- Review Article
- Abstract
- Introduction
- Industrial Chemicals and Intermediates: High Volume Caprolactam by Hydrocyanation of 1, 3-butadiene
- Caprolactam by carboalkoxylation of 1,3-butadiene
- Chloroprene by chlorination of 1,3-butadiene
- 4-Vinyl cyclohexene, ethyl benzene and styrene from 1,3-butadiene
- 1,5-Cyclooctadiene (COD) from 1,3-butadiene
- Dodecanoic acid (DDA) by cyclotrimerization of 1,3-butadiene
- Industrial Chemicals and Intermediates: Low Volume
- Diels-Alder Reaction of 1,3-butadiene
- Miscellaneous other Useful Reactions of 1,3-butadiene
- Conclusions
- Acknowledgements
- References
Industrial Chemicals and Intermediates from 1,3-Butadiene
Vivek K Srivastava*, Sukdeb Saha Preetom Sarkar , GS Srinivasa Rao and Raksh V Jasra
Reliance Research and Development Center, Vadodara Manufacturing Division, India
Submission: June 16, 2019; Published: November 04, 2019
*Corresponding author: Vivek K Srivastava, Reliance Research and Development Center, Vadodara Manufacturing Division, Reliance Industries Ltd., Vadodara-391346, Gujarat, India
How to cite this article: Vivek K Srivastava, Sukdeb Saha Preetom Sarkar, GS Srinivasa Rao, Raksh V Jasra. Industrial Chemicals and Intermediates from 1, 3-Butadiene. Recent Adv Petrochem Sci. 2018; 6(5): 555698. DOI: 10.19080/RAPSCI.2019.06.555698
- Review Article
- Abstract
- Introduction
- Industrial Chemicals and Intermediates: High Volume Caprolactam by Hydrocyanation of 1, 3-butadiene
- Caprolactam by carboalkoxylation of 1,3-butadiene
- Chloroprene by chlorination of 1,3-butadiene
- 4-Vinyl cyclohexene, ethyl benzene and styrene from 1,3-butadiene
- 1,5-Cyclooctadiene (COD) from 1,3-butadiene
- Dodecanoic acid (DDA) by cyclotrimerization of 1,3-butadiene
- Industrial Chemicals and Intermediates: Low Volume
- Diels-Alder Reaction of 1,3-butadiene
- Miscellaneous other Useful Reactions of 1,3-butadiene
- Conclusions
- Acknowledgements
- References
Abstract
1, 3-Butadiene (BD) is an important feedstock chemical of petrochemical industries for production of a variety of valuable products. The worldwide production of BD is estimated ~12 KTA in 2018 and is being produced as a by-product of ethylene production process from steam cracking of naphtha liquid feedstock. The elastomer plants are the largest consumer of BD to produce variety of synthetic elastomers, mainly for the tire industries. Therefore, the demand of BD mainly depends upon the market of automobile industries. Besides, the synthetic elastomer industries, ~22% of global BD demand is to produce several intermediates and industrial chemicals of various applications. The major industrial chemicals and intermediates are being produced from BD are Caprolactam, Adiponitrile, Adipic acid, Chloroprene, 1,4-Butanediol, C8- and C12-Cyclomers, Dodecanoic Acid, 2,6- Dimethyl naphthalene, Sulfolane, Hexadiene etc. The present review article attempts to cover the chemistry involved and production technologies behind these non-elastomeric industrial chemicals and intermediates produced from BD. Besides this, the present review article also covers miscellaneous synthetic strategies which were published recently while using BD as one of the precursor chemicals. A state of art review of the chemistry behind the production of various industrial chemicals from BD would be highly advantageous to a wide population of researches (Figure 1).
Keywords: 1,3-Butadiene Caprolactam Adiponitrile Adipic acid Chloroprene 1,4-Butanediol C8-and C12- Cyclomers Dodecanoic Acid; 2,6-Dimethyl naphthalene Sulfolane Hexadiene
Abbrevations: TPEs: Thermoplastic Elastomers; BD: Buatdiene; SBR: Styrene-Butadiene Rubber; ABS: Acrylonitrile-BD-Styrene; HIPS: High Impact Polystyrene; SB: Styrene-BD; HMDA: Hexamethylenediamin; ADN: Adiponitrile; ACN: 6-Aminocaprolacatm; CAPM: Caprolactam; PN: Pentenenitriles; HMI: Hexamethyleneimine; EB: Ethyl Benzene
- Review Article
- Abstract
- Introduction
- Industrial Chemicals and Intermediates: High Volume Caprolactam by Hydrocyanation of 1, 3-butadiene
- Caprolactam by carboalkoxylation of 1,3-butadiene
- Chloroprene by chlorination of 1,3-butadiene
- 4-Vinyl cyclohexene, ethyl benzene and styrene from 1,3-butadiene
- 1,5-Cyclooctadiene (COD) from 1,3-butadiene
- Dodecanoic acid (DDA) by cyclotrimerization of 1,3-butadiene
- Industrial Chemicals and Intermediates: Low Volume
- Diels-Alder Reaction of 1,3-butadiene
- Miscellaneous other Useful Reactions of 1,3-butadiene
- Conclusions
- Acknowledgements
- References
Introduction
The physical appearance of BD is a colorless gas with unpleasant odor and can be liquefied easily under pressure. BD is one the versatile chemical used in the production of a wide variety of synthetic elastomers and thermoplastic elastomers (TPEs) as well as other petrochemical intermediates and chemicals. 1, 3-Buatdiene (BD) is an important chemical feedstock of petrochemical industries and is being produced as a by-product of ethylene production process from steam cracking of liquid feedstock, usually naphtha. Steam cracking is highly endothermic pyrolysis reaction, during which a hydrocarbon feedstock is heated at elevated temperature and pressure for a fraction of second, which leads to C-C and C-H bonds breaking. As a result, mixture of olefins, aromatics, tar and gases are produced. These products are separated into specific boiling range cuts such as C1 to C4s etc. In this process, the overall yield of BD depends upon both, the process parameters and composition of feedstocks. Other than conventional steam cracking, BD can also be produced via “on-purpose” processes such as dehydrogenation and oxidative dehydrogenation and Catadiene processes [1].
The worldwide production of BD is estimated ~12 KTA in 2017 [2] and the synthetic elastomer plants are the largest consumer of BD to produce variety of synthetic elastomers mainly for the tire industries. The single largest application for BD is the production of Styrene-Butadiene rubber (SBR) and polybutadiene rubber (BR), which are principally used in the manufacturing of automobile tires [3]. The main outlet for BR is tire products but is also used as an impact modifier of polymers such as high impact polystyrene (HIPS). BD is also used in the manufacturing of acrylonitrile-BD-styrene (ABS), which is the largest-volume engineering thermoplastic resin. A growing outlet for ABS is the electronics industry where it is used in business machines, computers, radios, televisions and telephone handsets while another important market is the automobile industry
Other polymers made from BD include styrene-BD (SB) copolymer latex, which is used in paper coatings, carpet back coatings, foam mattresses and adhesives. Styrene-BD block copolymers have many applications ranging from asphalt modifiers in road and roofing construction to adhesives, footwear and toys. Nitrilerubbers, made by the copolymerization of acrylonitrile with BD, are used mainly in the manufacture of hoses, gasket seals and fuel lines for the automobile industry as well as in gloves and footwear. However, apart from synthetic elastomer industries, ~22% of global BD demand is to produce several intermediates and chemicals for various applications (Figure 2). For exampleadiponitrile is used to produce nylon fibers and polymers. Chloroprene is the monomer to produce polychloroprene, better known as Neoprene, which has a wide variety of uses such as wet suits, electrical insulation, car fan belts, gaskets, hoses, corrosion-resistant coatings and adhesives.
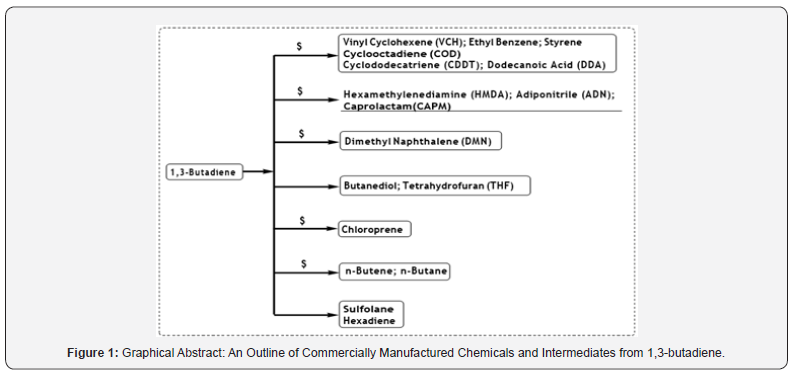
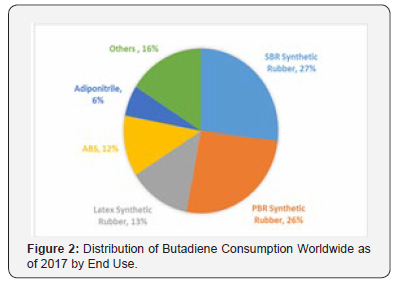
A variety of reviews are explicitly reported which enlightened the use of BD to produce various types of elastomers, polymers and co-polymers etc. However, literatures on the production, process and application of various industrial intermediate products and chemicals from BD is sparse. BD is a molecule with two reactive vinyl groups, offers a versatile chemistry which is industrially being exploited to produce various chemicals and intermediates. Hence, a state-of-the-art review of the chemistry behind the production of various industrial chemicals from BD would be highly advantageous to a wide population of researchers. Therefore, the aim of the present review is to provide a concise collection of the BD-based processes (excluding elastomer-based processes), which are currently being practiced in industries.For simplicity the present review is categorized based on the production volume of the chemicals from BD. The chemicals which are being practiced at industrial scale would be discussed under the heading of ‘high volume’ chemicals and which are at bench or pilot scale would be discussed under the heading of ‘low volume’. Moreover, the chemistry of the formation of an array of fine chemicals from BD, which were reported at lab scale, would also be discussed.
- Review Article
- Abstract
- Introduction
- Industrial Chemicals and Intermediates: High Volume Caprolactam by Hydrocyanation of 1, 3-butadiene
- Caprolactam by carboalkoxylation of 1,3-butadiene
- Chloroprene by chlorination of 1,3-butadiene
- 4-Vinyl cyclohexene, ethyl benzene and styrene from 1,3-butadiene
- 1,5-Cyclooctadiene (COD) from 1,3-butadiene
- Dodecanoic acid (DDA) by cyclotrimerization of 1,3-butadiene
- Industrial Chemicals and Intermediates: Low Volume
- Diels-Alder Reaction of 1,3-butadiene
- Miscellaneous other Useful Reactions of 1,3-butadiene
- Conclusions
- Acknowledgements
- References
Industrial Chemicals and Intermediates: High Volume Caprolactam by Hydrocyanation of 1, 3-butadiene
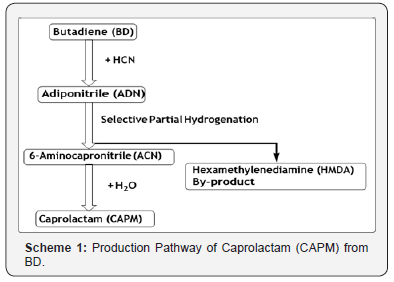
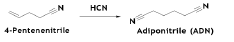
The reaction sequence for production of Caprolactam and/or Hexamethylenediamine (HMDA) via BD hydrocyanation reactionis shown below in Scheme 1. BD is hydrocyanated to adiponitrile (ADN) in two steps. Partial hydrogenation of ADN yields a mixture of HMDA and 6-aminocaprolacatm (ACN), which in turn might be converted to caprolactam (CAPM) by cyclization and complete hydrogenation of ADN results in HMDA [4].
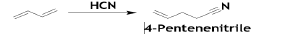
The first step is the hydrocyanation of BD yields a mixture of pentene and butenenitrile isomers. 4-Pentenenitriles (PN) are the main products and butenenitrile as a by-product
The reaction is conducted in the liquid phase at a temperature of 110°C and a pressure of 15 bar. The BD conversion is approximately 90%, with selectivity to mixture of penetenenitriles of approximately 97%. The reaction is catalyzed by combination of zero-valent Ni and tri-o-tolylphosphite ligands. The 3- and 4-pentenenitrile isomers are directly adaptable to ADN in the second hydrocyanation stage. The 2-pentenenitrile and butenenitriles are detrimental to catalyst efficiency and difficult to separate by distillation. They are removed from the system via conversion to adducts and then sent to waste treatment
In the second stage of the hydrocyanation, 4-pentenenitrile is further reacted with HCN to form adiponitrile (AND) [5]. Methyl- glutaronitrile and ethylsuccinonitrile by-products are also formed.
The reaction is conducted in the liquid phase at 50°C and 8 bar absolute pressure. PN conversion is 50%, with 92% selectivity to ADN. Glutaronitrile isomers are sent to waste treatment or may be recovered for the manufacture of beta-picoline.
Adiponitrile is fully hydrogenated to hexamethylenediamine (HMDA) in a fixed-bed reactor over reduced iron oxide catalyst in the presence of substantial ammonia (NH3/AND, molar ratio~35). Ammonia reduces the formation of hexamethyleneimine (HMI) in addition to facilitating the removal of reaction heat [6].

The reaction is conducted at 100°C and 310 bar pressure and ADN conversion is 100%, with 97% selectivity to HMDA. Major by-products in the reaction are ACN, HMI and Bis(hexamethylene) triamine. Other catalyst like Rh/Al2O3, was found to be effective for the ADN hydrogenation in very mild operating conditions in a three-phase stirred slurry reactor. Partial hydrogenation of ADN results 6-aminocapronitrile (ACN), which is a precursor of Caprolactam (CAPM). The reaction takes place at 50-80°C and approximately 1 to 7 Mpa catalyzed by Raney nickel promoted with Cr, Fe or Co. Selectivity to ACN decreases with increasing conversion, favoring formation of HMDA. HMI and small amounts of heavy amines are the principal by-products [7].
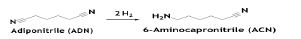
6-Aminocapronitrile might be converted to caprolactam (CAPM) by reacting it with water in either liquid or gas phase. Conductingthe reaction in the liquid phase over a TiO2 catalyst yields CAPM with 94% selectivity, with complete conversion of ACN. The reaction medium is an aqueous ethanol solution [8].
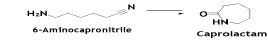
The reaction is conducted at 222°C and approximately 100 MPa. The by-products are open chain and cyclic nitriles. These by-products are different from those produced by conventional CAPM technology (Beckmann rearrangement of cyclohexanone oxime). The crude CAPM may be purified by hydrogenation followed by treatment with an acidic medium such as an iron-exchange resin. The purification is completed by distillation in an alkaline medium.
- Review Article
- Abstract
- Introduction
- Industrial Chemicals and Intermediates: High Volume Caprolactam by Hydrocyanation of 1, 3-butadiene
- Caprolactam by carboalkoxylation of 1,3-butadiene
- Chloroprene by chlorination of 1,3-butadiene
- 4-Vinyl cyclohexene, ethyl benzene and styrene from 1,3-butadiene
- 1,5-Cyclooctadiene (COD) from 1,3-butadiene
- Dodecanoic acid (DDA) by cyclotrimerization of 1,3-butadiene
- Industrial Chemicals and Intermediates: Low Volume
- Diels-Alder Reaction of 1,3-butadiene
- Miscellaneous other Useful Reactions of 1,3-butadiene
- Conclusions
- Acknowledgements
- References
Caprolactam by carboalkoxylation of 1,3-butadiene
Carboalkoxylation reaction refers to the reaction of CO and unsaturated hydrocarbon, in the presence of alcohol. The reaction sequence starting with carboalkoxylation of BD [9] and leading to either to ADA or CAPM is given below in Scheme 2;
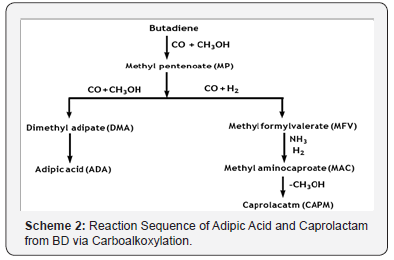
The products of the carboalkoxylation of BD are methyl pentenoate isomers, with the 3-isomer forming greatly
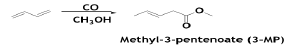
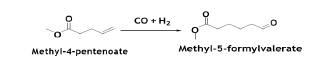
With a Pd-catalyst, the reaction is conducted in the liquid phase at 130°C and 950 psia in diphenyl ether solvent. The BD conversion is ~90%, with selectivities of ~96% and ~4% for the 3- and 2-isomers, respectively. Methyl-3-penetenoate isomerized to methyl- 4-penetenoate over a Pd-zeolite catalyst. The reaction proceeds at atmospheric pressure and at 135°C. The selectivity to methyl- 4-penetenoate is 100%. Methyl-4- pentenoate is hydroformylated to methyl formylvalerate isomers [10]. The conversion of methyl-4-pentenoate is ~94% with ~90% selectivity to 5-formylvalerate. The reaction is conducted at 150 psia and at 130°C in liquid phase with diphenyl ether solvent and Rh-based catalyst system.
The next step is to introduce an amine group into methyl- 5-formylavalearte by reacting it with ammonia in the presence ofH2. The reaction is catalyzed by Ru/alumina system. The reaction is conducted in the liquid phase at 130°C and 1470 psia, with xylene as a solvent. The conversion is >95% with 97% selectivity to methyl-6-aminocaproate [11].
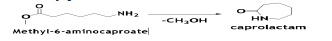
The last step is the hydrolytic cyclization of methyl-6-aminocaproate to Caprolactam (CAPM) in the presence of water. The uncatalyzed reaction is conducted at 270°C and 1470 psia with 93% conversion of methyl-6- aminocapraote [12].
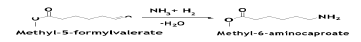
Important to mention here that methyl-3-penetenoate further reacts with CO and methanol at temperatures that are higher than those in the first stage carboalkoxylation in presence of Cobalt or Palladium based catalyst (pressure around 2175 psig and temperature 170°C). The primary product is dimethyl adipate, with some other by-products.
Methyl valerate is also formed in the hydrogenation of methyl- 3-penetenoate. Hydrogen is subjected to the reaction system with CO as an impurity but is also formed in the reaction between CO and methanol. The reaction is conducted in pyridine solvent in the liquid phase. The conversion is >90% with >78% selectivity to dimethyl adipate. Dimethyl adipate is hydrolyzed to Adipic acid over a strong acidic ion-exchange resin at 100°C and an autogenous pressure. The conversion of the diester is complete with 100% selectivity to adipic acid [13].

- Review Article
- Abstract
- Introduction
- Industrial Chemicals and Intermediates: High Volume Caprolactam by Hydrocyanation of 1, 3-butadiene
- Caprolactam by carboalkoxylation of 1,3-butadiene
- Chloroprene by chlorination of 1,3-butadiene
- 4-Vinyl cyclohexene, ethyl benzene and styrene from 1,3-butadiene
- 1,5-Cyclooctadiene (COD) from 1,3-butadiene
- Dodecanoic acid (DDA) by cyclotrimerization of 1,3-butadiene
- Industrial Chemicals and Intermediates: Low Volume
- Diels-Alder Reaction of 1,3-butadiene
- Miscellaneous other Useful Reactions of 1,3-butadiene
- Conclusions
- Acknowledgements
- References
1,5-Cyclooctadiene (COD) from 1,3-butadiene
The other isomeric product of cyclodimerization reaction is also of great interest from both academic and industrial viewpoint [24]. COD can be produced using the zero-valent nickel-based catalyst with bulky steric ligands such as tris-o-biphenyl phosphite. The production of COD by a catalyst prepared from Ni(CO)4 and phosphine the first report on cyclooligomerization of BD. Reduction of nickel(II)acetylacetonate with Al(C2H5)2(OC2H5) in the presence of phosphite forms a complex that catalyzed COD formation at 80°C and 1 atm. in 96% yield. The byproduct is usually VCH, a compound formed by thermal dimerization of BD. The favored ligand for selective COD formation is electron-withdrawing bulky phosphite while electron-releasing ligands such as tricyclohexyl phosphine coordinates more strongly to nickel and their steric effect favors the formation of the smaller VCH. A green and robust biphasic way of cyclodimerisation of BD into 1, 5-cyclooctadiene (COD) has been reported under fluorous–organic biphasic conditions using Ni(COD)2 as catalytic precursor and triarylphosphites bearing long perfluorinated alkyl chain separated from the aromatic ring by an ethylene spacer group as ligand [25]. In another example, BD cyclodimerization reaction was performed by an iron complex, prepared in-situ by the reduction of [Fe (NO)2Cl]2 with metallic zinc, diethylaluminum choride or n-butyllithium, dissolved in ionic liquids [26]. Recently, a detailed mechanistic understanding of structure– reactivity relationships for catalyst design has been reportedfor Iron-based complexes in the synthesis of cis, cis-1,5-cyclooctadiene (COD) from 1,3-butadiene [27].
COD is an intermediate for the manufacture of polyoctenamers, a wide variety of specialty products based on COD. Much of the current interest of COD is in ring opening metathesis polymerization (ROMP) reaction [28]. The ROMP of cyclooctene produces polyoctenylene, a unique elastomer also known as trans-polyoctenamer [29]. The, metathesis of COD, with ethylene gives a versatile intermediate 1, 5-hexadiene [30].
1, 4-Dichloro-2-butene, an intermediate in chloroprene manufacture, is employed to a limited extent as a starting material for adiponitrile, 1,4-butanediol (Scheme 5) and tetrahydrofuran. The most common application of 1, 4-butanediol is to production tetrahydrofuran via dehydration reaction. In the process, 1, 4-dichloro-2-butene is hydrolyzed at 110°C in the presence of an excess sodium formate (Scheme 6). After the hydrolysis, the free formic acid is neutralized with NaOH. The conversion is almost 100% with selectivity of ~90% with respect to 1, 4-butenediol. Direct hydrolysis lowers the selectivity due to enhanced polycondensation. The aqueous solution of 1, 4-butenediol is then hydrogenated to 1, 4-butanediol in the presence of Ni/Al catalyst at 1000C and 270 bars, which is further dehydrated to produce Tetrahydrofuran (THF) (Scheme 6).Recently, the experimental and theoretical investigations of the reaction of 1,3-Butadiene with Cl atom in the gas phase is also reported. Theoretical calculations were performed for the title reaction using CVT/SCT in combination with CCSD(T)/6-31+G (d,p)//MP2/6-311+G(2df,2p) level of theory [17]. C8- and C12-Cyclomers by cyclodimerization of 1,3-butadiene
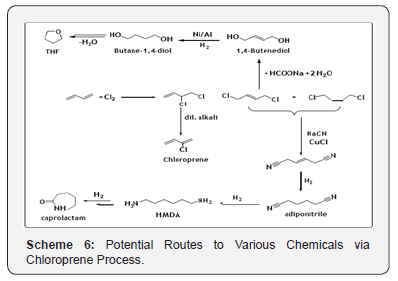
The formation of a variety of cyclic oligomers in the presence of different catalysts system is one of the unique and valuable features of BD. The possible commercially valued cyclomers of BD are shown below (Scheme 7) [18].
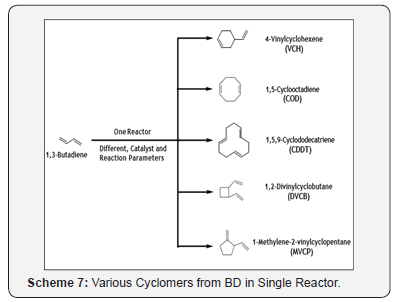
In the cyclodimerization reaction, the product distribution depends on the nature of the catalyst employed. A comprehensive theoretical and DFT investigation of the mechanism for cyclodimerization of BD has also been reported [19].
- Review Article
- Abstract
- Introduction
- Industrial Chemicals and Intermediates: High Volume Caprolactam by Hydrocyanation of 1, 3-butadiene
- Caprolactam by carboalkoxylation of 1,3-butadiene
- Chloroprene by chlorination of 1,3-butadiene
- 4-Vinyl cyclohexene, ethyl benzene and styrene from 1,3-butadiene
- 1,5-Cyclooctadiene (COD) from 1,3-butadiene
- Dodecanoic acid (DDA) by cyclotrimerization of 1,3-butadiene
- Industrial Chemicals and Intermediates: Low Volume
- Diels-Alder Reaction of 1,3-butadiene
- Miscellaneous other Useful Reactions of 1,3-butadiene
- Conclusions
- Acknowledgements
- References
4-Vinyl cyclohexene, ethyl benzene and styrene from 1,3-butadiene
Major commercial importance of BD cyclodimerization process is to produce VCH as it can be converted further to ethyl benzene (EB) and finally to styrene. The schematic representation of the pathway of styrene from BD is shown below in Scheme 8 [20].
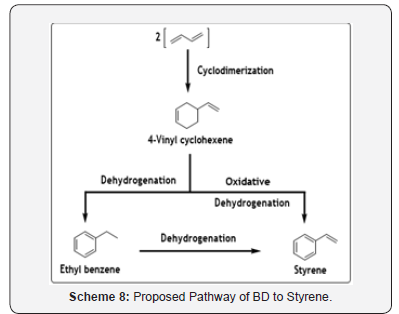
The gas-phase oxidative dehydrogenation of VCH to styrene in high selectivities was successfully carried out at moderate temperatures, 200–260°C, using a vanadium-based polyoxometalate, PV2Mo10O40 supported on carbon as catalyst [21]. In another report of oxidative dehydrogenation of 4-vinylcyclohexene to styrene using ZrO2- supported iron oxide catalysts, high styrene selectivity (88.9%) with VCH conversion of 80.1% at 400°C as well as a low rate of deactivation was achieved [22]. In one-pot protocol, it was reported that the large-pore NiII-phosphate, VSB-1, shows excellent selectivity (>80%) for the dehydrocyclodimerization of BD to ethylbenzene at 400°C [23].
- Review Article
- Abstract
- Introduction
- Industrial Chemicals and Intermediates: High Volume Caprolactam by Hydrocyanation of 1, 3-butadiene
- Caprolactam by carboalkoxylation of 1,3-butadiene
- Chloroprene by chlorination of 1,3-butadiene
- 4-Vinyl cyclohexene, ethyl benzene and styrene from 1,3-butadiene
- 1,5-Cyclooctadiene (COD) from 1,3-butadiene
- Dodecanoic acid (DDA) by cyclotrimerization of 1,3-butadiene
- Industrial Chemicals and Intermediates: Low Volume
- Diels-Alder Reaction of 1,3-butadiene
- Miscellaneous other Useful Reactions of 1,3-butadiene
- Conclusions
- Acknowledgements
- References
1,5-Cyclooctadiene (COD) from 1,3-butadiene
The other isomeric product of cyclodimerization reaction is also of great interest from both academic and industrial viewpoint [24]. COD can be produced using the zero-valent nickel-based catalyst with bulky steric ligands such as tris-o-biphenyl phosphite. The production of COD by a catalyst prepared from Ni(CO)4 and phosphine the first report on cyclooligomerization of BD. Reduction of nickel(II)acetylacetonate with Al(C2H5)2(OC2H5) in the presence of phosphite forms a complex that catalyzed COD formation at 80°C and 1 atm. in 96% yield. The byproduct is usually VCH, a compound formed by thermal dimerization of BD. The favored ligand for selective COD formation is electron-withdrawing bulky phosphite while electron-releasing ligands such as tricyclohexyl phosphine coordinates more strongly to nickel and their steric effect favors the formation of the smaller VCH. A green and robust biphasic way of cyclodimerisation of BD into 1, 5-cyclooctadiene (COD) has been reported under fluorous–organic biphasic conditions using Ni(COD)2 as catalytic precursor and triarylphosphites bearing long perfluorinated alkyl chain separated from the aromatic ring by an ethylene spacer group as ligand [25]. In another example, BD cyclodimerization reaction was performed by an iron complex, prepared in-situ by the reduction of [Fe (NO)2Cl]2 with metallic zinc, diethylaluminum choride or n-butyllithium, dissolved in ionic liquids [26]. Recently, a detailed mechanistic understanding of structure– reactivity relationships for catalyst design has been reportedfor Iron-based complexes in the synthesis of cis, cis-1,5-cyclooctadiene (COD) from 1,3-butadiene [27].
COD is an intermediate for the manufacture of polyoctenamers, a wide variety of specialty products based on COD. Much of the current interest of COD is in ring opening metathesis polymerization (ROMP) reaction [28]. The ROMP of cyclooctene produces polyoctenylene, a unique elastomer also known as trans-polyoctenamer [29]. The, metathesis of COD, with ethylene gives a versatile intermediate 1, 5-hexadiene [30].
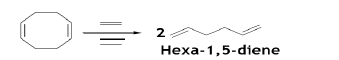
Additionally, the metathesis of cyclooctene with ethylene under similar conditions gives 1, 9-decadiene, which can be hydroformylated to produce undecenal, used to modify the fragrance of perfumes. The selective hydrogenation of COD yields an important chemical, cyclooctene. COD is also hydrogenated to cyclooctane which in turn oxidized to cyclooctanone and suberic acid for the production of Nylon 6/8 [31].
- Review Article
- Abstract
- Introduction
- Industrial Chemicals and Intermediates: High Volume Caprolactam by Hydrocyanation of 1, 3-butadiene
- Caprolactam by carboalkoxylation of 1,3-butadiene
- Chloroprene by chlorination of 1,3-butadiene
- 4-Vinyl cyclohexene, ethyl benzene and styrene from 1,3-butadiene
- 1,5-Cyclooctadiene (COD) from 1,3-butadiene
- Dodecanoic acid (DDA) by cyclotrimerization of 1,3-butadiene
- Industrial Chemicals and Intermediates: Low Volume
- Diels-Alder Reaction of 1,3-butadiene
- Miscellaneous other Useful Reactions of 1,3-butadiene
- Conclusions
- Acknowledgements
- References
Dodecanoic acid (DDA) by cyclotrimerization of 1,3-butadiene
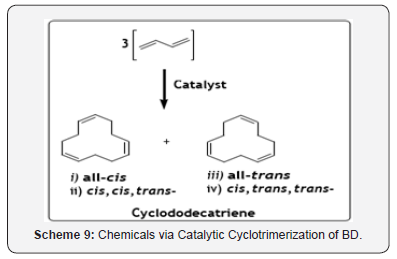
The cyclotrimerization products of BD are cyclododecatrienes (CDDT), which are also as important as other BD’s cyclodimer products (Scheme 9). The cyclotrimerization of BD to CDDT can be done efficiently with three different types of catalyst systems “Naked Nickel”, Titanium (IV)chloride/ethylaluminium sesquichloride and Chromyl chloride/triethyl aluminium. Generally, following four stereoisomers; all-cis, cis, trans- and all-trans CDDT, are produced. However, the product distribution essentially depends on the catalytic system and reaction condition employed [32].

A general observation is that with TiCl4/Et2Al2Cl3 as a catalyst, the main product is cis, trans, and trans- CDDT. The “naked nickel” will produce all-trans-CDDT as the main product while the systemCrOCl2/AlEt3 will produce mixture of all-trans and cis, trans, trans-CDDT [33]. During the process, COD and VCH were formed as side products due to cyclodimerization reaction of BD. The product CDDT finds application as an important intermediate in the manufacture of Nylon-12 via the cyclododecane-lauryl lactam route.
Moreover, CDDT is the starting martial for the manufacture of 1, 12-dodecanoic acid, also known as 1, 10- decanedicarboxylic mainly uses in polyamide and polyesters as its diester in lubricants. The CDDT can be converted into dodecanedioic acid in three-step reaction sequence [34]. Initially, cyclododecane is manufactured by liquid- phase hydrogenation at 200°C and 10-15 bars in presence of Ni-catalysts. Cyclododecane is then oxidized to a cyclododenol/ cyclododecanone mixture at 150-160°C and atmospheric pressure in the presence of boric acid in a manner analogous to the route involving cyclohexane and oxygen or air. With 25-30% conversion, the selectivity to alcohol/ketone mixture reaches an optimum 80- 82% and a ratio of 8 to 10:1. The next stage, involving oxidative ring cleavage of the alcohol/ketone, done commercially with HNO3 (Scheme 10).
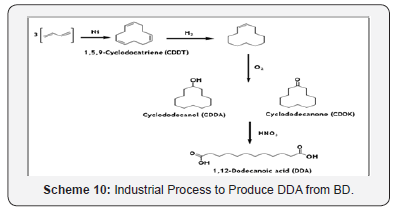
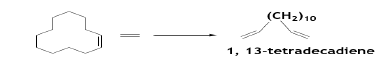
The selective hydrogenation of cyclododecatriene produces cyclododecene in high yields (>98%) using either heterogeneous or homogeneous catalysts [35]. The ROMP of CDDT with ethylene produces long chain terminal unsaturated 1, 13-tetradecadiene [36].
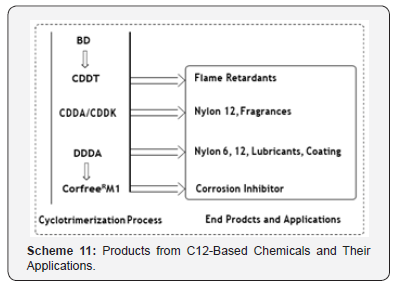
The triple-unsaturated character of the C12 ring permits a wide range of chemical reactions, making CDDT one of the most important starting materials for cyclic and linear compounds with 12 carbon atoms (Scheme 11).
- Review Article
- Abstract
- Introduction
- Industrial Chemicals and Intermediates: High Volume Caprolactam by Hydrocyanation of 1, 3-butadiene
- Caprolactam by carboalkoxylation of 1,3-butadiene
- Chloroprene by chlorination of 1,3-butadiene
- 4-Vinyl cyclohexene, ethyl benzene and styrene from 1,3-butadiene
- 1,5-Cyclooctadiene (COD) from 1,3-butadiene
- Dodecanoic acid (DDA) by cyclotrimerization of 1,3-butadiene
- Industrial Chemicals and Intermediates: Low Volume
- Diels-Alder Reaction of 1,3-butadiene
- Miscellaneous other Useful Reactions of 1,3-butadiene
- Conclusions
- Acknowledgements
- References
Industrial Chemicals and Intermediates: Low Volume
2,6-Dimethyl naphthalene (DMN) by alkenylation reaction of 1,3-butadiene and o-xylene
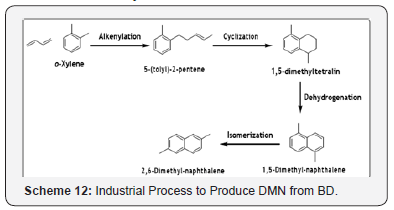
2, 6-Dimethyl naphthalene (DMN) is being produced on a commercial scale by a technology based on o-xylene and BD as the starting materials [37]. Four step processes are required to get final product DMN (Scheme 12). The alkenylation reaction is conducted with 5:1 molar excess of o-xylene. The catalyst is potassium immobilized on potassium carbonate. BD conversion is complete, with approximately, 70% selectivity to 5-(tolyl)-2-pentene. The by-products are multi-alkenylated xylenes. The cyclization may be carried out either in liquid or vapor phase. Liquid-phase cyclization is catalyzed by a zeolite catalyst yielding 1, 5-dimethyltetralin, with 93% yield. The dehydrogenation to 1, 5-dimethylnaphthalene is performed in the liquid phase at 400°C and 200 psig. The catalyst is noble metal, a mixture of Pt and Rh, on an alumina support. Both the conversion and selectivity are in the range of 99%. The isomerization and associated isomer separation are key to the economics of producing the preferred 2, 6- isomer. The isomerization may be conducted at 75 psig and 250°C. The reaction is catalyzed by a zeolite catalyst. The isomers may be separated by fractional crystallization or selective adsorption. DMN of 99% purity is obtained [38].
The DMN finds commercial application in the manufacture of polyethylene naphthalate (PEN). In this application, DMN is oxidized first to the corresponding dicarboxylic acid, which in turn is esterified to dimethyl-2, 6- naphthalenedicarboxylate to ensure the product purity. The ester is then reacted to ethylene glycol to produce PEN [39].
Sulfolane from 1,3-butadiene
BD reacts with SO2 in a reversible reaction via 1, 4-addition manner to form sulfolene, which can be hydrogenated to give thermally stable sulfolane [40].
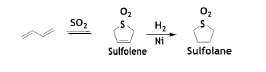
At approximately 125°C, sulfolene decomposes to BD and SO2. Such method could also be used to separate BD from a mixture of C4 olefins because the olefins do not react with SO2. Sulfolane is stable aprotic industrial water-soluble solvent used in aromatic distillation of aromatics or together with diisopropanolamine in the “Sulfinol Process” for gas purification process.
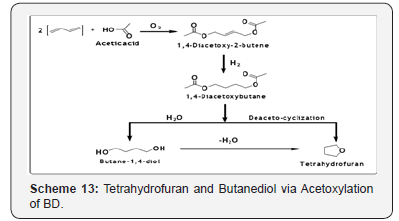
1, 4-Butanediol and tetrahydrofuran by acetoxylation reaction of 1,3-butadiene
The acetoxylation reaction of BD is conducted using acetic acid and oxygen contained in air which results formation of 1, 4-dicetoxy- 2-butene followed by hydrogenation and hydrolysis to 1,4-buatnediols (BDO) [41] and by deacetocyclization to tetrahydrofuran (THF) [42].
The acetoxylation reaction is conducted around 90-95°C and 850 psig. The catalyst is Pd and Te on active carbon. BD conversion is approximately 80-85% with a selectivity of 88%. The primary by-product is 3, 4-diacetoxy- 1-butene, which is recycled to the acetoxylation reactor (Scheme 13). The reaction is conducted with high concentration of acetic acid (approximately 12-times of BD). Hydrogenation is conducted at 1280 psig in two reaction zones at reaction temperature of 66°C and 110°C in zone one and two, respectively. The catalyst is Pd on active carbon in both the reaction zones. The conversion of 1, 4-diacetoxybut-2-ene is complete with 97% selectivity to corresponding butane derivative.
The by-product is n-butyl acetate. The hydrolysis is conducted in two stages: in the first-stage hydrolysis reactor, 70% of the diacetoxybutane is converted acetoxyhydroxybuatne with 100% selectivity. In the second stage, rest of the diacetoxybutane is converted to BDO. The effluent from the hydrolysis reactor is separated into a BDO fraction and BDO/acetoxyhydroxybutane fraction. The BDO content of the latter is the key to the BDO/THF output from the operation, since BDO is converted with 100% selectivity to THF, whereas the selectivity to THF from acetoxyhydroxy butane is only 50%. The hydrolysis is catalyzed with an ion-exchange resin. Alternatively, the 1, 4-diacetoxybuatne can undergo deacetocyclization to produce THF.
- Review Article
- Abstract
- Introduction
- Industrial Chemicals and Intermediates: High Volume Caprolactam by Hydrocyanation of 1, 3-butadiene
- Caprolactam by carboalkoxylation of 1,3-butadiene
- Chloroprene by chlorination of 1,3-butadiene
- 4-Vinyl cyclohexene, ethyl benzene and styrene from 1,3-butadiene
- 1,5-Cyclooctadiene (COD) from 1,3-butadiene
- Dodecanoic acid (DDA) by cyclotrimerization of 1,3-butadiene
- Industrial Chemicals and Intermediates: Low Volume
- Diels-Alder Reaction of 1,3-butadiene
- Miscellaneous other Useful Reactions of 1,3-butadiene
- Conclusions
- Acknowledgements
- References
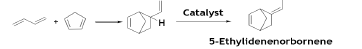
Diels-Alder Reaction of 1,3-butadiene
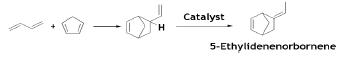
A classic example of Diels-Alder reaction of BD with cyclopentadiene for production of 5-ethylidenenorbornene (EDNBN) a monomer useful in the production of EPDM rubber [43].
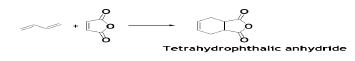
In another interesting example of D-A reaction, BD reacts with maleic anhydride to give tetrahydrophthalic anhydride which being used in polyester and alkyd resins and as an ingredient in the fungicide, Captan [44].
Fine chemicals from 1,3-butadiene at lab scale hydroxylation of BD
The hydroxylation of BD can be carried out by reaction of BD with water along with carbon dioxide (CO2) in presence a Palladium salt [45]. The product 2, 7-octadiene-1-ol which has a variety of applications; its main market involves hydrogenation to n-octanol, whose phthalate diester is utilized as a plasticizer with superior properties to the currently used branched octanol (2-ethylhexanol).
The reaction is carried out with BD in sulfolane/water in the presence of Pd (OAc)2 as a catalyst precursor and a soluble triarylphosphine (or its phosphonium bicarbonate, which is formed from octadienol itself and carbon dioxide) as ligand. The selectivity to 2, 7-octadien-1-ol is 92-94% (TOF > 1000), while the other isomers accounts for 3-5%. The product is extracted with hexane, while the aqueous sulfolane solution, containing the catalyst and triethylamine, is recycled. To increase the catalyst life and efficiency, the soluble phosphines or its phosphonium salt, used in a molar excess of about 50 over palladium, stabilizes the palladium complex in aqueous solution; the sulfolane-water solution ensures the solubility of the reactant, while extraction with hexane under CO2 pressure recovers the product with only less contamination by palladium, phosphorous or nitrogen. The phosphines or its phosphonium salt and the ammonium bicarbonate remain in the aqueous solution.
Epoxidation of 1,3-butadiene
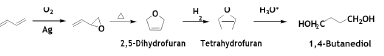
Another attractive and economical route for the production of 1, 4-butanediol was developed by Eastman Chemical [46]. In this process, BD is converted to 3, 4-epoxy-1-butene with air over silver catalyst. After thermal rearrangement to 2, 5-dihydrofuran and hydrogenation to tetrahydrofuran, the final hydrolysis gives 1, 4-butandiol. The elegance of the present technology is that the intermediate steps are also raw materials for other compounds.
Telomerization reaction of 1,3-butadiene and carbon dioxide
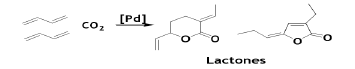
Furthermore, the Pd-catalyzed reaction of CO2 with BD givesvaluable products like five- and six-member lactones depending upon the ligands and reaction condition employed [47].
Inter-cyclodimerization of 1,3-butadiene
1, 4-Hexadiene is utilized as a third co-monomer in ethylene/ propylene elastomers. While the terminal double bond is incorporated into the polymeric chain, the internal one is not and can be cross-linked (vulcanized) to confer elasticity to the materials. 1, 4-Hexadiene (1, 4-HXD) in its E-form is manufactured from BD and ethylene. The Z- isomer must be kept to a minimum owing to its adverse effect on polymerization. Various metal salts (Ni, Co, Fe) were tested, but the best results were obtained with RhCl3.hydrate [48].
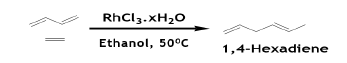
Optimal conversion to 1, 4-HXD is favored by a large excess of ethanol over RhCl3.xH2O. 3-Methyl-1, 4-pentadiene and 2, 4-hexadiene are also formed in low concentrations. BD and ethylene are used in excess to restrict isomerization to 2, 4-hexadiene as they compete with 1, 4-hexadiene for the coordination site involved. The reaction is stero selective, 1, 4-HXD being mainly obtained in the E-configuration; the Z-isomers are present only to a small extent and its formation can further decreased by donor ligands. The reaction is also regioselective: the positional isomer CH2=CHCH(CH3) CH=CH2, derived from ethylene insertion into the more substituted carbon-rhodium bond of the allylic system, is present less than 1% when ethanol is in large excess. It is interesting to note that the same a catalyst can dimerize ethylene at a comparable rate.
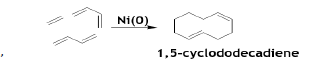
In addition to homo-oligomerization, BD undergoes cyclooligomerization with unsaturated bonds of alkenes and alkynes offering simple synthetic methods of cyclic compounds of various sizes. 1, 5-cyclododecadiene is formed by [4+4+2] cycloaddition of ethylene and BD in 1:2 ratio using Ni/P(OPh)3 based catalyst system [49].
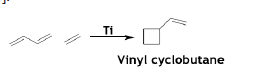
The reaction in the ratio of 1:1, catalyzed by Ti complex having bipyridyl ligands, produces vinyl cyclobutane in remarkable yield [50].
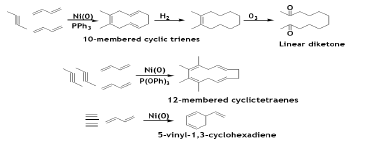
The 10-membered 1, 2-disubstituted cyclododecatriene can be prepared in high yield by the cooligomerization of alkyne and BD in 1:2 ratio. Furthermore, selective hydrogenation and subsequent ozonization afford the linear diketone, which off-course can further be converted to useful chemicals. Under the high concentration of alkynes, the cooligomerization of alkyne and BD in 2:2 ratio takesplace to give 12-membered cyclic tetraene. Using Ni/Bu3P catalytic system, 5-vinyl-1, 3-cyclohexadiene can be obtained by treating BD
- Review Article
- Abstract
- Introduction
- Industrial Chemicals and Intermediates: High Volume Caprolactam by Hydrocyanation of 1, 3-butadiene
- Caprolactam by carboalkoxylation of 1,3-butadiene
- Chloroprene by chlorination of 1,3-butadiene
- 4-Vinyl cyclohexene, ethyl benzene and styrene from 1,3-butadiene
- 1,5-Cyclooctadiene (COD) from 1,3-butadiene
- Dodecanoic acid (DDA) by cyclotrimerization of 1,3-butadiene
- Industrial Chemicals and Intermediates: Low Volume
- Diels-Alder Reaction of 1,3-butadiene
- Miscellaneous other Useful Reactions of 1,3-butadiene
- Conclusions
- Acknowledgements
- References
Miscellaneous other Useful Reactions of 1,3-butadiene
Octatrienes are used as industrial precursors for the manufacturing of butylene oligomers, such as, 1, 5- and 1, 6-octadienes for the synthesis of different bicyclic alcohols. These octatrienes could be synthesized at very high yield (yield 92%) by selective dimerization of BD using Pd-carbene based catalyst in in presence of 2-propanol and a base, sodium isopropoxide [52].
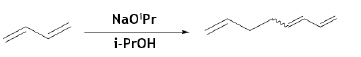
1, 6-hexanedial (adipic aldehyde) is an attractive precursor to the synthesis of many more valuable C6-compounds (e.g., adipic acid, hexamethylenediamine, ε-caprolactone, and 1, 6-hexanediol). BD could be used for the desired linear dual hydroformylation product (1, 6-hexanedial, also known as adipic aldehyde) using Rhodium catalyzed reaction with 50% selectivity. However, the reaction parameters (syngas pressure, H2 and CO partial pressures, temperature) do have a substantial effect on the selectivity for the products, resulting from the branched addition of the rhodium hydride to the C=C double bond [53]. The reaction mechanism of Rh-catalyzed hydroformylation of 1,3- butadiene to adipic aldehyde was also studied by in-situ NMR and IR experiments, kinetic measurements, and deuterioformylation. The rate law of the reaction was reported to be 2nd-order in syngas pressure and independent of the butadiene concentration [54].
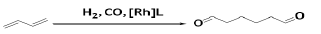
Carbon-Silicon bond formation reaction has excessive importance on the synthesis of organosilanes for various applications. However, example of such synthesis is rare in literature owing to difficulties in the oxidative addition of Si-Cl bond with low-valent transition metal complexes. Synthesis of disillylated dimer of BD could be performed with high regio- and stereo- selectivity when BD was reacted with chlorosilanes and Grignard reagents at 20°C in the presence of a catalytic amount of Pd-based metal [55].
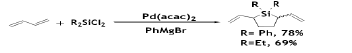
However, in a multicomponent coupling reaction, BD underwent Ni‐catalyzed dimerization and carbosilylation in the presence of a chlorosilane and a Grignard reagent. The coupling product was obtained regio‐ an stereoselectively if unsubstituted BD was used [56].
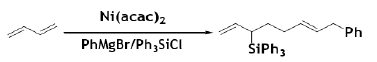
The C=N bonds of isocyanate and Schiff bases reacts with BD in the presence of Pd-based catalysts to give the piperidone and peperidiene [57].
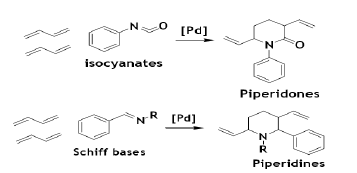
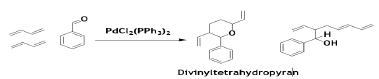
The Pd-catalyzed co-cyclization occurs with some C=O and –CN bonds to give heterocycles. The divinyltetrahydropyran and the unsaturated alcohol are obtained by the reaction aldehydes with BD. The unsaturated alcohol is obtained as a main product when a ratio of PPh3 and Pd is 1 to 2, and pyran is the main product when the ratio is larger than 3 [58].
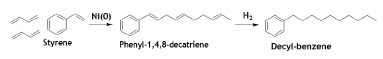
The Ni-catalyzed 1:2 addition of styrene and BD gives 1, 4, 8-phenyldecatriene which is then hydrogenated to get n-decyl benzene [59].
The phenol is a reactive substrate and smoothly gives octadienyl phenyl ether in high yield. Primary alcohols such as CH3OH react easily to form ethers [60].
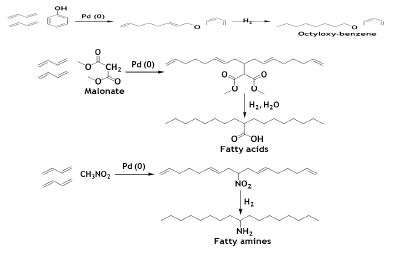
Malonate and nitro methane react with BD to give fatty acid and amines respectively after a hydrogenating the olefinic moiety [61].
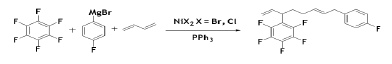
Organic fluorinated compounds have very high importance in the crop protection research. Synthesis of organo- fluorinated compounds could be performed in presence of anionic Ni (II) complex through a multicomponent coupling reaction of per-fluoroarenes with 1,3-butadiene and aryl Grignard Reagents [62]. In this case, use of fluorinated Grignard reagent might lead to highly fluorinated product by multi-component coupling reaction. The maximum yield (88%) of the reaction was obtained with NiCl2 with PPh3 ligand. The reaction was believed to occur via a Ni (II) anionic spices as an active catalyst which was isolated as orange solid by treatment of Ni (COD)2 with PhLi and 1,3-butadiene in dry ether at -78°C. The active catalyst was further trapped by the addition 12-crown-4.
Activation of BD under photochemical and discharge-driven pathways in gas-phase was also performed [63]. The reaction was initiated in photo-catalysis method by using a laser pulse of 218 nm wavelength in inert atmosphere in a counter-propagated the reaction tube. However, detection of products was performed either using single-photon vacuum ultraviolet (VUV) photoionization (118nm = 10.5eV) or resonant two-photon ionization (R2PI) spectroscopy in a time-of-flight mass spectrometer. According to the R2PI spectra, a total of about 15 aromatic products were recorded from the BD discharge. Amongst them, toluene, styrene, phenylacetylene, o-, m-, and p-xylene, ethylbenzene, indane, indene, β-methylstyrene and naphthalene were formed in significant amount.
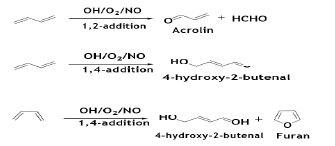
The gas-phase reaction of OH radicals with BD in the presence of NO has been investigated in a flow tube at room temperature in synthetic air of pressure 950 mbar pressure [64]. The reaction is dependent on the mode of addition of OH radical and O2. In case of 1, 2- addition, the major products were formaldehyde and acrolin while 1, 4-addition on trans-1, 3-butadiene produced 4-hydroxy- 2-butenal. The 1, 4-addition on cis-1, 3-butadiene produced furan along with 4-hydroxy-2-butenal by internal condensation
TThe Pd-catalyzed Carboxytelomerization of BD to form Linear α, ω-C10-Diester using phosphine or phosphite based ligand under CO atmosphere in methanol has been reported [65]. These types of compounds such as methoxycarbonylation and the carbonylation of allylic compounds certainly have importance in industrial and academic research. The reaction could be carried out in tandem pathway as well in stepwise pathway. In tandem catalysis pathway, formation of carboxytelomerization followed by acid-triggered methoxycarbonylation was successfully conductedyielding 22% of the desired C10-diester. The methane sulfonic acid (MSA) was charged after conducting the reaction for 2h to obtain the desire product. It was further isolated by distillation which was followed by acid treatment leading to the desired C10-diester (yield 84%).

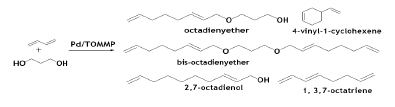
The Pd/TOMMP (tris-(o-methoxyphenyl) phosphine) system for the telomerization reaction of BD with different alcohols (primary and secondary alcohols) without using any solvent has also been reported [66]. The primary alcohols with monohydroxy group produced 80% yield of mono telomerized product with 100% selectivity. However, the selectivity lowered with increasing the numbers of hydroxyl groups. Reaction between BD and 1, 3- propanol under such condition produced mono telomerized product with 60% yield and 80% selectivity. The rate of the telomerization reaction was largely depend on the temperature. At 333K conversion of the above reaction was 20% after 90 minutes whereas at 353K, conversions over 70%.
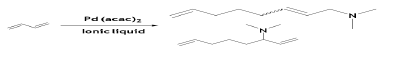
Telomarization of BD with dimethyl amine was also possible in presence of Pd(acac)2 and triphenylphosphate trisulfonate (TPPS) ligand in dimethylammonium dimethyl carbamate (dimcarb) ionic liquid at 80°C in 2h [67]. Cyclohexane was used as an extracting agent to overcome the miscibility effect of the product with the ionic liquid.
Pd-catalyzed telomerization of butadiene with methanol was also reported using various sterically encumbered and poorly electron-donating oxaphosphaadamantane ligands. The ligand 1,3,5,7-tetramethyl-6-(2-methoxyphenyl)- 2,4,8-trioxa-6-phosphaadamantane converted 96% of butadiene with 94% selectivity for 1-methoxy-2,7-octadiene at 70°C and is highly active for telomerization at a low reaction temperature (40°C) [68].
The Pd-catalyzed telomerization of butadiene with polyols has also been reported recently under aqueous and biphasic medium leading to the corresponding octadienylethers. It is reported that depending upon the types of organic co‐solvent and nature of the catalyst employed to promote the reaction, mono‐ to fully substituted ethers could be obtained with high yields [69].
The recycling of the homogeneous Pd-catalyst has been reported recently for the telomerization of 1,3- dienes with functionalized nucleophiles. The phase separation was achieved by a unique strategy of addition of 1,3- butadiene instead of an additional solvent which offered an elegant way of catalyst recycling. With this method an effective recycling of catalyst was reported atotal turnover number of >1400 [70]. A latest review on the advances and developments in the field of telomerization starting from 1,3‐dienes and over the last decade has been reported. The focus was on the conversion of renewable resources, the synthesis of promising new products and the development of efficient process concepts to combine these [71].
An enantioselective route for the synthesis of the Anti-Influenza Neuramidase Inhibitor Oseltamivir (Tamiflu) from BD and acrylic acid has been reported, which has great future potential [72].
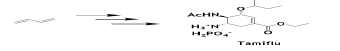
Additionally, the chiral tertiary alcohols are important building blocks for the synthesis of many pharmaceutical agents and biologically active natural products and the most common approach to synthesize them relies on stoichiometric organometallic reagents which are difficult to prepare, sensitive and uneconomical. Recently, a new mild and efficient protocol was reported using widely available 1,3-dienes which was applied to the preparation of an important drug, (R)-procyclidine and a key intermediate in the synthesis of several pharmaceuticals [73].

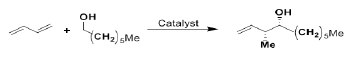
The Ru-catalyzed, catalytic asymmetric hydrohydroxyalkylation of butadiene to α-methyl homoallylic alcohols has been reported. The DFT calculations predicted that the product distribution is controlled by the kinetics of carbon–carbon bond formation [74-145].
- Review Article
- Abstract
- Introduction
- Industrial Chemicals and Intermediates: High Volume Caprolactam by Hydrocyanation of 1, 3-butadiene
- Caprolactam by carboalkoxylation of 1,3-butadiene
- Chloroprene by chlorination of 1,3-butadiene
- 4-Vinyl cyclohexene, ethyl benzene and styrene from 1,3-butadiene
- 1,5-Cyclooctadiene (COD) from 1,3-butadiene
- Dodecanoic acid (DDA) by cyclotrimerization of 1,3-butadiene
- Industrial Chemicals and Intermediates: Low Volume
- Diels-Alder Reaction of 1,3-butadiene
- Miscellaneous other Useful Reactions of 1,3-butadiene
- Conclusions
- Acknowledgements
- References
Conclusions
The present review covered the production technologies of various non-elastomeric industrially important intermediate and chemicals which are being produced from BD. It was also discussed that BD as a molecule with two reactive vinyl groups, offers a versatile chemistry which is industrially being exploited to produce various chemicals and intermediates. The aim of the present review was to provide a concise collection of the BDbased processes (excluding elastomer-based processes), which are currently being practiced in industries. For simplicity the present review is categorized based on the production volume of the chemicals from BD. The chemicals which are being practiced at industrial scale have been discussed under the heading of ‘high volume’ chemicals and which are at bench or pilot scale have been discussed under the heading of ‘low volume’. Moreover, the chemistry of the formation of several platform chemicals from BD, which were reported at lab scale, was also discussed in detail. Finally, we can conclude that BD will be remain as an attractivefeedstock chemical for the petrochemical industries not only to produce synthetic elastomers but also to various industrially important intermediates and chemicals for a variety of applications
- Review Article
- Abstract
- Introduction
- Industrial Chemicals and Intermediates: High Volume Caprolactam by Hydrocyanation of 1, 3-butadiene
- Caprolactam by carboalkoxylation of 1,3-butadiene
- Chloroprene by chlorination of 1,3-butadiene
- 4-Vinyl cyclohexene, ethyl benzene and styrene from 1,3-butadiene
- 1,5-Cyclooctadiene (COD) from 1,3-butadiene
- Dodecanoic acid (DDA) by cyclotrimerization of 1,3-butadiene
- Industrial Chemicals and Intermediates: Low Volume
- Diels-Alder Reaction of 1,3-butadiene
- Miscellaneous other Useful Reactions of 1,3-butadiene
- Conclusions
- Acknowledgements
- References
Acknowledgements
The authors are thankful to Reliance Industries Ltd. for the consent of publishing the review article.
- Review Article
- Abstract
- Introduction
- Industrial Chemicals and Intermediates: High Volume Caprolactam by Hydrocyanation of 1, 3-butadiene
- Caprolactam by carboalkoxylation of 1,3-butadiene
- Chloroprene by chlorination of 1,3-butadiene
- 4-Vinyl cyclohexene, ethyl benzene and styrene from 1,3-butadiene
- 1,5-Cyclooctadiene (COD) from 1,3-butadiene
- Dodecanoic acid (DDA) by cyclotrimerization of 1,3-butadiene
- Industrial Chemicals and Intermediates: Low Volume
- Diels-Alder Reaction of 1,3-butadiene
- Miscellaneous other Useful Reactions of 1,3-butadiene
- Conclusions
- Acknowledgements
- References
References
- Deb K (2014) Multi-objective optimization. Search methodologies, Springer, pp. 403-449.
- Skogestad S, Postlethwaite I (2007) Multivariable feedback control: analysis and design. Wiley New York, USA.
- Sharifzadeh M, (2013) Integration of process design and control: A review. Chemical Engineering Research and Design 91(12): 2515-2549
- Subramanian S, Georgakis C (2001) Steady-state operability characteristics of idealized reactors. Chemical engineering science 56(17): 5111-5130.
- Ekawati E, Bahri PA (2003) The integration of the output controllability index within the dynamic operability framework in process system design. Journal of Process Control 13(8): 717-727.
- Farsi M, Mazinani S, Jahanmiri A (2011) Steady state operability characteristics of an adiabatic fixed-bed reactor for methanol dehydration, Iran. J Chem Chem Eng Research Note 30(4).
- Santoso H, Rojas OJ, Bao J, Lee P (2007) Nonlinear process operability analysis based on steady-state simulation: a case study. Chemical Product and Process Modeling 2(2).
- Setiawan R, Hioe D, Bao J Plantwide operability analysis based on a network perspective A study on the Tennessee Eastman process Proc. Dynamics and Control of Process Systems 451-456.
- Yancheshmeh MS, Haghighi SS, Gholipour M, Dehghani O (2013) Modeling of ethane pyrolysis process A study on effects of steam and carbon dioxide on ethylene and hydrogen productions. Chemical Engineering Journal 215: 550-560.
- Keyvanloo K, Towfighi J, Sadrameli S, Mohamadalizadeh A (2010) Investigating the effect of key factors their interactions and optimization of naphtha steam cracking by statistical design of experiments. Journal of Analytical and Applied Pyrolysis 87(2): 224-230.
- Gao GY, Wang M, Ramshaw C, Li XG (2009) Optimal operation of tubular reactors for naphtha cracking by numerical simulation. Asia‐Pacific Journal of Chemical Engineering 4(6): 885-892.
- Masoumi M, Sadrameli S, Towfighi J, Niaei A (2006) Simulation optimization and control of a thermal cracking furnace Energy 31(4): 516- 527.
- Berreni M, Wang M (2011) Modelling and dynamic optimization of thermal cracking of propane for ethylene manufacturing. Computers & Chemical Engineering 35(12): 2876-2885.
- Seferlis P, Georgiadis MC (2004) The integration of process design and control. Elsevier.
- Madetoja E, Ruotsalainen H, Mönkkönen VM, Hämäläinen J, Visualizing multi-dimensional Pareto-optimal fronts with a 3D virtual reality system Proc. Computer Science and Information Technology (2008) IMCSIT (2008). International Multiconference on IEEE 907-913.
- Zou R, Lou Q, Mo S, Feng S (1993) Study on a kinetic model of atmospheric gas oil pyrolysis and coke deposition. Industrial & engineering chemistry research 32(5): 843-847.
- Etghani MM, Shojaeefard MH, Khalkhali A, Akbari M (2013) A hybrid






























