Convalescent Plasma Transfusion: A Life Saving Therapy for Covid-19 Patients
Shmmon Ahmad1>*, Shivani Thakur2>, Abdul Hafeez1>, Satish Kumar Sharma1>, Samera Ali Siddique1> and Isha Talwar1>
1 Glocal School of Pharmacy, Glocal University Saharanpur, India
2School of Pharmacy, Maharaja Agrasen University Solan, India
Submission: July 17, 2020; Published: July 24, 2020
*Corresponding author: Shmmon Ahmad, Glocal School of Pharmacy, Glocal University Saharanpur, India
How to cite this article: Shmmon A, Shivani T, Abdul H, Satish K S, Samera A S, et al. Convalescent Plasma Transfusion: A Life Saving Therapy for Covid-19 Patients. Palliat Med Care Int J. 2020; 3(4): 555619. DOI 10.19080/PMCIJ.2020.03.555619
Abstract
At present, there is no specific antiviral agent that have been found to be effective in the treatment of COVID-19. The disease in past few months was controlled by various measures, such as travel restriction and patient isolation but for a long duration these are not a permanent solution. As patients fight the virus, their bodies develop antibodies that will stay in their blood waiting for the same virus to attack again and fight against it. When those antibodies are administered to another infected person, they attack the outside virus and block the virus from penetrating human cells. Antibodies are extracted from blood plasma, and they can be used to possibly prevent or treat disease in another infected person. Convalescent plasma therapy involves transfusing certain components from the blood of recovered patients into people who are seriously infected. The therapy might prove to be a boon to patients suffering from COVID 19.
Keywords:COVID-19; Clinical Trials; Transfusion; Convalescent Plasma; Scientific Approach
Introduction
Coronavirus is a large family of viruses that may cause disease in humans and animals [1,2]. The pandemic caused by COVID-19, affected more than thirteen million people around the world. No specific treatment is completely effective against the novel coronavirus (SARS-CoV-2) and no vaccine is available yet to prevent the infection [3,4].
When no proven specific vaccine or drug is available for emerging infections Historically [5], Convalescent plasma immunotherapy has been used as a possible therapeutic option [3]. Some clinical researchers and doctors have started using plasma from the recovered patient of COVID-19 to treat others who have developed the disease [6]. The convalescent plasma administration, collected from the previously infected patient to passively-transfer antibodies. Convalescent plasma has previously been used to protect or treat humans, against viral illnesses such as polio, measles, influenza, rabies, hepatitis B, Ebola, and other pathogens [7].
In 2014, While there was no proven treatment available for Ebola (EVD), the WHO recommended the use of Ebola convalescent plasma (CP) transfusion as an empirical treatment with promising results in a small group of EVD cases [8]. To evaluate the efficacy of convalescent plasma therapy, during the SARS outbreak in 2002, various studies good outcome was defined. It was concluded that people treated with convalescent plasma therapy resulted in earlier recoveries and had a higher chance of being discharged from hospital compared to regular drug-treated patients [9].
An exploratory meta-analysis of 32 studies assessed the clinical benefit of the administration of convalescent plasma, hyperimmune immunoglobulin, or serum in the treatment of severe acute respiratory infections. It is found that the mortality rate was significantly reduced with no serious adverse effects [8].
Researchers or clinical scientists are testing the use of donated blood as a treatment for people with severe COVID-19 [10]. People who have recovered from COVID-19 have antibodies, developed by the individual against the disease. Scientists call this convalescent plasma. Researchers hope that convalescent plasma can be given to people with severe coronavirus infectious disease to boost their ability to fight the virus. Convalescent plasma could provide short-term immunity against the COVID-19 by providing antibodies (secreted by immune cell lymphocytes) that neutralize the novel coronavirus and prevent further damage. [10].
Working of CPT
In Convalescent Plasma Therapy, the blood is withdrawn from the patient who has recently recovered from COVID-19 [11]. Then the serum is separated to analyse the presence of virus neutralising antibodies. These antibodies are extracted and then administered to a a patient infected with Coronavirus. As per the Houston Methodologist, the donation process of Plasma takes around 1 hour just like blood donation. The donors of plasma are hooked up with a small device that removes the plasma and returns erythrocytes back to their bodies. The plasma rich in antibodies is extracted from a patient who has recently recovered from Coronavirus disease and infused into an infected patient. When the body gets infected with external pathogens, the body immediately starts releasing antibodies [12].
Researchers are expecting that convalescent plasma can be given to people suffering from severe COVID-19 to boost their ability to fight the virus. It also might help keep people who are moderately ill from becoming more ill and experiencing COVID-19 complications (Figure 1).
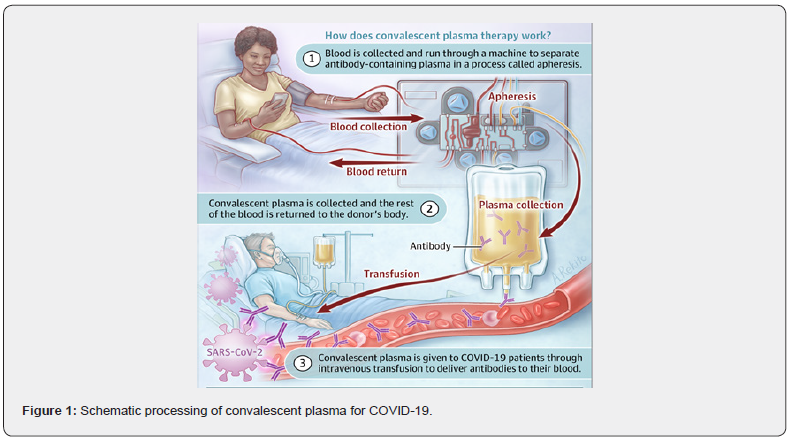
Setting up Clinical Trials
The U.S. Food and Drug Administration has outlined the requirements that individuals must meet to donate blood for this research. Before donated blood can be used, it must be tested for safety. It then goes through a process to separate blood cells so that all that’s left is plasma with antibodies [10,13]. The immediate goal of clinical trials is to determine whether the convalescent plasma approach can improve the chance of recovery for people with the most severe disease and secondary goals are to test whether convalescent plasma can help keep people who are moderately sick from getting sicker [14,10].
Passive immunization therapy has been successfully used to treat infectious diseases back to the 1890s. An individual who is sick with infectious diseases and recovers has blood drawn and screened for particular microorganism neutralizing antibodies [15]. Following identification of those with high titers of neutralizing antibody, convalescent plasma containing these neutralizing antibodies can be administered in individuals with specified clinical disease to reduce symptoms and mortality. Hence, Convalescent Plasma Transfusion (CPT) has been the subject of increasing attention, especially in the wake of largescale epidemics. It has recently been suggested by the Food and Drug Administration (FDA) that administration and study of investigational CPT may provide a clinical effect for the treatment of COVID-19 during the public health emergency [16].
The current treatment of COVID-19 caused by novel coronavirus SARS-CoV-2 has been limited to general supportive care, with provisions of critical care as no approved therapies or vaccines are available [16]. One of the hopeful treatments that has emerged is convalescent plasma (CP), or immune plasma. CP, which is plasma that is collected from an infected individual, such as by COVID-19 (i.e., human anti–SARS-CoV-2 plasma), is then transfused into infected patients as postexposure prophylaxis. 3 Unlike immunoglobulin (IgG)-derived antibodies such as plasma-derived monoclonal antibodies, CP is a passive antibody therapy that showed some success as a neutralizing antibody against other coronavirus epidemics, SARS-1 and the Middle East respiratory syndrome (MERS), in the first two decades of the 2000s. CP-derived antibodies can neutralize a virus by preventing replication (e.g., by complement activation or phagocytosis) or by binding without interfering with replication [17].
Convalescent plasma transfusion (CPT) therapy in COVID-19 patients appears safe, clinically effective, and reduces mortality. Well-designed large multicentre clinical trial studies should be conducted urgently to establish the efficacy of CPT to COVID-19 patients [16]. There was sufficient data that prove COVID-19 Patients can be treated With Convalescent Plasma [18]. Since late March, some U.S. patients have received a promising experimental treatment for the new coronavirus. All were critically ill and in hospitals. They didn’t get a new drug. Instead, doctors infused their blood with antibodies. These immune proteins were taken from the blood of people who had already recovered from COVID-19 [19].
A group of US academic researchers has sparked a nationwide effort to encourage people who have recovered from COVID-19 to donate plasma, which will be used to treat patients across the country. The convalescent plasma program was instigated by physicians and investigators from 40 institutions, including the Mayo Clinic, Johns Hopkins University, Washington University, Einstein Medical Center and the Icahn School of Medicine at Mount Sinai, among many others working closely with the US Food and Drug Administration (FDA) and industry partners. The UK’s National Health Service launched in April a program across its 23 main blood centers to collect convalescent plasma for testing in planned clinical trials. At the same time, a consortium of industry players Takeda, CSL Behring, Biotest, Bio Products Laboratory, LFB, Octapharma, and Microsoft have come together to develop an unbranded polyclonal antibody product: hyperimmune globulin (H-Ig) purified from the pooled plasma of donors who have recovered from COVID-19 [20].
The treatment is known as convalescent plasma. Plasma is a yellowish liquid in blood. It carries antibodies. The body’s immune system makes those antibodies special proteins in response to a virus or vaccine. Those proteins can bind to a virus and help to remove the infectious particles. It takes time for the body to make antibodies, around a week or two. But once they are available, the immune system can quickly respond to a particular virus the next time it confronts it. Antibodies are part of what is known as the body’s “active immunity.” For some viruses and vaccines, active immunity can last decades even the rest of your life [19].
In opposite, the new plasma therapy uses other person antibodies to fight infection. The immunity it offers may last for only weeks to a few months. But physician and scientist Jeffrey Henderson (Washington University School of Medicine) say it’s just possible that these proteins can prevent infection or treat infection in another patient [19,21].
Convalescent plasma is an experimental therapy for COVID-19. The U.S. Food and Drug Administration, or FDA, recently authorized it for emergency use. The United States now leading the world in confirmed cases of COVID-19 and no proven treatments known researchers are racing to set up clinical trials to test Convalescent plasma [19]. If the therapy works, the FDA might approve its wider use to treat infections with SARS-CoV-2, the virus that causes COVID-19. A vaccine is still needed to protect people from the infection. But such a vaccine for SARS-CoV-2 is at least a year away or more. Until then, scientists are searching for ways to treat the infection. John Roback. pathologist (Emory University School of Medicine in Atlanta) say, donated antibodies are one such treatment, he performs research on bloodtransfusion medicine [19].
Risks and Benefits
COVID-19 convalescent plasma can be used for either treatment of infection or treatment of disease. And plasma therapy is old techniques because it is used in many other conditions. Blood donors must have had documented SARS-CoV-2 infection, the right time to using plasma therapy is within the first 14 days of the symptoms. In a mode of action, the benefit of convalescent plasma administration is that it can prevent infection and subsequent disease in those who are at high risk for diseases, such as underlying chronic medical conditions, health care providers, and those with exposure to confirmed cases of COVID-19 [7]. Blood and plasma have been used many other conditions and may be caused minor risk which is involved in transfusion of blood. Being overweight is a major risk factor for people infected with coronavirus so we can say the obesity is a major COVID-19 risk factor. This risk is caused by age, pre-existing illness, or obesity. The virus can hit young people’s particularly obese young peoples. Another risk may be an allergic reaction. [22]. A passive antibody used to prevent COVID-19 infection. Such as patients exposed to hepatitis B are treated with hepatitis B immune globulin (HBIG) and patients exposed to rabies virus treated with human rabies immune globulin (HRIG). Besides, the passive antibody is used for the prevention of severe respiratory syncytial virus (RSV) disease in high-risk infants [23].
The utilization of plasma has increased over the last two decades, and Plasma transfusion is not without risk, Plasma transfusion has a greater risk than other blood components. Risks of passive use of convalescent plasma can be divided into two categories, known & theoretical. The risks associated with plasma transfusion is pathogenesis, diagnosis, and management. The common risk associated with fresh frozen plasma include are: [24]
a) Allergic or anaphylactic reactions.
b) Transfusion-related acute lung injury
c) Transfusion-associated circulatory overload
Other less common risks include
a) RBC alloimmunization,
b) Haemolytic transfusion reactions.
c) Transmission of infections,
d) Febrile non-haemolytic transfusion reactions,
With modern blood banking techniques and the risks of inadvertently transferring known infectious agents or transfusion reactions are low because of screening for blood-borne pathogens and match the blood type of donors and recipients [25]. Convalescent Plasma therapy provides some benefits over COVID-19. In a plasma, therapy immunity can be transferred from a healthy person to an infected person with the help of blood plasma. The idea behind this therapy is that antibodies produced by the patients who have had survived the virus will boost the immune system. And it also provides a medium for red blood cells and platelets to circulates through the body [26].
Deployment and Proposed Use
To use convalescent plasma administration for COVID-19 the following conditions must be considered;
1) Availability of assays, including virological assays to measure viral neutralization and serological assays, to detect SARS-CoV-2 in plasma,
2) Virology laboratory support to perform these assays
3) Availability of a population of donors who have recovered from the disease and can donate convalescent plasma;
4) Blood banking facilities to process the plasma donations;
5) Treatment and therapeutic protocols, which should ideally include randomized clinical trials to assess the efficacy of any intervention and measure immune responses;
6) Regulatory compliance, including institutional review board approval, which may vary depending on location. Ideally, the use of convalescent plasma would involve multiple points follow randomized control protocols, and have a single point as a governing body. Each of these conditions should be available in developed areas affected by COVID-19. At least one pharmaceutical company, Takeda, is gearing up to generate antibody preparations against SARS2-CoV-2 from COVID-19 convalescent sera [23,27].
We included 5 studies reporting CPT to COVID-19 patients. The main findings from available data are as follows:
1. Convalescent plasma may reduce mortality in critically ill patients
2. Increase in neutralizing antibody and disappearance of SARS-CoV-2 RNA was observed in almost all the patients after CPT therapy
3. Beneficial effect on clinical symptoms after administration of convalescent plasma [16]. General methods, which include existing broad-spectrum antiviral drugs. Use of these individual and combined form to treat with coronaviruses, including neuraminidase inhibitors such as oseltamivir, paramivir, zanamivir, etc. Commonly used antiviral drugs, other drugs possibly effective for 2019-nCoV include remdesivir, lopinavir/ ritonavir lopinavir/ritonavir, Neuraminidase inhibitors, peptide (EK1), RNA synthesis inhibitors (such as TDF, 3TC), anti- inflammatory drugs, Chinese traditional medicine, such as Lianhuaqingwen Capsule, and Shu Feng Jie Du Capsules could be the drug treatment options for 2019-nCoV. However, the safety and efficacy of these drugs for novel Coronavirus still under clinical experiments [28-30].
A Scientific Approach to Treat COVID-19
Liamas produce antibodies that block SARS-COV-2 from infecting cells this could help in developing treatment against COVID-19. Liamas produce a special type of antibodies when exposed to viruses. These antibodies are very small as compare to human antibodies and are known as single-domain antibodies or nanobodies. Researchers isolated two nanobodies from ilamas and engineered a new type antibody. That binds to the spike protein on the coronavirus that causes COVID-19 [31]. German biotech is testing its coronavirus vaccine candidate on human volunteers. BioNTech, a German pharmaceutical company launched its vaccine on human trials. The company is collaborating with Pfizer, a US-based pharmaceutical, on the vaccine called BNT162. After it had received regulatory approval from the US authorities 12 volunteers are currently participating in a clinical trial in Germany since April 2020 [32]. Clinical trials to evaluate the safety and efficacy of human Anti- SARS-CoV-2 (Severe Acute Respiratory Syndrome Coronavirus 2). These trials have been performed to evaluate the efficacy of convalescent plasma. At least five clinical trials have been proposed to evaluate human anti-SARS-CoV-2 plasma for the prevention and treatment of COVID-19 [33].
Improvement of Clinical Symptoms
The majority of patients who recover from COVID-19 illness develop circulating neutralizing antibodies to various SARS-CoV-2 proteins in 15-20 days. Transfer of patient’s plasma should be preventing further replication, neutralize the virus, and halting ongoing tissue damage. This theory would be predicted to work excellent in patients with less severe infection, or prophylactically in highly susceptible individuals such as exposed health care workers or family caregivers of SARS-CoV-2 patients. The COVID-19 may with mild, moderate, or severe illness. Among the severe clinical manifestations, there are ARDS, sepsis, septic shock, and severe pneumonia. The clinical course of the disease seems to predict a favourable trend in the majority of patients. For suspected cases, rapid antigen detection, and other investigations should be adopted for evaluating common respiratory pathogens and non-infectious condition [34]. COVID-19 patients who had established humoral immunity against the virus, contains a large quantity of neutralizing antibodies capable of neutralizing SARSCoV- 2 and eradicating the pathogen from blood circulation and pulmonary tissues. All investigated patients achieved plasma SARS-CoV-2 RNA negativity after CP transfusion, accompanied by an increase of oxygen saturation and lymphocyte counts, and the improvement of liver function and CRP [35]. The clinical symptoms were significantly overcome along with an increase in oxyhaemoglobin saturation. The Convalescent plasma therapy was well good and effective could potentially improve the clinical outcomes through severe COVID-19.
Conclusion
The convalescent plasma theory is an interim approach during the development of vaccines, hyperimmune globulin, and drug therapies. Human plasma from recovered novel coronavirus patients is to be a safe and potentially effective therapy for COVID-19 treatment. Hypothesize study suggests that the use of convalescent plasma will improve the clinical outcomes in patients with moderate Coronavirus infection. However, it is critically important to perform well-controlled clinical trials to confirm efficacy, Current estimates suggest a single donor collection could be used to treat 2-3 patients. Clinical researchers or Pharmacists can separate plasma, from the blood components that contain such antibodies, and administer it to people whose bodies are currently fighting an infectious disease novel coronavirus. This theory helps their immune systems reject the pathogen virus more efficiently.
To launch new Convalescent plasma therapy for the treatment of COVID-19 large pharmaceutical companies are racing to develop purification procedures to produce concentrated neutralizing antibodies from convalescent sera and immunizing in large animals such as Cow, Horse, and sheep with SARS-CoV-2 proteins to create antiserum. As well as biotech companies are trialling to develop cocktails of monoclonal antibodies active against the SARS-CoV-2 virus, but for finalized it take more months.
References
- Hafee A, Ahmad S, Siddqui S, Ahmad M, Mishra S, et al. (2020) A Review of COVID-19 (Coronavirus Disease-2019) Diagnosis, Treatments and Prevention. EJMO 4(2): 116-125.
- Scripps Research Institute (2020) COVID-19 coronavirus epidemic has a natural origin. ScienceDaily.
- Langhi DM, Santis GC, Bordin JO (2020) COVID-19 convalescent plasma transfusion. Hematol Transfus Cell Ther 42(2): 113-115.
- https://www.worldometers.info/coronavirus/?utm_campaign=homeAdTOA
- Wang L, Wang Y, Ye D, Liu Q (2020) Review of the 2019 novel coronavirus (SARS-CoV-2) based on current evidence. Int J Antimicrob Agents 55(6):105948.
- Malani AN, Sherbeck JP, Malani PN (2020) Convalescent Plasma and COVID-19. JAMA.
- Shaz B, Dunbar C, Hillyer C (2020) COVID-19 and Convalescent Plasma, American Society of Hematology.
- WHO (2014) Use of convalescent whole blood or plasma collected from patients recovered from Ebola virus disease for transfusion, as an empirical treatment during outbreaks.
- Cheng Y, Wong R, Soo YO, Wong WS, Lee CK, et al. (2005) Use of convalescent plasma therapy in SARS patients in Hong Kong. Eur J Clin Microbiol Infect Dis 24(1): 44‐46.
- Joyner MJ (2020) Convalescent plasma: Possible treatment for COVID-19? Mayoclinic.
- Pawar AY, Hiray AP, Sonawane DD, Bhambar RS, Derle DV, et al. (2020) Convalescent plasma: A possible treatment protocol for COVID- 19 patients suffering from diabetes or underlying liver diseases. Diabetes Metab Syndr 14(4): 665-669.
- https://www.jagranjosh.com/general-knowledge/what-is-convalescent-plasma-therapy-1587018676-1
- https://www.mayoclinic.org/tests-procedures/convalescent-plasma-therapy/about/pac-20486440
- https://www.fda.gov/vaccines-blood-biologics/investigational-new-drug-ind-or-device-exemption-ide-process-cber/recommendations-investigational-covid-19-convalescent-plasma
- Keller MA, Stiehm ER (2000) Passive immunity in prevention and treatment of infectious diseases. Clin Microbiol Rev 13: 602-614.
- Rajendran K, Narayanasamy K, Rangarajan J, Rathinam J, Natarajan M, et al. (2020) Convalescent plasma transfusion for the treatment of COVID-19: Systematic review. J Med Virol.
- Teixeira da Silva JA (2020) Convalescent plasma: A possible treatment of COVID-19 in India. Med J Armed Forces India 76(2): 236-237.
- Shen C, Wang Z, Zhao F, YangY, Jinxiu Li, et al. (2020) Treatment of 5 Critically Ill Patients With COVID-19 With Convalescent Plasma. JAMA 323(16): 1582-1589.
- Cunningham A (2020) Antibodies from former COVID-19 patients could become a medicine, Science News for Students.
- Sheridan C (2020) Convalescent serum lines up as first-choice treatment for coronavirus. Nature biotechnology.
- Leslie M (2020) Science, This cow’s antibodies could be the newest weapon against COVID-19.
- https://www.aljazeera.com/indepth/features/doctor-note-obesity-risk-factor-coronavirus-200406065716658
- Casadevall A, Pirofski LA (2020) The convalescent sera option for containing COVID-19. Journal of Clinical Investigation 130(1): 1545.
- Pandey S, Vyas GN (2012) Adverse effects of plasma transfusion. Transfusion 52(Suppl 1): 65S-79S.
- Gajic O, Rana R, Winters JL, Yilmaz M, Mendez JL, et al. (2007) Transfusion-related acute lung injury in the critically ill: prospective nested case-control study. Am J Respir Crit Care Med 176(9): 886‐891.
- Mora-Rillo M, ArsuagaM, Ramírez-Olivencia G, Calle FDL, Borobia AM, et al. (2015) Acute respiratory distress syndrome after convalescent plasma use: treatment of a patient with Ebola virus disease contracted in Madrid, Spain. Lancet Respir Med 3(7): 554-562.
- Mulangu S, Dodd LE, Davey RT Jr, Mbaya OT, Proschan M, et al. (2019) A Randomized, Controlled Trial of Ebola Virus Disease Therapeutics. N Engl J Med 381(24): 2293‐2303.
- Natsuko I, Dorigatti I, Cori A, Christl D, Steven R, et al. (2019) Estimating the potential total number of novel Coronavirus cases in Wuhan City China. Imperial College London, UK.
- Lu H (2020) Drug treatment options for the 2019-new coronavirus (2019-nCoV). Biosci Trends 14(1): 69‐71.
- Li H, Wang YM, Xu JY, Cao B (2020) Zhonghua Jie He He Hu Xi Za Zhi 43(3): 170‐172.
- Vaccine Research Center, National Institute of Allergy and Infectious Diseases, National Institutes of Health, Bethesda, Maryland, USA 20892.
- https://www.aljazeera.com/news/2020/04/german-company-begins-human-trials-coronavirus-vaccine 200429111230419
- Chun S, Chung CR, Ha YE, Han TH, Ki CS, et al. (2016) Possible Transfusion-Related Acute Lung Injury Following Convalescent Plasma Transfusion in a Patient with Middle East Respiratory Syndrome. Ann Lab Med 36(4): 393-395.
- Kogan A, Segel MJ, Ram E, Raanani E, Peled-Potashnik Y, et al. (2019) Acute Respiratory Distress Syndrome following Cardiac Surgery: Comparison of the American-European Consensus Conference Definition versus the Berlin Definition. Respiration 97(6): 518-524.
- Marano G, VaglioS, Pupella S, Facco G, Catalano L, et al (2016) Convalescent plasma: New evidence for an old therapeutic tool? Blood Transfus 14(2): 152-157.






























