Field Evaluation of the Diagnostic Efficacy of a New Designed Immunochromotographic Test (let) Comprasion with Serum Adenosine Deaminase (Ada) Enzyme and Tuberulin Skin Test (Tst) for Diagnosis of Active Pulmonary Tuberculosis (Tb) in Turkey
Gülnur Tarhan1*, Emel Kibaroğlu2, Ismail Ceyhan3 and Selçuk Kiliç4
1Department of Medical Microbiology, Adiyaman University, Turkey
2Refik Saydam Hygiene Center, Tuberculosis Dispensary, Turkey
3Ataturk Chest Diseases and Thoracic Surgery Education and Research Hospital, Turkey
4National Public Health Institute, Microbiology Reference Laboratory, Turkey
Submission: February 13, 2017; Published: April 13, 2017
How to cite this article: Gulnur T, Emel K, Ismail C, Selcuk K. Field Evaluation of the Diagnostic Efficacy of a New Designed Immunochromotographic Test (Ict) Comprasion with Serum Adenosine Deaminase (Ada) Enzyme and Tuberulin Skin Test (Tst) for Diagnosis of Active Pulmonary Tuberculosis (Tb) in Turkey. Adv Biotech & Micro. 2017; 3(1): 555605. DOI: 10.19080/AIBM.2017.03.555605.
Abstract
Objective: The objective of the present study was to investigate in order to determine the efficiency of a new designed ICT comparison with serum adenosine deaminase activity and TCT in active pulmonary tuberculosis patients, TB contacts and healthy subjects.
Materials and methods: In our study, we evaluated 99 active pulmonary TB (PTB) patients, 48 TB contacts and 153 healthy subjects. Study subjects were drawn from one hospital and one primary health care unit in Ankara, Turkey. ICT and ADA enzyme activity was evaluated by using serum samples.
Results: The results showed that a new designed ICT test was positive for 42 (42.42%) active pulmonary TB patients, 16 (33.33%) TB contacts and 13 (9.09 %). The ADA activity was (mean ± SD) 42.77 ± 6.72 U/L in pulmonary tuberculosis patients (n= 98), 13.36 ± 2.25 U/L in TB contacts (n= 48), 8.58±3.21 U/L in healthy control subjects (n= 153).
Conclusion: In conclusion, the serum ADA activity increases in pulmonary tuberculosis patients, therefore it may be supportive parameter for the diagnosis of pulmonary TB. A new designed ICT test was not found to be an effective diagnostic tool for pulmonary TB. However, further evaluation of these tests in specific epidemiologic settings may be warranted.
Keywords: Tuberculosis; ICT; TST; ADA
Introduction
Tuberculosis (TB) is one of the leading infectious causes of death and a major public health problem in the world. According to WHO report; Approximately 2 billion people globally are infected with TB each year. It has recently become more complex due to persistence in aging populations and the rise of drug-resistant strains for developing and developed countries [1,2]. The definitive diagnosis of tuberculosis acid fast bacilli (AFB) stains with smear microscopy and culture of sputum. The conventional Erlich-Ziehl-Neelsen (EZN) method on direct smears for acid fast bacteria AFB is widely used and plays a key role in the diagnosis and also for the monitoring of treatment, but it has low sensitivity. Mycobacterial culture is the gold standard method for detection of tubercle bacilli (70% to 80%), but it is time-consuming [3-5]. A delayed diagnosis of TB contributes to Mycobacterium tuberculosis transmission and mortality due to TB.
A sensitive and specific diagnostic test for the rapid, point- of-care identification of patients with active TB would facilitate early treatment and prevention of transmission.More sensitive and specific tests such as nucleic as it based amplification tests are too expensive for routine laboratory diagnosis of TB [5-7]. Rapid diagnostic tests for TB are needed to facilitate early treatment of TB and prevention of Mycobacterium tuberculosis transmission. Adenosine deaminase (ADA) is an enzyme group involved in the metabolism of purines that catalyzes irreversible hydrolytic deamination of adenosine and deoxyadenosine to inosine and deoxyinosine and is involved in the prolifration and differentiation of lymphocytes, particularly the T-cell subtype [8-10]. ADA acts in the differentiation of lymphoid cells, is secreted in biological fluids during the cellular immune response against intracellular pathogens such as Mycobacterium tuberculosis, HIV, EBV.etc., but can also be increased in other pathological processes.In human patients, the determination of ADA activity in several biological fluids is used as an auxiliary diagnostic tool for TB, mainly for extrapulmonary TB, demonstrating in those clinical forms high sensitivity and specificity [11-15].
The Mantoux tuberculin skin test (TST) is the standard method of determining whether a person is infected with Mycobacterium tuberculosis. It has been used for years as an aid in diagnosing latent tuberculosis infection (LTBI). TST remains the major tool used around the world for the diagnosis of TB infection because of well-established algorithms for test interpretation. In addition TST is easy to use and it has a good cost-effectiveness. Although widely used, TST has limitations; its sensitivity may be reduced by malnutrition, severe TB diseases and immunodeficiency. Decreased TST specificity might occur in settings where non-tuberculous mycobacteria (NTM) are prevalent and in populations who have received Bacillus Calmette-Guerin (BCG) vaccine post-infancy or via multiple vaccinations, although its effect on TST reactions could be modest after 10 years. Additionally, completing the TST requires two health care visits, resulting in loss of reading in approximately 10% of cases. This method is affected by interobserver variability and the positive result do not distinguish recent from remote infection [16,17].
The ICT Tuberculosis test is a rapid, card-based immunochromatographictest for detection of antibodies directed against M. Tuberculosis antigens. An antibody-based serological test for TB has long been sought. Such a test would be attractive because of its potential technical simplicity, rapidity, low cost, and lack of reliance on sputum. Until now, most serological tests developed for TB have had low sensitivities and low specificities. Studies that use recombinant culture filtrate antigens indicate that antibody responses. The combination of multiple antigens in a diagnostic test may therefore increase the sensitivity of an antibody-based test.For the antibody based tests evaluated to date, the presence of anti-M. Tuberculosis antibodies in persons with latent M. Tuberculosis infection, prior active TB disease, vaccination with Mycobacterium bovis bacillus Calmette-Guerin (BCG), or infection with mycobacteria other than M. Tuberculosis may have contributed to the low specificities of the tests for the diagnosis of active TB [18-21].
The new prototype ICT Tuberculosis test is a rapid card- based test for the detection of immunoglobulin G (IgG) antibodies directed against CFP8A, ORF3, CFP10: ESAT-6 and Acry: MPT83 purified M. Tuberculosis antigens immobilized in four lanes on a test strip and uses an anti-human IgG labeled with colloidal gold. Since the target proteins are secreted by actively growing organisms, the test has the potential to have a high degree of specificity for active TB disease [22]. The aim of the present study was to investigate in order to determine the efficiency of a new designed ICT comparison with serum adenosine deaminase activity and TCT in active pulmonary tuberculosis patients, TB contacts and healthy subjects.
Materials and Methods
Subjects
Sera samples from participants were collected from Atatürk Chest Diseases and Thoracic Surgery Education and Research Hospital and Tuberculosis Dispensary of Refik Saydam Hygiene Center in ANKARA, between March 2007 and September 2009. Approval for this study was granted by the institutional review boards of RSHNC, the Turkish Ministry of Health, and Atatürk Chest Diseases and Thoracic Surgery Education and Research Hospital. Written informed consent was obtained from all study subjects. The study included 90 consecutive adult patients with symptoms or signs suggestive of active PTB (patients suspected of having PTB), 8 extra pulmonary (5 smear-positive and 3 smear-negative cases) TB. Two control groups included 48 TB contacts and 153 BCG-vaccinated healthy controls. These control groups were selected from among healthy RSHNC and Atatürk Chest Diseases and Thoracic Surgery Education and Research Hospital staff.
Standard diagnosing and TST Test
From all participants in this program receive a chest X ray and a tuberculin skin test (TST) at entry and an annual evaluation that includes a symptom review for all participants and a TST for participants with a negative prior TST [23]. TB contact controls were no history of TB or vaccinated with BCG, no scar from vaccination with BCG, and a negative TST result. BCG-vaccinated healthy controls were no history of TB but with a scar from vaccination with BCG, a history of vaccination with BCG, or a positive TST result (in duration, -10 mm). A chest X ray and a sputum specimen were not obtained solely for study purposes for the controls since all controls had a prior chest radiograph not compatible with active TB and no symptoms at the most recent annual evaluation or at study entry. Persons less than 16 years of age were excluded from the study.Demographic and clinical data were collected for all study participants through a standardized questionnaire, interview, and physical examination. Venous blood was collected from all participants. Study participants underwent a two-step TST. An in duration of 10 mm in the first or second step was considered a positive TST result.
Patients suspected of having PTB were also given a post anterior and lateral chest X ray and were tested for HIV (by enzyme-linked immunosorbent assay with confirmation by Western blotting). The sputa of all patients suspected of having PTB were tested for acid-fast bacilli (AFB) by the Ziehl-Neelsen technique and were cultured in Lowenstein- Jensen medium. All specimens that were culture positive for mycobacteria were tested by standard biochemical methods to distinguish M. Tuberculosis from other nontuberculous mycobacteria. The reference standard for the diagnosis of PTB was a growth of M. Tuberculosis in a culture of sputum. Venous blood was drawn from the study participants and placed into glass tubes without preservative or anticoagulant for separation of serum and into EDTA- or heparin-coated tubes for whole blood and separation of plasma. The serum remaining after testing by ADA enzyme activity was frozen at 20°C for retesting at a later date by ICT.
ADA enzyme activity
ADA activity was determined in 1 ml serum sample using the colorimetric method described by Giusti and Galanti [24]. Adenosine is deaminated by ADA and the free ammonia is estimated by Berthelot's reaction. One unit of ADA is defined as the amount of enzyme required to release 1 mole of ammonia per minute from adenosine at standard assay conditions.A positive result was defined as a value >40 U/L based on the previous analysis of serum samples with proven TB and non-TB. A positive control sample and two negative control samples for which the ADA value was known were included in each group of clinical samples analyzed.
Protype Immmunochromotographic test (Lateral flow test)
The ICT Tuberculosis diagnostic kit is designed for the detection of antibodies to M. Tuberculosis. Briefly, four highly purified antigens (including ESAT6, CFP8A, Acryl:MPT83, ORF3) secreted by M. Tuberculosis during active infection are immobilized in four lines on the test strip. When serum or plasma applied, it flows past the antigen line. Bound antibody is detected by a goat anti-human IgG antibody conjugated to colloidal gold particles which produces one or more pink lines when bound to human antibody. The whole procedure is completed within 20 minutes. ICT Tuberculosis test doesn't require special equipment and technical skill [22].
Statistical Analysis
Data analysis was made by SPSS (SigmaStat; SPSS Inc; Chicago, IL). Mean, correlation coefficient, and group comparison were made with non parametric Mann-Whitney U test. A value of p<0.05 was considered significant.
Results
In our study, we evaluated 98 TB patients (90active pulmonary TB, 8 extrapulmonary TB), 48 TB contacts and 153 healthy control subjects. All samples were human immunodeficiency virus negative. Clinical signs and symptoms, demographic data, and radiologic results were recorded. The response to intra dermal purified protein derivative (PPD) was evaluated, and an in duration of >11 mm was considered to be a positive result. Among TB patients, 93 (93%) patients were both smear and culture positive, 5patients were smear-negative and culture-positive.
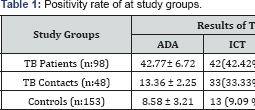
1ADA; Negative; <4OUl, positive≥4OUI
2TST; positive;<15mm,positive≥15mm
The results showed in (Table 1). The results showed that a new designed ICT test was positive for 42 (42.42%) TB patients, 16 (33.33%) TB contacts and healthy controls 13 (9.09 %). The ADA activity was (mean±SD) 42.77±6.72 U/L in TB patients (n= 99), 13.36±2.25 U/L in TB contacts (n= 48), 8.58±3.21 U/L in healthy control subjects (n= 153). TST test was positive for 93 (93%) TB patients, 48 (100%) TB contacts and healthy controls 7 (4.57 %). When compare all three test results each other, TST showed statistical difference compare with ICT and ADA in TB patients and TB contacts. There was not a statistical difference in healthy group for three test.
Discussion
The definitive diagnosis of M. Tuberculosis infection is based on isolation of the organism by culture; however, culture techniques are time-consuming and only provide such a diagnosis after 3 to 4 weeks' incubation. Despite low sensitivity, the acid fast smear plays an important role in the early detection of the presence of acid-fast bacilli in clinical specimens [25].
An increasing number of laboratories have established amplification techniques as an adjunct to standard methods for early diagnosis of tuberculosis, because they provide good rates of positive results with better turnaround times than culture (days versus weeks). In spite of the high sensitivity and specificity of the nested PCR system, its use has been limited by the tedious procedures and the requirement of highly specialised, experienced personnel, expensive instruments and separate rooms for reagents separation, specimen preparation and DNA amplification. These conditions are often lacking in small-scale laboratories and developing countries [26-28].
Various immunological parameters and diagnostic kits are used in the diagnosis of tuberculosis. However, a diagnostic method with high sensitivity and specificity, which can be taken as a substitute for microscopy and culture, cannot be developed [29,30]. Novel diagnostic approaches are urgently needed to improve TB diagnosis and TB control. Antigen detection methods for the diagnosis of TB have been developed as an alternative or a supplement to microscopy, growth based detection, antibody tests, analysis of volatile organic compounds or other bio markers, and to immunological assays. Rapid diagnosis can also be relied on detecting antibodies specific to M. Tuberculosis. The development of serological assays for tuberculosis lias been facilitated by intensive researches into the isolation and characterization of secreted antigens of actively growing M.tuberculosis [31,32].
In the present study, the diagnostic efficiency of the new prototype ICT designed with ESAT6, CFP8A, Acryl:MPT83, ORF3 antigens were compared with serum adenosine deaminase activity and TCT in active pulmonary tuberculosis patients, TB contacts and healthy subjects. Of the new prototype ICT sensitivity was found as 42% in TB patients and 33% in TB contacts. In previous studies reported that the sensitivity of ICT Tuberculosis tests for pulmonary TB is variable, ranging from 20% to 73% [33,34]. In this study ,our new prototype ICTwere found similar with previous studies in TB patients and TB contacts. When the three test (new prototype ICT, TST, ADA) results are compared with each other, TST sensitivity (93%) was higher than ICT and ADA.
Conclusion
In conclusion, serum ADA activity increases in TB patients, therefore it may be supportive parameter for the diagnosis of pulmonary TB. A new designed ICT test was not found to be an effective diagnostic tool for diagnosis of TB. TST is more useful and cheaper than ADA and ICT (42%) in TB patients and TB contacts.
References
- World Health Organization (2014) Global tuberculosis report, ISBN 978 92 4 156480 9.
- Wilson ML (2011) Recent advances in the laboratory detection of Mycobacterium tuberculosis complex and drug resistance. Clin Infect Dis 52(11): 1350-1355.
- World Health Organization (2014) Early detection of tuberculosis: an overview of approaches, guidelines and tools.
- Davis JL, Cattamanchi A, Cuevas LE, Hopewell PC, Steingart KR (2013) Diagnostic accuracy of same-day microscopy versus standard microscopy for pulmonary tuberculosis: a systematic review and meta-analysis. Lancet Infect Dis 13(2): 147-154.
- (2000) Diagnostic Standards and Classification of Tuberculosis in Adults and Children. Am J Respir Crit Care Med 161(4): 1376-1395.
- Greco S, Girardi E, Navarra A, Saltini C (2006) Current evidence on diagnostic accuracy of commercially based nucleic acid amplification tests for the diagnosis of pulmonary tuberculosis. Thorax 61(9): 783790.
- Ling DI, Flores LL, Riley LW, Pai M (2008) Commercial nucleic- acid amplification tests for diagnosis of pulmonary tuberculosis in respiratory specimens: meta-analysis and meta-regression. PLoS One 3(2): 1536.
- Tarhan G, Gumuslu F, Yilmaz N, Saka D, Ceyhan I, et al. (2006) Serum adenosine deaminase enzyme and plasma platelet factor 4 activities in active pulmonary tuberculosis, HIV-seropositive subjects and cancer patients. Journal of Infection 52(4): 264-268.
- Sharma SK, Banga A (2005) Pleural fluid interferon-gamma and adenosine deaminase levels in tuberculosis pleural effusion: a cost- effectiveness analysis. J Clin Lab Anal 19(2): 40-46.
- Greco S, Girardi E, Masciangelo R, Capoccetta GB, Saltini C (2003) Adenosine deaminase and interferon gamma measurements for the diagnosis of tuberculous pleurisy: a meta-analysis. Int J Tuberc Lung Dis 7(8): 777-786.
- Collazos J, Espana P, Mayo J, Martinez E, Izquierdo F (1998) Sequential evaluation of serum adenosine deaminase in patients treated for tuberculosis. Chest 114(2): 432-435.
- Oosthuizen HM, Ungerer JPJ, Bissbort SH (1993) Kinetic determination of serum adenosine deaminase. Clin Chem 39(10): 2182-2185.
- Canbolat O, Ulusoyduran S, O zgen G, Ceyhan I, Gumuslu F, et al. (1999) The comparison of adenosine deaminase activity values with polymerase chain reaction results in patient with tuberculosis. J Clin Lab Anal 13(5): 209-212.
- Baganha MF, Pego A, Lima MA, Gaspar EU (1990) Serum and pleural adenosine deaminase correlation with lymphocytic populations. Chest 97(3): 605-610.
- San JE, Valdes L, Sarendeses AL, Alvarez D, Choman B (1992) Diagnostic value of adenosine deaminase and lysozym in tuberculosis pleuresy. Clin Chem Acta 209(1-2): 73-81.
- Centers for Disease Control and Prevention (CDC) (2005) Guidelines for Preventing the Transmission of Mycobacterium tuberculosis in Health-Care Settings. MMWR 54(17): 1-141.
- Trajman A, Steffen RE, Menzies (2013) Interferon-Gamma Release Assays versus Tuberculin Skin Testing for the Diagnosis of Latent Tuberculosis Infection: An Overview of the Evidence. Pulm Med 2013: 601-737.
- Chan ED, Heifets L, Iseman MD (2008) Immunologic diagnosis of tuberculosis: a review. Tuber Lung Dis 80(3): 131-140.
- Al Zahrani K, Jahdali H, Poirier P (2000) Accuracy and utility of commercialy available amplification and serologic tests tests for the diagnosis of minimal pulmonary tuberculosis. Am J Respir Crit Care Med 162(4): 1323-1329.
- Chang l, Lee E, Son H (2000) Evaluating the usefulness of the ICT test kit for the diagnosis of tuberculosis. J Clin Pathol 53(9): 715-717.
- Pottumarty S, Wells V, Morris A (2000) A comparison of seven tests for serological diagnosis of tuberculosis. J Clin Microbiol 38(6): 22272231.
- Wassie L, Abebe M, Aseffa A, Bobosha K, Zewdie M, et al. (2013) Development of a proof of concept immunochromatographic lateral flow assay for point of care diagnosis of Mycobacterium tuberculosis. BMC Res Notes 6: 202.
- Centers for Disease Control and Prevention (CDC) (2016) Tuberculin Skin Testing, USA.
- Giusti G, Galanti B (1984) Adenosine deaminase: Colorimetric method. In: Bergmeyer HV (Ed.), Methods of enzymatic analysis, New York: Academic Press, USA, pp. 315.
- Daniel TM (1990) The rapid diagnosis of tuberculosis: laboratory techniques applicable in developing country settings. Rev Infect Dis 11(2): 471-478.
- Qanidge JE, Shawar RM, Shinnick TM, Plikaytis BB (1993) Large- scale use of polymerase chain reaction for detection of Mycobacterim tuberculosis in a routine inycobacterial laboratory. JClin Microbiol 31(8): 2049-2056.?
- Balasingham SV, Davidsen TI, Szpinda SA, Tonjum T (2009) Molecular diagnostics in tuberculosis: basis and implications for therapy. Mol Diagn Ther 13(3): 137-151.
- CDC (2009) Updated Guidelines for the Use of Nucleic Acid Amplification Tests in the Diagnosis of Tuberculosis. MMWR 58(1): 7-10.
- Daniel TM, Debanne SM (1987) The serodiagnosis of tuberculosis and other mycobacterial disease by enzyme-linked immunosorbent assay. Am Rev Respir Dis 135(5): 1137-1151.
- World Health Organization (WHO) (2011) Policy statement. Commercial serodiagnostic tests for diagnosis of tuberculosis, Geneva, Switzerland.
- Steingart KR, Flores LL, Dendukuri N, Schiller I, Laal S, et al. (2011) Commercial Serological tests for the Diagnosis of Active Pulmonary and Extrapulmonary Tuberculosis: An updated Systemic Review and Meta-Analysis. PLoS Medicine 8(8): 1001062.
- Bartolini A, Strohmeyer M, Bartalesi F, Messeri D, Tortoli E, et al. (2003) Evaluation of a rapid immuno chromatographic test for the serologic diagnosis of tuberculosis in Italy. Clin Microbiol Infect 9(7): 632-639.
- Khan N, Mian I, Zia-Ullah MJ (2004) Performance of ICT-TB test in thedetection of pulmonary and extra-pulmonary tuberculosis. J Ayub Med Coll Abbottabad 16(2): 55-56.
- Perkins MD, Conde MB, Martins M, Kritski AL (2003) Serologic diagnosis of tuberculosis using a simple commercial multiantigen assay. Chest 123(1): 107-112.






























