration system with pure solvents. The FLS protocol presented the lowest yield of extraction, affording only 26% lipids on average from the P. tricornutum dry biomass. Araújo and collaborators (2013) found a recovery of the Chlorella vulgaris’ lipidic fraction close (i.e., 15%) to the one herein described by using the FLS protocol (data not shown).
On the other hand, those authors achieved a lipid yield around 40% using the BD method as in this study. The low yield of FLS protocol could be related to the fact of being the only method that uses the recovered mixture to wash the filter in the second step. In the other methods the filter is washed with pure solvent which reduces the number of filtration steps. This additional stage of recovery and washing might influence negatively the extractions of small quantities of target compound(s) due to the volatility of the solvents used. As above described in our study, the FLS’s protocol was adapted with the addition of 3 mL chloroform for washing the paper filter, allowing to improve the recovery of metabolites and resembling the performance of the BD extraction method. This differences in the yield values observed may imply that when using a small amount of solvents, it is necessary to add a washing step of the filtration system with pure solvents to improve the recovering of the analyte of interest.
The yield extraction was lower with the SÖ protocol that afforded about 26% lipids from dry algal biomass, representing a reduction of almost 20% in relation to the highest yield method, i.e., FLS. [24] also adopted the SÖ protocol with modifications to extract lipids from four cultured microalga species (Scenedesmus dimorphus, Chlorella protothecoides, Selenastrum minutum and a polyculture) obtaining the highest income from the later one. This demonstrates the influence cell genotype on the yield of the lipid fraction recovered according to the microalga species. P. tricornutum’ s fatty acids profiles determined by gas chromatography showed to be in accordance to previous reports [29]. Table 1 shows the composition of fatty acids in the lipidic fractions recovered from the microalga samples according to the extraction protocols investigated. Nine saturated and fourteen unsaturated fatty acids were identified in the P. tricornutum’s metabolic profile, Interestingly, the fatty acid profiles detected seem to be protocol-dependent, since a more complete profile was got by recovering the lipidic fraction through the BD method.
The palmitoleic acid (C16:2n4), an omega-7 monounsaturated fatty acid, was the main compound detected in the lipidic fractions studied, varying from 29% to 35% depending on the extraction protocol adopted. Interestingly, palmitoleic acid and a minor compound also detected in the P. tricornutum’s lipidome, i.e. the hexadecatrienoic acid (C16:3n4 - HTA) were reported as antibacterial against Staphylococcus aureus, with IC50 of 10-20 μM and 20-40 μM, respectively [30]. The sum of these two FAs in Phaeodactylum tricornutum may represent at least 30% of the lipidic fration in this study. Since palmitoleic acid is active against S. aureus in micromolar concentrations [30], one could speculated that such high content of antibacterial fatty acids might confer a potential value of that microalga extract in the biotechnology market. Finally, following the extraction protocols investigated in this study, no meaningful differences were detected among BD, FLS, and SÖ methods regarding the recovery of palmitoleic acid (i.e., ~ 35%) from the microalgal biomass. However, the BD protocols when performed assisted with physical methods (ultrasound and microwave exposure) showed significantly lower yields (Table 1).
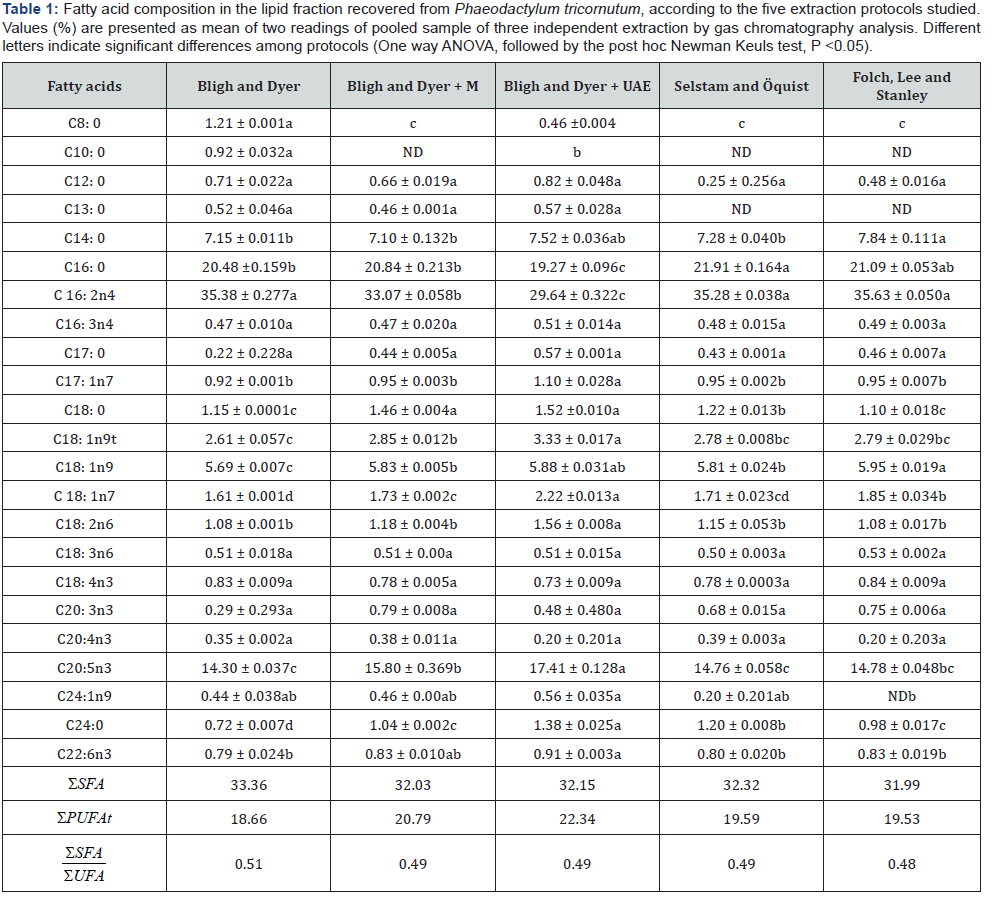
ND= not detected; SFA= saturated fatty acids; PUFA= polyunsaturated fatty acids; UFA= unsaturated fatty acids; M= microwave; UAE= Ultrasonic- assisted Extraction.
Significant amounts of palmitic (C16:0, > 19%) and eicosapentaenoic (C20: 5n3, > 14%) acids were also found in the P. tricornutum profile, with minor amounts of other saturated and unsaturated fatty acids. Palmitic acid is one of the most common saturated fatty acids found in animals, plants, and microorganisms, being the first fatty acid produced in the biosynthesis pathway of those primary metabolites and the precursor of longer fatty acids [31]. Consequently, it commonly appears as one of the major fatty acids in various organisms. The third most abundant fatty acid detected in microalgal samples was eicosapentaenoic acid (C20:5n3-EPA) with concentrations varying from 14% to 17% according to the extraction method (Table 1). The EPA extraction was positively influenced by the physically and chemically assisted extraction processes. Thus, samples exposure to UAE prior to the chemical extraction allowed to increase the yield of fatty acid recovery from 14% to 17%. For instance, the UAEassisted extraction of ω-3 DHA (C22:6n3) enabled the BD protocol yield to an increase from 0.79% (non-assisted) to 0.91%. [32] also observed increases of yield total lipids by gravimetry when using UAE-assisted extraction methods in studies with Nannochloropsis sp. extraction protocols. The data (Figure 1) infers that even when using a small amount of biomass and solvents, it seems advantageous to use a physical method to increase yield. The lipid extraction efficiency is augmented when cell membranes are lysed by the microwave or ultrasound exposure, facilitating the permeation of solvent and improving the recovery of the target analyte(s).
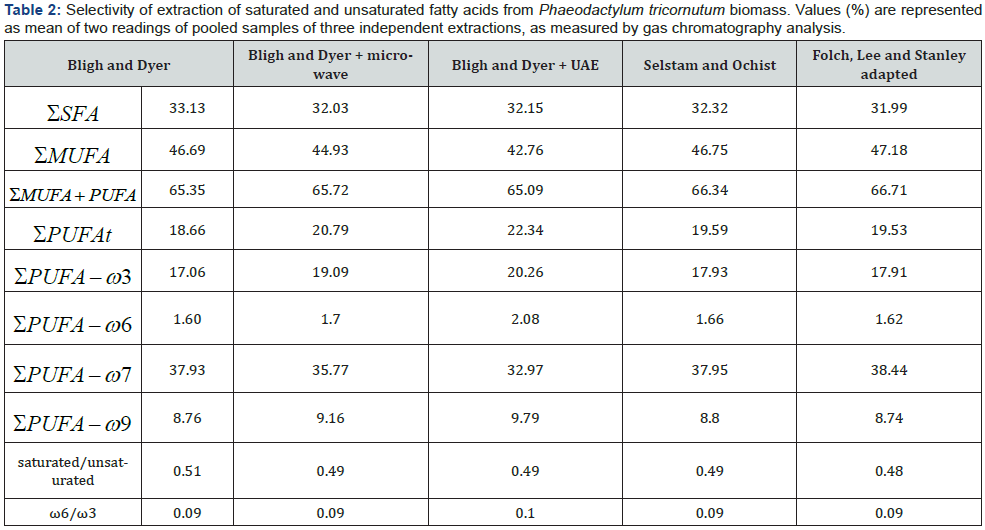
The extraction methods herein investigated demonstrated selectivity regarding the recovery of MUFAs and PUFAs (Table 2) from the microalgal biomass, while no significant differences were noted in the yield of saturated FA recovered. Methods that did not adopted physical treatment of the samples, i.e., microwave and ultrasonic bath previously to the chemical extraction afforded higher yield of MUFAs, 47.18%. Contrarily, the assisted methods using UAE and microwave and have lower yields, 44.93% for MUFA, respectively. In respect to the PUFA extraction, the addition of physical methods increased up to 4% the yield of the chemical extraction protocols. These results may infer that MUFAs are more sensitive than PUFA to the use of a physical method such as microwave and UAE, previously to the chemical extraction. The proportion of SFAs and PUFAs was around 0.48 to 0.51. The recommended ratio of PUFAs to SFAs for feed should be above 0.4 [33].
In a second approach, and taking into account the extraction protocols that resulted in higher efficiency of recovery of P. tricornutum’ s lipid fraction (i.e., FLS, BD, and BD + UAE), samples were analyzed regarding their fucoxanthin contents both in CHCl3 (lipid phase) and MeOH (non-lipid phase) extracts (Figure 2). As expected, due to the hydrophobic nature of this oxycarotenoid, the CHCl3 phase shown to be quite richer in fucoxanthin (0.6-1.4 mg g-1 +/- 0.003 dry mass) than the MeOH one, where negligible amounts were found (~0.10 mg g-1). For the CHCl3 phase, the extraction of fucoxanthin did not differ (p< 0.05) among the FLS and BD methods, recovering on average 1.22 mg g-1 of that pigment. However, in the methanol phase, the extraction of fucoxanthin was superior only in the Folch, Lee and Stanley method extracting 0.08 mg. g-1 fucoxanthin.
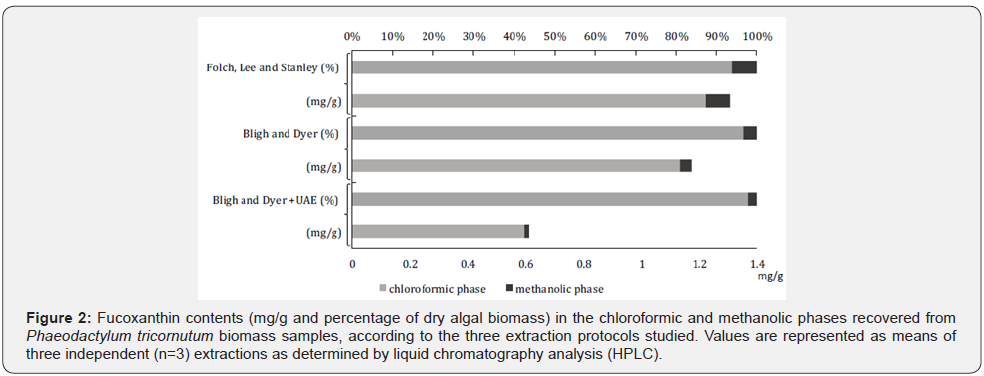
That resulted in higher efficiency of recovery of P. tricornutum’ s lipid fraction (i.e., FLS, BD, and BD + UAE), samples were analyzed regarding their fucoxanthin contents both in CHCl3 (lipid phase) and MeOH (non-lipid phase) extracts (Figure 2). As expected, due to the hydrophobic nature of this oxy carotenoid, the CHCl3 phase shown to be quite richer in fucoxanthin (0.6-1.4 mg g-1 +/- 0.003 dry mass) than the MeOH one, where negligible amounts were found (~0.10 mg g-1). For the CHCl3 phase, the extraction of fucoxanthin did not differ (p< 0.05) among the FLS and BD methods, recovering on average 1.22 mg g-1 of that pigment. However, in the methanol phase, the extraction of fucoxanthin was superior only in the Folch, Lee and Stanley method extracting 0.08 mg. g-1 fucoxanthin.
Conclusion
When choosing a method for oil and fucoxanthin extraction from Phaeodactylum tricornutum some protocols are available, eventually with distinct yield performances. The Folch, Lee and Stanley, Bligh and Dyer, and Bligh and Dyer + UAE methods afforded superior lipid recovery yields by gravimetric analysis. Importantly, the Folch, Lee and Stanley method showed to be more selective for MUFAs extraction. On the other hand, the Bligh and Dyer-based protocols are recommended due to their greater selectivity for PUFAs, increasing the nutraceutical value of extracts. Thus, the later protocols seem to be more suitable as effective methodologies for oil extraction, with potential application in food industry, for instance. In respect to the recovery of carotenoids from the microalgal biomass, it is noteworthy that similar results were detected in the relative extraction yield (%) of fucoxanthin in the chloroform phase among the methods studied, the same not being true regarding the extraction performance based on algal biomass (mg.g-1), where the Bligh and Dyer + UAE showed the lowest values. Finally, the microalga P. tricornutum is a rich source of nutraceutical substances as seen in the course of this work. It has been claimed that a diet based on fatty acids such as EPA and xanthophylls like fucoxanthin can help preventing various chronic diseases that affect millions of people worldwide. This work provides relevant information on the most efficient protocols for the extraction.
Acknowledgement
CS and CB were supported by a CAPES fellowships. The researcher fellowship from CNPq (process nº 307099/2015-6) on behalf of M. Maraschin is acknowledged.
References
- Haugan JA, Liaaen Jensen S (1994) Algal carotenoids 54. Carotenoids of brown algae (Phaeophyceae). Biochemical Systematic and Ecology 22(1): 31-41.
- Molinski TF, Dalisay DS, Lievens SL, Saludes JP (2009) Drug development from marine natural products. Nature reviews Drug discov 8(1): 69.
- Martins A, Vieira H, Gaspar H, Santos S (2014) Marketed marine natural products in the pharmaceutical and cosmeceutical industries: Tips for success. Marine drugs 12(2): 1066-1101.
- Bergé JP, Barnathan G (2005) Fatty acids from lipids of marine organisms: molecular biodiversity, roles as biomarkers, biologically active compounds, and economical aspects. Adv Biochem Eng Biotechnol 96: 49-125.
- Cooney M, Young G, Nagle N (2009) Extraction of bio‐oils from microalgae. Separation Purification Reviews 38(4): 291-325.
- Radmann EM, Costa JAV (2008) Lipid content and fatty acids composition variation of microalgae exposed to CO2, SO2 and NO. Química Nova 31(7): 1609-1612.
- Jorquera O, Kiperstok A, Sales EA, Embirucu M, Ghirardi ML (2010) Comparative energy life-cycle analyses of microalgal biomass production in open ponds and photobioreactors. Bioresource technology 101(4): 1406-1413.
- Kim SM, Jung YJ, Kwon ON, Cha KH, Um BH, et al. (2012) A potential commercial source of fucoxanthin extracted from the microalga Phaeodactylum tricornutum. Applied biochemistry and biotechnol 166(7): 1843-1855.
- Alessandra De Martino, Agnès Meichenin, Juan Shi, Kehou Pan, Chris Bowler (2007) Genetic and phenotypic characterization of Phaeodactylum tricornutum (Bacillariophyceae) accessions . Journal of Phycology 43(5): 992-1009.
- Ronga D, Biazzi E, Parati K, Carminati D, Carminati E, et al. (2019) Microalgal Bio stimulants and Bio fertilisers in Crop Productions. Agronomy 9(4): 192.
- Bowler C, Allen AE, Badger JH, Grimwood J, Jabbari K, et al. (2008) The Phaeodactylum genome reveals the evolutionary history of diatom genomes. Nature 456(7219): 239- 244.
- Ryckebosch E, Muylaert K, Foubert I (2012) Optimization of an analytical procedure for extraction of lipids from microalgae. Journal of the American Oil Chemists' Society 89(2): 189-198.
- Mullen A, Loscher CE, Roche HM (2010) Anti-inflammatory effects of EPA and DHA are dependent upon time and dose-response elements associated with LPS stimulation in THP-1-derived macrophages. J Nutr Biochem 21(5): 444-450.
- Simopoulos AP (2002) Omega-3 fatty acids in inflammation and autoimmune diseases. J Am Coll Nutr 21(6): 495-505.
- Simopoulos AP (2013) Dietary omega-3 fatty acid deficiency and high fructose intake in the development of metabolic syndrome, brain metabolic abnormalities, and non-alcoholic fatty liver disease. Nutrients 5(8): 2901-2923.
- Nishino H, Murakosh M, Ii T, Takemura M, Kuchide M, et al (2002) Carotenoids in cancer chemoprevention. Cancer Metastasis Rev 21(3-4): 257-264.
- Pangestuti R, Kim SK (2011) Biological activities and health benefit effects of natural pigments derived from marine algae. Adv Food Nutr Res 3(4): 255-266.
- Sugawara T, Yamashita K, Asai A, Nagao A, Shiraishi T, et al. (2009) Esterification of xanthophylls by human intestinal Caco-2 cells. Arch Biochem Biophys 483(2): 205-212.
- Sugawara T, Baskaran V, Tsuzuki W, Nagao A (2002) Brown algae fucoxanthin is hydrolyzed to fucoxanthinol during absorption by Caco-2 human intestinal cells and mice. The Journal of nutrition 132(5): 946-951.
- Guillard RR (1975) Culture of phytoplankton for feeding marine invertebrates. Culture of Marine Invertebrate Animals. Springer, Boston, MA, (pp. 29-60).
- Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and 382purification. Can J Biochem Physiol 37(8): 911-917.
- Folch J, Lees M, Sloane Stanley GH (1957) A simple method for the isolation and purification of total lipides from animal tissues. J biol Chem 226(1): 497-509.
- Selstam E, Öquist G (1985) Effects of frost hardening on the composition of galactolipids and phospholipids occurring during isolation of chloroplast thylakoids from needles of scots pine. Plant science 42(1): 41-48.
- Axelsson M, Gentili F (2014) A single-step method for rapid extraction of 374total lipids from green microalgae. PloS one 9(2): 1-6.
- O fallon JV, Busboom JR, Nelson ML, Gaskins CT (2007) A direct method for fatty acid methyl ester synthesis: application to wet meat tissues, oils, and feedstuffs. J Anim Sci 85(6): 1511-1521.
- Ranjan A, Patil C, Moholkar VS (2010) Mechanistic assessment of microalgal lipid extraction. Industrial & Engineering Chemistry Research 49(6): 2979-2985.
- Suslick KS, Didenko Y, Fang MM, Hyeon T, Kolbeck K J, et al. (1999) Acoustic cavitation and its chemical consequences. Philosophical Transactions of the Royal Society of London Series A: Mathematical, Physical and Engineering Sciences 357(1751): 335-353.
- Araujo GS, Matos LJ, Fernandes JO, Cartaxo SJ, Gonçalves LR, et al. (2013) Extraction of lipids from microalgae by ultrasound application: Prospection of the optimal extraction method. Ultrasonics sonochemistry 20(1): 95-98.
- Yang YH, Du L, Hosokawa M, Miyashita K, Kokubun Y, et al. (2017) Fatty acid and lipid class composition of the microalga Phaeodactylum tricornutum. J Oleo Sci 66(4): 363-368.
- Desbois AP, Lebl T, Yan L, Smith VJ (2008) Isolation and structural characterisation of two antibacterial free fatty acids from the marine diatom, Phaeodactylum tricornutum. Appl microbiol Biotechnol 81(4): 755-764.
- Wishart DS, Feunang YD, Marcu A, Guo AC, Liang K, et al. (2018) HMDB 4.0 - The Human Metabolome Database for 2018. Nucleic Acids Res 46(D1): D608-D617.
- Balasubramanian RK, Doan TT Y, Obbard JP (2013) Factors affecting cellular lipid extraction from marine microalgae. Chemical Engineering Journal 215: 929-936.
- Milićević D, Vranić D, Mašić Z, Parunović N, Trbović D, et al. (2014) The role of total fats, saturated/unsaturated fatty acids and cholesterol content in chicken meat as cardiovascular risk factors. Lipids in health dis 13(1): 42.






























