Prevalence of Gastrointestinal Infections Among Human Population in Some Communities in Akwanga Local Government Area, Nasarawa State, Nigeria
Ali AA*, Pam VA, Uzoigwe NR, Ombugadu A and Maikenti JI
Department of Zoology, Faculty of Science, Federal University of Lafia, PMB 146, Lafia, Nasarawa State, Nigeria
Submission: November 16, 2022; Published: January 20, 2023
*Corresponding author: Ali AA, Department of Zoology, Faculty of Science, Federal University of Lafia, PMB 146, Lafia, Nasarawa State, Nigeria E-mail: alimesut2110@gmail.com
How to cite this article: Ali AA, Pam VA, Uzoigwe NR, Ombugadu A and Maikenti JI. Prevalence of Gastrointestinal Infections Among Human Population in Some Communities in Akwanga Local Government Area, Nasarawa State, Nigeria. Trends Tech Sci Res. 2023; 5(5): 555674. DOI: 10.19080/TTSR.2023.05.555674
Abstract
Parasitic diseases constitute the greatest thread to the health and socio-economic status of the people particularly in the rural communities. This study determined the prevalence of gastrointestinal infections and the associated risk factors among human population in some communities in Akwanga Local Government Area, Nasarawa State, Nigeria. A total of 202 volunteers were administered with a structured questionnaire and their urine and stool samples were collected. Wet mount and sedimentation methods were used for urine sample examination while stool samples were examined microscopically using direct smear saline preparation and concentration methods using formal-ether sedimentation technique. Overall prevalence of 55 (27.2%) was recorded in this study with hookworm having the highest prevalence of 9.4% out of the six (6) parasites identified followed by Ascaris lumbricoides 7.7% while S. haematobium 1.48% had the least prevalence. Prevalence of gastrointestinal parasites between the communities showed no significant difference (P>0.05). But a significant difference (P<0.05) was observed in gastrointestinal infections prevalence in relation to age groups as well as sexes of the subjects respectively. The predisposing risk factors responsible for gastrointestinal parasite infections in the communities is due to lack of latrines and poor access to potable water sources. Hence, this study provides a new insight into the burden of gastrointestinal parasites among the people in the study areas and the prevailing risk factors associated with their transmission and spread.
Keywords: Gastrointestinal parasites; Human population; Predisposing risk factors; Akwanga
Abbreviations: LGA: Local Government Area; LGC: Local Government Council
Introduction
Intestinal parasites infections are a global public health challenge. The problem is highly experienced in tropical and sub-tropical developing countries where safe and adequate water and sanitation are lacking due to poverty [1]. The infections are caused by intestinal worms such as Ascaris lumbricoides, Trichuris trichiura, Strongyloides stercoralis, Necator americanus and Ancyclostoma duodenale. These are also referred to as geohelminths or soil-transmitted helminths because they are transmitted through contaminated soil [2].
More than 1.5 billion people, or 24% of the world’s population, are infected with soil-transmitted helminth worldwide [2]. Infections are widely distributed in tropical and subtropical regions, with the greatest numbers occurring in sub-Saharan Africa, the Americas, China and East Asia [2]. Other intestinal helminths are Enterobius vermicularis and cestodes such as Taenia saginata, Taenia solium, Diphyllobothrium latum and Hymenolepis nana [3,4]. Therefore, this assessed the prevalence of gastrointestinal infections and the associated risk factors among human population in some selected communities in Akwanga Local Government Area, Nasarawa State, Nigeria.
Materials And Methods
Study Area
The study was conducted in Akwanga Local Government Area (LGA) of Nasarawa State in North Central Nigeria. Akwanga LGA lies between Latitude 8°54' 22.65"N and Longitude, 8°24' 30.57"E. It has an area of 996km2 and a population of 113,430 at the 2006 census [5]. Historically, Nasarawa State has been an agricultural State which is known for the production of Groundnut, Yam, Maize, etc. as well as minerals. Akwanga people are predominantly farmers and civil servants with few businessmen and Women.
Ethical Approval
Ethical approval for the study was granted on the 22nd of March 2021 by the Akwanga Local Government Council (LGC) through the Nasarawa State Primary Healthcare Development Agency Department. Also, informed consent was sought from the volunteer subjects. But those who did not sign the consent were, however, not recruited into the study.
Sample Size Determination
The sample size was determined by the formula given by Yamane [6] as shown below.
n = N/ (1 + N (e)2)
Where: n signifies the sample size
N signifies the population under study
e signifies the margin error (it could be 0.10, 0.05 or 0.01)
Sample Collection
Stool and urine samples were collected aseptically from 202 individuals between the ages of 20 and 70 years old from five different communities which includes Akwanga East, Andaha, Agyaga, Ancho-Nighaan and Ancho-baba.
Stool
Stool samples were collected from each participant into labeled sterile bottles. Screw capped bottles with wide mouths were given to each participant, with instructions on how much stool to put inside, and it was emphasized that only fresh, early morning stool should be collected. The stool samples were preserved in 10% formalin solution, and then transported to primary healthcare clinic Andaha laboratory and primary healthcare clinic Wamba Road Akwanga laboratory, for examination depending on the community and proximity.
Urine
Urine samples were collected from each participant in a labeled clean, dry screw capped specimen bottles.
Questionnaire Administration
Well–structured questionnaire (in English Language) was administered to consenting participants, to obtain information on socio-demographic characteristics such as age, sex, sanitary habits and other predisposing factors.
Samples Processing
The wet mount and Sedimentation method as described by Dawet et al. [7] were used to examine the urine samples collected. Each urine sample was thoroughly mixed. About 10ml was transfer into a centrifuge tube and spun at 5,000 rpm for 5 minutes. The supernatant was decanted, and a drop of the sediment was placed on a clean grease free slide, covered with a cover slip and view under a microscope using x10 and x40 objectives [7].
The stool samples were visually examined to note the consistency, presence of abnormal features whether watery, bloody with mucus, formed or unformed [8]. Direct smear saline preparation of stool was examined for ova of parasites under the microscope within 24 hours of collection. On a clean grease free slide a drop of normal saline was placed and emulsified with a small quantity of faeces and covered with a cover slip and view under microscope using both x10 and x40 objectives lenses [9]. Stool samples were also subjected to concentration method using formol-ether sedimentation technique; formal ether concentration is rapid and can concentrate a wide range of faecal parasites from fresh or preserved stool [9].
Data Analysis
Data obtained was analyzed using R console version 4.1.1. Descriptive simple percentage was used to determine proportions of infectivity among subjects. Pearson’s Chi-square test was used to analyze the difference in infection rate in relation to age, gender as well as across communities respectively. Also, it was used to compare the packed cell volume levels among infected and uninfected individuals in the study. Response to predisposing factors to parasitic infection was also analyzed using Pearson’s Chi-square test. The level of significance was set at P<0.05.
Results
Occurrence of Gastrointestinal Parasites in Human Population
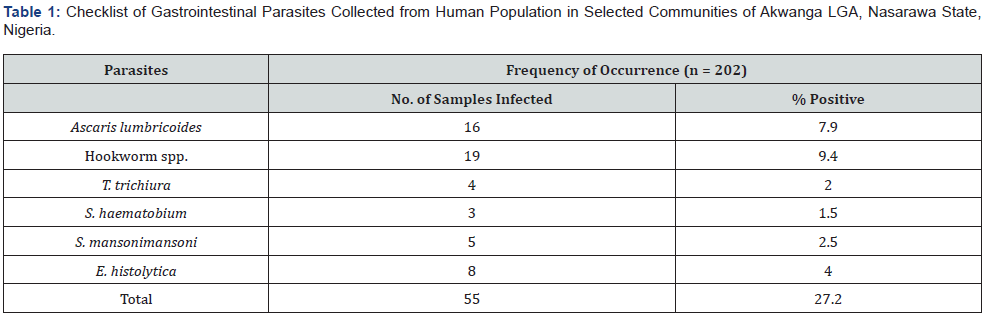
Out of the 202 subjects screened for gastrointestinal parasites, only 55 (27.2%) of them were infected as shown in table 1. The prevalence between the six species of gastrointestinal parasites recorded differed significantly (χ2 = 12.1, df = 5, P = 0.03344). Hookworm was the most prevalent 19 (9.4%) followed by Ascaris lumbricoides 16 (7.7%), while S. haematobium had the least prevalence of 3 (1.48%) (Table 1).
The Prevalence of Parasites Across Selected Communities
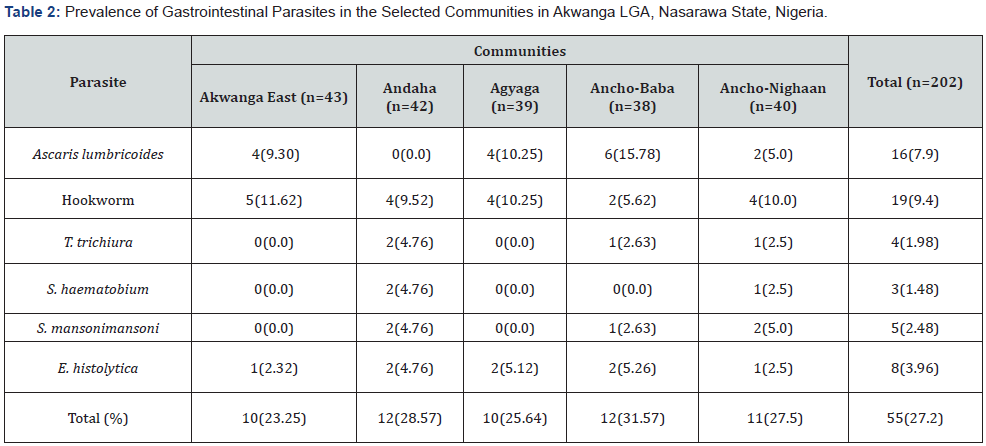
Ancho-Baba had the highest burden of infection 12 (31.57%). All the six parasites were endemic in Ancho Nighaa and Andaha while Ancho-Baba was endemic to five of the parasites, whereas Akwanga-East andAgyaga showed endemicity for three parasite species (Table 2). However, the differences in number of infected individuals between communities showed no significant difference (χ2 = 0.4542, df = 4, P = 0.9778).
Prevalence of Gastrointestinal Infections in Relation to Age and Sex of the Subjects
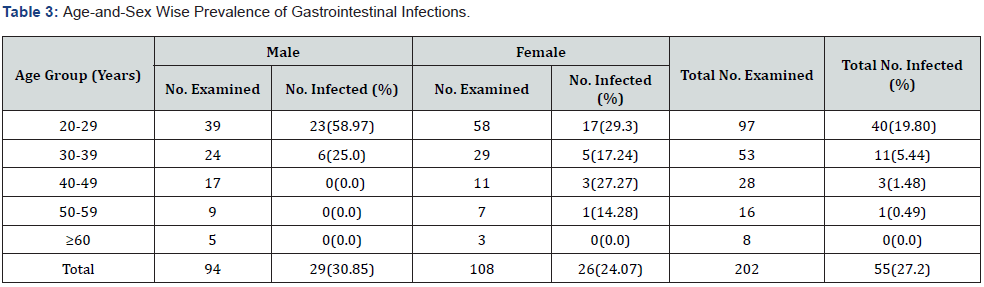
Individuals aged 49 years and below were more infected than 50 years and above subjects. Thus, this accounted for the age-wise significant difference (χ2 = 12.7709, df = 4, P = 0.0124) recorded among the subjects. The highest prevalence of 58.97% and 29.3% in male and female subjects respectively was observed in individuals aged between 20 to 29 years followed by those aged 30 to 39 years with 25.0% and 17.24% for male and female respectively (Table 3). Interestingly, the age group 60 years and above subjects were not infected. Apparently, infections occurred more in males (30.85%) than females (24.07%). Therefore, the prevalence of gastrointestinal infection across sex varied significantly (χ2 = 0.6643, df = 1, P= 0.0191).
Predisposing Risk factors for Gastrointestinal Infections in the study Area
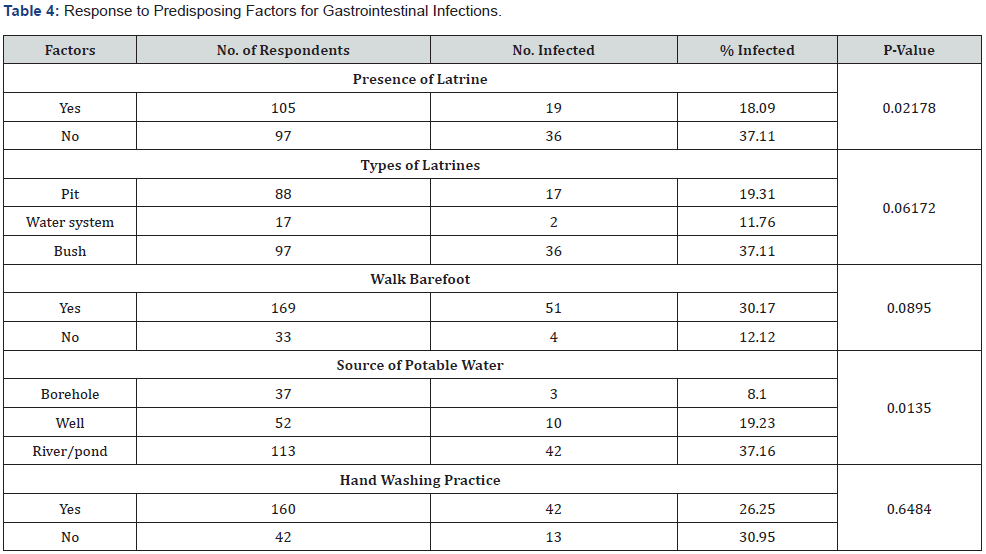
Based on the respondent’s feedback, the predisposing risk factor between subjects who use and don’t use latrines showed a significant difference (χ2 = 5.2626, df = 1, P = 0.0217). Those without latrine were more infected 37.11% than those with latrines 18.09%. However, there was no significant difference (χ2 = 5.5695, df = 2, P=0.06172) in prevalence across the type of latrines in the study area. Prevalence in relation to sources of potable water significantly differed (χ2 = 8.6090, df = 2, P = 0.0135). Factors such as walking bare footed and hand washing practice among the subjects were not statistically significant (walking bare footed: P = 0.0895; hand washing: P = 0.6484) respectively (Table 4).
Discussion
Exactly 27.2% of the population sampled had gastrointestinal parasitic infections. This level of prevalence is high and could be attributed to lack of regular potable water and poor refuse disposal systems observed in the study area. Parasites spread fast through poor personal hygiene and poor sanitary environment contributes to the spread of gastrointestinal infections among the study subjects. The result of this study is similar to the findings of Abah and Arene [10] who recorded a prevalence rate of 27.66% in Rivers State, Nigeria. Lower prevalence rate of 0.7% and 17% were reported in study conducted in Edo State Nigeria and Uganda by Mordi and Ngwodo [11] and Aschalew et al. [12] respectively while a higher prevalence of 46.3%, 47.5%, 77.8% and 48.7% respectively was reported by Ojurongbe et al. [13], Tongjura et al. [14], Owaka et al. [15] and Udensi et al. [16] in Ile-Ife, Lafia, Ebonyi and Imo state.
This occurrence of hookworm species in this study could be attributed to practices of open defecation and the use of human and animal faeces as fertilizer on the farm. Poor sanitary disposal of human faeces and indiscriminate defecation are the principal factors in the spread of hookworm infections. The prevalence value of 9.4% in our study is similar to the 10% prevalence reported by Mordi and Ngwodo [11] in Edo State. This value is very low when compared with the value from other previous studies in various parts of the country. Tongjuraa et al. [14], Egwunyega et al. [17], and Babatunde et al. [18] reported infection rate of 14%, 22.5%, and 13.9% respectively at Lafia in Nasarawa State, Eku in Delta State, and Kwara State, Nigeria. However, lower infection rate of 2.7% and 5.6% were reported by Okike-Osisiogu et al. [19] and Ojurongbe et al. [13] in Aba, Abia State and Ile-Ife in Nigeria while Sammy et al. [20] reported 2.9% prevalence in Ghana.
Ascaris lumbricoides was the second most common parasite in the study with a prevalence of 7.9%. Higher prevalence of 11.1%, 11.5%, 20.7%, 27.7% and 46% were reported by Babatunde et al. [18], Tongjura et al. [14], Oluwarotimi and Ifeanyi [21], Muoneke et al. [22] and Owaka et al. [15] in Kwara State, Lafia- Nasarawa State, Ifetedo- Osun State, Lagos State and Ebonyi State respectively. A lower prevalence was reported by Okike-Osisiogu et al. [19] in Aba, Abia State. The findings of this study could be attributed to lack of potable water supply, poor personal hygiene and lack of sanitary disposal system which enhance transmission in the communities.
The prevalence of E. histolytica 3.96% in this study agrees with the 4% prevalence reported by Mordi and Ngwodo [11] in a study in Edo State. Studies done elsewhere in Nigeria had much higher prevalence values, 7.5%, 7.6%, and 14.1% was reported by Tongjura et al. [14], Okike-Osisiogu et al. [19] and Babatunde et al. [18] in Lafia, Aba and Kwara State respectively. Amoebiasis occurs both in tropical and temperate climates, but infections are more rampant in the tropics. This parasite, though low in prevalence was found in all the selected communities. The findings can be attributed to transmission of viable cysts by direct contact with contaminated foods such as raw vegetables fertilized with human faeces and through the intermediary of filthy flies and contaminated hands of human cyst carriers.
S. mansoni recorded a relatively high prevalence value of 2.48% in this study is in conformity with Junior et al. [23] who reported a higher prevalence value of 6.4% in Kinshasha, Democratic Republic of Congo. On the other hand, Ojurongbe et al. [13], Babatunde et al. [18] and Sammy et al. [20] reported a less value of 0.6%, 0.99% and 1.8% in Ile-Ife, Kwara State, Nigeria and Ghana respectively. The prevalence of S. haematobium in this study characterized by haematuria can be attributed to poor sanitary habits, contact with water bodies infested with parasites and the availability of suitable moluscan intermediate hosts. The 1.48% prevalence of S. haematobium infection we recorded is in accordance with the report by Mordi and Ngwodo [11] who recorded a 2% prevalence of S. haematobium in Edo State. On the contrary, higher prevalence of 21.5% and 32.7% was reported by Obadiah et al. [24] and Adekola et al. [25] in Benue and Oyo State respectively.
In this study, T. trichiura prevalence (1.98%) is like the report by Muoneke et al. [22] in Lagos State. Also, Sammy et al. [20] reported lower prevalence rate of 0.1% in Ghana. The prevalence of T. trichiura in this study is low in comparison with previous studies by Tongjura et al. [14], Owaka et al. [15] and Oluwarotimi and Ifeanyi [21] who obtained a prevalence of 4%, 13.4% and 12.1%in Nasarawa, Ebonyi, and Osun States respectively T trichiura popularly known as whipworm because of the whip-like form of the adult worm has a cosmopolitan distribution. It is, however, prevalent in the warm humid tropics. Soil pollution is a major factor in the transmission of the infection in a community. Transmission occurs through poor sanitary habits of indiscriminate defecation. Infections usually occur through ingestion of infective ova from contaminated hands, food or drinks. Flood and copraphagous animals play some part in the transportation of the ova to locations other than the defecation site.
Hookworm and E histolytica were distributed in in all the five communities surveyed. Ascaris lumbricoides was found in 4 out of the 5 areas that were investigated S. mansoni and S. haematobium occurred in 3 and 2 areas respectively. This finding means that all the communities in the study area are endemic for gastrointestinal parasitic infections and that the study subjects are potentially susceptible to such infections. The sex-wise absence of variation in the prevalence of gastrointestinal parasitic infections clearly suggests that that both sexes are susceptible to parasitic infections and that the parasites have no preferred sex. Although, a higher prevalence of the infections was observed in males 30.85% than females 24.07%. Males are often seen walking bare foot both at home and on the farm and also swim, bath and fish in infested water bodies. This is in agreement with Oluwarotimi and Ifeanyi [21] and Muoneke et al. [22] but in contrast with Eleni et al. [4], Tongjura et al. [14] and Owaka et al. [15] who recorded higher prevalence in females than in males.
With respect to age, the younger age groups had the highest prevalence rate than the older age groups. Younger age groups are more at risk of infections than the older age groups. There was decline in prevalence with increased in age because the people become more conscious of personal hygiene, and it may also be as a result of acquired immunity after been exposed at a younger age. The finding in this study concurs with the findings of other researchers from other parts of the country [15, 21, 22]. The main predisposing factor to gastrointestinal infections in the study area is lack of laterine by most subjects in which such individuals were more infected 37.11% than those with latrines 18.09%. Though infectivity across laterine types was the same.
Furthermore, lack of access to potable water sources accounted for parasitic infections most especially on the subjects who accessed water from rivers/ponds. Pam et al. [26] reported similar findings in Doma, Nasarawa State. Rivers/ponds are easily contaminated by people who tend to dump refuse in them compared to other sources of potable water such as borehole and wells. The lack of association of walking barefooted and hand washing practices with gastrointestinal parasitic infection possibly suggests that the subjects got infected through contaminated water, indiscriminate defecation and poor sanitary disposal.
Conclusions
The prevalence of gastrointestinal parasites was quite high, and such poses a major public health problem in the selected communities in Akwanga LGA of Nasarawa State. This study generated a checklist of six parasite species and their prevalence across the selected communities. All the six parasites were endemic in Ancho-Nighaa and Andaha communities. Hookworm and E. histolytica were widely distributed in all the selected communities. The data obtained from this study provides information on the various parasitic diseases associated with gastrointestinal tract infections of humans in the communities studied. The main predisposing risk factor to gastrointestinal parasite infections in the communities is due to lack of latrines. Also, lack of access to potable water sources predisposes people to parasitic infections especially around river/pond areas. Hence, there is the need to aggressively educate the people in the selected communities and Akwanga LGA as whole on public health implications of gastrointestinal parasitic infections. Control measures such as provision of deworming programmes coupled with provision of improved water supplies and sanitation should be put in place in order to bring about drastic reduction in the prevalence level of parasitic infections.
References
- Ngui R, Ishak S, Chuen CS, Mahmud R, Lim YAL (2011) Prevalence and Risk Factors of Intestinal Parasitism in Rural and Remote West Malaysia. PLoS Negl Trop Dis 5(3): e974.
- World Health Organization (2020) Soil transmitted helminthes.
- Bogitsh B, Carter CE, Oeltmann NT (2013) Human Parasitology. Academic Press. Massachusetts, United States, pp. 1-430.
- Eleni K, Sissay M Ameha K, Mulugeta D (2014) Prevalence of intestinal Parasitic Infections and their association with arthropometric measurements of school children in selected primary Schools, Wukro Town, Eastern Tigray, Ethiopia. International Journal of Current Microbiology and Applied Sciences 3(3): 11-29.
- National Bureau of Statistics (2006) 2006 Population Census, Federal Republic of Nigeria.
- Yamane T (1967) Statistics: An Introductory Analysis, 2nd Edition, New York: Harper and Row.
- Dawet A, Benjamin CB, Yakubu DP (2012) Prevalence and intensity of Schistosoma haematobium among residents of Gwong and Kabong in Jos North Local Government Area, Plateau State, Nigeria. International Journal of Tropical Medicine 7(2): 69-73.
- WHO (2003) World Health Organization Prevention and control of schistosomiasis and soil-transmitted helminthiasis: report of a WHO expert committee. Geneva, Switzerland.
- Cheesbrough M (2013) District Laboratory Practice in the Tropical Countries. 2nd edition, Cambridge University press, UK, Part 1, pp. 178-219.
- Abah AE, Arene FOI (2015) Status of intestinal parasitic infections among primary school children in Rivers State, Nigeria 2015: 1-7.
- Mordi RM, Ngwodo POA (2007) A study blood and gastrointestinal parasites in Edo State. African Journal of Biotechnology 6(19): 2201-2207.
- Aschalew G, Belay A, Bethel N, Betrearon S, Atnad Y, et al. (2013) Prevalence of intestinal Parasitic infections and risk factors among schoolchildren at the University of Gondar Community School, Northwest Ethiopia: A cross-sectional study. Journal of Parasitology Research 13(304).
- Ojurongbe O, Oyesiji KF, Ojo JA, Odewale G, Adefioye OA, et al. (2014) Soil transmitted helminth infections among primary school children in Ile-Ife Southwest, Nigeria: A cross-sectional study. International Research Journal of Medicine and Medical Sciences 2(1): 6-10.
- Tongjura JDC, Ombugadu JR, Abdullahi MM, Blessing MA, Amuga GA, et al. (2019) Intestinal Parasites amongst primary school children attending Ta'al Model Primary School in Lafia Local Government Area of Nasarawa State, Nigeria. Nigeria Journal of Parasitology 40(1): 92-96.
- Owaka EE, Njoku OO, Uhuo CA, Odikamnoro OO (2016) Survey of Intestinal Helminth Infection amongst School Children in Rural Communities of Ebonyi State Nigeria. International Journal of Scientific and Research Publications 6(5): 2250-3153.
- Udensi JU, Mgbemena IC, Emeka Nwabunnia I, Ugochukwu MG, Awurum IN (2015) Prevalence of intestinal Parasites among primary school children in three geopolitical zones of Imo State, Nigeria. Science Journal of Public Health 3(5-1): 25-28.
- Egwuyenga OA, Ataikiru DP, Nmorsi QPG (2004) Studies on intestinal helminthes infection in Eku, Delta State, Nigeria. 28th annual conference Abstract (20) from the Nigeria Soc Parasitol 20: 24
- Babatunde SK, Kolawole DO, Majekodunmi RA, Ajiboye AE, Ojo S, et al. (2019) Profile of intestinal parasites among nomadic Fulani in Kwara State, Nigeria. Nigeria Journal of Parasitology 40(1): 56-60.
- Okike Osisiogu FU, Nwoke BEB, Ukaga CN, Amaechi AA, Ezeigbo OR, et al. (2018) Prevalence of intestinal parasites and bacteria among school pupils in Aba, Abia State. Nigerian Journal of Parasitology 39(1): 13.
- Sammy CKT, Stephen YG, Thomas KG (2011) High Prevalence of Hookworm Infections and apparent absence of Ascaris lumbricoides: A Case Study at the Komfo Anokye Teaching Hospital in Ghana. International Journal of Pharmaceutical Sciences and Research 2(5): 1217-1224.
- Oluwarotimi AO, Ifeanyi EO (2019) Prevalence and Intensity of Soil Transmitted Helminths among School Children in Ifetedo, Osun State, Nigeria. Journal of Bacteriology Parasitology 10(1): 352.
- Muoneke PD, Akindele AA, Akanni RA, Sanusi TO, Adefioye OA, et al. (2019) Epidemiology of Intestinal Parasitic Pathogens, Plasmodium Falciparum and their Contribution to Anaemia among School Children in Lagos State, Southwest, Nigeria. IOSR Journal of Dental and Medical Sciences (IOSR-JDMS) 18(7): 58-64.
- Junior MR, Doua JY, Linsuke S, Madinga J, Van Geertruyden JP, et al. (2014) Malaria, Schistosomiasis and Soil Transmitted Helminth Burden and Their Correlation with Anemia in Children Attending Primary Schools in Kinshasa, Democratic Republic of Congo. PLoS ONE 9(11): e110789.
- Obadiah HI, Idu ME, Omudu EA, Shenge MF, Ameh MO, et al. (2018) Studies on Schistosoma haematobium infection in school-aged children in some parts of Benue State, Nigeria. Nigeria Journal of Parasitology 39 (1): 9.
- Adekola SS, Samuel OA, Oluyomi AS (2014) Co-infections with Schistosoma haematobium and soil-transmitted helminths among school-aged children in Saki, Oyo State, Nigeria. Journal of Public Health and Epidemiology 6(12): 417-423.
- Pam VA, Idris AA, Adejoh VA, Pam RG, Ombugadu A, et al. (2019) Studies on Parasitic contamination of soil and local drinking water sources, Doma Local Government Area, Nasarawa State, Nigeria. Nigeria Journal of Parasitology 40(1): 51-55.






























