Understanding the Morphogenesis and Pathogenicity of Candida Albicans: A Way to Interfere and Develop New Antifungals Candida Albicans Morphogenesis and Pathogenicity: Understanding to Interfere
Lais Cavalcanti1,2,3, Letícia Saules1,2, Gabriela Saramago1,3, Bruno Leal1,3, Andrea Regina S Baptista2, Vanessa Gremião dos Santos1, Marcos Kalil3, Alba Regina de Magalhães2, Paula Alvarez Abreu4* and Helena C Castro1*
1 Programa de Pós-graduação em Ciências e Biotecnologia, Instituto de Biologia, Universidade Federal Fluminense, Campus do Valonguinho, Niterói, RJ, Brasil
2Laboratório de Micologia Médica e Molecular, Instituto Biomédico, Universidade Federal Fluminense, Niterói, RJ, Brasil
3Laboratório de Microbiologia oral, Faculdade de Odontologia, Universidade Federal Fluminense, Niterói, RJ, Brazil
4Núcleo em Ecologia e Desenvolvimento Sócio-Ambiental de Macaé (NUPEM), Universidade Federal do Rio de Janeiro, Macaé, RJ, Brazil
Submission: June 03, 2019; Published:June 26, 2019
*Corresponding author: Helena Carla Castro, Instituto de Biologia, Universidade Federal Fluminense, Campus do Valonguinho, Outeiro de São João Baptista, s/n, Niterói, RJ, Brazil
*Paula Alvarez Abreu, Núcleo em Ecologia e Desenvolvimento Sócio-Ambiental de Macaé (NUPEM), Universidade Federal do Rio de Janeiro, Av. São José do Barreto, Macaé, RJ, Brazil
How to cite this article:Lais Cavalcanti, Letícia Saules, Paula Alvarez Abreu, Helena C Castro, et al. Understanding the Morphogenesis and Pathogenicity of Candida Albicans: A Way to Interfere and Develop New Antifungals Candida Albicans Morphogenesis and Pathogenicity: Understanding to Interfere. Adv Biotechnol Microbiol. 2019; 14(2): 555885. DOI: 10.19080/AIBM.2019.14.555885
Abstract
Fungi from Candida genus can be found in the human mouth, skin, and gastrointestinal microbiota. These microorganisms are present in more than half of the world population and may act as an opportunistic pathogen. Among this genus, C. albicans is the main causative agent of hospital systemic and bloodstream fungal infections. This species presents pathogenic mechanisms that include biofilm formation that act as barriers against the antifungal drugs. The increased mortality involving Candida-related infections, the higher incidence of opportunistic infections, the emergence of resistant fungi strains and the limited number of antifungal agents, demand the search for new effective antifungals. In that matter, it is important to know the morphology and pathogenicity of C. albicans to design new and better drugs able to penetrate the natural and resistance barriers and act against this pathogen. Therefore, this study aimed to review the literature about morphology, virulence factors, resistance mechanism and clinical features that can be a target for antifungals to help with designing new compounds. Besides that, we described the state of the art of the innovations on antifungals for Candida, through the perspective of the recent patents available in Thomson Reuters Integrity platform which is a very useful database for health science research and drug design.
Keywords: Candida albicans; Morphogenesis; Drug Resistance; Antifungal; Virulence factors
Abbrevations: HIV: Human Immunodeficiency Virus; ROS: Reactive Oxygen Species; PRR: Path Recognition Receptors; CYP51 - sterol 14α-demethylase cytochrome P450
Introduction
Currently about 600 fungal species are pathogenic for humans [1,2]. The genus Candida are composed of a pathogenic fungus of great relevance on the world. The genus Candida is currently composed of about 200 species, but only approximately 10% are pathogenic. Among them, the species of greatest clinical importance are C. parapsilosis, C. dubliniensis, C. tropicalis, C. glabrata, C. krusei, C. guilliermondii, C. lusitaniae, C. auris and C. albicans which is the most prevalent species, isolated in invasive and superficial infections in several anatomical sites [3,4]. Studies report that Candida spp invasive infections have increased over the past two decades. Candidiasis are the high incidence of opportunistic infections mainly in immunocompetent patients with a high mortality rate [5,6].
Candida is part of oral commensal microbiota of healthy individuals [3,7]. But is also, the leading causes of hospital systemic fungal infections and responsible for candidemia (bloodstream infections), mainly affecting immunocompromised patients [6]. Candida albicans is the most prevalent species in hospital, which occupy different anatomical sites [8-10]. Other Candida species also have great clinical importance as they have been related to bloodstream infections (candidemias) and account for about 50% of non-superficial infections (Candida tropicalis, Candida parapsiloisis, Candida glabrata, Candida krusei, Candida dubliniensis, Candida guilliermondii and Candida lusitaniae) [11].
Also, Candida auris is an emerging pathogen with high clinical importance as it is a multidrug resistant species, identified from a Japanese patient for the first time in 2009. Outbreaks of infections caused by C. auris have been reported in several countries such as Kuwait, Pakistan, United Kingdom, Spain and Venezuela. This species can cause ventriculitis, osteomyelitis, intra-abdominal infections, pericarditis, vulvovaginitis, pleural effusion, bloodstream infection among others. C. albicans is commensal in various anatomical sites (e.g. oral cavity, gastrointestinal and urogenital tracts) (Table 1).
The main common manifestation is the vaginal infections and is generally found causing infections in immunocompromised individuals, mainly patients with Human Immunodeficiency Virus (HIV), neutropenic patients undergoing chemotherapy or transplanted and diabetes mellitus patients. Besides the mucocutaneos infections, this microorganism may cause invasive mycosis in internal organs like kidneys, liver, spleen and brain of these patients [8-10]. Candidiasis present different denominations according to the anatomical site (Table 1) [12,13]. In this work we briefly reviewed the literature, addressing different aspects including morphological and pathogenic characteristics of the Candida albicans, as well the related mechanisms of resistance to antifungals, which may be important to the development of new antifungals.
Morphogenesis of Candida albicans and relation with infection and control
Morphology studies show that infections caused by C. albicans generally involve more than one cellular form, in which the filaments (hyphae) are responsible for the penetration of tissues. Interestingly yeast form is important in the early dissemination and less invasive infections. Several virulence factors are present in C. albicans, such as adhesins, transition from the yeast form, to pseudo hyphae or hyphal, biofilm formation and hydrolytic enzymes such as proteases, lipases, and phospholipases, among others [8,14,15].
C. albicans species exhibit polymorphism that varies according to the adaptation to the environment. Pseudo hyphae, chlamydospores and hyphae are important in the invasive process since they are resistant to the action of phagocytes and overcome the epithelial barrier [11]. Yeast cells are well adapted to hematogenic dissemination, allowing them to spread as a systemic infection. Therefore, the polymorphism observed in this species is an important virulence factor [16].
The chemical composition of the cell wall of these fungi is very complex, consisting mainly of polysaccharides, whether or not linked to proteins or lipids, and polyphosphates and inorganic ions forming a matrix. The most frequently components found are Quitins, Glucans, Galactomannans, mannans and proteins, however the quantity of these components varies according to the fungal species. Candida spp cell wall is a complex structure of approximately 100 to 300 nm thick, composed of 5 to 8 distinct layers of carbohydrate (80-90%), besides proteins and lipids. The main constituent of the wall is a highly branched polysaccharide called mannan and this polysacharide is composed of mannose residues linked by alpha-1,6 and alpha-1,2 (alpha-1,3 rarely) and phosphate.
The glucans are important polymers present in Candida, cell wall, being α-glucans and β-glucans its main representatives. β-glucans are highly immunogenic, and present great importance in therapeutic medicine and also in the production of vaccines, while the α-glucan are less immunogenic structures found in greater quantity in the yeast stage of many dimorphic fungi contributing to evade host’s immune system [1]. The mannans present in the fungal wall are polymers, important for the fungal cell wall architecture and also in the degradation of macromolecules (e.g. enzymatic manoproteins). Chitin is another polymer of the fungal cell wall, present in lesser quantity in yeasts. These structures are recognized by the host’s immune system by the Pathern Recognition Receptors (PRR) of dendritic cells, macrophages, among others. It is worth pointing out that large fragments of chitin are not immunogenic, so an intense production of chitin is an evasion mechanism carried out by fungi. Studies show that Candida albicans increases production of chitin once exposed to antifungals [17,18].
Antifungal Agents and Mechanisms of Resistance
Candida albicans, as other fungi, subsequent to antifungal treatment, can undergo microevolution, leading to drug resistance [17]. The knowledge about these adaptations is very important for directing the treatment and to develop new antifungals. The main antifungal agents used for treating candidiasis are the class of polyenes and azoles. However other classes such as echinocandins and nucleosides analogs are also used to treat these infections. Currently there are few options of antifungal agents on the market and in recent years the isolation of strains resistant to these antifungal agents has increased greatly [18]. A microorganism is resistant when it is not inhibited by the prescribed concentration of the antimicrobial in vitro. The primary resistance is observed when a strain is naturally resistant before exposed to the drug whereas the secondary resistance or acquired is when the resistance is caused by acquisition or modification of genetic material, allowing such microorganisms to survive and reproduce even in the presence of the drug [18]. Besides this, the clinical resistance is observed when antifungal therapy failure. It may occur due to other factors besides the fungal resistance (e.g. the immune status of the host, pharmacokinetics of the antifungal agent) [19].
The emergence of strains resistant to the current drugs has increased recently. Currently, the antifungal used in clinical practice have at least one resistant strain report which, is of great concern. It has been emphasized the need to control the proper use of these agents, and the importance of the discovery of new drugs with antifungal action [18,20].
Antifungal Therapy
Polyenes
The family of polyenes is represented by Amphotericin B and nystatin [21,22]. They are macrolides of great importance, with broad spectrum of activity. The polyenes act in superficial and invasive fungal infections, however, they present high toxicity mainly related to kidney cells [23]. Amphotericin B is the main representative of this family, introduced in clinics in 1958, currently it remains as the first choice in the treatment of systemic fungal infections. amphotericin B can also be found as a lipid complex and in liposomal form, that are both formulations developed to reduce the toxicity, and the doses to be administered, thus increasing the efficacy of this drug. The mechanism of action is triggered by one of the components of the cell membrane, ergosterol, leading to the formation of pores (transmembrane channels), causing a disturbance of the influx of ions mainly potassium, leading to cell death.) [21-25]. There are less reports of resistance for the polyenes in comparison to other antifungals; the mechanism is related to ergosterol binding in fungal membranes. Another mechanism is the increase in the catalase production, reducing oxidative damage promoted by amphotericin B [18-20].
Azoles
The drugs belonging to the class of heterocyclic azoles (imidazoles and triazoles) have a broad-spectrum of oral bioavailability and low toxicity. [5,25], The triazoles are more slowly metabolized with less effect on the synthesis of sterols in humans compared to imidazole. The family of imidazoles include ketoconazole, miconazole, econazole, clotrimazole, whereas the triazoles family is composed of fluconazole, itraconazole, voriconazole, posaconazole [5,23].
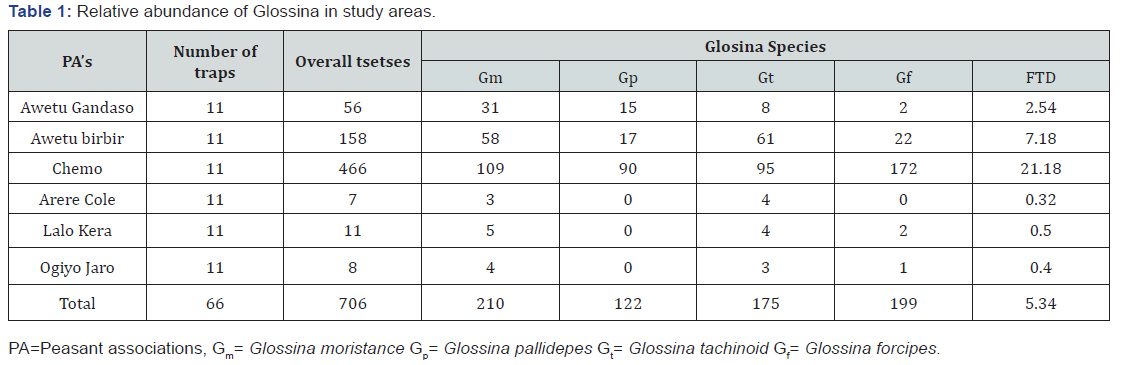
These antifungal agents generally exhibit fungistatic action and are the first choice for the treatment of infections related to Candida spp [5,25]. They act by inhibiting lanosterol 14-α demethylase (CYP51), an enzyme of the ergosterol biosynthesis pathway of, the, this inhibition results in modification of the membrane permeability facilitating the efflux of potassium ions for example [5,26]. The resistance to azoles is mainly related to the mutation in the gene ERG11encoding the target of these drugs. It alters the azoles binding domain. It is noteworthy that there are many reports of strains resistant to azole drugs as these are the first-choice treatment for Candida infections. Another mechanism of resistance is the change in expression of the drug efflux pump, CDR1, CDR2, MDR1 [20,27]. These genes code the ATP Binding Cassette (ABC) carrier family of transmembrane proteins Cdr1 and Cdr2, which act as efflux pumps. Once overexpressed, these genes are associated with the azolic resistance observed in some fungal strains [28] (Figure 1).
Echinocandins
Echinocandins are used as the first-choice treatment of C. glabrata infections due to this specie reduced sensitivity to azoles. The class of echinocandins is composed of semi-synthetic lipopeptides caspofungin, micafungin and anidulafungin. Caspofungin was originally obtained from the species Glarea lozoyensis, micafungin from Coleophoma empetri, and anidulafungin from Aspergillus nidulans. The mechanism of action involves inhibition of the enzyme β-1,3-glucan synthase, which participates in the biosynthesis of fungal cell wall glycans, this enzyme prevents the maintenance of the integrity and rigidity of the cell wall, resulting in a cellular lysis [29-33]. Strains resistant to these drugs were reported and they are described as directly related to FKS genes family (FKS1, FKS2, FKS3). These genes codes proteins involved in the biosynthesis of β-1,3-glucan, so, the mutation of these genes leads to the resistance observed against echinocandins. Mutations in the CDC6 and CDC55 genes are also associated with resistance to these drugs [24,28,34].
Nucleoside Analogues
Flucytosine or 5-fluorocytosine inhibits fungal metabolism by interfering in the RNA and DNA synthesis. This drug is a fluorinated pyrimidine analogue and acts entering the fungal cell by a cytosine permease. Within the cytoplasm 5-fluorocytosine is converted to 5 fluoruridil and competes with uracil in RNA synthesis resulting in a defective RNA and interfering with protein synthesis. High rates of acquired resistance to these drugs were reported [35-37]. Association of fluconazole or itraconazole with flucytosine have been described to be efficacious in Candidiasis, with synergistic effects and reducing the risk of resistance to these drugs. The acquired or innate resistance to flucytosine occurs due to mutations in the genes FCY 1, FCY 2 that codes cytosine deaminase and FUR 1 that codes uracil phosphoribosyl transferase [38].
Virulence factors
Virulence factor are defined as the process or components that participates directly causing infection or damage to the host tissues [39]. When exposed to sub inhibitory concentrations of antifungals, Candida spp promotes the stimulation of virulence factors, such as production of hydrolytic enzymes to improve adherence to tissues and survival. Since fungi are eukaryotic, only limited number of selective targets in relation to humans can be explored for drug design [40,41]. The antifungal drugs actually in the market act by inhibiting fungal growth or killing the fungal cells; in these cases, a selective pressure occurs which leads to the emergence of resistance Targeting virulence factors can be an interesting strategy to avoid resistance and to maintain the microbiome, important for the host. The suppression of C. albicans by antifungals may lead to the growth of other species less susceptible to antifungals (e.g. C. krusei and C. glabrata) [38,42]. Many virulence factors have been described in Candida albicans such as filamentation, biofilm production, adhesins, phospholipases, proteases and toxins and they may be explored as target for the development of new antifungals.
Polymorphism
To survive inside the host, C. albicans needs to adapt to changing environments and different stresses condition. The polymorphism of Candida albicans is an important virulence factor [43,44]. Several environmental stimuli such as pH affect the morphology of C. albicans. The pH less than 6 favors the growth of yeasts, while a pH higher than 7 favors the growth of hyphae. Other conditions, such as physiological temperature, the presence of carbon dioxide, the presence of N- acetylglucosamine or serum also favor the formation of hyphae [16,44,45]. Hyphae formation is important for adhesion to epithelial and endothelial cell, besides the invasion and damage.
The polymorphism of Candida is also regulated by the Quorum Sensing (QSM). This communication mechanism detects high cell densities (>107 cells per ml) stimulating the growth of yeast whereas low cell density (<107 cells por mL), promotes the hyphae growth. Farnesol, tyrosol and dodecanol are the key molecules related to quorum morphogenesis [16], in particular farnesol, as it decreases the viability of macrophages by induction of reactive oxygen species (ROS), protecting the C. albicans from the oxidative stress [1,13].
Candida albicans yeast forms are recognized easily by the host immune system and readily phagocytosed [1,44]. Within the macrophage Candida induces the cAMP-dependent protein kinase A and the mitogen-activated protein kinase pathways responsible for the activation of the main yeast transcription factor, EFG 1p and Cph1 respectively. In this way, the yeast-to-hypha transition occurs within the macrophage, causing the death of these cells, outwitting an important mechanism of defense of the host immunological system [16,17,46,47].
Biofilm formation
According to the literature, biofilms are microbial community structures coated with a matrix material that adhere surfaces through adhesins and extracellular polymers [2, 47,48]. Some Candida spp. have the ability to form biofilm in host tissues and medical devices. This is regulated by chemical signals among cells, the quorum sensing. More than 50 transcriptional regulators have been described related to the C. albicans biofilms production [48- 50]. Studies show that farnesol is the main molecule quorum sensing molecule, it acts as a transcription inhibitor between yeast for hyphae in certain concentrations [3,15,49]. This occurs, due to the Farnesol act keeping the biofilm in the stationary phase, not allowing its maturation because the yeasts do not present good adhesion, so it seems that inhibiting this morphological transition may cause the dispersion of the biofilm. This dispersion can lead to the dissemination and formation of new biofilms [25]. Literature reports that biofilm is associated with increased resistance to antimicrobial. For example, C. albicans biofilm compared to planktonic cells may be 200 times more resistant to antifungal agents [3,25,50-52].
In contrast to other species of the genus, C. albicans presents a basal layer of blastoconidia with a dense matrix of exopolysaccharides and hyphae [52,53]. Yeast are released when biofilms are mature, thereby spreading infection, while hyphae express many adhesins and are therefore most likely responsible for the biofilm integrity [14,50]. Biofilm formation is an important C albicans virulence factor as this mechanism is associated with antifungal resistance. Biofilm acts as a barrier, avoiding the permeation of the antifungal therapeutic agent. Resistance is commonly observed against azole antifungals, which are the first choice in treating candidiasis [3,15,49,54,55].
Adhesins
The adherence of microorganisms to the host tissue is a primary virulence factor, as it allows invasion and subsequent infection. C. albicans presents a set of proteins, commonly known as adhesins that interact with receptors on epithelial cells of the host or abiotic surfaces. The adhesins represent about 6 to 25% of the wall weight, and comprises some manoproteins with molecular mass of 60, 68, 200 and greater than 200 kDa. The cellular wall of Candida yeasts is comprised of about 60-70% of glucan, manoprotein and chitin, which in addition to owning structural property; they begin the process of interaction of microorganism and the environment. These adhesins assist the adherence of the micro-organism to extracellular receptors such as fibronectin, Fibrinogen and laminin, present in the tissues of the host. It is worth pointing out that most of the proteins present on the wall are called glycosylphosphatidylinositol (GPI). These are present on the outside of the wall, also responsible for regulating interactions between the host and the pathogen and are covalently linked to the carbohydrate β-1.6-glucan [16,56].
Current reports show three main families of genes related to the codification of adhesins in C. albicans, including Aglutinine- Like Sequence (ALS), Hyphal Wall Protein (HWP) and hyphal upregulated protein (IFF/HYR) [16,56]. The ALS forms a family of eight members (ALS1-ALS7 and ALS9). The adhesin ALS3 is important in adhesion regulated positively during vaginal or oral infections Hwp1 and Hwp2 are only expressed in filamentous phase helping biofilm formation, cell-cell aggregation, adhesion proteins, and cell surface coupling [56]. The Hwp1 protein has an important role in epithelial attachment and is linked to GPI [16]. The IFF/HYR family consists of twelve genes with high sequence similarity and they are of great clinical importance. The IFF11 gene, plays an important role in organizing and modificating the cell wall. IFF4 protein increases adhesion to epithelial cells and plastic surface while the Hyr1 gene encodes protein that has a relationship with increased resistance to death by neutrophilsmediated immunity [24,56].
Enzymes and Toxins
C. albicans produces enzymes known as proteases (proteinases, peptidases, or proteolytic enzymes) which can degrade IgA present in the saliva, thus reducing the protective effect of these antibodies in the oral mucosa. In addition, protease activity is directly related to degradation, not only of hemoglobin but also albumin, casein, keratin and collagen. Studies show that proteases are related to adhesion, changes in the immune response, tissue damage, hyphal invasion of oral epithelium and epithelial cell apoptosis [16,57,58]. These enzymes are of great importance to the pathogenicity of C. albicans as they facilitate the hydrolysis of the host’s cellular membranes, favoring the adhesion and invasion of the tissue. They can also cause damage to the host’s immune system cells and molecules [16]. The proteinases are classified according to their catalytic mechanism in Serine proteases, cysteines, metalloproteases and aspartil proteases. The proteolytic activity of C. albicans is related to the family of 10 aspartic proteinases (SAP). This family is the best characterized and studied in relation to the expression patterns during the infection by Candida spp. They present variable molecular weight and may be anchored in different locations (SAP 1-8 means extracellular and sap 9-10 are surface proteins) [57,58].
All Candida species of secreted Saps, however, this expression can vary among the species, and the expression of Saps in Candida species with greater pathogenicity potential is greater. The SAPs are related to the degradation of various proteins present in the host`s mucous, for example Vimentin, collagen, keratin, mucin, fibronectin and laminin. The removal of the barrier of the host enable the adhesion, colonization and penetration in the host tissue and dissemination of the Candida spp to the bloodstream. SAPS are related to several physiological functions, according to each gene, such as phenotypic changes (SAPS 1 and 3), dimorphism (SAPS 4 and 6 in hyphae), biofilm production (SAPS 6 and 9), adhesion (SAPS 1-3), interaction with the host´s immunological system (SAPS 2, 4-6), Invasion (SAPS 1-3 during the invasion of the mucosa and SAPS 4-6 during systemic infection), internalization (SAP 1 in the skin) and nutrient acquisition. Exposure to subinhibitory concentrations of antifungal agents promotes the development of resistant strains with an increased expression of SAP genes [16,57,58].
The phospholipase enzymes (PL) and lipase (LIP) play accessory functions, on epithelial invasion and nutrients acquisition by Candida spp. Ten genes have been identified that belongs to the family of lipases (LIP1 - LIP10) and seven phospholipase genes (PLA, PLB1, PLB2, PLC1, PLC2, PLC3 and PLD1). The production of both enzymes are related to facilitating the penetration in the host tissue, and they are concentrated on the hyphae tips [16,57,58]. Also, C. albicans produce proteins called canditoxin that present antibiotic effect to other fungi and bacteria. They act on the cytoplasmic membrane leading to increased permeability, inhibition of amino acids active transport, acidification decrease and potassium ions within the cells, resulting in cell death. The, important in competition situations where C. albicans needs to settle in the canditoxin is a protein present in the cytoplasm of the cell, that too involved in releasing histamine from the mast cells. Nitrosamine also is a carcinogen for the oral tissue cells and is produced by some C. albicans biotypes [16].
Innovations related to antifungals for candida albicans: analysis in the integrity plat form
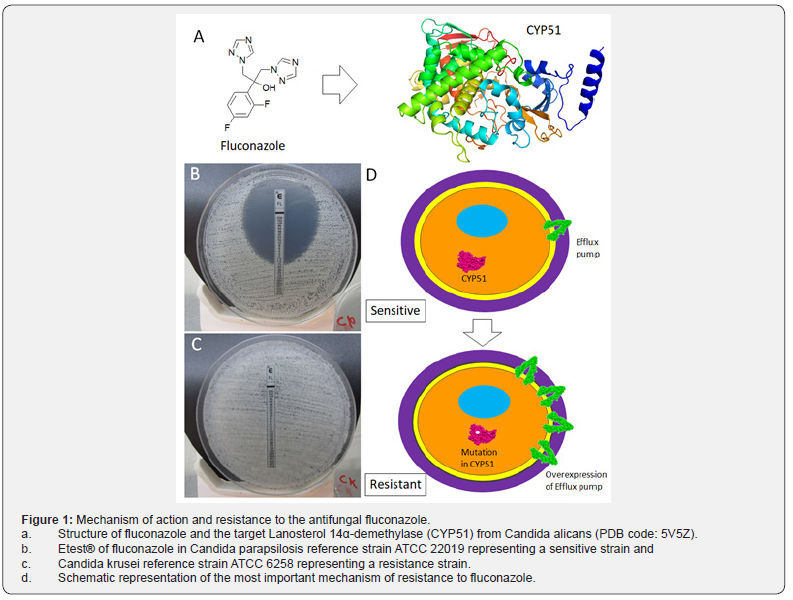
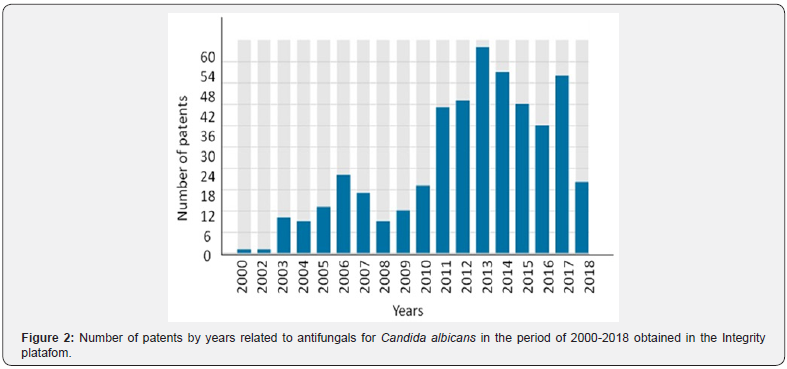
In order to demonstrate the innovations related to Candida albicans, we used the Integrity Clarivate Platform from Thomson Reuters. This platform enable to search for patents related to some theme in a specific period. We used the keywords antifungal and Candida albicans and the search was done in June 2018, regarding the period of 2000 to 2018. We obtained 340 patents, the highest number of patents was in 2013, but it is important to highlight that the search was done in June 2018, and more patents are expected to be deposited in 2018 (Figure 2). China was the country with most patents related to this theme, followed by the United States and Japan. More than fifteen categories were found (Figure 3).
The main subject matter was drug substances (with more than a half of the results), followed by natural products. Both subject matter seems to be directly related to antifungals. Despite the search using the keywords antifungal and “Candida albicans” some of the results may not be related to antifungals for C. albicans (Figure 4). We performed a more detailed analysis specifically in the 2017 year for exemplification and found 35 patents produced (Table 2).
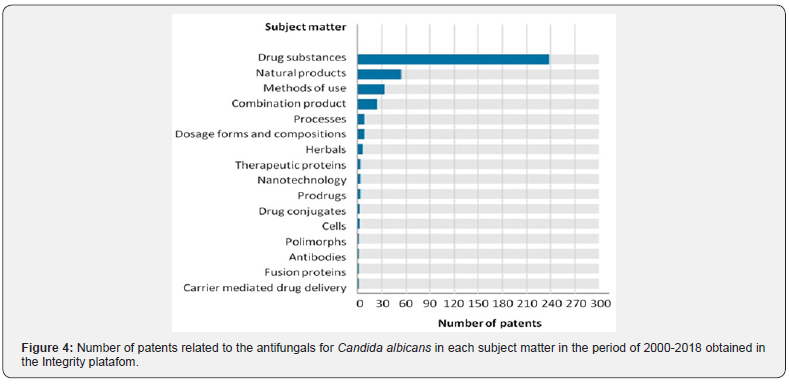
Most of which are research in the category of drug substances, following a tendency indicated from 2000 to 2018. The country with most patents in 2017 was also China, followed by Korea. The evaluation of the title of the patents in 2017 seems that most of them are really related to antifungals, Natural products and synthetic derivatives were found in the text, besides the drugs already in the market such as azoles. In three of the patents the word resistant appears in the title of the production, highlighting the interest of some researchers for the resistant microorganisms. In main patents, the mechanism of action of the product generated was not specified but in most of those with the mechanism described, it was related to Cytochrome P450 Inhibitors, it is interesting to highlight that the azolic class of antifungals are Cytochrome P450 Inhibitors, which suggests that many products are still under development to target these enzymes, besides other mechanisms described were antioxidant, Signal Transduction Modulator, kinase inhibitor, DNA-intercalating drugs, and others [59-65].
Conclusion
Understanding the morphology and pathogenic Candida albicans is essential due to its high prevalence. Considering their virulence factors, the mechanisms of action of the antifungal agents available in the market and resistance mechanism is essential for the development of new drugs with novel mechanism of action. New strategies are being studied as targeting virulence factors and the number of patents related to antifungals for C. albicans has increased in the last years.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be considered as a potential conflict of interest.
Acknowledgement
This study was funded by Coordination for the Improvement of Higher Education Personnel (CAPES) – Finance Code 001 for Postgraduate scholarships and National Council for Scientific and Technological development (CNPq) for Research scholarships.
References
- Cheng SC, Joosten LAB, Kullberg BJ, Netea MG (2012) Interplay between Candida albicans and the mammalian innate host defense. Infect Immun 80 (4): 1304-1313.
- Morales DK, Grahl N, Okegbe C, Dietrich LE, Jacobs NJ, et al. (2013) Control of Candida albicans metabolism and biofilm formation by Pseudomonas aeruginosa phenazines. mBio 4(1): e00526-00512.
- Barros PP, Ribeiro FC, Rossoni RD, Junqueira JC, Jorge AO (2016) Influence of Candida krusei and Candida glabrata on Candida albicans gene expression in in vitro Arch Oral Biol 64: 92-101.
- Jain C, Pastor K, Gonzalez AY, Lorenz MC, Rao RP (2013) The role of Candida albicans AP-1 protein against host derived ROS in in vivo models of infection. Virulence 4(1): 67-76.
- Álvarez-Pérez S, García ME, Cutuli MT, Fermín ML, Daza MÁ, et al. (2016) Acquired multi-azole resistance in Candida tropicalis during persistent urinary tract infection in a dog. Med. Mycol. Case Rep 11: 9-12.
- Pérez JC, Kumamoto CA, Johnson AD (2013) Candida albicans commensalism and pathogenicity are intertwined traits directed by a tightly knit transcriptional regulatory circuit. PLoS Biol 11(3): e1001510.
- Cho T, Nagao J, Imayoshi R, Tanaka Y (2014) Importance of Diversity in the Oral Microbiota including Candida Species Revealed by High-Throughput Technologies. Int J Dent 2014: ID 454391.
- Brown AJP, Budge S, Kaloriti D, Tillmann A, Jacobsen MD, et al. (2014) Stress adaptation in a pathogenic fungus. J Exp Biol 217(pt 1): 144-155.
- Jha A, Kumar A (2016) Development and targeting of transcriptional regulatory network controlling FLU1 activation in Candida albicans for novel antifungals. J Mol Graph Model 69: 1-7.
- Theill L, Dudiuk C, Morano S, Gamarra S, Nardin ME, et al. (2016) Prevalence and antifungal susceptibility of Candida albicans and its related species Candida dubliniensis and Candida africana isolated from vulvovaginal samples in a hospital of Argentina. Rev Argent Microbiol 48(1): 43-49.
- Chen YL, Yu SJ, Huang HY, Chang YL, Lehman VN, et al. (2014) Calcineurin Controls Hyphal Growth, Virulence, and Drug Tolerance of Candida tropicalis. Eukaryot Cell 13(7): 844-854.
- Tong Y, Tang J (2017) Candida albicans infection and intestinal immunity. Microbiol Res 198: 27-35.
- Mucci MJ, Cuestas ML, Landanburu MF, Mujica MT (2017) Prevalence of Candida albicans, Candida dubliniensis and Candida africana in pregnant women suffering from vulvovaginal candidiasis in Argentina. Rev Iberoam Micol 34(2): 72-76.
- Fox EP, Bui CK, Nett JE, Hartooni N, Mui MC, et al. (2015) An expanded regulatory network temporally controls Candida albicans biofilm formation. Mol Microbiol 96(6): 1226-39.
- Tan Y, Leonhard M, Ma S, Schneider Stickler B (2016) Influence of culture conditions for clinically isolated non-albicans Candida biofilm J Microbiol Methods 130: 123-128.
- Mayer FL, Wilson D, Hube B (2013) Candida albicans pathogenicity mechanisms. Virulence 4(2): 119-128.
- Wartenberg A, Linde J, Martin R, Schreiner M, Horn F, et al. (2014) Microevolution of Candida albicans in Macrophages Restores Filamentation in a Nonfilamentous Mutant. PLoS Genet 10(12): e1004824.
- Africa CWJ, Abrantes PM (2016) Candida antifungal drug resistance in sub-Saharan African populations: A systematic review. Version F1000Res 5: 2832.
- Mayers DL, Lerner SA, Ouellette M, Sobel JD (2009) Antimicrobial Drug Resistance: Mechanisms of Drug Resistance, In: O Shaughnessy EM, Lyman CA, Walsh TJ(Eds) Amphotericin B: Polyene Resistance Mechanisms. Humana Press, New York, USA, (1): pp. 295-305.
- Campoy S, Adrio JL (2017) Antifungals. Biochem Pharmacol 133: 86-96.
- Alvarado Socarras J, Rojas Torres JP, Vargas Soler JA, Guerrero C (2016) Kodamaea ohmeri infection in a newborn with a mediastinal mass. Arch Argent Pediatr 114(5): e319-322.
- Magalhães YC, Bomfim MR, Melônio LC, Ribeiro PC, Cosme LM, et al. (2015) Clinical significance of the isolation of Candida species from hospitalized patients. Braz J Microbiol 46(1): 117-123.
- Vandeputte P, Ferrari S, Coste AT, Blanco JL (2011) Antifungal Resistance and New Strategies to Control Fungal Infections. Int J Microbiol ID713687: 1-26.
- Pfaller MA (2012) Antifungal drug resistance: mechanisms, epidemiology, and consequences for treatment. Am J Med 125(1): S3-13.
- Husain A, Drabu S, Kumar N, Alam MM, Bawa S (2013) Synthesis and biological evaluation of di- and tri-substituted imidazoles as safer anti-inflammatory-antifungal agents. J Pharm Bioallied Sci 5(2): 154-161.
- Eddouzi J, Parker JE, Vale-Silva LA, Coste A, Ischer F, et al. (2013) Molecular mechanisms of drug resistance in clinical Candida species isolated from Tunisian hospitals. Antimicrob Agents Chemother 57(7): 3182-3193.
- Salari S, Khosravi AR, Mousavi SA, Nikbakht-Brojeni GH, (2016) Mechanisms of resistance to fluconazole in Candida albicans clinical isolates from Iranian HIV-infected patients with oropharyngeal candidiasis. J Mycol Med 26(1): 35-41.
- de Oliveira Santos GC, Vasconcelos CC, Lopes AJO, de Sousa Cartágenes MDS, Filho AKDB, et al. (2018) Candida Infections and Therapeutic Strategies: Mechanisms of Action for Traditional and Alternative Agents. Front Microbiol 9: 1351.
- Azanza JR, Montejo M (2008) Farmacocinética y farmacodinamia. Interacciones y efectos secundarios. Comparación con otras equinocandinas. Enfermedades Infecc Microbiol Clínica 26: 14-20.
- Azanza Perea JR (2016) Equinocandinas: aspectos aplicados de la farmacologí Rev Iberoam Micol 33: 140-144.
- Shen YC, Liang CY, Wang CY, Lin KH, Hsu MY, et al. (2014) Pharmacokinetics and safety of intravitreal caspofungin. Antimicrob Agents Chemother 58(12): 7234-7239.
- Singh-Babak SD, Babak T, Diezmann S, Hill JA, Xie JL, et al. (2012) Global analysis of the evolution and mechanism of echinocandin resistance in Candida glabrata. PLoS Pathog 8(5): e1002718.
- Gil Alonso S, Jauregizar N, Eraso E, Quindós G (2016) Postantifungal effect of caspofungin against the Candida albicans and Candida parapsilosis clades. Diagn Microbiol Infect Dis 86(2): 172-177.
- Biswas C, Chen SC, Halliday C, Kennedy K, Playford EG, et al. Identification of genetic markers of resistance to echinocandins, azoles and 5-fluorocytosine in Candida glabrata by next generation sequencing: a feasibility study. Clin Microbiol Infect 23(9): 676.e7-676 e10.
- Wani MY, Ahmad A, Kumar S, Sobral AJFN (2017) Flucytosine analogues obtained through Biginelli reaction as efficient combinative antifungal agents. Microb Pathog 105: 57-62.
- Morace G, Perdoni F, Borghi E (2014) Antifungal drug resistance in Candida species. J Glob Antimicrob Resist 2(4): 254-259.
- Barbaro G, Barbarini G, Di Lorenzo G (1996) Fluconazole vs itraconazole-flucytosine association in the treatment of esophageal candidiasis in AIDS patients. A double-blind, multicenter placebo-controlled study. The Candida Esophagitis Multicenter Italian Study (CEMIS) Group Chest 110(6): 1507-1514.
- Casadevall A, Pirofski LA (2003) The Damage-Response Framework of Microbial Pathogenesis. Nat Rev Microbiol 1(1): 17-24.
- Ostrosky Zeichner L, Casadevall A, Galgiani JN, Odds FC, Rex JH (2010) An Insight into the Antifungal Pipeline: Selected New Molecules and Beyond. Nat Rev Drug Discov 9(9): 719-727.
- Fekete Forgacs K, Gyure L, Lenkey B(2000) Changes of virulence factors accompanying the phenomenon of induced fluconazole resistance in Candida albicans. Mycoses 43(7-8): 273-279.
- Mores AU, Souza RD, Cavalca L, de Paula e Carvalho A, Gursky LC, et al. (2009) Enhancement of secretory aspartyl protease production in biofilms of Candida albicans exposed to sub-inhibitory concentrations of fluconazole. Mycoses 54(3): 195-201.
- Vila T, Romo JA, Pierce CG, McHardy SF, Saville SP, et al. (2017) Targeting Candida albicans filamentation for antifungal drug development. Virulence 8(2): 150-158.
- Chauvel M, Nesseir A, Cabral V, Znaidi S, Goyard S, et al. (2012) A versatile overexpression strategy in the pathogenic yeast Candida albicans: identification of regulators of morphogenesis and fitness. PloS One 7(9): e45912.
- Whibley N, Gaffen SL (2015) Beyond Candida albicans: Mechanisms of immunity to non-albicans Candida species. Cytokine 76(1): 42-52.
- Grumaz C, Lorenz S, Stevens P, Lindemann E, Schöck U, et al. (2013) Species and condition specific adaptation of the transcriptional landscapes in Candida albicans and Candida dubliniensis. BMC Genomics 14: 212.
- Lo HJ, Kohler JR, DiDomenico B, Loebenberg D, Cacciapuoti A, et al. (1997) Nonfilamentous albicans mutants are avirulent. Cell 90(5): 939-949.
- Stoldt VR, Sonneborn A, Leuker CE, Ernst JF (1997) Efg1p, an essential regulator of morphogenesis of the human pathogen Candida albicans, is a member of a conserved class of bHLH proteins regulating morphogenetic processes in fungi. Embo J 16(8): 1982-1991.
- Nobile CJ, Johnson AD (2015) Candida albicans Biofilms and Human Disease. Annu Rev Microbiol 69: 71-92.
- Guembe M, Cruces R, Peláez T, Muñoz P, Bouza E (2017) Assessment of biofilm production in Candida isolates according to species and origin of infection. Enferm Infecc Microbiol Clínica 35(1): 37-40.
- Lohse MB, Gulati M, Johnson AD, Nobile CJ (2018) Development and regulation of single- and multi-species Candida albicans Nat Rev Microbiol 16(1): 19-31.
- Sapaar B, Nur A, Hirota K, Yumoto H, Murakami K, et al. (2014) Effects of extracellular DNA from Candida albicans and pneumonia-related pathogens on Candida biofilm formation and hyphal transformation. J Appl Microbiol 116(6): 1531-1542.
- Shinde RB, Chauhan NM, Raut JS, Karuppayil SM (2012) Sensitization of Candida albicans biofilms to various antifungal drugs by cyclosporine A. Ann Clin Microbiol Antimicrob11: 27.
- Mathé L, Van Dijck P (2013) Recent insights into Candida albicans biofilm resistance mechanisms. Curr Genet 59(4):
- Desai JV, Bruno VM, Ganguly S, Stamper RJ, Mitchell KF, et al. (2013) Regulatory role of glycerol in Candida albicans biofilm formation. M Bio 4(2): e00637-00612.
- Tsang PWK, Bandara H MHN, Fong WP (2012) Purpurin suppresses Candida albicans biofilm formation and hyphal development. PloS One 7(11): e50866.
- de Groot PWJ, Bader O, de Boer AD, Weig M, Chauhan N (2013) Adhesins in human fungal pathogens: glue with plenty of stick. Eukaryot Cell 12(4): 470-481.
- Pammi M, Holland L, Butler G, Gacser A, Bliss JM (2013) Candida parapsilosis is a Significant Neonatal Pathogen: A Systematic Review and Meta-Analysis. Pediatr Infect Dis J 32(5): e206-e216.
- Wiebusch L, Lonchiati D, Rodrigues L, Rodrigues A, Almeida A, et al. (2014) Production of virulence factors by species of Candida albicans isolated from urine culture. BMC Proc 8: 19.
- Williams DW, Jordan RPC, Wei X-Q, Alves CT, Wise MP, et al. (2013) Interactions of Candida albicans with host epithelial surfaces. J Oral Microbiol 5.
- Mattede M das GS, Piras C, Mattede KDS, Ferrari AT, Baldotto LS, et al. (2015) Urinary tract infections due to Trichosporon in severely ill patients in an intensive care unit. Rev Bras Ter Intensiva 27(3): 247-251.
- Misme-Aucouturier B, Albassier M, Alvarez-Rueda N, Le Pape P (2016) Specific Human and Candida Cellular Interactions Lead to Controlled or Persistent Infection Outcomes during Granuloma-Like Formation. Infect Immun 85(1): e00807-816.
- Campois TG, Zucoloto AZ, Araujo EJ de A, Svidizinski TIE, Almeida RS, et al. (2015) Immunological and histopathological characterization of cutaneous candidiasis. J Med Microbiol 64(8): 810-817.
- Pappas PG, Kauffman CA, Andes DR, Clancy CJ, Marr KA, et al. (2016) Clinical Practice Guideline for the Management of Candidiasis: 2016 Update by the Infectious Diseases Society of America. Clin Infect Dis 62(4): e1-50.
- Bianchi CMP De C, Bianchi HA, Tadano T, De Paula CR, Hoffmann-Santos HD, et al. (2016) Factors related to oral candidiasis in elderly users and non-users of removable dental prostheses. Rev Inst Med Trop São Paulo 58: 17.
- Mucci MJ, Cuestas ML, Landanburu MF, Mujica MT (2017) Prevalence of Candida albicans, Candida dubliniensis and Candida africana in pregnant women suffering from vulvovaginal candidiasis in Argentina. Rev Iberoam Micol 34(2): 72-76.






























