Promethazine an Environmental Friendly Reagent for Novel SpectrophotometricMethod Estimation of Hypochlorite inDifferent Environmental Samples
Nief Rahman Ahmed*
Department of Environmental Technology, University of Mosul, Iraq
Submission: July 18, 2019; Published: November 05, 2019
*Corresponding author: b Nief Rahman Ahmed, Department of Environmental Technology, College of Environment, University of Mosul, Iraq
How to cite this article: Nief Rahman Ahmed. Promethazine an Environmental Friendly Reagent for Novel Spectrophotometric Method Estimation of Hypochlorite in Different Environmental Samples. Recent Adv Petrochem Sci. 2018; 6(5): 555700. DOI: 10.19080/RAPSCI.2019.06.555700
Abstract
A simple, rapid and accurate spectrophotometric method has been developed for the determination of hypochlorite using promethazine. The proposed method reports the reaction of hypochlorite with promethazine in acid medium to form a red colored product with an absorption maximum at 517 nm. Beer’s Law was obeyed in the range of 1-10 μg/ml with molar absorptivity of 0.382×104 L.mol-1.cm-1 and sandell sensitivity 0.0195μg/cm2 .The relative standard deviation of the method was less than 2% and accuracy (average recovery ) was 100±1.7% .The optimum conditions for all color development are described and the proposed method has been successfully applied for the determination of hypochlorite in drinking tap water and household bleaching samples.
Keywords:Hypochlorite ion Spectrophotometric Drinking tap water Household bleaching samples Iodimetric Potentiometric Promethazine Water chlorination Microorganisms Beam spectrophotometer
Introduction
Sodium hypochlorite (NaOCl) is a compound that can be effectively used for water treatment and purification. It is used on a large scale for odor removal, bleaching, disinfection, and surface purification [1]. Water chlorination has been the most common disinfectant methods used by waters suppliers [2]. Chlorine is an efficient agent for inactivating several types of microorganisms, it has been preferred as a disinfecting agent to assure the bacteriological quality of the drinking water [3]. The water bacterial contamination also occur in the distribution network; therefore, to prevent this occurrence, a free chlorine residual in excess of 0.2 mg L−1 must be maintained throughout the distribution lines [3]. Various analytical methods have been utilized for the determination of NaOCl .Some of the examples were colorimetric [4]. iodimetric [5-7]. chemiluminisces [8,9]. potentiometric [10]. spectrophotometric methods [11-14]. potentiometric [15]. and flow injection methods [16,17]. In this work, we intend to develop a spectrophotometric procedure for the determination of residual hypochlorite in drinking tap water and household bleaching samples using promethazine.
Experimental
Apparatus
Spectro Uv / visible dual beam spectrophotometer [UVS-2700, Labomed, INC] with 1.0 cm quartz cells and Genway 3310 pH meter were used.
Reagents
All chemicals used were of analytical purity grade and all solutions were prepared in distilled water. A sodium hypochlorite stock solution (0.01%) was prepared by dilution of 0.25 ml of 4 % sodium hypochlorite to 100 ml by distilled water, store in a dark bottle and standardized every 4-5 days. This solution stored in a dark bottle [18,19]. Standard solution of sodium hypochlorite (10ppm). This solution was prepared by diluting 10 ml of the stock solution to 100 ml by distilled water in a volumetric flask. Promethazine hydrochloride solution 0.01% was prepared by dissolving 0.01g of Promethazine hydrochloride in 100ml distilled water in a volumetric flask. Sulfuric acid 0.1M, this solution was prepared by diluting 1.4 ml of 18 M H2SO4 solution to 250 ml by distilled water in a volumetric flask.
Recommended Procedure
Aliquots of standard solution of hypochlorite (1-10μg) were transferred into a series of 25 ml calibrated flasks, added 1 ml of 0.1M sulfuric acid solution and 5 ml of 0.01% Promethazine hydrochloride solution, dilute the solution to the mark with distilled water. The absorbance of the red-colored product was measured at 517 nm against a reagent blank
Sampling
Drinking water samples were taken from tap water in Mosul city. After the water samples were transported to the laboratory, hypochlorite ion analyses were performed immediately. The household bleach samples of different brands were purchased from local markets.
Procedure for the determination of hypochlorite in tap water samples
An aliquot of tap water samples containing not more than 10μg of hypochlorite was transferred to a 25 mL calibrated flaskand the hypochlorite content was determined as mentioned under recommended procedure.
Procedure for the determination of hypochlorite in household bleach
1.0 ml of the household bleach solution was transferred to a 100 mL calibrated flask, dilute the solution to the mark with distilled water. From this solution take 1 mL and diluted to 100 ml by distilled water in a volumetric flask, and aliquot of this solution was treated as described above for recommended procedures.
Results and Discussion
Promethazine hydrochloride is oxidized in sulfuric acid medium with sodium hypochlorite solution instantaneously at room temperature to form a red-colored product which is believed to be a radical cat ion and is irreversibly oxidized to a colorless sulfoxide with loss of electron [20]. as shown below.
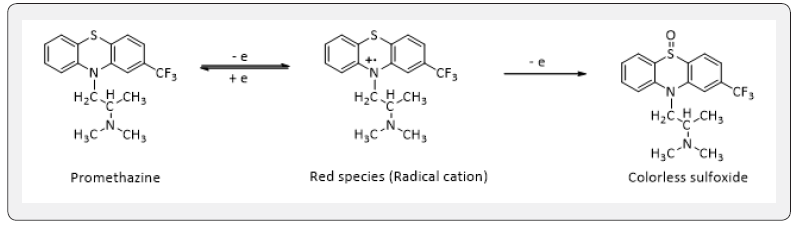
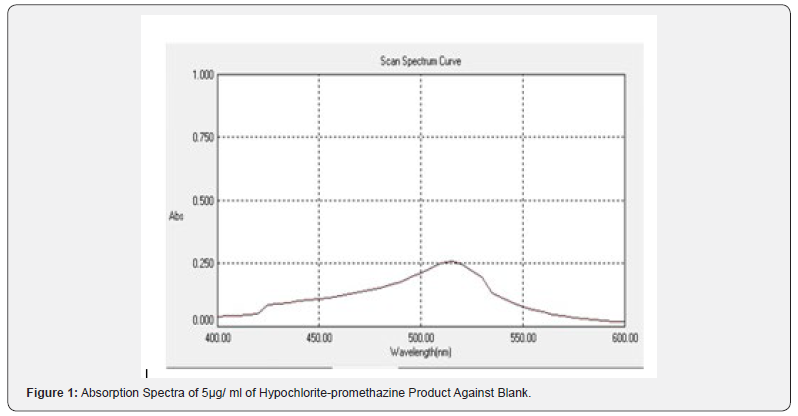
The red –colored radical cat ion shows maximum absorbance at 517 nm, where the blank does not absorb appreciably, as shown in (Figure 1).
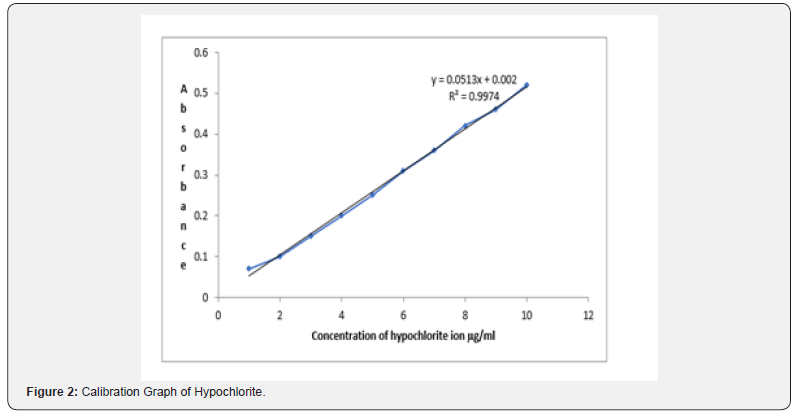
The reaction variables were optimized by varying each variable while keeping others constant for obtaining maximum absorbance. The oxidation reaction was found to be quantitative in acetic acid medium. It was found that 1 ml of o.1 M sulfuric acid solution give high sensitivity and this amount has been used for subsequent experiments. The effect of the amount promethazine on the absorbance was investigated. A maximum and constant absorbance was found with 3 to 6 ml of 0.01% promethazine solution and 5 ml has been used for subsequent experiments. The color reaction occurred at room temperature immediately and remained stable for at least 24h. and a reaction time of 5 min was selected for reproducible results. under the experimental conditions described, Beer ‘s law is obeyed over the concentration range 1-10 μg /ml (Figure 2). with correlation coefficient of 0.9974, intercept of 0.002 and slope of 0.0513. The conditional molar absorptivity of the product formed and sandell ҆s sensitivity was found to be 0.382×104 L.mol-1.cm-1 and 0.0195μg/cm2 respectively. The accuracy and precision of the method was established by analyzing the standard hypochlorite solution at three different levels. The average recovery which is a measure of accuracy is 100 ± 1.7 revealing high accuracy of the method. The relative standard deviation (RSD), which is an indicator of precision is better than ±2%. The results are compiled in (Table 1).
Application to Real Samples
To demonstrate the practical applicability of the proposed method, drinking tap water samples collected from left stationof Mosul water-supply network of Mosul city. The samples were collected in plastic canes of 1 Liter capacity without of any air bubbles. The results were also compared statistically by student t-test with those obtained by standard method at 95% confidence level. The calculated t- values did not exceed the theoretical values indicating that there were no significant differences between the precision of the proposed and literature method as cited in (Table 2). For the determination of hypochlorite ion in household bleach samples. twelve type of household bleach samples of different brands were purchased from local markets and analyzed by the proposed and standard methods; the results were cited in (Table 3).
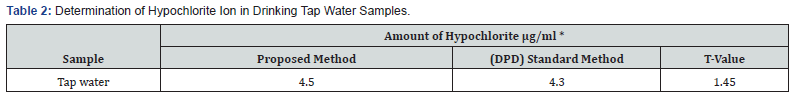
*Average of Ten Determinations.
T values (n=10, at 95% Confidence Level Tabulated Value 2.101).
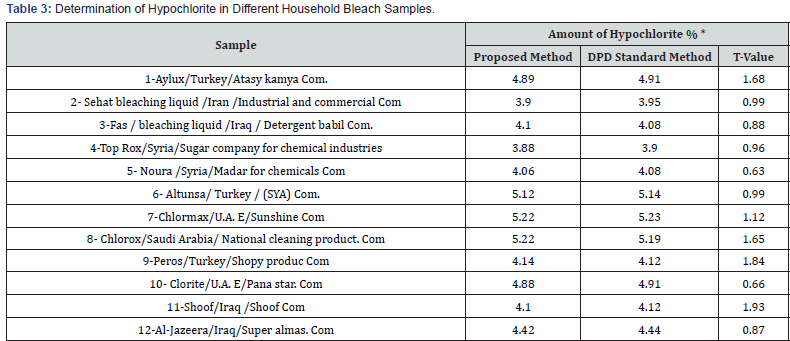
*Average of Ten Determinations.
T values (n=10, at 95% Confidence Level Tabulated Value 2.101).
Conclusions
For the first time, promethazine has been used as a chromogenic reagent for the spectrophotometric determination of hypochlorite. The proposed method, which is simple and rapid, offers the advantages of sensitivity and wide range of determinations without the need for extraction or heating. The method does not involve any stringent reaction conditions and can be compared favorably with the other methods. The proposed method has been successfully applied to the determination of hypochlorite in various samples.
References
- Somani SB, Dr Ingole NW, Dr Kulkarni NS (2011) Disinfection of water by using sodium chloride (NaCl) and sodium hypochlorite (NaOCl). Journal of Engineering Research and Studies (11): 40-43.
- Wang Y, Claeys L, Vander Ha D, Verstraete W, Boon N (2010) Effects of chemically and electrochemically dosed chlorine on Escherichia coli and Legionella beliardensis assessed by flow cytometry. Applied Microbiology and Biotechnology (87): 331–341.
- Al-Jasser AO (2007) Chlorine decay in drinking-water transmission and distribution systems: pipe service age effect. Water Research (41): 387–396
- Zhang Jia, Xiaolei Wang, Xiurong Yang (2012) Colorimetric determination of hypochlorite with unmodified gold nanoparticles through the oxidation of a stabilizer thiol compound Analyst (137): 2806- 2812.
- (2010) (2016) The Japanese Pharmacopoeia,17th edn, English Version, The Ministry of Health, Labor and Welfare. The United State Pharmacopeia Convection, Inc,33-NF, (28): 4698
- Sreekanth B, Jonnalagadda, Prabhashini Gengan (2010) Titrimetric and Photometric methods for determination of hypochlorite in commercial bleaches. Journal of Environmental Science and Health Part A (45): 917-922.
- Ballesta-Claver J, Valencia-Miron MC, Capitan-Vallvey LF (2004) Determination of hypochlorite in water using a chemiluminescent test strip Anal. Chim. Acta, 522:267–273.
- Qin W, Zhang Z, Liu S (1997) Flow-injection chemiluminescence sensor for the determination of free chlorine in tap water. Analytical Letters (30): 11-19.
- Saad B, Wai T, Ali AM, Saleh MI (2006) Sequential flow injection determination of chlorine species using a tri iodide- selective electrode detector. Analytical Sciences (22): 45-50.
- Fernanda HS, Viviane GB, Geiser G (2008) Spectrophotometric Multi Commutated Flow System for the Determination of Hypochlorite in Bleaching Products. Analytical Letters (41): 3187–3197.
- Nief Rahman, Farha KO (2018) New Indirect Spectrophotometric Method for Determination of Hypochlorite using Nile blue. Journal of Enhanced Research in Science Technology & Engineering (7): 13-16.
- Gengan P, Jonnalagadda SB (2005) New Spectrophotometric method with KMnO4 for determination of hypochlorite in commercial. Bulletin of the Chemical Society of Ethiopia (19): 1-8.
- Nief Rahman Ahmad, Sufyan Hisham (2019) Novel spectrophotometric method for estimation of hypochlorite in tap water and bleaching product samples. World Journal of Parmacy and Pharmaceutical Sciencs 8(5): 1450-145.
- Soundappan Thiagarajan, Zhi-Yuan Wu, Shen-Ming Chen (2011) Amperometric Determination of sodium hypochlorite at Mn TAPPnano Au film modified electrode. Journal of Electro analytical Chemistry (661): 322-328
- Ramos LA, Prieto KR, Cavalheiro ETG, Cavalheiro CCS (2005) Determination of hypochlorite in bleaching products with flower extracts to demonstrate the principles of flow injection analysis. J.Chem. Education (82): 1815-1819.
- Sivanildo S Borges, Boaventura F Reis (2011) An Environmental Hypochlorite in Tap Water Employing a Miniaturized Multi commuted Flow Analysis Setup. Journal of Automated Methods and Management in Chemistry 1-6.
- Nief Rahman Ahmed (2011) An Indirect Spectrophotometric Determination of Mesna in Pharmaceuticals and Environmental Samples. Iraqi National Journal of Chemistry (44): 492-500.
- Theia’a Najim, Al-Sabha, Nief Rahman Ahmad, Mona Ismael Ibrahim (2006) Spectrophotometric determination of promethazine hydrochloride via Oxidative coupling reaction with sulfanic acid. University of Sharjah Journal of Pure & Applied Sciences 3(1): 1-12.
- Theia’a Najim, Al-Sabha, Nief Rahman Ahmad, Mona Ismael Ibrahim (2006) Spectrophotometric determination of promethazine hydrochloride via Oxidative coupling reaction with sulfanic acid. University of Sharjah Journal of Pure & Applied Sciences 3(1): 1-12.
- APHA, AWWA, WPCF (1985) Standard Methods for the Examination of Water and Wastewater. APHA, 16th Ed. Washington, DC, USA, p.309.






























