A Comparison Study of Serological Test, Culturing Technique and Molecular Detection of Salmonella Typhi Among Febrile Patients in Khartoum State, Sudan
Ali M Badri1*, Hiba M Ahmed2, Nahla Y Ahmed2, Hala H Ahmed2, Sara M Ali2, Sahar M Sid3, Nasr M Nasr3 and Mohamed H Arbab3
1Department of Microbiology, Sudan International University, Sudan
2Department of Microbiology, International University of Africa, Sudan
3Department of Microbiology, Omdurman Ahlia University, Sudan
Submission: March 08, 2019; Published: March 27, 2019
*Corresponding author: Ali M Badri, Department of Microbiology, Faculty of Medical Laboratory Sciences, Sudan International University, Khartoum, Sudan
How to cite this article: Ali M B, Hiba M A, Nahla Y A, Hala H A Sara M A, et al. A Comparison Study of Serological Test, Culturing Technique and Molecular Detection of Salmonella Typhi Among Febrile Patients in Khartoum State, Sudan. Adv Biotech & Micro. 2019; 13(4): 555866. DOI: 10.19080/AIBM.2019.13.555866
Abstract
Typhoid fever, a systemic disease caused by Salmonella enterica serovar typhi (S. typhi) it is one of the most important pathogens involved in human food-borne illness. A Cross-sectional descriptive study was conducted to detect the Salmonella typhi among patients suspected of having enteric fever using serological test, Isolation based technique and Molecular based test. The results revealed that the Widal test, isolation (Blood, Stool) of the bacteria and PCR detection showed 64%, (12%, 18%) and 27% respectively the sensitivity and specificity of different diagnostic tests were calculated using PCR as a reference method. Higher sensitivity was observed in stool culture (77.8%) and lower sensitivity was observed in Widal test (33.3%). The blood culture showed the highest specificity (97.2%), whereas Widal test showed the lowest specificity (27.4%) We suggest that the PCR technique could be used as a novel diagnostic method of typhoid fever, particularly in culture-negative cases..
Keywords: Widal Test; Stool Culture; Blood culture; Molecular Identification; S. typhi, Khartoum; Sudan
Abbrevations: PCR: Polymerase Chain Reaction, RCLB: Red Cell Lysis Buffer, WCLB: White Cell Lysis Buffer
Introduction
Typhoid fever and salmonellosis are public health problems in developing countries, Salmonella enterica subspecies enterica serovar Typhi (S. typhi) causes typhoid fever, a systemic febrile illness in humans [1]. The clinical presentation of typhoid fever varies from a mild illness with low-grade fever, malaise, and slight dry cough to a severe clinical picture with abdominal discomfort and multiple complications [2]. The transmission of the infection is by faecal-oral route, when contaminated food or water is consumed or from contaminated hands [3]. S. typhi can be isolated using cultures of bone marrow, blood, stool and intestinal secretion in more than 90% of the patients [4]. The Widal test had been one of the most commonly used diagnostics to date despite its limited sensitivity of only 39% [5,6], and false positives have been reported in certain non-typhoid infections [7-9]. Typhidot and Tubex, both serological assays, are also available for use [10,11], and have a reported sensitivity of around 70% [12,13], blood culture can detect only 45 to 70% of patients with typhoid fever, depending on the amount of blood sampled, the bacteremia level of S. typhi, the type of culture medium used, and the length of incubation period [4,14], if the culture of the mononuclear cell-platelet layer of blood is combined with cultures of bone marrow aspirate and rectal swab, the positive rate of detection can increase up to 100% [15].
Molecular detection methods are most suitable to identify pathogens in human excretions because these methods are highly specific and sensitive. In particular; the Polymerase Chain Reaction (PCR) is capable of detecting minute quantities of DNA of specific pathogens through amplification of a defined DNA segment and discriminating in one reaction between different organisms even if they are closely related, in combination with the appropriate sample preparation method, PCR can be applied on almost any specimen including whole blood, stool, and urine [16], thus; PCR seems to be suitable to identify those patients actively excreting the organism and to investigate the carrier state through the specific detection the DNA of S. typhi in urine and feces samples [6]. However; these methods require a well-trained staff and sophisticated equipment which generally are not available in developing countries or point-of-care testing facilities [1,17]. Typhoid fever has an important socio-economic impact, so accurate diagnosis of the disease at an early stage is important not for etiological diagnosis but also for identifying individuals that may serve as potential carrier who may be responsible for acute typhoid fever outbreak [18,19].
Materials and Methods
Study design
A Cross-sectional descriptive study was carried out in the period from January to July 2018 in Bashair Teaching hospital at International University of Africa, Khartoum state, Sudan. to detect the S. typhi among patients suspected of having enteric fever.
Sample collection
A total of 100 blood samples and 100 stool samples were collected from febrile patients suspected of having enteric fever attendant Bashair Teaching hospital. A volume of 7 ml of blood was collected and inoculated directly in blood culture bottles, 3 ml of blood was collected in plain container and centrifuged to obtain serum for Widal test and another 2.5 ml of blood sample was collected in EDETA container for DNA extraction. The stool samples were collected in clean and dry, free-disinfectant container with spoon and inoculated in selenite F broth for transportation.
Data collection
A structured questionnaire and referring to the patient clinical sheet were being used; demographic data and other Data (Previous typhoid, Symptoms, if using any drug and) verbal consent was obtained from each patient enrolled in this study.
Laboratory Work and Protocols
Widal test (Serological test): 3ml of whole venous blood was collected in plain container. The collected specimens were centrifuged at 3000 rpm for 5min the serum was separated and stored at -20 °C until tested. Perfectly clean and dry test tubes were made and prepared dilutions beginning with 1:20 and doubling through 1:320. 0.1 ml of serum were added to 0.9 ml of physiological saline and then diluted serially by mixing 0.5 ml diluted serum with 0.5 ml saline and discarding 0.5 ml from the last tube. From specimen submitted to detect possible rise in titer, series of 5 dilutions were prepared, ending with 1: 320. The prepared tubes were incubated at 37 °C for 24 hours, after incubation period observe the agglutination reaction within the tubes. Each serial dilution had been interpreted according to [3].
Blood culture
Sterile syringe was used in drowning the blood samples, the caps of the culture bottle were disinfected with 70% ethanol, afresh sterile needle was used to puncture the cap of the culture bottles to dispense the blood into the brain heart infusion broth. The blood was gently mixed with the broth. Blood culture samples were labelled and incubated aerobically at 37OC for 7 days but were examined daily for signs of bacterial growth. Subcultures was made on MacConkey agar (Liofilchem, Italy) every other day for seven days. The subcultures on MacConkey agar was incubated aerobically at 37 °C. Bacterial growths with colonies of 2-3mm in diameter and pale non-lactose fermenting were suspected as S. typhi but required full identification.
Stool culture and Identification
Patients was instructed on the collection and handling of the stool specimen according to (20), portions of the stool samples were inoculated into 5% Selenite F broth for enrichment and transport from which a new subculture were did within 6 hours. Then a loop full was sub-cultured on Xylose lysine Deoxycholate (XLD). The identification of Salmonella typhi was done using standard conventional biochemical tests (Kligler Iron agar: slant /Alkaline, butt/ Acid, H2S production / weak, Gas /-; Motility test /+; Indole /-; Urease /-; Citrate /-) according to [20].
Molecular detection
DNA extraction was done by following SDS and Proteinase K extraction method [21], 300 μl of blood samples were suspended in 1.5 ml Eppendorf’s tube with 1000 μl Red Cell Lysis Buffer (RCLB), mixed well and centrifuged at 5000 rpm for 10 min, Supernatant was discarded and 300 μl of white cell lysis buffer (WCLB) was added, 10 μl of 10% SDS and 10 μl of proteinase K solution were then added and the mixture was incubated for 1 h at 65°C. Then 100 μl of 6 M NaCl was added followed by 200 μl of cold chloroform and centrifuged at 11000 rpm for 6 min supernatant containing the DNA was then transferred to a new tube and absolute ethanol was added and centrifuged at 11000 rpm for 5 min. The supernatant was then discharged, and the pellet was washed with 600 μl 70% ethanol and centrifuged at 6000 rpm for 5 min, the ethanol was discarded and the purified DNA was dissolved in100 μl TE buffer and stored at -20 ºC until used.
Polymerase Chain Reaction (PCR)
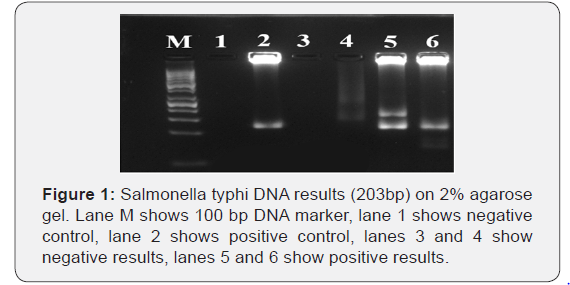
PCR was performed and the test was carried out using following primers: forward: 5-TTACC CCACA GGAAG CACGC-3and reverse: 5-CTCGT TCTCT GCCGT GTGGG-3. Each reaction was performed in total volume of 25 μl, containing 5 μl master mix (Solis Bio dyne master mix), 2 μl of primer, 5 μl of DNA and 13 μl of distilled water. The PCR was performed for 30 cycles at 94°C for 45 s for denaturation, 67°C for 30 s for annealing and 72°C for 30s for extension; the final extension at 72°C for 7 min. [22]. 5 μl of the PCR product was analyzed using 2% Agarose gel electrophoresis and stained with 0.15% Ethidium bromide and the product was visualized using UV gel documentation system. The expected size of S. typhi DNA amplicon is 203bp.
Data analysis
The Statistical Package for Social Sciences (SPSS 25) was used for statistical analysis.
Results
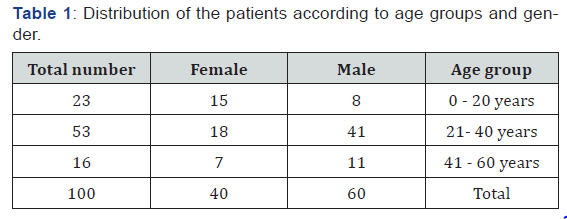
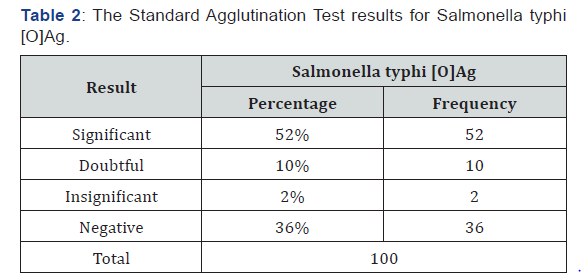
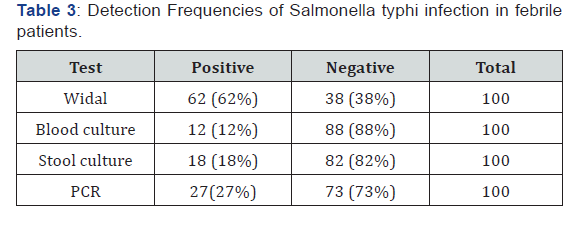
100 blood samples and 100 stools were enrolled in this study, males were 60 (60%) and females were 40 (40%) (Table 1). The reactive sera for Widal test of total sample were 62 (62%) samples and non-reactive sera for Widal test were 38 (38%) of total sample. The titration results of Typhoid fever among febrile patients for S. typhi [O] Ag were (significant, doubtful, insignificant and negative) as 52(52%), 10 (10%),2(2%), 36(36%) respectively (Table 2). The percentage of titer values of Widal test positive patients selected for the study includes ≥ 1/160. Out of the 100 blood and stool samples collected, Salmonella enterica serovar typhi were isolated from 12(12%) and 18 (18%) of the samples respectively (Table 3). PCR was performed for all recruited patients, EDTA blood samples were collected from 100 patients. had a sufficient volume (≥300 μL) to extract DNA for PCR amplification. 27(27%) DNA samples were positive for S. typhi by PCR (Figure 1).
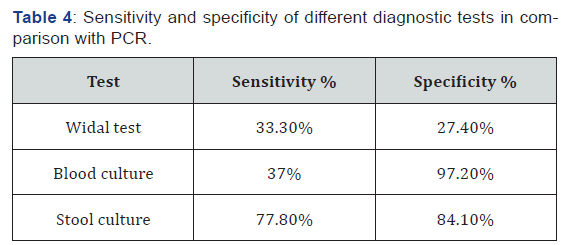
The sensitivity and specificity of different diagnostic tests were calculated using PCR as a reference method. Higher sensitivity was observed in stool culture (77.8%) and lower sensitivity was observed in Widal test (33.3%). The blood culture showed the highest specificity (97.2%), whereas Widal test showed the lowest specificity (27.4%) (Table 4).
Discussion
S. typhi is the causative agent of typhoid fever. Typhoid fever is more common in developing countries where there is limited safe water and health care system. The ‘gold standard’ for identifying the cause of an infection is the isolation and identification of the causative agent of disease. In the absence of a viable bacterium, antibody tests can give evidence of infection provided that suitable immunoassays, based on well-characterized antigens, are used. In this study all the isolates were confirmed and identified as S. typhi based on cultural, morphological and biochemical tests. Further identification was done by molecular technique. In the current study, the magnitude of typhoid fever was 64%, 27%, 18% and 12% by Widal titration, PCR technique, stool culture and blood culture, respectively. This indicates that there was a great variation among those tests. Widal test has been used for over a century in developing countries for diagnosing typhoid fever but it has been reported to have low sensitivity and specificity [23,24]. In the present study, Widal test was positive in 62 %(62/100) of the patients.
Widal test was positive in 62 of the 27 positives by PCR. Thus, the test had sensitivity of 33.3% and specificity of 27.4%. Depending on this results, we suggest that significant titer of antibodies detected in patient serum by Widal test is not specific for having typhoid fever our findings were supported to the findings from study conducted by [25-28], other study has been reported the Widal test has a sensitivity of 54% and specificity of 81% where there may be high levels of specific and cross reacting antibodies, also in typhoid fever-suspected Indian children, sensitivity of Widal test was 64.6% [29], and in Iranian children, it was 75.86% [30]. Decreased sensitivity is due to the long latent period after which the test may become positive, negative result in early infection or due to prior antibiotic therapy, while decreased specificity is attributed to cross reactivity with other Gram negative bacteria and non-typhoidal salmonella, pre-existing base line antibodies in endemic areas, anamnestic reactions in unrelated infections and prior TAB or oral typhoid vaccination within two weeks before coming for the diagnosis during this study [31].
Blood culture has the promise of diagnosis in the first week and is very specific, but its sensitivity is poor due to various factors. Out of total 100 clinically suspected having typhoid fever cases enrolled in the study, 12 cases were positive by blood culture, which reports sensitivity of 37 % and specificity of 97.2, while culture positivity in other studies have quoted sensitivity ranging from 8.9-43% [32-34]. The relative low rate of isolation from blood culture had been attributed to delay in diagnosis, widespread and irrational use of antibiotics and low volume of blood obtained for cultures especially among children. on the other hand studies have report the 65-68% of sensitivity [35,36]. The high rate of isolation in those studies could be attributed to awareness among people leading to early presentation to secondary or tertiary health care facility, adequate quantities of blood could be taken for diagnostic test and improvements in blood culture methods especially use of automated systems.
In our study, stool culture had very good sensitivity 77.8% and specificity 84.1% this finding supports an earlier study by [37,38]. But there are a number of factors that should be considered while using stool culture for diagnosis of typhoid fever-suspected cases. Stool culture is mostly positive after 3 weeks of infection with S. typhi, in early infection, the stool culture is positive only in 30%-40% of cases [39]. The other factor that may affect the stool culture is its proneness to contamination that may result in misdiagnosis. Additionally, increasing the quantity of stool used for culture has been shown to increase the sensitivity with culturing two grams of stool rather than the standard one gram increasing isolation by 10% [40].
Nucleic acid-based detection of S. typhi was developed in earlier studies using different target gene sequences [41]. All the isolates were further identified as S. typhi by molecular technique. The samples showed positive results for 27%. The finding of positive PCR results for blood samples from culture-negative typhoid patients is consistent with the previous observations. Although detection of Salmonella typhi by using labeled probes is 99% specific [41]. We conclude that PCR is much superior to blood culture, stool culture and Widal test. It has great discriminating value due to its very high sensitivity and specificity [42], and reasonably quick diagnostic modality. Even 1-5 bacteria/mL can be detected with absolute specificity within 1-2 days. Therefore, it can be of singular importance for the detection of early cases of typhoid, which is not only important for the treatment of patients but is also necessary for control of the disease. Due to the need for extensive infrastructure and specialized skills, the PCR facility cannot be made available everywhere, especially in developing countries.
Conclusion
This study suggests that the true burden of S. typhi cannot be estimated by culture alone, clinical history coupled with using PCR in addition to routine diagnosis of suspected typhoid fever cases in hospitals are highly recommended.
Ethics approval and consent to participate
This study was approved by the ethical committee of International University of Africa, Faculty of Medical Laboratory Sciences and verbal consent was obtained from each patient enrolled in this study.
Availability of data and materials
The datasets analysed during this current study are available from the corresponding author on reasonable request.
Funding
This work is partially supported by International University of Africa, Faculty of Medical Laboratory Sciences, Khartoum, Sudan.
Author’s contribution
AMB conceived the idea for this paper and wrote the manuscript, HBA and HHA conducted the analysis, SMA and SMS participated in project oversight. MHA and NMN contributed to the design of the work, drafting and revision of the manuscript. All authors contributed to drafting the manuscript and approved the final submission.
Acknowledgment
The author wishes to thank the Bashair Teaching hospital. Thanks, are also extended to Dr. Khalid A. Enan, Dr. Sara M. Ali and Mr. Yousof S. Yousof.
References
- Abdullah J, Saffie N, Sjasri FAR, Husin A, Abdul-Rahman Z, et al. (2014) Rapid detection of Salmonella Typhi by loop-mediated isothermal amplification (LAMP) method. Brazilian Journal of Microbiology 45(4): 1385-1391.
- (2003) World Health Organization, et al. Background document: the diagnosis, treatment and prevention of typhoid fever.
- (2006) Cheesbrough, Monica. District laboratory practice in tropical countries. Cambridge university press, USA.
- Gilman RH, Terminel M, Levine MM, Hernandez-Mendoza P, Hornick RB (1975) Relative efficacy of blood, urine, rectal swab, bone-marrow, and rose-spot cultures for recovery of Salmonella typhi in typhoid fever. The Lancet 1(7918): 1211-1213.
- Levine MM, Grados O, Gilman RH, Woodward WE, Solis-Plaza R, et al. (1978) Diagnostic value of the Widal test in areas endemic for typhoid fever. Am J Trop Med Hyg 27(4): 795-800.
- Hatta M, Smits HL (2007) Detection of Salmonella Typhi by nested polymerase chain reaction in blood, urine, and stool samples. Am J Trop Med Hyg 76(1): 139-143.
- Wilson (1975) Topley and Wilson’s principles of bacteriology, virology and immunity.
- Kumar A, Arora V, Bashamboo A, Ali S (2002) Detection of Salmonella typhi by polymerase chain reaction: implications in diagnosis of typhoid fever. Infect Genet Evol 2(2): 107-110.
- Reynolds DW, Carpenter RL, Simon WH (1970) Diagnostic specificity of Widal’s reaction for typhoid fever. JAMA 214(12): 2192-2193.
- Lim PL, Tam FC, Cheong YM, Jegathesan M (1998) One-step 2-minute test to detect typhoid-specific antibodies based on particle separation in tubes. Journal of clinical microbiology 36(8): 2271-2278.
- Prakash P, Sen MR, Mishra OP, Gulati AK, Shukla BN, et al. (2007) Dot enzyme immunoassay (Typhidot) in diagnosis of typhoid fever in children. J Trop Pediatr 53(3): 216-217.
- Dutta S, Sur D, Manna B, Sen B, Deb AK, et al. (2006) Evaluation of newgeneration serologic tests for the diagnosis of typhoid fever: data from a community-based surveillance in Calcutta, India. Diagn Microbiol Infect Dis 56(4): 359-365.
- Al-Emran HM, Eibach D, Krumkamp R, Ali M, Baker S, et al. (2016) A multicountry molecular analysis of Salmonella enterica serovar Typhi with reduced susceptibility to ciprofloxacin in sub-Saharan Africa. Clin Infect Dis 62(1): 42-46.
- Guerra-Caceres JG, Gotuzzo-Herencia E, Crosby-Dagnino E, Miro- Quesada M, Carrillo-Parodi C (1979) Diagnostic value of bone marrow culture in typhoid fever. Trans R Soc Trop Med Hyg 73(6): 680-683.
- Rubin FA, McWhirter PD, Burr D, Punjabi NH, Lane E, et al. (1990) Rapid diagnosis of typhoid fever through identification of Salmonella typhi within 18 hours of specimen acquisition by culture of the mononuclear cell-platelet fraction of blood. J Clin Microbiol 28(4): 825-827.
- Boom R, Sol CJ, Salimans MM, Jansen CL, Wertheim-van Dillen PM, et al. (1990) Rapid and simple method for purification of nucleic acids. Journal of clinical microbiology 28(3): 495-503.
- Curtis KA, Rudolph DL, Owen SM (2008) Rapid detection of HIV-1 by reverse-transcription, loop-mediated isothermal amplification (RTLAMP). J Virol Methods 151(2): 264-270.
- Zailani SB, Aboderin AO, Onipede AO (2004) Effect of socio-economic status, age and sex on antibody titre profile to Salmonella typhi/ paratyphi in Ile-Ife, Nigeria. Niger J Med 13(4): 383-387.
- Akinade SF (2015) Molecular Identification of Salmonella enterica serovar typhi from Stool Culture of Widal Test Positive Patients in Selected Hospitals in Abeokuta, Nigeria. International Journal of Scientific 4(3): 228-235 .
- McCartney JE, Collee JG, Mackie TJ (1989) Practical medical microbiology. Charchil Livingstone, UK.
- Nishiguchi MK (2002) DNA isolation procedures techniques in molecular systematics and evolution. Pp, 247-287.
- Park SH, Kim HJ, Cho WH, Kim JH, Oh MH, et al. (2009) Identification of Salmonella enterica subspecies I, Salmonella enterica serovars Typhimurium, Enteritidis and Typhi using multiplex PCR. FEMS microbiology letters 301(1): 137-146.
- Sherwal BL, Dhamija RK, Randhawa VS, Jais M, A Kaintura, et al. (2004) A comparative study of Typhidot M and widal test in patients of Typhoid fever. Journal of Indian Academy of Clinical Medicine 5(3): 244-246.
- Wain John, Hosoglu S (2008) The laboratory diagnosis of enteric fever. The Journal of Infection in Developing Countries 2(6): 421-425.
- Andualem G, Abebe T, Kebede N, Gebre-Selassie S, Mihret A, et al. (2014) A comparative study of Widal test with blood culture in the diagnosis of typhoid fever in febrile patients. BMC research notes 7: 653.
- Itah AY, Uweh EE (2005) Bacteria isolation from blood, stool and urine of typhoid patient in a developing in a developing country. Southeast Asian J Trop Med Public Health 36(3): 673-677.
- Ley, Mtove G, Thriemer K, Amos B, von Seidlein L, et al. (2010) Evaluation of the Widal tube agglutination test for the diagnosis of typhoid fever among children admitted to a rural hdospital in Tanzania and a comparison with previous studies. BMC infect dis 10: 180.
- Olsen, Pruckler J, Bibb W, Nguyen TM, Tran MT, et al. (2004) Evaluation of rapid diagnostic tests for typhoid fever. J clinical Microbiology 42(5): 1885-1889.
- Beig, FarazAhmad, Mohd Ekram, Indu Shukla (2010) Typhidot M and Diazo test vis-à-vis blood culture and Widal test in the early diagnosis of typhoid fever in children in a resource poor setting. Brazilian Journal of Infectious Diseases 14(6): 589-593.
- Noorbakhsh S, Rimaz S, Rahbarimanesh AA, Mamishi S (2003) Interpretation of the widal test in infected children. Iranian Journal of Public Health 32(1): 35-37.
- Bhutta, Zulfiqar Ahmed, Mansurali, Naseem (1999) Rapid serologic diagnosis of pediatric typhoid fever in an endemic area: a prospective comparative evaluation of two dot-enzyme immunoassays and the Widal test. Am J Trop Med Hyg 61(4): 654-657 .
- Narayanappa D, Sripathi R, Jagdishkumar K, Rajani HS, et al. (2010) Comparative study of dot enzyme immunoassay (Typhidot-M) and Widal test in the diagnosis of typhoid fever. Indian pediatrics 47(4): 331-333.
- Abucejo PE, Capeding MR, Lupisan SP, Arcay J, Sombrero LT, et al. (2010) Blood culture confirmed typhoid fever in a provincial hospital in the Philippines. Southeast Asian journal of tropical medicine and public health 32(3): 531-536.
- Saha, Malay Ranjan (2003) A note on incidence of typhoid fever in diverse age groups in Kolkata, India. Japanese journal of infectious diseases 56(3): 121-122.
- Sanjeev H, Sweetha Nayak, Pai Asha KB, Rai Rekha, Vimal Karnaker, et al. (2013) A systematic evaluation of rapid dot-EIA, blood culture and Widal test in the diagnosis of typhoid fever. Nitte University Journal of Health Science 3(1): 21.
- Sherwal BL, RK Dhamija, VS Randhawa, M Jais, A Kaintura, et al. (2004) A comparative study of Typhidot M and widal test in patients of Typhoid fever. Journal of Indian Academy of Clinical Medicine 5(3): 244-246.
- Hohman EL (2013) Approach to patient with nontyphoid salmonella in a stool culture.
- Muthoni, Gitonga Ciriaka (2016) performance of widal test and stool culture as diagnostic methods for salmonella typhi infection in chuka general hospital, tharaka nithi county Thesis. Kenyatta University, Kenya.
- Edelman, Robert Levine, Myron M (1986) Summary of an international workshop on typhoid fever. Reviews of infectious diseases 8(3): 329- 349.
- Bhanu, Shrivastava, Vandana, Shrivastava, Archana, et al. (2011) Comparative study of the diagnostic procedures in salmonella infection, causative agent. An overview study. International research journal of Pharmacy 2230-8407.
- Rubin FA, McWhirter PD, Punjabi NH, Lane E, Sudarmono P, A et al. (1989) Use of a DNA probe to detect Salmonella typhi in the blood of patients with typhoid fever. Journal of clinical microbiology 27(5): 1112-1114.
- Song JH, Cho H, Park MY, Na DS, Moon HB, et al. (1993) Detection of Salmonella typhi in the blood of patients with typhoid fever by polymerase chain reaction. J Clinical Microbiol 31(6): 1439-1443.






























