Diagnosis and Treatment of Hepatitis-C in Bhalwal : Case Report
Asif Bilal* and Urooj Fatima
Department of Zoology, University of Lahore, Pakistan
Submission:June 14, 2021; Published: June 25, 2021
*Corresponding author: Asif Bilal, Department of Zoology, University of Lahore, Pakistan
How to cite this article: Asif B, Urooj F. Diagnosis and Treatment of Hepatitis-C in Bhalwal : Case Report. Palliat Med Care Int J. 2021; 4(1): 555630. DOI: 10.19080/PMCIJ.2021.04.555630
Abstract
In Pakistan, around 10 million people are affected with the hepatitis C virus (HCV). Hepatitis B and C are infectious liver disorders caused by the hepatitis B (HBV) and hepatitis C (HCV) viruses. They can be acute or chronic. There are five identified pathogens, A-E, as well as non-A-E hepatitis viruses. This is a case report of 54 years old housewife from Bhalwal city. . About 14 years before, she was detected HCV. She was tested by different ways like CBC, LFT, PCR, thyroid test and electrolyte test. She was treated by different medicine in different time like levooxacin, ribovarin, sofosbuvir etc. After 4 - 6 weeks she got negative HCV.
Keywords: HCV; CBC; LTF; Sofosbuvir; Ribovarin
Background
The worldwide hepatitis B and C epidemics are a severe public-health issue. In Pakistan, around 10 million people are affected with the hepatitis C virus (HCV). The majority of individuals acquire chronic hepatitis, with only a few occurrences of spontaneous hepatitis clearing. In Pakistan, however, little is known about multidrug-resistant virus strains. Hepatitis B and C are infectious liver disorders caused by the hepatitis B (HBV) and hepatitis C (HCV) viruses, accordingly. HBV is a partly double-stranded DNA virus with a diameter of 42 nanometers that belongs to the Hepadnaviridae family. There are eight primary HBV genotypes. HCV is a 55-nanometer encapsulated positive-strand RNA virus that belongs to the Flaviviridae family’s Hepacavirus genus; there are at least six primary HCV genotypes. Hepatitis B and C are two different types of hepatitis. They can be acute or chronic. The acute type of HBV or HCV infection is a short-term sickness that develops within the first six months of infection. The diseases can become chronic, but this does not always happen, and the likelihood of chronicity, particularly in the case of hepatitis B, is dependent on a person’s age at the time of infection. Chronic active hepatitis, cirrhosis, and primary liver cancer are among the chronic effects of hepatitis B virus (HBV), hepatitis D virus (HDV), and hepatitis C virus (HCV) infection. There are five identified pathogens, A-E, as well as non-A-E hepatitis viruses, which encompass a variety of unrelated and frequently odd human infections [1-5].
Case Presentation
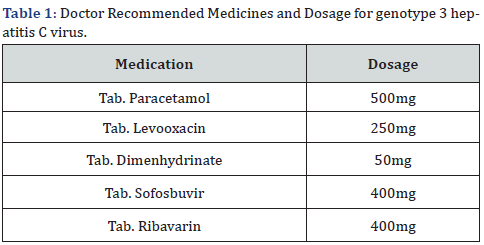
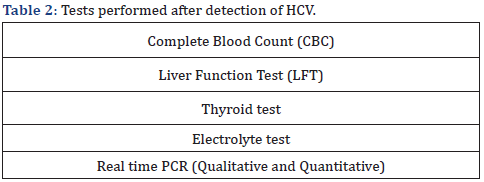
This is a case report of 54 years old housewife from Bhalwal city. Her qualification is Bachelor of Arts and now she is employed at non-government institute. She has no disability and does not use of any drug of abuse. About 14 years before, when she was 40 years old and at her job, she felt a fever, which was accompanied by body ache and sporadic vomiting. She went to visit her family’s doctor. Following an inspection, the doctor suggested paracetamol 500mg three times a day, levooxacin 250mg twice a day, and dimenhydrinate 50mg if needed for seven days. After a week, the patient returned to the doctor and inquired about her health. She had a fever that hadn’t gone away and pale skin. The doctor recommended that the patient get a complete blood count (CBC) test, a liver function test (LFT), a typhoid test, and an electrolyte test. Following the lab tests, which revealed that the values of the liver functional test had risen to a dangerous level? [6-10].
Test Reports and Medicine
After a detection of genotype 3 hepatitis C virus, the doctor recommended sofosbuvir 400mg once a day, ribavarin 400mg twice a day, and paracetamol (500mg) when the patient feels feverish, or their body temperature rises. Patient was completely informed by its disorder. The HCV was detected by real time PCR qualitative and quantitative. Various types of adverse medication reactions have been recorded during treatment, including nausea, lack of appetite, muscle pain, diarrhoea, fever, stomach pain, sickness, vomit, and anxiety. After vaccination and complete course of treatment she was tested again by real time PCR. This time there is HCV was detected because she was vaccinated [11- 15] (Table 1). And following tests were performed (Table 2).
Discussion
About 1 in 12 people (480-520 million people) are chronically infected with HBV or HCV over the world (Lavanchy, 2008; WHO, 2009). Hepatitis caused by chronic viral infection is a silent killer. Many chronically infected people are unaware that they are infected until symptoms of severe liver disease occur, due to a lack of infection testing. Globally, an estimated 130–170 million people have chronic HCV infection, with 350,000 people dying each year from HCV-related liver disease (Perz et al., 2006). Each year, roughly 2.3-4.7 million new HCV infections are caused solely by nosocomial transmission (Lavanchy, 2009).
Hepatitis C is a serious liver disease that can be lethal and cause death. Many people do not realise they have hepatitis C until they have significant liver damage. The hepatitis C virus is the cause of hepatitis C. It is transferred by coming into touch with the blood of an infected individual. The proportion of needles and other injecting equipment used to inject illegal substances. It can also be spread through unsterilized surgical instruments, contaminated water, and contaminated blades, as well as through intercourse. The reported results of the patient’s condition are that the patient was cured by regular treatment, medication, and regular checkups. During treatment, the patient experienced a variety of side symptoms, although these were all resolved over time. It is concluded that hepatitis C used to be more lethal than it is now, because rational medicine is now available that is more useful in the fight against the disease. To avoid contracting the sickness, we should drink only pure water, use untainted syringes, avoid smoking, and eat only fresh foods. There should be general awareness about the disease among patients, thus physicians can play an important role in disease prevention by teaching patients about the risks of Hepatitis C [16-19].
Patient Declaration Patients
The authors claim to have gotten all of the necessary parental consent paperwork. By filling out the form, the patient(s) has/ have granted his/her/their permission for his/her/their pictures and other clinical facts to be published in the journal. The parents are aware that their names and initials will not be published, and that every effort will be taken to keep their identities anonymous, although perfect anonymity cannot be guaranteed.
References
- AASLD/IDSA HCV Guidance Panel (2015) Hepatitis C guidance: AASLD‐IDSA recommendations for testing, managing, and treating adults infected with hepatitis C virus. Hepatology 62(3): 932-954.
- Altekruse SF, McGlynn KA, Reichman ME (2009) Hepatocellular carcinoma inci-2009. Hepatocellular carcinoma incidence, mortality, and survival trends in the United States from 1975 to 2005. J Clin Oncol 27(9): 1485-1491.
- American Cancer Society (2009) Cancer facts & figures, 2009. Atlanta, Georgia.
- Amon JJ, Garfein RS, Ahdieh-Grant L, Armstrong GL, Ouellet LJ, et al. (2008) Prevalence of hepatitis C virus infection among injection drug users in the United States, 1994-2004. Clin Infect Dis 46(12): 1852-1858.
- Armstrong GL, Wasley A, Simard EP, McQuillan GM, Kuhnert WL, et al. (2006) The prevalence of hepatitis C virus infection in the United States, 1999 through 2002. Ann Intern Med1 44(10): 705-714.
- Beasley R, Hwang L (1991) Overview of the epidemiology of heptocellular carcinoma. In Viral hepatitis and liver disease. Proceedings of the 1990 international symposium on viral hepatitis and liver disease. In: Hollinger FB, Lemon S, Margolis HS, Baltimore (Eds.), Williams & Wilkins. Pp. 532-525.
- Bonacini M, Groshen MD, Yu MC, Govindarajan S, Lindsay KL, et al. (2001) Chronic hepatitis C in ethnic minority patients evaluated in Los Angeles county. Am J Gastroenterol 96(8): 2438-2441.
- Boutwell AE, Allen SA, Rich JD (2005) Opportunities to address the hepatitis C epidemic in the correctional setting. Clin Infect Dis 40(suppl 5): S367-S372.
- But DY, Lai CL, Yuen MF (2008) Natural history of hepatitis-related hepatocellular carcinoma. World J Gastroenterol 14(11): 1652-1656.
- Buti M, Gordon SC, Zuckerman E, Lawitz E, Calleja JL, et al. (2016) Grazoprevir, elbasvir, and ribavirin for chronic hepatitis C virus genotype 1 infection after failure of pegylated interferon and ribavirin with an earlier‐generation protease inhibitor: final 24‐week results from C‐SALVAGE. Clin Infect Dis 62(1): 32-36.
- CDC (Centers for Disease Control and Prevention) (1998) Recommendations for prevention and control of hepatitis C virus (HCV) infection and HCV-related chronic disease. Morbidity and Morality Weekly: Recommendations and Reports 47(RR-19): 1-39.
- European Association for Study of Liver (2015) EASL recommendations on treatment of hepatitis C 2015. J Hepatol 63(1): 199-236.
- Friborg J, Zhou N, Han Z, Yang X, Falk P, et al. (2015) In vitro assessment of re‐treatment options for patients with hepatitis C virus genotype 1b infection resistant to daclatasvir plus asunaprevir. Infect Dis Ther 4(1): 137-144.
- Karino Y, Toyota J, Ikeda K, Suzuki F, Chayama K, et al. (2013) Characterization of virologic escape in hepatitis C virus genotype 1b patients treated with the direct‐acting antivirals daclatasvir and asunaprevir. J Hepatol 58(4): 646-654.
- Kumada H, Suzuki F, Suzuki Y, Toyota J, Karino Y, et al. (2016) Randomized comparison of daclatasvir + asunaprevir versus telaprevir + peginterferon/ribavirin in japanese HCV patients. J Gastroenterol Hepatol 31(1): 14-22.
- Manns M, Pol S, Jacobson IM, Marcellin P, Gordon SC, et al. (2014) All‐oral dual therapy with daclatasvir and asunaprevir in patients with HCV genotype 1b infection: phase 3 HALLMARK‐DUAL study results. J Hepatol 60: S524-S525.
- Marks K, Weinberg E, Kumar S, Down C, de Jong Y, et al. (2015) Sofosbuvir, simeprevir, ± ribavirin in HCV G1 protease inhibitor‐experienced patients.
- McPhee F, Friborg J, Levine S, Chen C, Falk P, et al. (2012) Resistance analysis of the hepatitis C virus NS3 protease inhibitor asunaprevir. Antimicrob Agents Chemother 56(7): 3670-3681.
- McPhee F, Suzuki Y, Toyota J, Karino Y, Chayama K, et al. (2015) High sustained virologic response to daclatasvir plus asunaprevir in elderly and cirrhotic patients with hepatitis C virus genotype 1b without baseline NS5A polymorphisms. Adv Ther 32(7): 637-649.






























