Positive Association of Mutations in Genes Related Vascular Endothelial Dysfunction in Relation to High Altitude Pulmonary Edema (HAPE): A Case Control Study
Swati Srivastava1*, Shuchi Bhagi2 and Rajinder Kumar Gupta1
1Department of Genomics, Defence Institute of Physiology & Allied Sciences (DIPAS), India
2Department of Medicine, Institute of Nuclear Medicine and Allied Sciences (INMAS), India
Submission: January 08, 2019; Published: April 01, 2019
*Corresponding author: Swati Srivastava, Department of Genomics, Defence Institute of Physiology & Allied Science, Timarpur, Delhi, India
How to cite this article: Swati S, Shuchi B, Rajinder KG. Positive Association of Mutations in Genes Related Vascular Endothelial Dysfunction in Relation to High Altitude Pulmonary Edema (HAPE): A Case Control Study. Adv Biotech & Micro. 2019; 13(4): 555870. DOI: 10.19080/AIBM.2019.13.555870
Abstract
Introduction: High Altitude Pulmonary Edema (HAPE) is a potentially fatal condition, occurring at altitudes greater than 3000m and effecting rapidly ascending, non-acclimatized healthy individuals. It is characterized by excessive pulmonary vasoconstriction and is associated with decreased concentrations of nitric oxide (NO) in the lungs. Hypoxic conditions at high altitude (HA) markedly impairs vascular endothelial function in the systemic circulation in HAPE sensitive subjects due to a decreased bioavailability of NO. Impairment of the NO pathway could contribute to the enhanced hypoxic pulmonary vasoconstriction that is central to the pathogenesis of HAPE. Considering the pivotal role of endothelium in maintaining homeostasis in the body, we thought it would be interesting to study the common mutations in various genes related to endothelial functioning such as Angiotensin Converting Enzyme (ACE), vascular endothelial growth factor (VEGF), endothelin-1 (EDN) and endothelial nitric oxide synthase (eNOS) gene; that could be playing a significant role in pathology of HAPE.
Methodology: Peripheral blood was collected from two groups of volunteers (n~=40 in each group): HAPE susceptible (HAPE-s) with history of at least one episode of HAPE in past and controls (rCON), with no past history of HAPE. Six mutations including insertion/deletion (I/D) polymorphism in ACE (rs1799752), C(936)T and C(-634)G single nucleotide polymorphisms (SNPs) in VEGF, G(8002)A and 3A/4A mutations in EDN and Glu(298)Asp in eNOS gene were selected based on their functional relevance in the published literature. Specific regions were amplified and genotyping was done using polymerase chain reaction restriction fragment length polymorphism (PCR-RFLP). The results were analysed statistically to assess their association with HAPE.
Results: Genotypic and allelic frequencies for each mutation were calculated amongst the two groups. Prism 5.0 was used to assess χ2 differences and also Fisher’s exact test for allelic frequency variation. Odds ratio (OR) (with 95% confidence interval) was used to compare the difference in of genotype between HAPE-s and rCON. For all tests, a p value of ≤ 0.05 was considered significant. Amongst the six polymorphisms studied, ACE (I/D), EDN G(8002)A and eNOS Glu(298)Asp showed a significant association with HAPE. The other three polymorphisms did not show statistically significant difference between the two groups under study.
Conclusion: In conclusion, our results indicate the possible role of ACE I/D, EDN G(8002)A and eNOS Glu(298)Asp mutations with HAPE susceptibility in Indian population. Our preliminary finding needs to be further substantiated by genotyping larger number of samples.
Keywords: Pathophysiology; Diagnosis; Endothelium; Cardiovascular
Abbrevations: HAPE: High-Altitude Pulmonary Edema; ACE: Angiotensin Converting Enzyme; PVR: Pulmonary Vascular Resistance; PAP: Pulmonary Artery Pressure; VEGF: Vascular Endothelial Growth Factor
Introduction
High-Altitude Pulmonary Edema (HAPE) is a severe form of altitude illness and a life-threatening condition that occurs in healthy persons ascending to altitudes in excess of 2500 meters above sea level [1,2]. Over several decades of research, the exact genetic mechanism underlying the development of HAPE remains unclear. An exaggerated pulmonary hypertension has been suggested to play a crucial role in pathogenesis of HAPE [1-5]. It is a multifactorial complex disease and is typically characterised by uneven vasoconstriction and over-perfusion which causes stress failure of pulmonary capillaries leading to alveolar flooding. HAPE pathophysiology involves both environmental and genetic risk factors and it depends on multiple factors including altitude attained, initial diagnosis and treatment and access to medical care [6,7].
Endothelial cells play a pivotal role in regulating blood flow and maintaining haemostatic balance as they provide surface for interactions of several coagulant and anticoagulant molecules [8]. Dysfunction of endothelium is characterized by altered vasodilation and a pro-inflammatory state. Previous studies report that improper functioning of vascular endothelium plays a central role in pathogenesis of vast spectrum of human diseases like cardiovascular diseases, chronic heart failure, peripheral vascular disease, diabetes, kidney failure and severe viral infections etc. [9].
Since uneven pulmonary vasoconstriction appears to play an important role in the development of HAPE, the genes involved in maintaining pulmonary vascular tone such as Angiotensin Converting Enzyme (ACE) and endothelin-1 (EDN-1) could be possible candidates for HAPE (Figure 1). EDN1, a potential vasoconstrictor, stimulates nitric oxide (NO) release, which is an essential endogenous vasodilator, regulates pulmonary vascular tone and maintains physiological low pulmonary vascular resistance (PVR) [10].
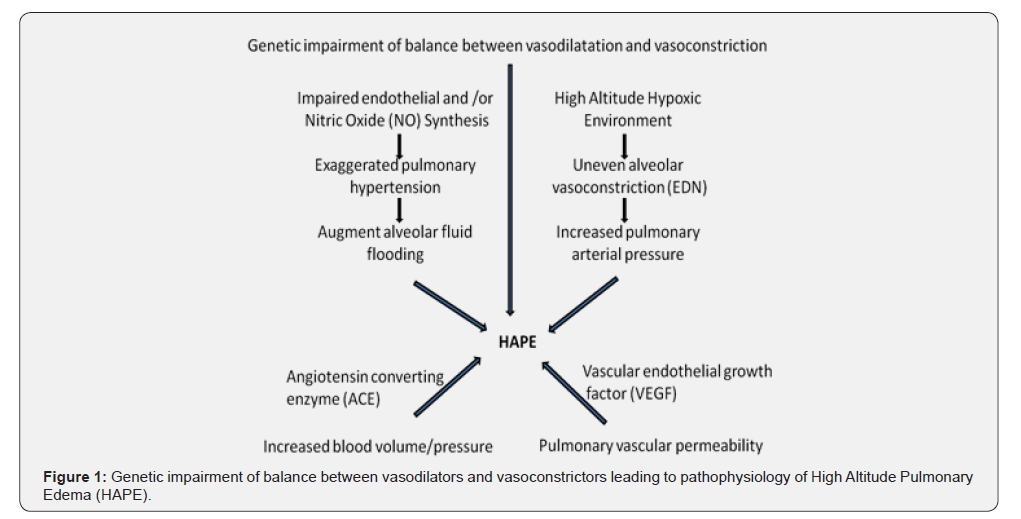
It is synthesized locally by endothelial nitric oxide synthase (eNOS or NOS3) in vascular endothelial cells, which is a key catalytic enzyme responsible for providing basal pulmonary NO release [11]. Previous studies have reported that the exhaled NO from the respiratory tract of HAPE-susceptible subjects is lower than that from controls both at altitude as well as sea level conditions under hypoxic breathing and it is in inverse relationship with pulmonary artery pressure (PAP) [12,13]. Moreover, Glu(298)Asp polymorphism in eNOS gene, causing amino acid substitution of glutamic acid (Glu) by aspartic acid (Asp) at the 298th position in exon 7, has been positively associated with essential hypertension [14,15].
Another gene apelin, plays a significant role in inducing endothelium induced vasodilation by activating endothelial nitric oxide synthase [16]. It plays a crucial role in physiological response of vascular endothelium and smooth muscles during hypoxia. Mishra and co-workers recently reported that amongst several variants of apelin, rs3761581G and rs2235312T have been associated with low levels of apelin in individuals who suffered from HAPE at HA [17]. Also, reduced levels of serum apelin have been reported in patients of Pulmonary Hypertension (PH) [18].
The gene encoding Vascular Endothelial Growth Factor (VEGF) is considered as endothelial cell-specific mitogen, an angiogenic inducer and a mediator of vascular permeability [19], and its inhibition has been correlated with suppression of tumour growth and angiogenesis [20-22]. Functionally active polymorphisms within VEGF gene influence production of VEGF protein and are reportedly involved in susceptibility to various disorders wherein angiogenesis is a critical factor [23,24]. VEGF also increases the capillary permeability and stimulate vascular endothelial cell proliferation promoting angiogenesis. CC genotype of C(936)T polymorphism in the 3’-untranslated region (3-UTR) of the VEGF gene has been associated with an increased serum VEGF level as compared with the CT and TT genotypes [25].
Gupta and co-workers demonstrated that HAPE susceptible (HAPE-s) individuals show exaggerated hypoxic response and baseline hypoxia compared to controls [26,27]. They further showed that HAPE-s group of volunteers have increased PH and vascular remodelling making them hypoxia sensitive and could demonstrate exaggerated PVR to hypoxia in them. We hypothesize that HAPE-s individuals may have some constituted abnormality which make them hypoxia sensitive.
In the present study, we selected genes involved in maintaining pulmonary vascular tone such as eNOS and VEGF and in the same cohort we studied ACE and EDN-1 gene polymorphisms, whose abnormal function can lead to PH and may contribute to HAPE susceptibility. The subjects included in the study were truly HAPE susceptible, as they suffered from HAPE despite of following acclimatization schedule at high altitude (HA) [28]. Thus we aimed to identify endothelial risk factors predisposing individuals to HAPE, by studying common genetic mutations in ACE, VEGF, EDN1 and eNOS genes.
Materials and Methods
Study design
The study protocol was approved by institutional ethical committee and a written consent was obtained from the all participating volunteers. The subjects recruited for study were age and body weight matched unrelated males volunteers. Study population consisted of Army troops, healthy non-mountaineers, who were categorized based on their resistance and susceptibility to HAPE. Both the groups were air inducted from sea level at an altitude of 3500m and followed acclimatization schedule of six days [28]. The sample collection was retrospective in nature i.e., the volunteers had history of HAPE and were clinically diagnosed with the same upon acute induction to HA (HAPE-s).
HAPE-s group was compared to equal number of controls (rCON), those who remained healthy after exposure to same altitude in similar conditions. At plains, they were all healthy and had normal BMI, heart rate and blood pressure (BP) (systolic and diastolic), with no significant difference between controls and patients group. Blood samples were collected in K2 EDTA vacutainers with Na-citrate as anticoagulant (Becton and Dickinson, NJ, USA); for subjects having established history of HAPE upon rapid induction to Lehi, Ladakh (Himalayan region of India), situated at an altitude of 3524m (11,562ft) and those who remained healthy after exposure to HA conditions upon rapid induction at the same altitude and similar duration. Blood was stored at -20 °C till the time of processing in the lab situated at sea level.
Isolation of DNA and genotyping
For genetic analysis whole blood samples were collected in EDTA. High molecular weight DNA was extracted from peripheral blood by QIAamp DNA isolation kit (Qiagen Midi kit, Germany), using manufacturer’s protocol. Quantitative check of genomic DNA was done using DNA/RNA nanodrop (Thermo Fischer, USA). For qualitative analysis, samples (100ng/μl) were loaded on 0.7% agarose gel containing ethidium bromide and run for ~20min and visualized under UV. Gel images were visualized under UV (Fusion Fx5, Viler Lour mat, France) and concentration was checked using Nanodrop 2000 spectrophotometer (Thermo Fisher Scientific, USA).
The present study evaluated six genotypic polymorphisms in four candidate genes involved in angiogenesis and endothelial functioning in view of their probable functional relevance in development of HAPE. Specific gene regions were amplified using primer sequences as detailed in (Table 1) and were subsequently subjected to restriction digestion (Figure 2). A negative control containing no genomic DNA and a positive control with a known genotype was always included in all experiments.
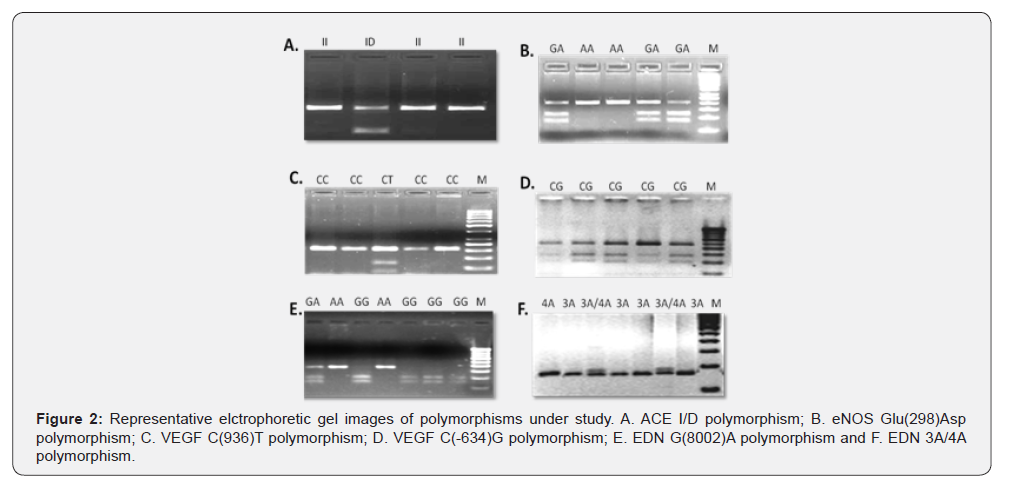
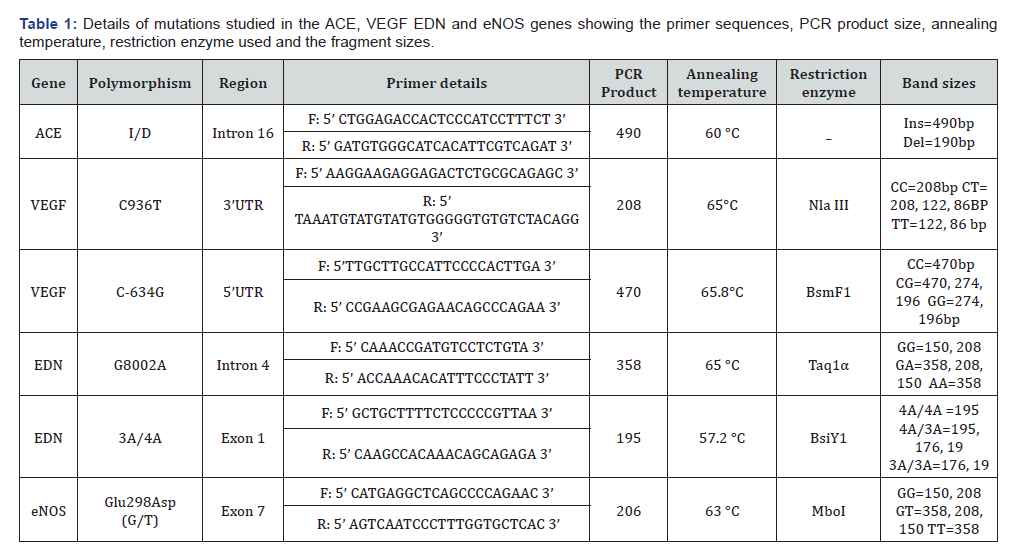
The digested PCR products were resolved on 2-3% agarose gels stained with ethidium bromide. Gel images were visualized under UV ((Fusion Fx5). (Table 1) shows the primers used for RFLP method, PCR cycling conditions, restriction endonucleases and the digested restriction fragment size products. An independent observer read and confirmed all the genotypes, discrepancies, if any, were resolved by repeated PCR-RFLP. Allelic sizes were determined by comparison of bands with molecularweight markers. Specific alleles were determined by observing the presence or absence of digested bands.
Statistical analysis
Genotypic and allelic frequencies were computed for each locus by gene counting and comparing by 3X2 and 2X2 contingency table respectively and their distribution in HAPE and rCON group was analysed. These frequencies were also tested for expected and observed genotype difference on Hardy Weinberg’s equilibrium (HWE; 1 degree of freedom) (www.tufts.edu/~mcourt01/ Court%20lab%20-20 HW %20 calculator.xls) (Table 2). Chi square (χ2) test for genotypic significance (degrees of freedom=2) and Fisher’s exact test for allelic significance along with odds ratio to the level of 95% confidence interval (95%) was calculated using Prism 5.0 (GraphPad, USA) (Table 3). Also, genotypic distribution for all six mutations was statistically tested for dominant and recessive models (Table 4). For all tests a p value of ≤ 0.05 was considered significant. Percentage heterozygosity i.e, difference between expected and observed heterozygosity was measured using analysis for ascertainment bias for dominant/recessive models (www.oege.org/software/hwe-mr-calc.shtml) (Figure 3). This calculator indicated the expected counts under HWE if gains or losses have occurred in one genotype group in addition to the conventional analysis which distributes gains and losses across all three genotype groups. To describe the relationship between haplotypes and disease status, haplotype frequency estimation and association of these haplotypes with disease, was carried out for two mutations of EDN gene (i.e., G(8002)A and 3A/4A) and VEGF gene (i.e., C(936)T and C(-634)G) (Table 5).
The haploid frequencies with significant difference in their distribution between HAPE and Control group are marked in bold. *represents the C allele of C-634G SNP.
Results
Case control characteristics
The study cohort comprised of all unrelated male subjects. They were healthy individuals serving Indian Army. The major inclusion criteria for cases was that the subjects had a history of HAPE upon HA rapid induction. Since the present study is retrospective and the sample collection was done at plains, physiological parameters did not show any statistically significant difference between their heart rate, blood pressure and haemoglobin.
Single locus analysis identified polymorphisms associated with HAPE
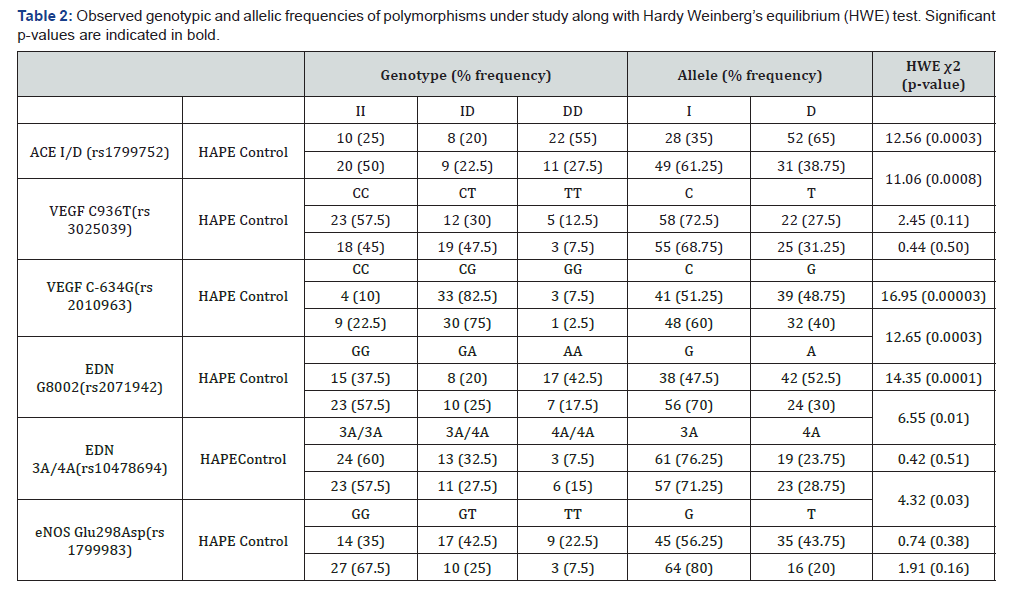
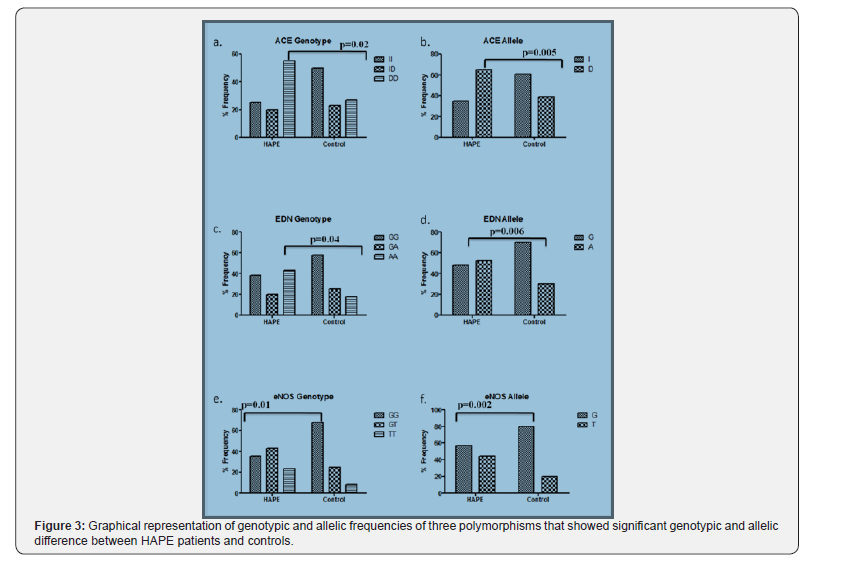
In the present investigation, three of the six polymorphisms showed a statistically significant difference between the HPAE-s and rCON. ACE I/D polymorphism was significantly associated with HAPE and showed a higher percentage of DD genotype in HAPE (55%) compared controls (27.5%) (Table 2) (χ2=7.05, p=0.02) (Table 3). Also, at the allelic level, percentage occurrence of I allele was only 35% in HAPE-s compared to 61.25% in rCON whereas % occurrence of D allele was as high as 65% in HAPE-s compared to 38.75% (Table 2) in rCON (p=0.005, OR=0.34, CI=0.17-0.64) (Table 3, Figure 3).
Also, G8002A polymorphism of EDN gene showed a significant association with HAPE susceptibility both at genotypic and allelic level. A higher percentage of GG genotype was observed in rCON (57.5%) compared to HAPE-s (37.5%). Moreover, the frequency of mutants i.e., AA genotype was lower in rCON (17.5%) compared to HAPE-s (42.5%) (Table 2) (χ2=6.07, p=0.04) (Table 3). Statistically significant difference was also evident at the allelic level (p=0.06, OR=0.38, CI=0.20-0.74) (Table 3), wherein the percentage occurrence of both G and A alleles were comparable in HAPE-s group (G=47.5% and A=52.5%), however a vast difference occurred in rCON group (G=70% and A=30%) (Table 2, Figure 3).
The third SNP that showed significant association with HAPE was Glu298Asp in eNOS gene. The wild type GG genotype appeared in statistically higher percentages in rCON (67.5%) compared to HAPE-s (35%) (Table 2). The graphical representation of these polymorphisms with significant genotypic and allelic differences is shown in Figure 3. Also, the mutant genotype, i.e., TT showed higher percentage occurrence in HAPE-s (22.5%) compared to controls (7.5%). Thus, the genotypic frequency distribution amongst the two groups was significant (χ2=8.93, p=0.01) (Table 3). Also, since G allele was predominating in rCON (80%) compared to HAPE-s (56.25%) and T allele was predominating in HAPE-s (43.75%) compared to rCON (20%) (Table 2, Figure 3), the allelic difference was also significant (p=0.002, OR=0.32, CI=0.15-0.64) (Table 3). The two polymorphisms studied in VEGF gene i.e., C(936)T and C(-634)G and 3A/4A polymorphism in EDN- 1 gene did not show any statistical significance both at genotypic and allelic level between the two groups under study.
Dominant and Recessive model testing
Since each genotype is comprised of two alleles, the risks associated with each genotype depend upon the number of variant allele copies carried. For studying the dominant model, it was assumed that heterozygous and mutant homozygous genotypes have similar risk and hence they were compared in combination with the homozygous wild type. This model yielded a significant difference in ACE I/D polymorphism (II vs. DD+ID) with χ2=4.32, p=0.03, OR=0.33 (CI=0.12-0.85); and eNOS Glu(298)Asp polymorphism (GG vs. GT+TT) with χ2=7.2, p=0.007, OR=0.25 (CI=0.10- 0.65) (Table 4).
For recessive model study, it was assumed that both wild type homozygous and heterozygous genotypes produce same effect and to modify the gene product, two copies of risk allele (homozygous mutant) are essential. Recessive model yielded a significant difference in case of ACE (I/D) polymorphism (II+ID vs. DD) with χ2=5.15, p=0.02, OR=0.31 (CI=0.12-0.78) and EDN- 1 G(8002)A polymorphism with χ2=4.82, p=0.02, OR=0.28 (CI=0.10-0.80) (Table 4).
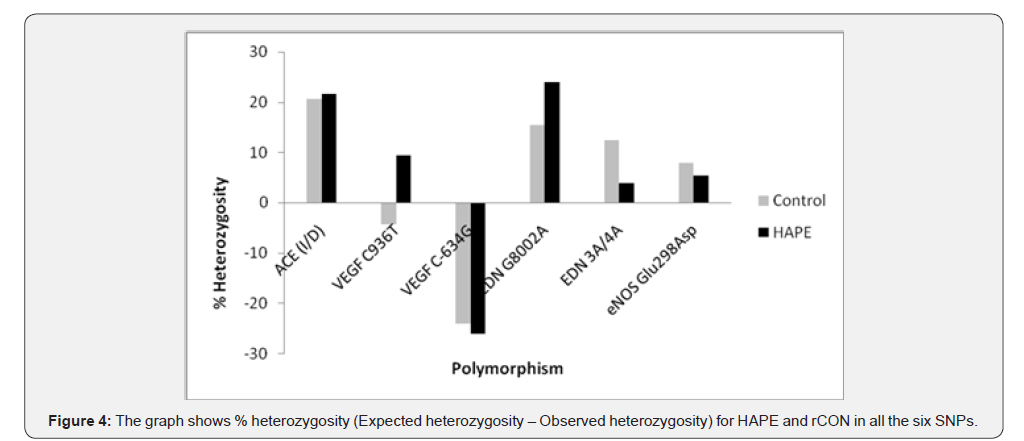
Analysis of Heterozygosity
In case of VEGF C(-634)G SNP, the observed heterozygosity was much higher than expected, both in HAPE-s and rCON. This might be due to predominance of CC genotype in the study population. Percentage heterozygosity for ACE I/D and eNOS Glu(298)Asp polymorphism were comparable between both the groups under study. However, striking differences were observed between the two SNPs in EDN gene, wherein observed heterozygosity was much higher in case of controls compared to HAPE-s group in G(8002)A polymorphism whereas observed heterozygosity was much lower in controls compared to HAPE-s group in case of 3A/4A polymorphism (Figure 4). These differences could be due to ascertainment bias in biological sample which may in turn lead to gains or losses in observed counts.
Haplotype analysis
Frequency occurrence of A-3A haplotype of EDN gene in HAPE-s was significantly higher compared to rCON (Table 5). The other haplotypes in EDN and VEGF genes did not show a significant difference between the two groups under study.
Discussion
The occurrence of HAPE in our Indian subcontinent is a common phenomenon because of global attraction to the Himalayas for tourism, induction by Army volunteers as duty and the poor awareness of the knowledge about the predisposing factors that can lead to HA related amongst un-acclimated visitors. There is a fine balance maintained between vasoconstrictors and vasodilators under the extreme conditions of hypoxia at HA and imbalance between them could lead to serious pathologic conditions such as HAPE. Endothelial dysfunction leading to unusual vasoconstriction forms characteristics of HA disorders such as HAPE and also the clinical features of cardiovascular disease [12].
Regulators of endothelin function (ET-1) as well as vascular tone are altered significantly in HAPE and contribute to its pathophysiology [29]. The present study examines the possible association of six polymorphisms present in four different genes playing a crucial role in endothelium functioning with individual’s susceptibility towards the development of high altitude pulmonary edema. The most noteworthy finding of this study was a significant association of I/D, G(8002)A and Glu(298) Asp variants in ACE, EDN-1 and eNOS genes respectively with susceptibility towards HAPE.
It is interesting to note that EDN1 interacts with other endothelium derived substances such as NO and ACE. Regarding the ACE I/D polymorphism, we observed a significantly higher percentage of DD genotype and D allele in HAPE patients compared to resistant controls. D allele has been shown to be associated with elevated ACE activity [30], and is considered beneficial for sprint performance, whereas I allele has been shown to contribute to enhanced physical performance and elite endurance [31,32]. I allele was first reported to be associated with endurance capacity in elite British male mountaineers who could manage to reach beyond the height of 7,000m without the use of supplemental oxygen [33].
Subsequently in an independent study Woods and co-workers also demonstrated that I allele genetically corresponds to lower ACE activity and is associated with the maintenance of the higher oxygen saturation (SaO2) at HA [34]. Thus, the current study also demonstrates the genetic predominance of I allele in rCON compared to HAPE-s which could be helping these individuals in maintaining higher SaO2 at HA. Also, since both dominant and recessive models show significant p value (<0.05), it seems that both these alleles play an equally important role in genetic makeup (Table 4).
Hypoxia-induced pulmonary hypertension plays a major role in pathophysiology of HAPE, and endothelin-1 being a potent vasoconstrictor could be responsible for the rise in Pulmonary Artery Pressure (PAP) during hypoxic exposure. Both, ET-1 gene variants and plasma levels have been found to be associated with hypoxia adaptation for high-altitude natives [35]. Out of a study of various polymorphisms of EDN gene, G2288T polymorphism has been shown to be significantly associated with HAPE susceptibility [36]. We studied two common polymorphisms of EDN gene (G (8002)A and 3A/4A) and found significant association of G(8002) A SNP with HAPE.
Frequency occurrence of 4A/4A genotype and 4A allele was significantly lowered in case of HAPE-s compared to rCON. Moreover, two copies of 4A allele seems to play a significant role in modifying gene product (p<0.05 in recessive modelling, Table 4). Haplotype analysis for polymorphisms in EDN gene identified haplotype A-3A in EDN gene to be associated with increased risk of HAPE. Haplotyping of SNPs in VEGF gene could not yield any predictive haplotype for HAPE susceptibility.
Another interesting finding from the present study was significant association of Glu(298)Asp (G/T) polymorphism in eNOS with HAPE. Significantly higher GG genotype frequency was observed in rCON group compared to HAPE-s who showed higher percentage of mutant genotype TT. Our results also demonstrated that heterozygous GA and homozygous mutant AA might be playing a significant role in controlling expression of eNOS gene (p<0.05 for dominant model, Table 4). eNOS gene has widely studied gene and shown to play essential genetic role in maintaining local vascular NO homeostasis and thus helps in maintaining low PAP, which in turn improves lung ventilation [13,16].
The available literature suggests the benefits of elevated NO levels for the survival at HA under hypoxic condition [37]. NO inhalation therapy improves the localized pulmonary hypoxic state of HAPE patients [38,39]. Reduced expression of eNOS and apelin, as well as increased expression of ET-1 have been reported previously in HAPE patients [17, 40], thereby suggesting evidence of PH in HAPE. We have found evidence of subclinical PH in HAPE-s at sea level which can manifest as HAPE at HA. One of the first report of positive association of eNOS Glu (298) Asp polymorphism with HAPE appeared in 2002 by Droma and co-workers in Japanese population [41] and later by Ahsan et al. [42] in Indian population, demonstrating significantly lower frequencies of GG genotype and G allele in HAPE group compared to resistant controls.
Our results are also in concurrence with these reports. A contrasting report came later in 2012 wherein researchers found no significant correlation of this polymorphism in eNOS gene with HAPE incidence [43].
Vascular endothelial growth factor (VEGF) gene plays a central role in the process of angiogenesis, being the most potent angiogenesis factor and the mutations in this gene have been studied in relation to various disorders. In a study by Hanoka and coworkers, C(936)T polymorphism in 3’UTR region along with C(-2578)A, G(-1154)A and T(-460)C in promoter region have not been found to be associated with HAPE susceptibility in Japanese population [44].
However in a contrasting report, C(936)T polymorphism of VEGF gene has not shown any significant correlation with HAPE in Chinese population [45]. Additionally, Awata and co-workers (2005) demonstrated VEGF C(-634)G polymorphism as a genetic risk factor for muscular edema as well as diabetic retinopathy [46]. However, results of present investigation show no statistically significant genotypic and allelic variation both in SNPs (C(936) T and C(-634)G) between the two groups under study. Whether the gene polymorphisms reported in the present study are cause or having effect on hypoxia sensitivity at HA in non-mountaineer population cannot be established by this study, however results of present investigation shows that these gene polymorphisms may contribute to HAPE susceptibility.
Conclusion
Polymorphisms such as I/D, G(8002)A and Glu(298)Asp in ACE, EDN-1 and eNOS genes respectively, seems to play a significant role in determining HAPE sensitivity. Thus, we conclude that mutations in these genes related to impaired pulmonary endothelial function with reduced nitric oxide and enhanced endothelin-1 (ET-1) bioavailability could be important genetic factors for HAPE susceptibility. Bringing some new insights in the genetic predisposition to HAPE, the findings of the present investigation may find application in tourist industry, mountaineering and even official recruitment of soldiers for high altitude duties.
Acknowledgement
The authors are extremely thankful to all the volunteers who participated in the study. Also, authors are grateful to scientists and staff of Heat Physiology Division of DIPAS for their cooperation and logistic support in collection of blood samples. We extend our thanks to Defence Research and Development organization (DRDO), India for funding this research work.
Funding
This study was funded by Defence Research and Development Organization (DRDO), India.
References
- Menon ND (1965) High altitude pulmonary edema. N Engl J Med 273: 66-73.
- Hultgren HN, Spickard W, Hellriegel K, Houstan CS (1961) High altitude pulmonary edema. Medicine 40: 289-313.
- Hultgren HN (1996) High-altitude pulmonary edema: current concepts. Annu Rev Med 47: 267-284.
- Gibbs JS (1999) Pulmonary hemodynamics: implications for high altitude pulmonary edema (HAPE). A review. Adv Exp Med Biol 474: 81-91.
- Mortimer H, Patel S, Peacock AJ (2004) The genetic basis of highaltitude pulmonary oedema. Pharmacol Ther 101(2): 183-192.
- Bhagi S, Srivastava S, Singh SB (2014) High-altitude pulmonary edema: review. J Occup Health 56(4): 235-43.
- Jacob D Jensen, Andrew L Vincent (2017) Altitude Illness, Pulmonary Syndromes, High Altitude Pulmonary Edema (HAPE).
- Lerman A, Zeiher AM (2005) Endothelial function: cardiac events. Circulation 111(3):363-368.
- Wildlansky ME, Gokce N, Keaney JF, Vita JA (2003) The clinical implications of endothelial dysfunction. J Am Coll Cardiol 42(7): 1149- 1160.
- Hampl V, Herget J (2000) Role of nitric oxide in the pathogenesis of chronic pulmonary hypertension. Physiol Rev 80: 1337-1372.
- Förstermann U, Closs EL, Pollock JS, Nakane N, Schwarz P, et al. (1994) Nitric oxide synthase isozymes: characterization, purification, molecular cloning, and functions. Hypertension 23(6 Pt 2): 1121-1131.
- Duplain H, Sartori C, Lepori M, Egli M, Allemann Y, et al. (2000) Exhaled nitric oxide in high altitude pulmonary edema: role in the regulation of pulmonary vascular tone and evidence for a role against inflammation. Am J Respir Crit Care Med 162(1): 221-224.
- Busch T, Bärtsch P, Pappert D, Grünig E, Hildebrandt W, et al. (2001) Hypoxia decreases exhaled nitric oxide in mountaineers susceptible to high-altitude pulmonary edema. Am J Respir Crit Care Med 163(2): 368-373.
- Miyamoto Y, Saito Y, Kajiyama N, Yoshimura M, Shimasaki Y, et al. (1998) Endothelial nitric oxide synthase gene is positively associated with essential hypertension. Hypertension 32(1): 3-8.
- Shoji M, Tsutaya S, Saito R, Takamatu H, Yasujima M (2000) Positive association of endothelial nitric oxide synthase gene polymorphism with hypertension in northern Japan. Life Sci 66(26): 2557-2562.
- Japp AG, Cruden NL, Amer DA, Li VK, Goudie EB, et al. (2008) Vascular effects of apelin in vivo in man. J Am Coll Cardiol 52(11): 908-13.
- Mishra A, Kohlia S, Duaa S, Thinlasd T, Mohammadd G, et al. (2015) Genetic differences and aberrant methylation in the apelin system predict the risk of high-altitude pulmonary edema. Proc Natl Acad Sci 122(19): 6134-6139.
- Chandra SM, Razavi H, Kim J, Agrawal R, Kundu R, et al. (2011) Disruption of the Apelin-APJ System Worsens Hypoxia-Induced Pulmonary Hypertension. Arterioscler Thromb Vasc Biol 31(4): 814- 820.
- Ferrara N, Davis-Smyth T (1997) The biology of vascular endothelial growth factor. Endocr Rev 18(1): 4-25.
- Jain RK (2002) Tumor angiogenesis and accessibility: role of vascular endothelial growth factor. Semin Oncol 29 (6 Suppl 16): 3-9.
- Ferrara N, Alitalo K (1999) Clinical applications of angiogenic growth factors and their inhibitors. Nat Med 5(12): 1359 -1364.
- Kim KJ, Li B, Winer J, Armanini M, Gillett N, et al. (1993) Inhibition of vascular endothelial growth factor-induced angiogenesis suppresses tumour growth in vivo. Nature 362(6423): 841-844.
- McCarrson SL, Edwards S, Evans PR, Gibbs R, Dearnaley DP, et al. (2002) Influence of cytokine gene polymorphism on development of prostate cancer. Cancer Res 62(1): 3369-3372.
- Krippl P, Langsenlehner U, Renner W, Yazdani-Biuki B, Wolf G, et al. (2003) common 936C/T gene polymorphism has been associated with decreased breast cancer risk. Int J Cancer 106(4): 468-471.
- Renner W, Kotschan S, Hoffmann C, Obermayer-Pietsch B, Pilger EA (2000) common 936 C/T mutation in the gene for vascular endothelial growth factor is associated with vascular endothelial growth factor plasma levels. J Vasc Res 37(6): 443-448.
- Soree P, Gupta RK, Singh K, Desiraju K, Agrawal A, et al. (2016) Raised HIF1α during normoxia in high altitude pulmonary edema susceptible non-mountaineers. Scientific Reports 6: 26468.
- Gupta RK, Soree P, Desiraju K, Agrawal A, Singh SB (2017) Subclinical pulmonary dysfunction contributes to high altitude pulmonary edema susceptibility in healthy non-mountaineers. Scientific Reports 7(1): 14892.
- Gupta RK, Himashree G, Singh K, Soree P, Desiraju K, et al. (2016) Elevated pulmonary artery pressure and brain natriuretic peptide in high altitude pulmonary edema susceptible non-mountaineers. Scientific Reports 6: 21357.
- Barker KR, Conroy AL, Hawkes M, Murphy H, Pandey P, et al. (2016) Biomarkers of hypoxia, endothelial and circulatory dysfunction among climbers in Nepal with AMS and HAPE: a prospective case-control study. J Travel Med 23(3).
- Rigat B, Hubert C, Corvol P, Soubrier F (1992) PCR detection of the insertion/deletion polymorphism of the human angiotensin converting enzyme gene (DCP1) (dipeptidyl caroxypeptidyase 1). Nucleic Acids Res 20(6): 1433.
- Gayagay G, Yu B, Hambly B, Boston T, Hahn A, et al. (1998) Elite endurance athletes and ACE I allele-the role of genes in athletic performance. Hum Genet 103(1): 48-50.
- Williams AG, Rayson MP, Jubb M, World M, Woods DR, et al. (2000) The ACE gene and muscle performance. Nature 403(6770): 614.
- Montgomery HE, Marshall R, Hemingway H, Myerson S, Clarkson P, et al. (1998) Human gene for physical performance. Nature 393(6682): 221-222.
- Woods DR, Pollad AJ, Collier DJ, Jamshidi Y, Vassiliou V, et al. (2002) Insertion/deletion polymorphism of the angiotensin I-converting enzyme gene and artery oxygen saturation at high altitude. Am J Respir Crit Care Med 166(3): 362-366.
- Rajput C, Najib S, Norboo T, Afrin F, Pasha QMA (2006) Endothelin-1 gene variants and levels associate with adaptation to hypobaric hypoxia in high-altitude natives. Biochem Biophys Res Commun 341(4): 1218-1224.
- Charu R, Stobdan T, Ram RB, Khan AP, Pasha QMA, et al. (2006) Susceptibility to high altitude pulmonary oedema: role of ACE and ET-1 polymorphisms. Thorax 61(11): 1011-1012.
- Tesauro M, Thompson WC, Rogliani P, Qi L, Chaudhary PP, et al. (2000) Intracellular processing of the endothelial nitric oxide synthase isoforms associated with differences in severity of cardiopulmonary diseases: cleavage of proteins with aspartate vs glutamate at position 298. Proc Natl Acad Sci 97(6): 2832-2835.
- Beall CM, Laskowski D, Stohl KP, Soria R, Villena M, et al. (2001) Pulmonary nitric oxide in mountain dwellers. Nature 14: 411-412.
- Anand IS, Prasad BAK, Chugh SS, Rao KRM, Cornfield DM, et al. (1998) Effects of inhaled NO and O2 in high-altitude pulmonary edema. Circulation 98(22): 2441-2445.
- Sartori C, Vollenweider L, Löffler BM, Delabays A, Nicod P, et al. (1999) Exaggerated endothelin release in high-altitude pulmonary edema. Circulation 99(20): 2665-2668.
- Droma Y, Hanaoka M, Ota M, Katsuyama Y, Koizumi T, et al. (2002) Positive association of the endothelial nitric oxide synthase gene polymorphisms with high-altitude pulmonary edema. Circulation 106(7): 826-830.
- Ahsan A, Mohd G, Norboo T, Baig MA, Pasha MAQ (2006) Heterozygotes of NOS3 polymorphisms contribute to reduced nitrogen oxides in highaltitude pulmonary edema. CHEST 130(5): 1511-1519.
- Luo Y, Chen Y, Zhang Y, Zhou Q, Gao Y (2012) Association of endothelial nitric oxide synthase (eNOS) G894T polymorphism with high altitude pulmonary edema susceptibility: a meta-analysis. Wilderness Environ Med 23(3):270-274.
- Hanaoka M, Droma Y, Ota M, Ito M, Katsuyama Y, et al. (2009) Polymorphism of human vascular endothelial growth factor gene in high altitude pulmonary oedema susceptible subjects. Respirology 14(1): 46-52.
- Wu AL, Xiong YS, Li ZQ, Liu YG, Quan Q, et al. (2015) Correlation between single nucleotide polymorphisms in hypoxia-related genes and susceptibility to acute high-altitude pulmonary edema. Genet Mol Res 14(3): 11562-11572.
- Awata T, Kurihara S, Takata N, Neda T, Iizuka H, et al. (2005) Functional VEGF C-634G polymorphism is associated with development of diabetic macular edema and correlated with macular retinal thickness in type 2 diabetic. Biochem Biophys Res Commun 333(3): 679-685.






























