Solubility and Toxicity Level of Monascus Pigments
Anna Yuliana1,2*, Marlia Singgih Wibowo1 and Elin Julianti1
1School of Pharmacy, Bandung Institute of Technology, Indonesia
2Departement of Pharmacy, STIKes Bakti Tunas Husada, Indonesia
Submission:June 13, 2018; Published: July 23, 2018
*Corresponding author: Anna Yuliana, School of Pharmacy, Bandung Institute of Technology, Ganesa 10 Bandung, West Java, Indonesia; Tel.: +6281222903376; Email: anna_yuliana@stikes-bth.ac.id
How to cite this article: Anna Yuliana, Marlia S W, Elin J. Solubility and Toxicity Level of Monascus Pigments. Trends Tech Sci Res. 2018; 2(4): 555594. DOI: 10.19080/TTSR.2018.02.555594
Abstract
Research on Monascus pigments (MPs) has been progressing very rapidly. The progresses include the discovery of new pigments and methods used for isolating and identifying the new pigments. Currently, nearly fifty-seven new pigments have been found as derivations of the six main pigments existed. The process of new pigments discovery is unable to separate from that of fermentation or isolation process. In several new pigments, there was no complete information relating to solubility and level of toxicity. This research covers data of all fifty-seven new pigments including color, molecular weight, water solubility, log Kow (octanol-water partition coefficient), and baseline toxicity level by Ecological Structure Activity Relationships (ECOSAR) Program. These results are expected to give more comprehensive data about Monascus pigments.
Keywords: Monascus pigments; Solubility; Toxicity; Ecosar
Introduction
Monascus Pigments (MPs) has long been used as a natural food colorant, especially in some Asian countries such as South China. Monascus produces red yeast rice, which is rice covered with a red Monascus sp. Angkak can be used as a dye for yoghurt, bacon, and sausage and also for the preservative of fruits, vegetables, and fish products [1].
The Ecological Structure Activity Relationships (ECOSAR) is a computerized predictive system that estimates aquatic toxicity. The program estimates a chemical's acute (short-term) Result toxicity and chronic (long-term or delayed) toxicity to aquatic organisms, such as fish, aquatic invertebrates, and aquatic plants, by using computerized Structure Activity Relationships (SARs) [2].
Materials and Methods
Data collected from various studies that have been done before [3-5] and added with some of the data from ECOSAR results. This toxicity test on ECOSAR program was used to determine the toxicity against water organisms.
Results
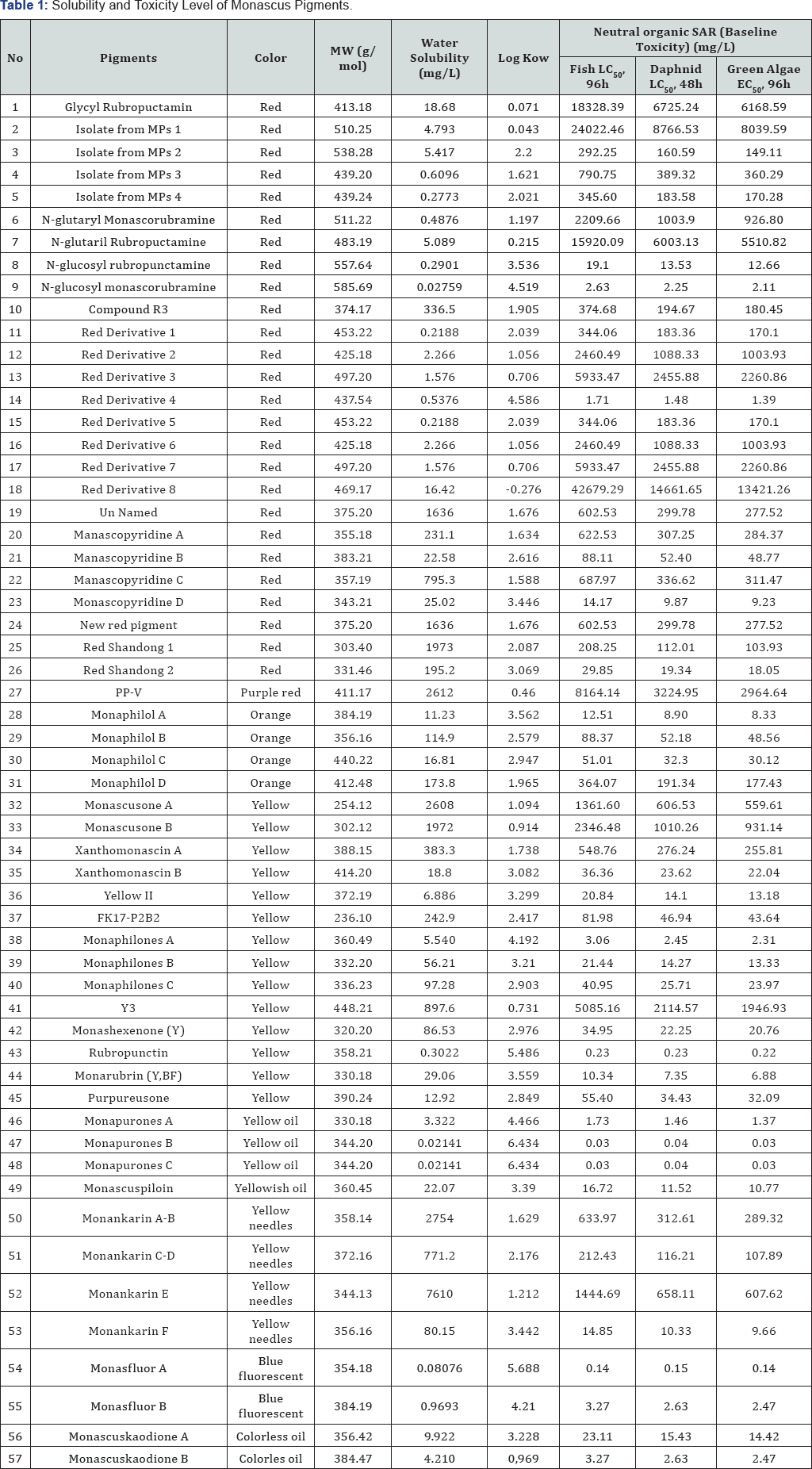
As shown in Table 1, data showed that the lower log Kow of a compound, the lower of toxicity. And from the toxicity point of view, those of compounds to categorize having lower toxicity than others were LC50 and EC50 values>100mg/L. This was because the lower binding energy of a compound, the stronger bond between the receptor and that compound. It made the compound attach to biological membranes longer and more toxicable [2].
Conclusion
Based on log Kow value of fifty-seven pigments of Monascus were determined, Red Pigment 8 has the lowest toxicity while Monapurones (B and C) have the highest toxicity.
References
- Singgih W, Dan Julianti E (2015) Food colorant from microorganisms. In: Liong MT (Edt.) Beneficial Microorganism in Food and Nutraceuticals, Microbiology Monographs 27.
- Benfenati E, Gini G, Piclin N, Roncaglioni A, Vari MR (2003) Predicting logP of pesticides using different software. Chemosphere 53(9): 11551164.
- Feng Y, Shao Y, Chen F (2012) Monascus pigments. Appl Microbiol Biotechnol 96(6): 1421-1440.
- Patakova P (2013) Monascus secondary metabolites: production and biolitical activity. J Ind Microbiol Biotecnol 40(2): 169-181.
- Mostafa ME, Abbady MS (2014) Secondary metabolites and Bioactivity of the Monascus pigments review article. Global Journal of Biotechnology & Biochemistry 9(1): 1-13.






























