Mini Review: A Case Review of Bovine Lymphosarcoma in a 6-Year-Old Angus Cow and the Economic Impact of BLV
Cassandra M Powers* and Frederick Tippett
Department of Pathobiology, Tuskegee University College of Veterinary Medicine, USA
Submission: July 21, 2022; Published: September 07, 2022
*Corresponding author: Cassandra M Powers, Department of Pathology, Purdue University College of Veterinary Medicine, West Lafayette, USA
How to cite this article: Cassandra M P, Frederick T. Mini Review: A Case Review of Bovine Lymphosarcoma in a 6-Year-Old Angus Cow and the Economic Impact of BLV. Arch Anim Poult Sci. 2022; 1(5): 555575. DOI: 10.19080/AAPS.2022.01.555575
Abstract
Bovine lymphosarcoma is a neoplasm of lymphocytes that is commonly associated with bovine leukemia virus (BLV). BLV can be transmitted both vertically and horizontally, and transmission is highly dependent on farm control measures. BLV-induced tumors usually arise from the CD5+ IgM+ B-cell subpopulations. Clinical signs associated with development of lymphosarcoma are highly variable. This is because the clinical symptoms observed depend on the location of the tumors and the affected organs. BLV has been eradicated in over 20 countries via traditional antibody testing and slaughter methods. This case review looks at a 6-year-old, angus beef cow that presented to the hospital for sudden death. Necropsy and histopathological examination revealed a marked lymphadenopathy and lymphocytic infiltration of multiple organs, consistent with bovine lymphosarcoma.
Keywords: Cattle; Bovine; Lymphosarcoma; Lymphoma; Leukosis; BLV
Introduction
Bovine lymphosarcoma (aka: lymphoma) is a neoplasm of lymphocytes. It is commonly associated with bovine leukemia virus (BLV). BLV is an oncogenic member of the Deltaretrovirus genus and is closely related to the human T-cell leukemia virus [1,2]. Bovine leukemia virus (BLV) is transmitted horizontally to cows via arthropods such as the horsefly. It can also be transmitted to calves either transplacentally or via the ingestion of colostrum from a BLV-infected cow [1]. Cattle can also be infected iatrogenically, usually via surgical instruments or sleeves contaminated with infected blood during rectal palpation [3]. It may be sporadic and unrelated to BLV. Sporadic lymphosarcoma can manifest in three different but similar forms: juvenile, thymic, and cutaneous [4].
Only a small fraction, about 1.4%, of all BLV infected cattle actually develop clinical symptoms, such as malignant lymphosarcoma [5]. The majority of infected cattle are asymptomatic. Bovine lymphosarcoma, however, is still the most common neoplasm of dairy cattle in general. Both beef and dairy cattle are susceptible to BLV infection and BLV-associated lymphosarcoma. However, due to variation in management practices the disease is more commonly detected in dairy herds [6].
BLV has been eradicated in over 20 countries via traditional antibody testing and slaughter methods. However, the United States has been experiencing an increasing prevalence of BLV in the absence of efforts to control transmission [6-9]. BLV has a significant impact on the bovine multiple immune system, affecting both cellular and humoral immunity, and leading to decreased dairy production and productive lifespan [7,9,10]. Traditional control methods for BLV are not economically feasible for many nations where the average herd antibody prevalence is about 50%. ELISA screening coupled with qPCR helps prioritize the most infectious cattle for segregation or culling. Breeding cattle for resistance to BLV can also be performed [7,9,10,11]. At present, no treatment or vaccine has proven effective for the control of BLV. Due to the marked effect BLV has on cattle population, the industry, and overall human health, there is an urgent need for the industry as a whole to review and implement control measures and try to eradicate this disease from the United States’ cattle population [6-8].
The case reviewed in this paper is an example of bovine lymphosarcoma in a herd with marked pathologic lesions that caused acute death in an angus cow.
Case Summary
A 6-year-old, angus beef cow presented to the Tuskegee University Teaching Hospital for sudden death. The only clinical abnormality that the clinician noted prior to necropsy was multiple enlarged, palpable lymph nodes.
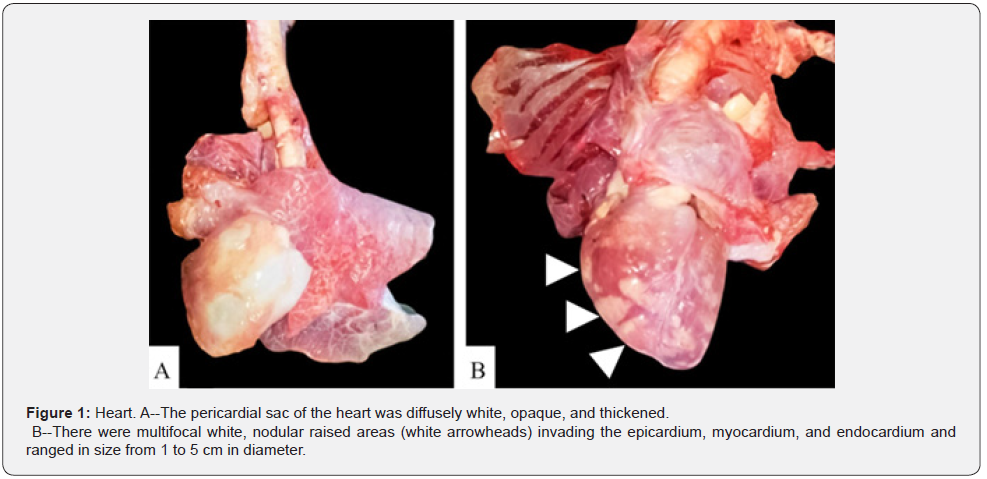
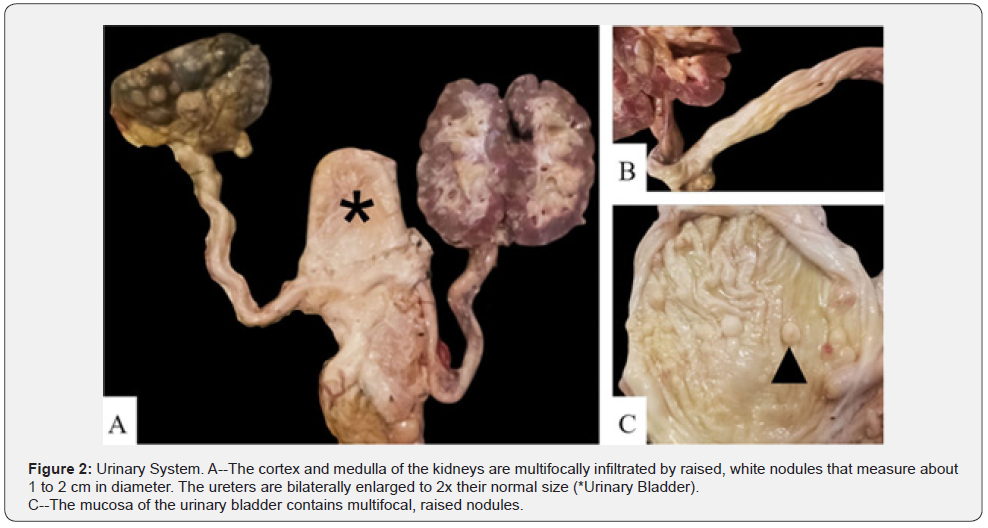
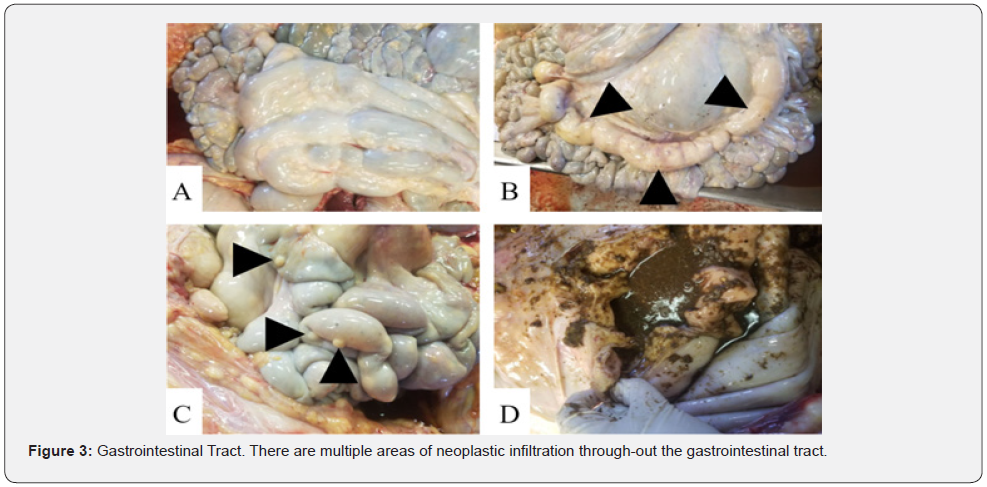
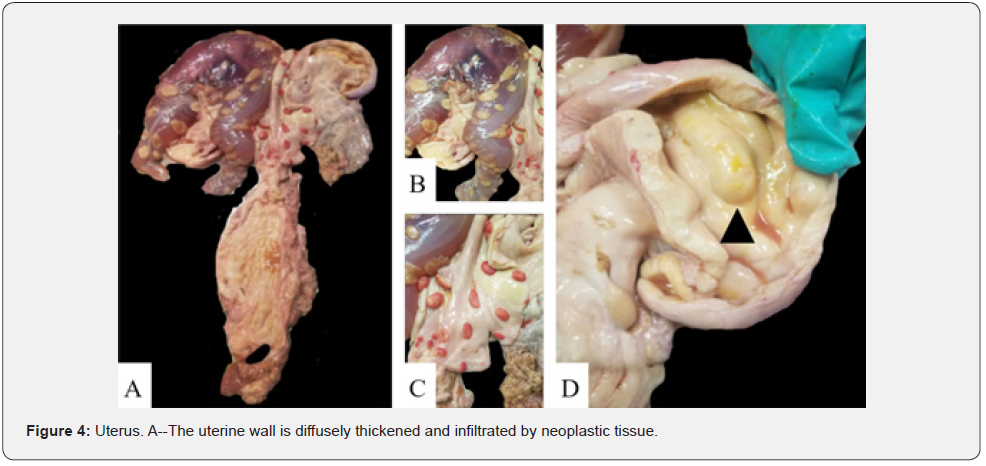
The carcass had a BCS of 4/9 based on adequate internal and external adipose stores. Upon dissection, multiple subcutaneous lymph nodes were enlarged 3-10x their normal sizes. The heart had multifocal white, nodular raised areas invading the epicardium, myocardium, and endocardium and ranged in size from 1 to 5 cm in diameter (Figure 1). The kidneys bilaterally contained multifocal white, raised nodules that invaded into both the cortex and medulla. The ureters were distended 2x their normal size and had raised nodules within their mucosa that measured 1 to 2 cm in diameter. The urinary bladder also contained similar nodules infiltrating its mucosa (Figure 2). There was a chain of enlarged and coalescing lymph nodes within the jejunal mesentery, with the largest measuring 30 x 10 x 5.5 cm (Figure 3). The vaginal mucosa had raised mucosa nodules and the caruncles were diffusely reddened. The right horn of the uterus contained a fetus measuring 24 cm from head to rump (Figure 4).
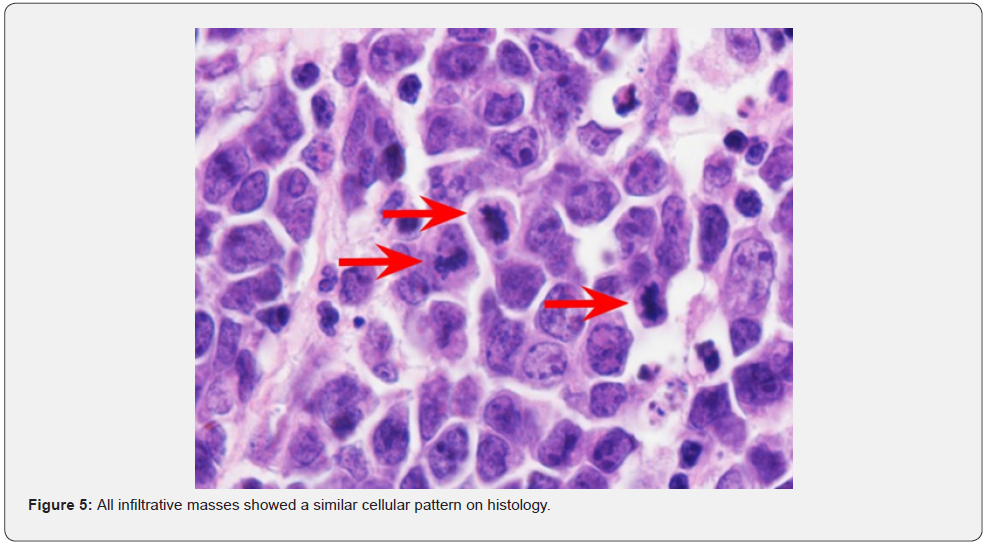
Histopathology of the masses was performed following the necropsy. All infiltrative masses showed a similar cellular pattern. Infiltrating normal tissue, are multifocal well-demarcated and non-encapsulated densely cellular masses composed of sheets of round cells on scant eosinophilic fibrovascular stroma. The cells have distinct cell borders and scant basophilic cytoplasm with a low nuclear to cytoplasm ratio. The nuclei are round and stippled with 1-4 nucleoli. There are 14 mitotic figures in a 2.37 mm2 area. There is marked anisocytosis and anisokaryosis (Figure 5).
Based on the gross and histological results a diagnosis of bovine lymphosarcoma was given to this animal. Additional testing for BLV was not performed. Although this case was submitted as a herd health concern, no other animals on the farm were affected so it is highly likely that this was a sporadic case of bovine lymphosarcoma. Due to financial constraints, additional ancillary testing, like a molecular PCR, could not be performed.
Discussion
Bovine Leukemia virus (BLV), a single-stranded diploid RNA, that occurs mainly in cattle [1]. It has also been reported to occur naturally in zebus, buffalos, and capybaras. BLV can induce B-cell lymphosarcoma and can infect various immune cell populations. However, BLV-induced tumors usually arise from the CD5+ IgM+ B-cell subpopulations. Lymphocyte counts are not usually elevated in BLV-infected but clinically normal cattle, but CD5+ IgM+ B-cells subpopulations are still increased. Also, BLV-infected carriers who appear clinically normal may still exhibit signs of reduced milk production, a high incidence of infectious disease, and reproductive inefficiency [2].
In practice, most animals remain persistently infected with no outward signs of infection. Cattle that do develop lymphosarcoma often develop lesions in the heart, uterus, and abomasum [12]. However, lesions can technically manifest anywhere in the body. Clinical signs associated with development of lymphosarcoma are highly variable. This is because the clinical symptoms observed depend on the location of the tumors and the affected organs [4,12]. Ultimately, the clinical signs of BLV-induced tumors primarily involve digestive disturbance, weight loss, weakness, paresis, reduced milk production, loss of appetite, enlarged lymph nodes, and possibly death [2,4]. Grossly, lymphosarcoma often appears as yellow to tan, discrete nodular masses or a diffuse tissue infiltrate. Histologically, the tumor masses are composed of densely packed, monomorphic lymphocytic cells.
Transmission of BLV is usually through the transfer of blood and blood products that contain infected lymphocytes. These include both medical products and procedures, and normal farm practices like tattooing and dehorning. In addition to transplacental infection, calves fed colostrum from BLV-positive cows can also become infected as well. However, since colostrum contains a large amount of antibodies that have a protective effect, these antibodies dampen the infectious potential of BLV. Most BLV transmission is horizontal. Nose to nose contact between BLV-negative and BLV-positive cattle is a major risk factor. Insect vectors also transmit the virus between cattle. Once infected, cattle develop a lifelong antibody response [1,4].
Viral infection of BLV is diagnosed by serology or virology. Serology is the most common and reliable way to diagnose infection with BLV [1,4]. PCR is a sensitive and specific assay for diagnosis of BLV infection in peripheral blood lymphocytes. It should be noted that a positive serology or virology for BLV confirms the presence of the virus, but does not directly indicate the presence of lymphosarcoma. The diagnosis of lymphosarcoma must be made by cytology or histopathology [4].
The prevalence of BLV infection varies around the world. The virus has been eradicated in many European countries, Australia, and New Zealand. However, the prevalence of the disease is still fairly high in the U.S. The most recent surveys in the USA estimate that 44% of dairy and 10% of beef cattle are infected with the virus. Prevalence tends to increase on dairies with increasing herd size [4,7,8,11]. Steps can be taken to reduce the spread of disease, including: creating and enforcing adequate cleaning and disinfecting protocols; Clean and disinfecting dehorning, castration, and tattooing equipment; feeding calves whose mothers are BLV positive replacement milk; changing rectal sleeves in between cows; implementing artificial insemination or embryo transfer techniques to limit herd contact; single use, disposable needles for blood and IM injections; and implementing adequate fly control measures [4,7,10,11].
There is no set treatment against BLV or lymphosarcoma in cattle. However, an infected patient may be treated with parenteral corticosteroids to help alleviate the severity of clinical signs. Possible BLV vaccines are currently being worked on. One group has developed an attenuated vaccine by targeted mutations and deletions and is seeking regulatory approval for use [9-11].
References
- Balić D, Lojkić I, Periškić M, Bedeković T, Jungić A, et al. (2012) Identification of a new genotype of bovine leukemia virus. Arch Virol 157(7): 1281-1290.
- Polat M, Takeshima SN, Aida Y (2017) Epidemiology and genetic diversity of bovine leukemia virus. Virol J 14(1): 209.
- Oguma K, Suzuki M, Sentsui H (2017) Enzootic bovine leukosis in a two-month-old calf. Virus Res 233: 120-124.
- Nagy DW (2016) Overview of Bovine Leukosis: (Bovine lymphosarcoma, Leukemia, Malignant lymphoma). Merck Veterinary Manual.
- Tsutsui T, Kobayashi S, Hayama Y, Yamamoto T (2016) Fraction of bovine leukemia virus-infected dairy cattle developing enzootic bovine leukosis. Prev Vet Med 124: 96-101.
- Juliarena MA, Barrios CN, Lützelschwab MC, Esteban EN, Gutiérrez SE (2017) Bovine leukemia virus: current perspectives. Virus Adaptation and Treatment 2017(9): 13-26.
- Frie MC, Coussens PM (2015) Bovine leukemia virus: a major silent threat to proper immune responses in cattle. Vet Immunol Immunopathol 163(3-4): 103-114.
- LaDronka RM, Ainsworth S, Wilkins MJ, Norby B, Byrem TM, et al. (2018) Prevalence of bovine leukemia virus antibodies in US dairy cattle. Vet Med In 2018.
- Rodríguez SM, Florins A, Gillet N, De Brogniez A, Sánchez-Alcaraz MT, et al. (2011) Preventive and therapeutic strategies for bovine leukemia virus: lessons for HTLV. Viruses 3(7): 1210-1248.
- Bartlett PC, Ruggiero VJ, Hutchinson HC, Droscha CJ, Norby B, et al. (2020) Current Developments in the Epidemiology and Control of Enzootic Bovine Leukosis as Caused by Bovine Leukemia Virus. Pathogens 9(12): 1058.
- Miller JM, Vander Maaten MJ (1982) Bovine Leukosis--Its Importance to the Dairy Industry in the United States. J Dairy Sci 65(11): 2194-2203.
- Valli T, Kiupel M, Bienzle D (2016) Chapter 3: Hematopoietic System. In: Jubb, Kennedy, and Palmer's Pathology of Domestic Animals, vol 3, Elsevier, St. Louis, MO, USA, Pp. 235-237.






























