Pharmacokinetics of Cefepime after Single Intravenous and Intramuscular Administrations in Broiler Chickens
Elsayed MGA, Aboubakr M* and Rabea S
Department of Pharmacology, Faculty of Veterinary Medicine, Benha University, Egypt
Submission: July 23, 2019; Published:December 03, 2019
*Corresponding author: Aboubakr M, Department of Pharmacology, Faculty of Veterinary Medicine, Benha University, 13736, Moshtohor, Toukh, Qalioubeya, Egypt
How to cite this article: Elsayed MGA, Aboubakr M, Rabea S. Pharmacokinetics of Cefepime after Single Intravenous and Intramuscular Administrations 0051 in Broiler Chickens. Arch Anim Poult Sci. 2019; 1(3): 555561. DOI: 10.19080/AAPS.2019.01.555561
Abstract
The pharmacokinetic parameters of cefepime were studied following single intravenous and intramuscular injection in healthy broiler chickens. Following a single intravenous injection of 100 mg cefepime/kg b. wt in healthy chickens, cefepime could be detected therapeutically for 24 hours post intravenous injection with mean value 0.21μg/ml. The serum concentration–time curve of cefepime following intravenous injection showed that the drug obeyed a two compartments open model with elimination half- life (t0.5(β) =4.54 h), volume of distribution (Vdss = 3.41L/kg) and total body clearance of the drug (CLtot= 0.916L/hr/kg). Following a single intramuscular administration of 100mg/kg body weight cefepime in healthy chickens, the peak plasma concentration (Cmax) was 14.42μg/ml was achieved at a maximum time (Tmax) of 2.12 h. The intramuscular bioavailability of cefepime in healthy chickens was 85.22%. The serum concentration of cefepime following intravenous and intramuscular administration could be detected till 24 hours and remaining above MIC for a majority of cefepime sensitive bacteria, this indicate that cefepime is the drug of choice for treatment of infection in broiler chickens.
Keywords: Pharmacokinetics; Cefepime; Broiler chickens
Introduction
Cefepime, is a parenteral fourth-generation cephalosporin antibiotic with an extended spectrum of antimicrobial activity and a pharmacokinetic profile similar to that of ceftazidime [1]. It is active against many Gram-positive and Gram-negative bacteria, including most members of the family Enterobacteriaceae, Pseudomonas aeruginosa, and Staphylococcus aureus [2], with reduced susceptibility to extended-spectrum β-lactamases [3]. The chemical structure of cefepime allows it to bind to penicillin-binding proteins and to penetrate through the outer membrane of Gram-negative bacteria more rapidly than most cephalosporins. In humans, it is approved for treatment of lower respiratory tract, intra-abdominal, complicated and uncomplicated urinary tract infections, and uncomplicated skin and skin structure infections [1].
Cefepime have been described in various animals’ species as in rabbits [4], Foals and dogs [5], buffalo calves [6], cow calves [7], goats [8], sheep [9] and horses [10]. Currently, there are little data on the pharmacokinetics of cefepime in broiler chickens. Therefore, the aim of present work was undertaken to study the pharmacokinetic parameters of cefepime after intravenous and intramuscular injection in chickens. Also, the bioavailability of cefepime was calculated after intramuscular administration in healthy chickens.
Material and Methods
Drugs
Cefepime was used in this study under trade name (Cefepime®, sterile vial) each vial contains cefepime hydrochloride monohydrate 1019mg equivalent to cefepime 1000mg. Each ml of reconstituted solution contains cefepime hydrochloride monohydrate equivalent to 1000mg cefepime, which was manufactured by Pharco B International Company, Alexandria, Egypt.
Experimental Birds"
Six clinically healthy Hubbard chickens of four weeks of age weighting about 1500 to 2000gm, each chosen randomly from poultry farm, Qalubia government, Egypt, were used in investigation. Chickens were feed balanced ration free from antibacterial for two weeks to ensure complete excretion of any drugs from their bodies. Water and feed free from antibacterial additives were provided ad- libitum.
Experimental Design
Six healthy chickens were administered intravenously into the wing vein with single dose of 100mg cefepime/kg b.wt. These chickens were left for 15 day after the intravenous injection to ensure complete elimination of cefepime from their bodies andthen administered intramuscularly with 100mg cefepime/kg b.wt, to determine the bioavailability of cefepime in healthy chickens.
Collection of Samples
About one milliliter of blood was taken from the right-wing vein, following injection of the drug. Blood samples were collected at 5, 10, 25, 30 minutes, 1, 2, 4, 6, 8, 12 and 24 h after single intravenous and intramuscular injection of cefepime.
All blood samples were collected in sterilized centrifuged tubes and allowed to clot. Serum was separated by centrifugation for 15 minutes at 3000 r.p.m. Sera were kept frozen until assayed.
Analytical Procedure
Cefepime were assayed using microbiological method of antibiotic [11], using E. coli ATCC 6633 as test organism for cefepime. The test organism was obtained from Department of Microbiology, Animal Health Research Institute, Dokki, Giza, Egypt. Three plates were used for each sample. One well in each plate was filled with reference concentration (6.25μg/ml of cefepime in distilled water or healthy chickens’s serum). The plates were incubated at 37 ͦC for 24 h, and then the diameter of inhibitory zones was measured. The average diameter of inhibition zone of the samples was corrected by using the diameter of the reference concentration as mentioned previously in the preparation of the standard curve. From the standard curve, the concentration corresponding to the correct values of the zone diameter were obtained.
Pharamacokinetic analysis
The pharmacokinetics parameters were calculated by Winnonlin program, version 4.1.
Results
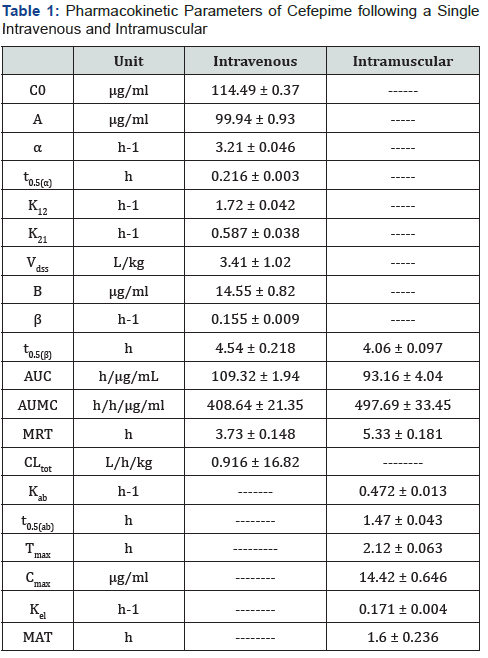
Following a single intravenous injection of 100mg cefepime/ kg b. wt in healthy chickens, cefepime could be detected therapeutically for 24 hours post intravenous injection. The plasma concentration-time curve of cefepime following intravenousinjection showed that the drug obeyed two compartments open model. The disposition kinetics of cefepime following a single intravenous and intramuscular injection were recorded in Table 1 and showed in Figure 1.
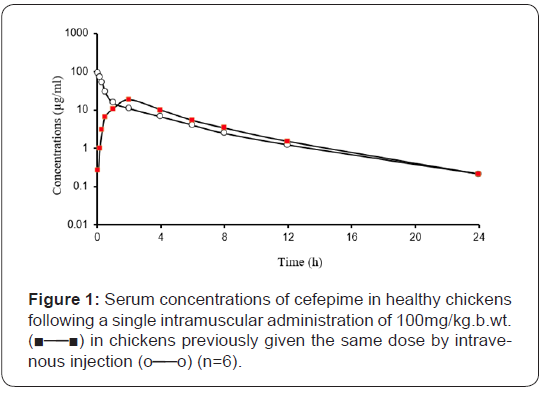
Adminstration of 100Mg/Kg B.Wt. in Healthy Chickens (N=6).
Discussion
In the present investigation, intravenous injection of 100 mg of cefepime /kg b.wt in healthy chickens showed that the disposition best fitted a two compartments open model. The obtained result was consistent with those reported for cefepime in horses [11], in ewe [12], in sheep [9], in goats [13] and in broiler chickens [14]. Also, this phenomena is in agreement with those reported for other cephalosporine, as ceftazidime in a rabbit [15], ceftriaxone in cats [16] and in goats [17], cefuroxime in dogs [18], cefquinome in chickens [19] and cephradine in broiler chicken [20,21].
Following a single intravenous injection of 100 mg cefepime/ kg b.wt. in healthy chickens, the drug showed high serum level (90.77μg/ml) at 5 minutes post injection, then its concentration decreased gradually till reached its minimum level at 24 hours post injection (0.21μg/ml).
The Vdssvalue was shorter than the data reported after intravenous administration of cefepime in rabbit 1.168L/kg [4]. On the other hand, volume of distribution was higher than these recorded for cefepime in calves 0.21L/kg [22] and ewes 0.32L/kg [12], and cefepime in goats 0.44 L/kg [13]. for cefepime was 3.41L/kg suggesting higher penetration through biological membranes and tissue distribution after intravenous administration in broiler chickens. The obtained
The elimination half-life (t0.5(β)) of cefepime following single intravenous injection of 100 mg/kg b.wt was equal to 4.54 h. This observation agreed with the data reported after intravenous administration of ceftazidime in dromedary camels 2.85 h, [23] and cefepirome in cow calves 2.41 h [24] and cephradine in chicken 2.79 h [20]. On contrast, the obtained value was longer than those recorded in cefuroxime in goats 1.48 h [25], ceftazidime in rabbits 2.22 h [15], cefepime in ewes 1.76 h [12], ceftriaxone in cats 1.73 h [16], cefquinome in piglets 1.85 h [26], cefquinome in ducks 1.57 h [27], ceftriaxone in goats 1.82 h [17], and cefoperazone in goats 1.97 h [14]. On the other hand, it was shorter than those showed in cephradine in goats 4 h [28], and cefepime in goats 3.34 h [13].
Such differences in pharmacokinetics parameters are relatively common and frequently related to inter-species variations assay methods used, the time between blood samplings, and/or the health status and age of the animals [29].
The rate of total body clearance (CLtot) of cefepime following intravenous injection was 0.916 L/kg/h. This value was shorter than the clearance values of cefepime in goats 0.098 L/Kg/h [13]. This value was longer to these values reported in other cephalosporins as ceftriaxone in calves 0.31 L/Kg/h [30], ceftriaxone in cats 0.37 L/Kg/h [16], cefquinome in ducks 0.22L/Kg/h [27], cefquinome in chickens 0.35 L/Kg/h [19] and ceftriaxone in goat 0.23L/Kg/h [17]. The variation in total body clearance might be attributed to specific interspecies variation [31].
Following a single intramuscular administration of 100mg/ kg b.wt, the drug reached its maximum concentration (18.35μg/ ml) at 2 hours and could be detected in serum in therapeutic level (0.262μg/ml) for 24 hours. On the bases of cefepime concentration for avian pathogenic microorganisms, it was suggested that intramuscular administration of 100mg/kg b.wt with 24 hours intervals should be adequate for control of avian bacterial diseases. These concentrations exceeded the minimum inhibitory concentrations for a majority of cefepime sensitive bacteria is in the range of 0.01 to 4.0 μg ml-1 [32].
In the present study, cefepime was rapidly absorbed from site of injection in chickens after single intramuscular administration with a short absorption half-life (t0.5(ab) = 1.47 h). This value was nearly similar to that reported for cefuroxime in goats 1.379 h [25], ceftiofur in fresian calves 1.01 h [33], and cefotaxime in muscovy ducks 1.58 h [34]. On contrast, this value was higher than that reported for ceftazidime in rabbit 0.779 h; [15], cephradine in goat 0.64 h [28], and cefquinome in chickens 0.07 h [19]./p>
Maximal serum concentration (Cmax) was (14.42μg/ml) achieved at (tmax) (2.12 h). These value were higher than to those recorded for cefepime in cow (Cmax) (8.93 μg/ml) and (tmax) (4.5hours) [35] and lower than to those recorded for cefepime in calves and ewe (cmax) (31.9 and 21.7μg/ml), (tmax) (1.1 h) [12], in sheep (cmax) (26.34μg/ml), (tmax; 0.75 h) cand (max; 28.76 μg/ ml), (tmaxsub>; 0.27 h) [9, 36], respectively. In comparison with other cephaloporins, these values were higher than that reported in ceftiofur in parrots (cmax) (11 μg/ml) and (tmax) (1 hour) [37], in camels (cmax) (10.34 μg/ml ) and (d) (1.22 hours ) [38] and in cow (cmax) (7.83 μg/ml ) and (d) (1.55 hours ) [39]. Also, these values were lower than that reported for cefepime in calves and ewe (cmax) (21.7μg/ml ) , (d) (1.1 hours ) and (cmax) (31.9μg/ml) , (tmax) (1.1 hours) [22, 12], ceftriaxone in goat and calves (cmax) (23.6 μg/ml ) , (tmax) (0.7 hours ) and (cmax) (54.4 μg/ml ) , (tmax) (0.33 hours ) [40, 16], respectively). These variationcs in max and tmax are common and might be attributed to anatomical differences between species, healthy status and those administered in each case interspecies variation, assay methods used, age, breed and health status of the animal and formulation of the drug used [41].
The elimination half-life (t0.5(β)) expresses the overall rate of elimination of the drug and allows the predication of drug accumulation. The t0.5(β) after intramuscular administration was 4.06 ± 0.097 hours. These value was lower than to that reported for cefepime in cefepime in sheep 5.17 hours [9], but higher than cefepime in calves and ewe (3.02 ± 0.18 and 2.06 ± 0.11 hours) [22, 12]. In comparison with other cephaloporins, was higher than cefuroxime in goats = 2.088 hours [25], ceftazidime in rabbit = 2.12 hours [15].
The bioavailability of cefepime in healthy chickens, which estimated the rate and extent of the dose entered the systemic circulation after oral administration was 85.22%. This percent indicated a good absorption of cefepime after intramuscular administration. This value was nearly similar to those recorded for cefepime in ewe [12], ceftriaxone in goats and calves (85% and 85.72 %) [40, 16], cefoperazone in camels (89.42%) [42]. On the other hand, this value was lower than the bioavailabilities recorded for ceftazidime in rabbits (96.6%) [15], cefquinome in ducks and chicken (93.28% and 95.81 %) [27, 19] and ceftazidime in camel (93.72%) [23]. But higher than these values recorded in cephradine in goats (73.9%) [28].
In this study, the in vitro plasma protein binding experiment showed that cefepime displayed a low level of binding to plasma proteins (13.89 %) to broiler chicken plasma. The results of in vitro protein binding of antibiotics vary considerably depending upon the method and experimental conditions. This value was nearly similar to these reported values of 13.3% for ceftazidime in rabbits [15], and 15.65% for cefquinome in sheep and 14.42% in goats [13]. On contrast, this value was lower than those reported values of 39.68% for ceftiofur sodium in friesian calves [33].
Conclusion
From the present study it could be concluded that, the intramuscular bioavailability of cefepime is excellent and this value revealed a better absorption from its site of intramuscular administration. The serum concentration of cefepime following intravenous and intramuscular administration could be detected till 24 hours and remaining above MIC for a majority of cefepime sensitive bacteria is in the range of 0.01 to 4.0 μg ml-1, this indicate that cefepime is the drug of choice for treatment of infection in broiler chickens.
References
- Okamoto MP, Nakahiro RK, Chin A, Bedikian A (1993) Cefepime clinicalpharmacokinetics. Clin. Pharmacokinet 25(2): 88 102.
- Thornsberry C, Brown SD, Yee YC, Bouchillon SK, Marler JK, et al. (1993) In-vitro activity of cefepime and other antimicrobials: survey of European isolates. J Antimicrob Chemother 32: 31 53.
- Jacoby GA, Carreras I (1990) Activities of beta-lactam antibioticsagainst Escherichia coli strains producing extended-spectrum betalactamases.Antimicrob. Agents.Chemother 34(5): 858 862.
- Abd El Aty AM, Goudah A, Mouneir SM, Sunwoo YE, Jang JH, et.al. following intravenous administration to rabbits. Vet Res Commun
- 5. Gardner SY, Papich MG (2001) Comparison of cefepime pharmacokinetics in neonatal foals and adult dogs. J Vet Pharmacol Ther 24(3): 187-192.
- 6. Joshi B, Sharma SK (2007) Pharmacokinetic disposition and bioavailability of cefepime in buffalo calves. J. Vet. Pharmacol. Ther 30(5): 500-502.
- 7. Patel UD, Bhavsar S, Thaker AM (2006) Pharmacokinetics and dosage regimen of cefepime following single dose intravenous administration in calves. Iran J Pharmacol Ther 5: 127-130.
- 8. El Rabbat AN, Abdel wadood MH, Sayed M, Mousa SH (2010) High performance Liquid chromatographic determination and Pharmacokinetic study of cefepime in goat plasma and milk after precolumn derivatization with Hg(1). J Sep Sci 33: 2599-2609..
- 9. Patel PN, Patel UD, Bhavsar SK, Thaker AM (2010) Pharmacokinetics of cefepime following intravenous and intramuscular administration in sheep. Iran J Pharmcol Ther 9(1): 7-10..
- 10. Guglick MA, Mac ALisster CG, Clarke CR, Pollet R, Hague C, Clarke JM (1998) Pharmacokinetics of cefepime and comparison with those of ceftiofur in horses. Am. J. Vet. Res 59(4): 458-463..
- 11. Arret B, Johnson DP, Kirshbaum A (1971) Outline of details for microbiological assay of antibiotics: Second revision. J Pharm Sci 60(11): 1689-1694..
- 12. 12. Ismail MM (2005) Pharmacokinetics of cefepime administered by intravenous and intramuscular routes to ewes. J. Vet. Pharmacol. Therap 28 (6): 499-503..
- 13. El-Hewaity M (2014) Influence of Flunixin on the disposition kinetics of cefepime in goats. Adv. Pharmacol. Sci 2014: 471517..
- 14. Attia AT, Hanan AA, Mohamed HS (2018) Disposition kinetics of cefepime in healthy and experimentally Salmonella typhimurium infected broiler chicken SVU-IJVS 1 (1): 66-81..
- 15. Abd El Aty AM, Goudah A, Abo El Sooud K (2001) Pharmacokinetics, intramuscular bioavailability and tissue residue profiles of ceftazidime in a rabbit model. Dtsch Tier Wochen 108(4): 168-171..
- 16. Albarellos GA, Kriel VE, Landoni MF (2007) Pharmacokinetics of ceftriaxone after intravenous, intramuscular and subcutaneous administration to domestic cats J Vet Pharmacol Therap 30(4): 345- 352..
- 17. Prashant MN, Karmakar UK, Suman S, Mishra AK, Datta BK et.al. (2014) Disposition kinetics of ceftriaxone and sulbactam (1:1) in black bengal goats with experimental mastitis. Asian J Pharmacol Toxicol 2(7): 7-17..
- 18. Zaho L, Li Q, Li X, Yin R, Chen X, et.al. (2012) Bioequivalance and population pharmacokinetic modeling of two forms of antibiotic, cefuroxime lysine and cefuroxime sodium, after intravenous infusion in beagle dogs. J Biomed Biotechnol 2012: 507294..
- 19. Xie W, Zhang X, Wang T,Du S (2013) Pharmacokinetic analysis of cefquinome in healthy chickens. Br. Poult. Sci 54(1): 81-86..
- 20. Elsayed MGA, Aboubakr M Rabea S (2016) Pharmacokinetics and tissue residues of cephradine in healthy and experimentally Salmonella Enteritidis infected chickens. World J Pharm Pharm Sci (WJPPS) 5(6):61-74..
- 21. Aboubakr M, Elbadawy M (2017) Bioavailability, pharmacokinetics and tissue residues of cephradine (Atocef Forte®) in healthy and colisepticemic broiler chickens. Int J Pharmacol Toxicol 5(1): 57-60..
- 22. Ismail MM (2005) Bioavailability, pharmacokinetics and tissue residues of cephradine (Atocef Forte®) in healthy and colisepticemic broiler chickens. Int J Vet Res Commun 29(1): 69-79..
- 23. Goudah AM, Hasabelnaby SM (2013) Pharmacokinetics and distribution of ceftazidime to milk after intravenous and intramuscular administration to lactating female dromedary camels (Camelus dromedaries). J. Am. Vet. Med. Assoc 243(3): 424-429..
- 24. Patel RB, Bhavsar SK, Solanki PF, Patel JH, Varia RD, et.al. (2013) Pharmacokinetics of cefpirome following intravenous and intramuscular administration in cow calves. Sci. Int 1(11): 371-374..
- 25. Abo El Sooud K, El Banna HA, Hanafy MS, Goudah A (2000) Pharmacokinetics and intramuscular bioavailability of cefuroxime sodium in goats. Res Vet Sci 69(3): 219-224..
- 26. Li XB, Wu WX, Su D, Wang ZJ, Jiang HY, et al. (2008) Pharmacokinetics and bioavailability of cefquinome in healthy piglet. J. Vet. Pharmacol. Therap 31(6): 523-527..
- 27. Yuan L, Sun J, Wang R, Sun L, Zhu L, et.al. (2011) Pharmacokinetics and bioavailability of cefquinome in healthy ducks. Am. J. Vet. Res 72(1): 122-126..
- 28. El Sayed MG, Atef M, El Komy AA (1994) Disposition kinetics of cephradine in normal and Escherichia coli infected goats. Dtsch. Tier. Wochen 101(2): 56-60..
- 29. Haddad NS, Pedersoli WM, Ravis WR (1985) Pharmacokinetics of gentamicin at steady state in ponies: serum, urine, and endometrial concentrations. Am J Vet Res 46(6): 1268-1271..
- 30. Johal B, Srivastava AK (1999) Disposition kinetics and dosage regimen of ceftriaxone in crossbred calves. Acta Vet Hung 47(2): 243-248..
- 31. Baggot JD (1980) Distribution of antimicrobial agents in normal and diseased animals. JAVMA 19(176): 1085-1090..
- 32. Cynamon MH, Palmer GS, Song TB (1987) Comparative in vitro activities of ampicillin, BMY-28142 and imipenem against Mycobacterium avium complex. Diag Microbial Infect Dis 6(2): 151-155..
- 33. El Gendy AAM, Tohamy MA, Ismail M (2007) Comparative pharmacokinetic and renal clearance study of ceftiofur in cross breed Freisian and buffalo calves. Beni-Suef Vet MedJ 17(1): 69-77..
- 34. Aboubakr M (2016) Bioavailability and pharmacokinetics of cefotaxime in Muscovy ducks. Int J Pharmacol Toxicol 4(1): 93-95..
- 35. Patel NN, Patel HB, Patel SD, Patel JH, Bhavsar SK, et.al. (2012) Effect of ketoprofen co-administration or febrile state on pharmacokinetics of cefepime in sheep. Vet Arhiv 82(5): 473-481..
- 36. Patel AJ, Bhavsar SK, Patel HB, Patel NN, Patel SD et.al. (2012) Effect of ketoprofen co-administration or febrile state on pharmacokinetics of cefepime in goats. Asian J. Anim Vet. Adv 7(1): 46-53..
- 37. Tell L, Harrenstien L, Wetzlich S, Needham M, Nappier J et.al (1998) Pharmacokinetics of ceftiofur sodium in exotic and domestic avian species. J. Vet. Pharmacol. Ther 21(2): 85-91..
- 38. Goudah A (2007) Pharmacokinetics ofceftiofur after single intravenous and intravenous and intramuscular administration in camels (Camelus dromedarius). J. Vet. Pharmacol. Therap 30(4): 371-374..
- 39. Tohamy MA (2008) Pharmacokinetic of ceftiofur sodium administered concomitantly with dipyrone in healthy and feverish cows. J Egypt Soc Pharmacol Exp Therap 29(2): 539-550..
- 40. Ismail MM (2005) Pharmacokinetics, urinary and mammary excretion of ceftriaxone in lactating goats. J Vet Med A 52(7): 354-358..
- 41. El Sayed MG, Hatem ME, El Komy AA (1989) Disposition kinetics of gentamicin in normal and endometric cow using microbiological assay. Dtsch Tier Wochen 96(8): 412-415..
- 42. Aboubakr M, Soliman A, Uney K, Elmas M (2018) Plasma disposition of cefoperazone after single intravenous and intramuscular administrations in camels (Camelus dromedarius). Acta Vet Hung 66(3): 444-450..






























