The Influence of Morphine on Cognitive Functions in Cancer Patients
Magdalena Osowicka*
Medical University of Gdańsk, Poland
Submission: November 13, 2019;Published: November 21, 2019
*Corresponding author: Magdalena Osowicka, Medical University of Gdańsk, Poland
How to cite this article: Magdalena Osowicka, The Influence of Morphine on Cognitive Functions in Cancer Patients. Palliat Med Care Int J. 2019; 3(2): 555606. DOI:10.19080/PMCIJ.2019.03.555606
Perspective
Morphine is one of the most commonly used painkillers in cancer patients. Unfortunately, the myths about opioids that have survived over the years, often make effective analgesia difficult to achieve [1].
Aim: The aim of our work was to evaluate the effect of morphine on the cognitive function of cancer patients, taking into account the effect of the clinical status and dose of the drug.
Materials and Methods: The computerized CDR (cognitive drug research) system and paper tests were used for the study. Patients with hepatic and renal insufficiency and patients receiving corticosteroids are excluded from the study. We examined patients: in advanced stage of cancer with good mental performance determined by the Minimental State scale untreated with strong opioids, papaverine, codeine, corticosteroids in the last 4 weeks preceding the study. We’ve examined 68 patients, 40 patients have completed the study.
Results & conclusions: In the morphine-treated group, the speed of attention (selection response time), quality and speed of long-term memory (word memory) deteriorated, and the quality of memory (spatial memory and number memory) decreased. Statistical inference proved that the dominant factor affecting cognitive functions is the general state of the patients.
Keywords: Computerized CDR; Cognitive Drug; Opioids; Papaverine; Codeine; Corticosteroids; Deteriorated; Cancer; Morphine Myths; Socio-Demographic Factors; Drug; Urine Test; VAS Scale; CDR System; ECOG Scale; Sensitivity; Hepatic Failure; Blood; Morphine Consumption
Introduction
Pain is an inevitable sign of cancer. According to the WHO guidelines, opioids are the basic treatment of moderate to severe pain. Despite the lack of strong scientific evidence, opioids are attributed a series of negative effects on the psyche and cognitive abilities of man. This results in fear of opioid use, especially morphine. The most famous fact about morphine is that it is addictive [2]. The second well-known fact is that morphine causes respiratory depression [3]. Despite the lack of evidence showing that morphine has a negative effect on mental performance, in many countries its taking results in the inability to take legal action, to operate machines and to drive vehicles [4].
Morphine myths, which have survived for years, often hinder effective analgesia [1]. The decisions made by patients taking opioids are questioned and their validity is questioned in the context of legal action. Many patients at the end of their lives receive strong analgesia, while at the same time making important life decisions, such as writing down a will. The results of previous studies on the influence of morphine on cognitive functions are not clear. Studies are usually conducted on small groups of patients or on healthy volunteers, using a variety of not always complementary research tools. It is known that not only drugs but also a pain shape the cognitive ability of man. It is more appropriate to evaluate the effects of analgesics in sick people than in healthy people [5]. Although cancer patients are the largest group using opioid analgesics, there are only a single study of them. The reason is probably the research difficulties of this particular group of patients.
Aim
The main objective of this research is a detailed assessment of the effect of morphine on the cognitive function of patients, taking into account the influence of socio-demographic factors, general condition of patients and dose of the drug.
Materials and Methods
Patients who met the following criteria were enrolled to the study:
a) Over the age of 18,
b) In advanced stage of cancer,
c) With good mental performance determined by the Minimental State scale,
d) Untreated with strong opioids in the past 4 weeks
e) Untreated with papaverine, codeine, corticosteroids in the last 4 weeks preceding the study,
f) Expressing a written consent to the study.
Criteria to exclude the patient from the study were
g) Bad general condition not allow to conduct the test,
h) The presence of metastases to the central nervous system
i) Renal or liver failure
The Study was Conducted in Two Stages
Stage I
a) Qualification in accordance with the criteria
b) Blood and urine laboratory tests : sodium and potassium levels, creatinine level, bilirubin level, urea, calcium, liver enzymes (Aspat, Alat), glucose level, INR value, general urine test, urine test for opioids.
c) After a structured interview and a medical examination, a study was conducted with:
1. VAS scale for pain assessment,
2. Computerized CDR (Cognitive Drug Research) test,
3. ECOG scale for general condition examination.
Stage II
a) After inclusion morpine according to standards of analgesic treatment, we assess the efficacy of analgesia with VAS scale for consecutive days after inclusion. The patient was reexamined with paper tests and a computerized CDR test on the third day after achieving analgesic effect (VAS up to 2).
b) Cognitive Evaluation - CDR-P Cognitive Drug Research Program Automated cognitive assessment tests are a recognized way of assessing cognitive ability. In numerous studies of the effects of drugs are more sensitive compared to paper tests.
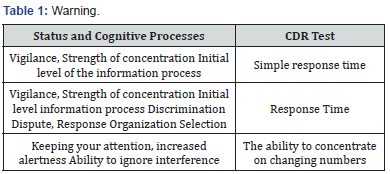
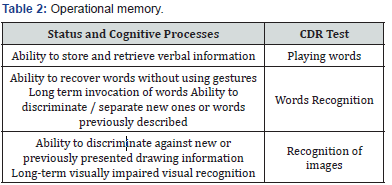
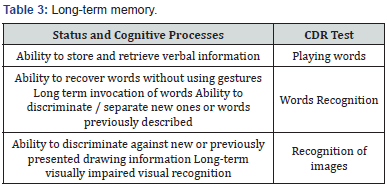
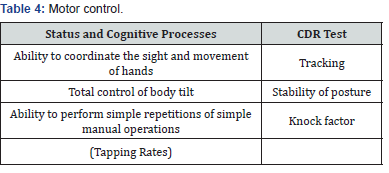
Tests Composing the CDR System
(Table 1-4) Some authors propose to analyze the CDR system in terms of the quality and speed of cognitive functions [6,7]. These 6 factors served to interpret the study in this paper [3,6].
a) Note - the quality of the test
b) Note - the speed of the test
c) Operational memory - test execution quality
d) Operational memory - test execution speed
e) Long-term memory - the quality of the test
f) Long-term memory - the speed of the test
The clinical status was assessed on the basis of medical documentation and ECOG scale. We used VAS scale to assess the pain.
Statistical Analysis
Collected data is saved in Excel spreadsheet. The obtained results were subjected to statistical analysis using statistical computer package STATISTICA PL v.8. Statistical description of the variables was made with calculating mean value (arithmetic mean) and standard deviation. Quality data was presented in the form of frequency and interest tables. The statistical inference for verifying the hypothesis of mean equality was performed on the basis of the Student’s t test, after checking the conditions of application of the parametric tests. The relationships between the variables studied were estimated R-Pearson’s product moment correlation coefficient. All hypotheses for the null hypothesis criteria were assumed to be p <0.05 [8].
Result
According to the criteria for inclusion 68 patients were included and tested (1st stage of the study). Finally, the study was completed by 40 patients, in home and stationary hospice.
Lots of patients included in the study
a) Number of patients included in the study 68
b) Patients who completed the study 40
c) Patients who died during the study 7
d) Patients who did not require morphine treatment 9
e) Patients whose general condition has deteriorated sufficiently to make it impossible to conduct the second stage of the study 11
f) Patients who resigned from stage II of study 1
Place of study: 21 patients (52.5%) covered by home hospice care and 19 patients (47.5%) in stationary hospices.
Sex: The study involved 19 women (47.5%) and 21 men (52.5%).
Age: The mean age of the subjects was 69.6 years (min.38,max.86) SD±10.3
In females an average of 71.8 years (min 38, max 86) SD±11.5 Whereas in men 67.7 years (53 min, max 81) SD±9
Patients were divided into 2 age groups (Table 5)
Socio-demographic factors and changes in cognitive functions
Patients treated with morphine did not show statistically significant effects of age or sex and education for cognitive performance.
Comparative study of cognitive function - before and after treatment
After achieving the therapeutic effect by morphine statistically significant changes in the quality and speed of all areas of cognitive function were noted (attention, memory and long-term memory). Morphine-treated patients performed worse than before treatment. Mainly memory-related processes have been impaired. Taking the drug was correlated with less accuracy and sensitivity in terms of spatial working memory (SPMOACCI and SPMSI tests), less accurate identification of numbers. There was a negative impact of the drug on the quality and the long-term memory speed, patients slower and less accurately recognize the presented words (DRECOAC and DRECSI test). In addition, attention-related processes (CRTs) were released (Figure 1&2).
Effect of morphine treatment time on cognitive function
There was a correlation between the size of cognitive improvement and the time needed to achieve the pain effect. Cognitive changes in patients with short-term pain treatment (less than 4 days) are much faster.
In patients treated with morphine over 10 days there’s no or just little cognitive improvement. This concerns two aspects of word recognition. Namely, in a group with a faster therapeutic effect, the rate of improvement in the rate of speech recognition is higher than in those treated for longer.
The same relationship also applies to the overall rate of word recognition. Simple response time improves in patients treated shortly but worsens in patients treated over 10 days.
Dose and cognitive function
There was no statistically significant relationship between dose and cognitive performance. Morphine treated patients received from 10 to 120 mg orally or in subcutaneous injections. To objectify the answer to the above-mentioned thesis, the criteria for qualifying patients for the study were rigorously observed. Exclusion of the patients with renal and hepatic failure has eliminated the factor that increases blood and urine drug levels.
The exclusion of papaverine and codeine reduced the errors in biochemical determination of concentration of drugs and their metabolites in the blood. The ban on steroid use was linked to the fact that these drugs could affect cognitive decline [9].
The effect of clinical status (according to ECOG) on cognitive function
The coefficients of correlation between ECOG values on the one hand and cognitive measurements on the other were calculated. The results indicate that the cognitive functioning of the patients studied remains correlated with their general state. This relationship is statistically significant for many variables, but the correlation values are not large. These statistically significant relationships occurred in all major areas of cognitive function, namely attention, memory and long-term memory. Patients with poorer general condition of attention quality have made more mistakes in detecting stimuli (changing numbers) and their reaction time has increased. Patients in the worse general state exhibited significantly longer response times to the presented stimuli - word recognition in the original and new stimulation variant, and longer response time to the stimulus in the form of digits. Long-term memory was also found in the presence of statistically significant relationships between the general state and the cognitive functions of the group. Patients in worse general condition were less likely to recognize words and images after the postponement of time and the performance of these tasks took place in extended time.
Discussion
In our study, we thoroughly evaluated the effect of morphine on patients’ cognitive function. Analysis showed that after morphine consumption, statistically significant deterioration has changed for all major areas of cognitive decline. Morphine has slow down processes and long-term memory and has compromised memory performance and long-term memory. What’s essential - we indicate that the cognitive functioning of patients remains correlated with their general state. We’ve used computer tests because of their superiority over paper tests.
Wesnes, in rivastigmine studies [10], and Walker in interleukin 2 studies [11,12] have demonstrated that the results of automated tests are more sensitive to drug-induced changes. The advantage of automated testing over traditional methods has also been demonstrated in previous observations by Corani [13] and Brook [14] on morphine.
For a thorough analysis of the problem, 68 patients were examined and the study was completed by 40 people. These data support the fact that patients with advanced cancer are a difficult research group, and their eligibility for clinical trials is difficult. Severe general condition of patients often makes it impossible to carry out time-consuming research and testing. For example, the first stage of study (VAS scale, socio-demographic history, CDR) performed a total of 45 to 90 minutes. Definitely better general condition of the patients facilitated the conduct of the study and increased the chances of completing the second stage of the study. This is probably due to the relatively short predicted survival time of the patients studied. In addition, the available literature provides, as a rule, the following:
a) Groups of small numbers. Probably a small number of the examined groups is caused by reasons mentioned above. Available studies typically involve over a dozen people [15-18].
Difficulty in completing the research group is also due to the fact that patients with advanced cancer are undergoing other medical conditions, often using pharmacotherapy that disqualifies them from the study.
b) The reason for doing the above research were no clear data of the effect of morphine on the cognitive functions of patients with advanced cancer.
Although numerous publications on morphine pharmacokinetics are available, these are only single observations for patients with advanced cancer. The available literature refers very ambiguously to this problem.
Long-term follow-up of Tassain’s results, indicates that morphine therapy does not impair cognitive ability and even improves it to a small extent [19]. However, taking long-term study time (up to 12 months), it can be assumed that the patients observed by Tassain were in significantly better clinical conditions than the patients tested by us. Against Tassain’s research, the results of Sjogren’s analysis [20], which evaluated non-cancer patients receiving chronic morphine for pain. 40 patients were treated with regular, regular doses of morphine. Cognitive tests used continuous reaction time (CRT) tests, which evaluated the FTT (finger tapping test), a psychomotor test; And PASAT (paced auditing serial addition task), which evaluates working memory. The results showed that patients performed tests worse than the control group of volunteers. This test confirms our results, especially since it uses similar neuropsychological tests. Wood has examined hospice patients with equivalent research tools as we [15]. The results of his analysis did not show memory impairment, but the quality of attention and the ability to process information were impaired. Similarly to our observation, long-term memory was disturbed.
Vainio evaluated the long-term effects of controlled-release morphine on cancer patients cognitive and psychomotor functions. He used neurological, psychological and special tests for drivers. The results were compared with those in the control group (cancer patients without pain). There were no statistically significant differences between the groups in terms of vigilance, attention and liquidity in performing psychomotor tasks. The results of the tests in “morphine” patients were slightly worse than in the control group [21].
In contrast to our observations, Morita’s findings show that confusion occurs at the start of opioid treatment and disappear after about one week of use [2,17,22]. Wood and Morita are the only researchers of opioid treatment in the late stages of life [15,17]. Their value raises the fact that they evaluate by specific psychological tests variables such as short-term memory and longterm concentration. These tests showed a lower level of intellectual function than the mean, although there was no impairment in clinical judgment. Wood et al. [17] used psychological tests to examine hospice patients during morphine treatment. They did not find fresh memory disturbances in them, but attention was concentrated and the ability to process information. The longterm memory was also significantly impaired. In this study no relationship was observed between the route of administration (p.o. and s.c.), dose and concentration of morphine metabolites and cognitive impairment [18]. Similar memory disturbances have been reported by Cull in his study of patients with remitting lymphoma [23]. It was found that when using morphine at doses prescribed by the patient as being sufficient to control pain, cognitive impairments caused by somnolence were not observed. Cognitive improvement after pain relief is most likely to compensate for morphine drowsiness [24] and cognitive function is more impaired by pain than morphine.
Conclusion
Verifying the research on the effect of opioids on cognitive function and getting answers to the question of how they affect cognitive performance is difficult. In the morphine-treated group, the speed of attention (selection response time), quality and speed of long-term memory (word memory) deteriorated, and the quality of memory (spatial memory and number memory) decreased. Negative importance of morphine seems to be overestimated. statistical inference points that dominant factor affecting cognitive function is the general condition of the patient.
References
- Davis MP, Srivastava M (2003) Davis, manish srivastava demographics, assessment and management of pain in the elderly. Drugs Aging 20(1): 23-57.
- Attard A, Ranjith G, Taylor D (2008) Delirium and its treatment. CNS Drugs 22(8): 631-644.
- Porter J, Jick H (1980) Addiction, rare in patients treated with narcotics. N Engl Med 302(2): 123.
- Buss T, Modlinska A, Janiszewska J (2003) Morfina a funkcje poznawcze. Polska Medycyna Paliatywna 2(4): 235-240.
- De Walden, Gałuszko K (2007) Poznanie a ból Psychoonkologia 12(1-2): 1-5.
- Majkowicz M, Walden-Gałuszko DK, Zdun-Ryżewska A, Bartosińska A (2007) Badania wpływu leków na funkcje poznawcze w Modelu Cognitive Drug Research Psychoonkologia 12(1-2): 6-11.
- Nicholl CG, Lynch S, Kelly CA, White L, Simpson PM, et al. (1995) The cognitive drug research computerized assessment system in the evaluation of early demention is speed of the essence? International Journal of Geriatric Psychiatry 10(3): 199-206.
- StatSoft Inc (2007) STATISTICA (data analysis software system), version 8.0. (www.statsoft.com).
- Kostowski W (2001) Farmakologia, podstawy farmakoterapii PZWL. Warszawa 177-188: 898-929
- Wesnes KA, Mckeith IG, Terrara R, Emre M, Del ST, et al. (2002) Effects of rivastigmine on cognitive function in dementia with levy bodies: A randomised placebo-controlled international study using the cognitive drug research computerised assessment system. Dement Geriatr Cogn Disord 13(3): 183-192.
- Walker LG, Wesnes KP, Heys SD, Lolley J, Wesnes K, et al. (1996) The cognitive effects of recombinant interleukin 2 (rIL-2) therapy: A controlled clinical trial using computerized assessments. Eur J Cancer 32A(13): 2275-2283.
- Walker LG, Walker MB, Heys SD, Lolley J, Wesnes K, et al. (1997) The psychological and psychiatric effects of rIL2 therapy. A controlled clinical trial. PsychoOncology 6(4): 290-301.
- Corani G, Edgar C, Marshall I, Wesnes K, Zaffalon M, et al. (2006) Classification of dementia types from cognitive profiles data. Lecture Notes in computer science 4213: 470-477.
- Brooke C, Enhange A, Transson B (1998) The effects of intravenous morphine on cognitive function in healthy volunteers. J Psychopharmacol 12(A): 45.
- Wood MM, Ashby MA, Somogyi AA, Fleming BG (1998) Neuropsychological and pharmacokinetic assessment of hospice inpatients receiving morphine. J Pain Symptom Manage 16(2): 112-120.
- Hanks GW, O Neill WM, Simpson P (1995) The cognitive and psychomotor effects of opioid analgesics II. A randomized controlled trial of single doses of morphine, lorazepam and placebo in healthy subjects. Eur J Clin Pharmacol 48(6): 455-460.
- Morita T, Tei Y, Tsunoda J, Inoue S, Chihara S, et al. (2002) Increased plasma morphine metabolites in terminally ill cancer patients with delirium; an intra-individual comparison. J Pain Symptom Manage 23(2): 107-113.
- Bruera E, Macmillan K, Hanson J (1987) Delirium and severe sedation in patients with terminal cancer. Can Treat Rep 71(7-8): 787-788.
- Tassain V, Attal N, Fletcher D, Brasseur L, Dégieux P, et al. (2004) Long term effects of oral sustained release morphine on neuropsychological performance in patients with chronic non-cancer pain. Pain 107(1-2): 191.
- Sjøgren P, Thomsen AB, Olsen AK (2000) Impaired neuropsychological performance in chronic nonmalignant pain patients receiving long-term oral opioid therapy. J Pain Symptom Manage 19(2): 100-108.
- Vainio A, Ollila J, Mantikainen E (1995) Driving ability in cancer patients receiving long term morphine analgesia. Lancet 346(8976): 667-670.
- Stoutz N, Tapper M, Fasinger R (1995) Reversible delirium in terminally ill patients. J Pain Symptom Manage10(3): 249-253.
- Cull A, Hay C, Love SB, Mackie M, Smets E, et al. (1996) What do cancer patients mean when they complain of concentration and memory problems? Br J Cancer 74(10): 1674-1679.
- Zacny JP (2006) Chronic pain and driving: Proceed with caution. Pain 122(1-2): 6-7.






























