Oligometastatic Squamous Cell Carcinoma of the Head and Neck Treated with Standard Dose Chemoradiation Therapy: A Valid Option for Selected Patients?
Toms Vengaloor Thomas*, Ashley Albert, Eldrin Bhanat, Sanjay Joseph, Eswar Kumar Mundra, Anu Abraham, Divyang Mehta, Sophy Mangana, Madhava Kanakamedala and Srinivasan Vijayakumar
Department of Radiation Oncology, University of Mississippi Medical Center, USA
Submission:July 27, 2019; Published: August 14, 2019
*Corresponding author: Toms Vengaloor Thomas, Department of Radiation Oncology, University of Mississippi Medical Center, USA
How to cite this article: Toms Vengaloor Thomas, Ashley Albert, Eldrin Bhanat, Sanjay Joseph, Eswar Kumar Mundra,et.al. Oligometastatic Squamous Cell Carcinoma of the Head and Neck Treated with Standard Dose Chemoradiation Therapy: A Valid Option for Selected Patients?. Palliat Med Care Int J. 2019; 2(5): 555599. 10.19080/PMCIJ.2019.02.555599
Abstract
Background: The purpose of this study was to explore the feasibility and tolerability of delivering standard dose chemoradiation therapy to patients with oligo-metastatic squamous cell carcinoma of the mucosal surfaces of the head and neck (Stage IV C) and to evaluate their survival outcomes. Methods: We retrospectively analyzed 10 well-performing patients with newly diagnosed oligo-metastatic carcinoma who underwent concurrent chemoradiation to a dose of 70 Gy. Overall survival curves were generated using the Kaplan-Meier method. Result: All patients completed their chemoradiation treatment; mean radiation duration was 53 days. Only 10% experienced Grade 3 or higher toxicity. The median overall survival was 12.5 months. Conclusion: Well-performing head and neck cancer patients with oligo-metastatic disease who were treated aggressively, tolerated their treatment with acceptable morbidity and had improved survival compared to historical controls
Keywords: Head; Neck; Fluro deoxy glucose; Positron emission; Intensity modulated; Radiation Therapy
Abbrevations: SCCHN: Squamous Cell Carcinoma Of The Head And Neck; IRB: Institutional Review Board; UMMC: University of Mississippi Medical Center; CT: Computed Tomography; FDG: Fluro Deoxy Glucose; PET: Positron Emission Tomography; IMRT: Intensity Modulated Radiation Therapy; SBRT: Stereotactic Body Radiation Therapy; RTOG: Radiation Therapy Oncology Group; NCCN: National Cancer Comprehensive Network; ECOG: Eastern Cooperative Oncology Group; KPS: Karnofsky Performance Score; TKI: Tyrosine Kinase Inhibitor; PMH: Princess Margaret Hospital; IHF2SQ: Irradiation Hypo Fractionnee 2 Séances Quotidiennes; NCDB: National Cancer Data Base; 3DCRT: 3-Dimensional Conformal Radiation Therapy; EXTREME: Erbitux in First-Line Treatment of Recurrent or Metastatic Head and Neck Cancer
Introduction
Squamous cell carcinoma of the head and neck mucosal sites (including the nasopharynx, oropharynx, larynx, and hypopharynx; SCCHN) is the 6th most common cancer diagnosed globally. Approximately, 3% of these patients present with metastatic disease at diagnosis with the lung being the most common site of metastasis [1]. The standard of care for patients in this setting is palliative multi-agent chemotherapy and their median survival is only about 10 months [2].
Generally, patients with locally advanced head and neck cancer also suffer from pain, dysphagia or odynophagia, hoarseness, otalgia, cough, and respiratory distress, significantly impairing their quality of life. As such, any local treatment which improve locoregional disease control is likely to improve their quality of life. Traditionally, because they have already stage IV disease, locoregional treatment usually involves the use of a palliative course of radiation therapy. However, there is currently no consensus for the appropriate dose and fractionation schedule for radiation therapy. it is hypothesized that selected patients with good performance status and oligometastatic disease may benefit from using standard fractionation chemoradiation therapy to improve their locoregional controlsurvival, and quality of life. To date, there are few data on standard chemoradiation therapy for patients with metastatic head and neck cancer [3-6].
We retrospectively analyzed our patients who had been treated with standard chemoradiation therapy to explore the tolerability of this treatment option in well-performing patients with oligometastatic disease to determine if this pathway would result in improved locoregional control and overall survival compared to historical studies.
Material and Methods
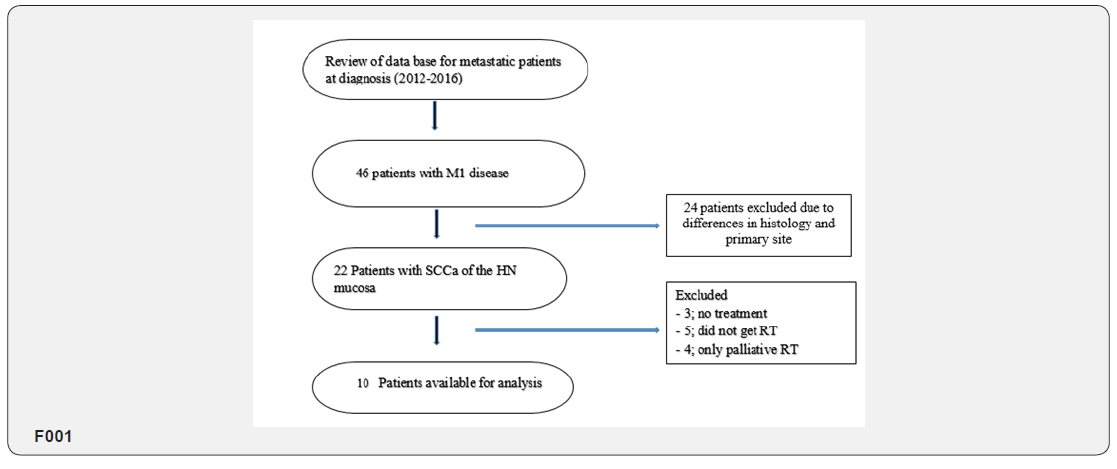
We obtained Institutional Review Board (IRB) before the retrospective collection of patient information. A review of our institutional head and neck database identified 46 patients who had been diagnosed with metastatic disease at the time of their presentation between 2012 and 2016. We included only patients with squamous cell carcinoma of the mucosal surfaces of head and neck, including oropharynx, oral cavity, larynx, and hypopharynx. We excluded 24 patients who had squamous cell carcinoma of the skin, melanoma, or carcinoma of the salivary or thyroid gland. We excluded 3 patients who did not receive any treatment as well as 5 patients who did not undergo any radiation treatment as part of their therapy. Four patients who received a lower dose of radiation for purposes of palliation were excluded as well. Ten patients were available for the final analysis.
All these patients had biopsy-proven head and neck squamous cell carcinoma. Their diagnostic evaluations included a thorough physical examination and nasopharynx golaryngoscopy, if indicated. Contrast enhanced computed tomography (CT) scans of the neck and chest, complete blood counts, and stand chemistry panels were also obtained for each patient. The performance status of each patient was also assessed based upon the Karnofsky Performance Scale. Sites of metastatic disease were either biopsy-proven or fluorodeoxyglucose-avid (FDG) on positron emission tomography (PET) scans. Patient data collected included identity of the primary disease site, duration of radiation therapy, use of chemotherapy, and treatment of the sites of metastatic disease (Flow diagram).
Patient characteristics
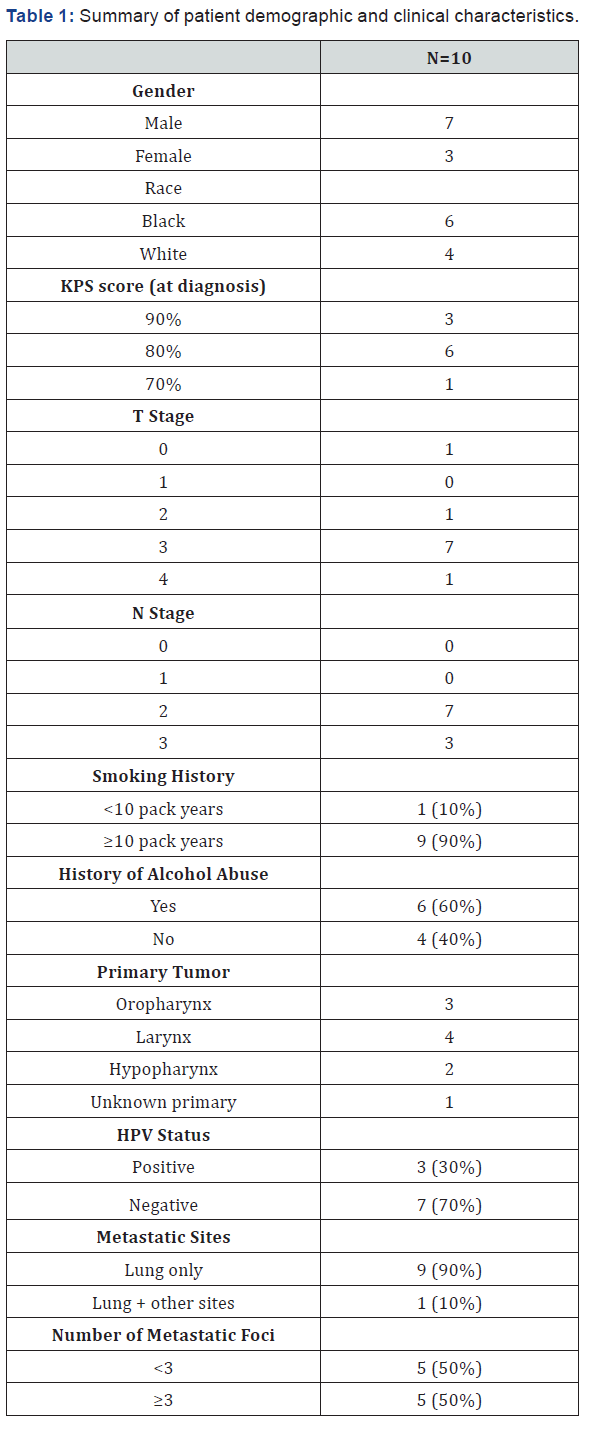
A total of 10 patients with metastatic head and neck cancer at diagnosis and who received standard chemoradiation therapy at UMMC between January 2012 and December 2016 were identified. All of them had metastatic disease at diagnosis (stage IVC), before the initiation of any therapy. Three patients had an oropharyngeal primary, four patients had a laryngeal primary, one patient had a hypo-pharyngeal primary, and one patient had a squamous cell carcinoma from an unknown primary. All but one patient also had a history of tobacco use and six patients had a history of alcohol use. Three of the patients had (human papilloma virus) HPV positive cancer. All but one patient had metastatic disease only in the lungs at the time of diagnosis. Among the patients, five patients had less than 3 metastatic foci. All the patients had a KPS score of at least 70 (range-70 to 90%). Two patients had feeding tubes inserted starting their chemoradiation treatment. Table 1 summarizes pertinent patient demographics and characteristics.
Treatment details
Three patients received 2 or 3 cycles of systemic chemotherapy using Paclitaxel, Cisplatin, and Cetuximab and were found to have stable or improved disease before starting concurrent chemoradiation treatment. Other patients who did not get upfront systemic chemotherapy their started concurrent chemoradiation therapy immediately. All treated patients underwent CT simulation using thermoplastic mask immobilization and a Philips big-bore CT scanner. Decisions regarding the radiation therapy including fractionation and treatment technique were decided upon by the treating radiation oncologist but were approved in pre-treatment peer review conferences before the patients started treatment [7,8].
All the ten patients were treated to a dose of 7,000 cGy to the primary disease and involved nodes and to 5,600 cGy to the subclinical nodal regions. Intensity Modulated Radiation Treatment (IMRT) was used for all patients. Most patients also received 40mg/m2 of Cisplatin weekly for 5-7 cycles. Two patients also received Stereotactic Body Radiotherapy (SBRT) to their sites of oligometastatic disease in the lungs after completion of their head and neck therapy.
Patient outcomes including median survival, overall survival, local and regional control were statistically evaluated. Survival curves were generated using the Kaplan Meier method. Survival was determined from the date of diagnosis to either the date of death or the date of the last follow-up for living patients. Survival data, including the date of death, were obtained from institutional and state cancer registries. All statistical analyses were performed using SPSS version 24 (IBM Inc, Armonk, NY, USA)
Result
Tolerability of the treatments
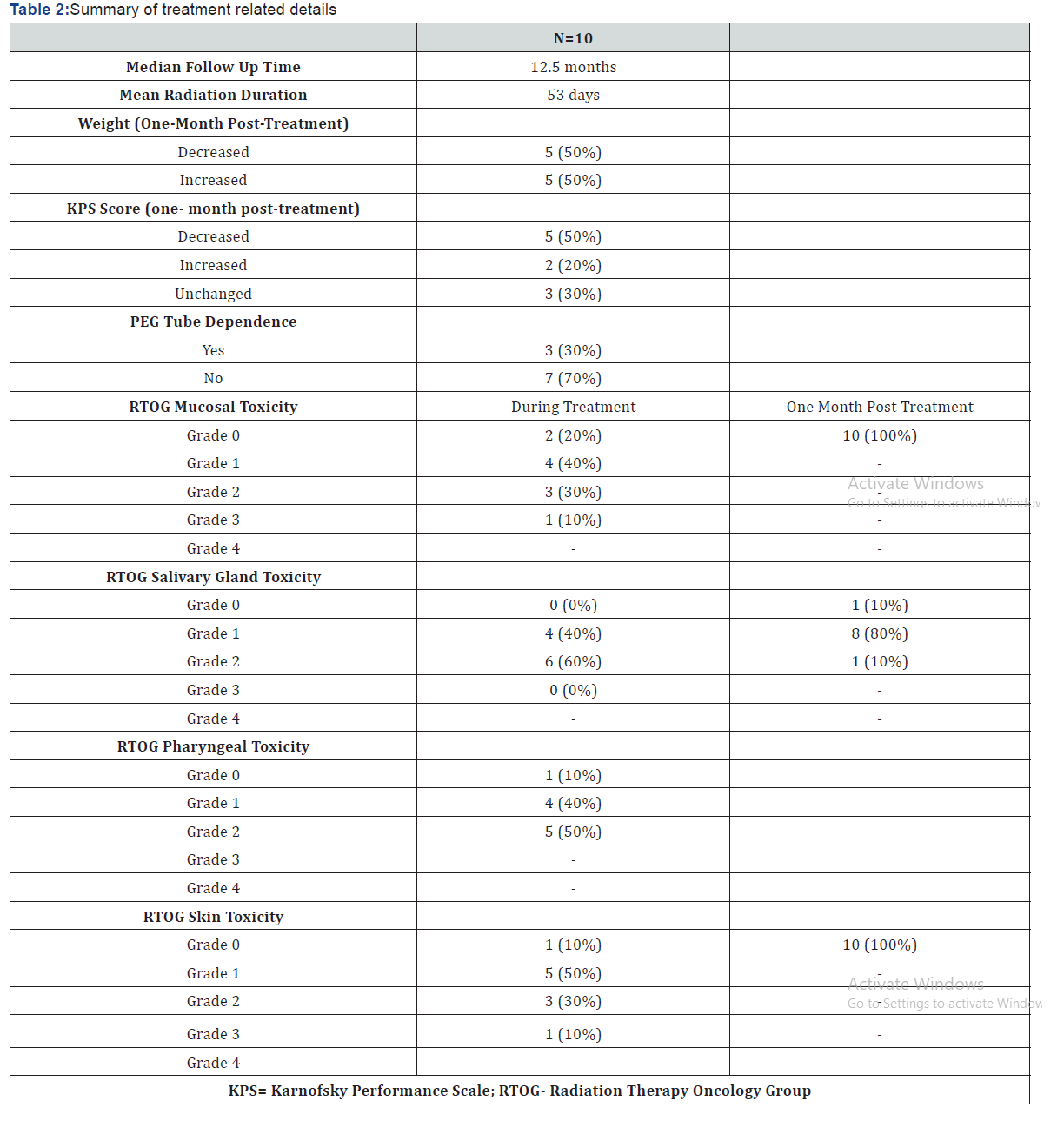
All patients completed their intended chemoradiation treatment as prescribed and the mean duration of radiation therapy was 53.5 days. Toxicity was assessed at weekly followup using the toxicity criteria defined by the Radiation Therapy Oncology Group (RTOG) [9]. Ninety percent of patients developed grade 2 or lower mucositis; all patients had grade ≤2 xerostomia and grade ≤2 acute pharyngeal toxicity. Ninety percent of them developed grade ≤2 radiation dermatitis. Two patients had feeding tubes inserted before the initiation of their treatment. Only one patient required intra-treatment feeding tube insertion. No patient developed grade ≥4 toxicity of any type. A summary of treatment and toxicity details for the 10 patients is shown in Table 2.
All patients had complete resolution of mucositis and dermatitis within one month of completing therapy. Only 10 percent of the patients had persistent grade 2 xerostomia at one-month post-treatment. Forty percent of the patients began to gain weight within a month following completion of therapy. Their KPS remained stable or improved in 50 percent of patients at one month after treatment. Two of the three patients who had had a feeding tube placed either before or during treatment remained feeding tube dependent.
Survival analysis
The median follow-up of this cohort of patients was 12.5 months. All of them eventually progressed and died of their disease. Ninety percent of them developed metastatic disease as the site of first progression after their definitive treatment. Only 30% had local or regional progression of disease along with metastatic disease progression. Forty percent of the patients underwent palliative chemotherapy after disease progression and two patients had salvage neck dissection for their regionally recurrent disease
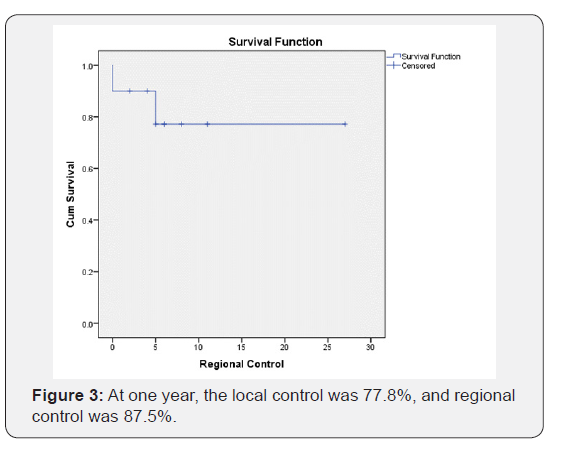
The median overall survival was 12.5 months (range 9-15 months). The overall survival at six months was 90% and at one year was 50% (Figure 1). At one year, the local control was 77.8%, and regional control was 87.5% (Figures 2&3). In patients with ≤3 metastatic foci, the one-year survival was 60% compared to 40% for patients with >3 metastatic lesions (p=0.60). For the patients who received induction chemotherapy, their 1-year survival was 67% compared to 43% for the patients who did not receive induction chemotherapy (p=0.90). There was no difference in survival between patients who underwent SBRT to the oligometastatic lesions in the lungs, compared to those who did not (p=0.465). A summary of treatment outcomes is summarized in Table 3.



Discussion
The current standard of care treatment for patients with metastatic squamous cell carcinoma of the head and neck as recommended by National Cancer Comprehensive Network (NCCN) guidelines is palliative multi-agent chemotherapy using Cisplatin, Cetuximab, and 5 Fluro-uracil for patients with good performances status [10]. Alternatively, Platinum-based doublet [11-14] or single-agent systemic therapy may be administered to patients with Eastern Cooperative Oncology Group (ECOG) Performance status of 0-1. Nivolumab (Opdivo®) [15] and Pembrolizumab (Keytrudra®) [16] are approved as Category 1 and Category 2A NCCN recommendations, respectively, for the patients who have progressed on or following Platinum-based regimens. Alternatively, Afatinib, a tyrosine kinase inhibitor has been approved as second-line therapy for those who have progressed following the use of Platinum agents. This is based on a phase III trial in which Afatinib was compared to Methotrexate [17].
Radiation doses
Our patient population was treated with definitive concurrent chemoradiation treatment using 70 Gy in 35 fractions to the gross disease and involved nodes and 56 Gy in 35 fractions to the subclinical nodal stations, concurrently with chemotherapy. Currently, there is no consensus regarding radiation regimens or dose in the setting of metastatic SCCHN. A few phase II studies and several retrospective studies have explored the optimal treatment for such patients 5, [18-28]. Most of these studies have included patients with newly diagnosed disease who were ineligible for curative treatment due to comorbid conditions, poor performance status, and metastatic disease at diagnosis. The regimens include the QUAD SHOT18 fractionation of treating the patients with 14 Gy in four fractions (3.5 Gy given twice a day at least six hours apart, for two consecutive days; repeated at four weekly intervals for a further two courses if there was no tumor progression). Another fractionation schedule called the Hypo Trial 20 used a hypofractionated course of radiotherapy consisting of 30 Gy in 5 fractions (6 Gy/fraction) at two fractions per week, with an optional 6 Gy boost for small volume disease (<3cm). A recent Canadian study 19 reported the use of 25 Gy in 5 daily fractions using IMRT. The Princess Margaret Hospital (PMH) has used 50 Gy in 2.5 Gy fractions over six weeks (2 cycles of 25 Gy in 10 fractions given within two weeks, separated by a 2-week break). Two reports from India have documented the use of 20 Gy in 5 fractions using 4 Gy daily over one week24 and 40 Gy in 16 fractions at 2.5 Gy daily with a possible boost to total 50 Gy22. The Radiation Therapy Oncology Group (RTOG) protocol 85-02 [29-31] was designed to deliver 370 cGy twice a day for 2 consecutive days at 2- to 3-week intervals for 3 total cycles to a total dose of 4,440 cGy25 (described as QUAD SHOT in the United States); 70 Gy in 2 Gy fractions, 30 Gy in 3 Gy fractions, 3,750 cGy in 2.5 Gy per fraction are other fractionation schedules that have been reported from the University of California, Davis. The Christie Hospital scheme 26 of palliative radiation treatment delivers 50 Gy in 3.125 Gy per fractions daily over 16 fractions. In all these reports, however, radiation was used alone for palliation without any concurrent chemotherapy. In some of these studies, the authors document a dose response relationship, with a better and longer tumor control with higher doses of radiation, consistent with the findings in our analysis [23,25].
The use of concurrent chemotherapy with radiation
The use of concurrent chemoradiation treatment in the metastatic setting is not well studied. In a special regimen named “IHF2SQ (Irradiation Hypo Fractionnee 2 Séances Quotidiennes), the patients received radiation to a dose of 3 Gy twice daily on days 1 and 3, during the first, third, fifth, and seventh weeks for a total of 48 Gy, concurrently with chemotherapy. Gamez et al. [5] reported a modified “QUAD- SHOT” regimen using hypo-fractionated palliative radiotherapy with concurrent chemotherapy using Carboplatin (AUC 2) with each cycle of the QUAD- SHOT regimen. Another study compared the QUAD-SHOT regimen with and without concurrent Paclitaxel and reported an improvement in clinical response without a significant difference in toxicity [6]. An analysis of National Cancer Data Base (NCDB) [32] also noted that aggressive local treatment and systemic therapy improves survival.
Radiation planning modality
In our patient population, we used IMRT as the planning modality, as compared to 3-Dimensional Conformal Radiation Therapy (3D CRT) planning in the aforementioned reports, apart from a Canadian study reported by Fortin et al. [19]. We believe that the use of IMRT will help dose escalate without a corresponding increase in toxicity, making it easier for the patients to tolerate the treatment.
Local therapy directed to the metastatic lesions
A few of our patients were treated with Stereotactic Body Radiation Therapy (SBRT) to the single metastatic lesions in the lungs. Both metastasectomy and SBRT have been described in the literature in this setting and have reported to improve patient survival [33-35]. Due to the limited number of patients, we were not able show any statistically significant benefit among the patients who received SBRT to the metastatic lesions in the lungs.
Survival
The median overall survival across the studies with palliative radiation treatment alone for squamous cell carcinoma of the head and neck region is 6-7 months. But this is a very inhomogeneous cohort of patients as these studies include patients with metastatic disease at diagnosis, patients with recurrent disease and patients with the advanced locoregional disease who are not candidates for curative therapy due to their performance status or medical co- morbidities. In comparison, patients who were eligible to undergo palliative chemotherapy had a median overall survival of 10.1 months as reported in the EXTREME TRIAL (Erbitux in First-Line Treatment of Recurrent or Metastatic Head and Neck Cancer) [2] by receiving palliative multi agent chemotherapy alone, the standard of care. Compared to all of these studies, our cohort of patients with metastatic disease diagnosis who underwent treatment with a curative radiation dose had a median overall was 12.5 months. Most of our patients had been treated prior to the use of Immunotherapy for the patients with metastatic squamous cell carcinoma of the head and neck. So, use of standard dose chemoradiation for this cohort of patients are to yet to be determined in the era of immunotherapy.
It is very important to carefully select patients for aggressive multi-modality treatment in the oligometastatic setting and it should preferably be verified in a multi-disciplinary setting. The increased morbidity of the aggressive local therapy should also be carefully weighed against long-term benefits. It is essential to discuss long-term goals with the patient and family and the treatment plan should be individualized
Limitations
The limitations of our study include its retrospective nature and the limited number of patients. The selection of the patients was also subjective. The added use of induction chemotherapy and SBRT to the oligo-metastatic lung lesions with concurrent chemoradiation therapy in a very limited cohort made it difficult to tease out the benefits of each of these treatments individually. There was no standardized evaluation of the palliative response to the treatment. Incomplete documentation hindered an accurate analysis of the late toxicity profile as well
Conclusion
In conclusion, carefully selected patients with oligometastatic head and neck cancer with good performance status who underwent aggressive treatment with standard dose chemoradiation therapy tolerated their treatment with acceptable morbidity. Their survival was prolonged in comparison to the use of chemotherapy alone, suggesting that this approach may be an additional option for selected patients who have limited metastatic disease and a good performance status. In addition, on-going advances in systemic therapy options leading to longer patient survival make improvements in local control more imperative. Future prospective studies are needed to better define the role of standard dose chemoradiation in patients presenting with metastatic head and neck squamous cell carcinoma. The use of induction chemotherapy and localized treatment of the oligo-metastatic lesions should also be addressed in such studies.
References
- Kuperman DI, Auethavekiat V, Adkins DR, Nussenbaum B, Collins S, et al. (2011) Squamous cell cancer of the head and neck with distant metastasis at presentation. Head Neck 33(5): 714-718.
- Vermorken JB, Mesia R, Rivera F, et al. (2008) Platinum-based chemotherapy plus cetuximab in head and neck cancer. N Engl J Med 359(11): 1116-1127.
- Monnier L, Touboul E, Durdux C, Lang P, Guily SJL, et al. (2013) Hypofractionated palliative radiotherapy for advanced head and neck cancer: the IHF2SQ regimen. Head Neck 35(12): 1683-1688.
- Minatel E, Gigante M, Franchin G, Gobitti C, Mascarin M, et al. (1998) Combined radiotherapy and bleomycin in patients with inoperable head and neck cancer with unfavourable prognostic factors and severe symptoms. Oral Oncol 34(2): 119-122.
- Gamez ME, Agarwal M, Hu KS, Lukens JN, Harrison LB, et al. (2017) Hypofractionated Palliative Radiotherapy with Concurrent Radiosensitizing Chemotherapy for Advanced Head and Neck Cancer Using the "QUAD-SHOT Regimen". Anticancer Res 37(2): 685-691.
- Dubey M (2018) Comparative Evaluation of Palliative Radiotherapy with Chemotherapy vs. Palliative Radiotherapy Alone in Locally Advanced Head and Neck Cancer. Journal of Cancer Research and Immuno-Oncology.
- Duggar WN, Bhandari R, Yang CC, Vijayakumar S (2018) Group consensus peer review in radiation oncology: commitment to quality. Radiat Oncol 13(1): 55.
- Bhandari RP, Duggar WN, Yang C, Kanakamedala MR, Packianathan S, et al. (2018) A Sustainable Model for Peer Review and Utility of At-a-Glance Analysis of Dose Volume Histogram in Radiation Oncology. J Am Coll Radiol 15(2): 310-312.
- Cox JD, Stetz J, Pajak TF (1995) Toxicity criteria of the Radiation Therapy Oncology Group (RTOG) and the European Organization for Research and Treatment of Cancer (EORTC). Int J Radiat Oncol Biol Phys 31(5): 1341-1346.
- NCCN (2019) National Comprehensive Cancer Network Guidelines.
- Samlowski WE, Moon J, Kuebler JP, Nichols CR, Gandara DR, et al. (2007) Evaluation of the combination of docetaxel/carboplatin in patients with metastatic or recurrent squamous cell carcinoma of the head and neck (SCCHN): A Southwest Oncology Group Phase II study. Cancer Invest 25(3): 182-188.
- Gibson MK, Li Y, Murphy B, Hussain MH, DeConti RC et al. (2005) Randomized phase III evaluation of cisplatin plus fluorouracil versus cisplatin plus paclitaxel in advanced head and neck cancer (E1395): an intergroup trial of the Eastern Cooperative Oncology Group. J Clin Oncol 23(15): 3562-3567.
- Burtness B, Goldwasser MA, Flood W, Mattar B, Forastiere AA, et al. (2005) Phase III randomized trial of cisplatin plus placebo compared with cisplatin plus cetuximab in metastatic/recurrent head and neck cancer: An Eastern Cooperative Oncology Group study. J Clin Oncol 23(34): 8646-8654.
- Guigay J, Fayette J, Dillies AF, Sire C, Kerger JN, et al. (2015) Cetuximab, docetaxel, and cisplatin as first-line treatment in patients with recurrent or metastatic head and neck squamous cell carcinoma: a multicenter, phase II GORTEC study. Ann Oncol 26(9): 1941-1947.
- Ferris RL, Blumenschein G, Fayette J (2016) Nivolumab for Recurrent Squamous-Cell Carcinoma of the Head and Neck. N Engl J Med 375(19): 1856-1867.
- Chow LQM, Haddad R, Gupta S, Mahipal A, Mehra R, et al. (2016) Antitumor Activity of Pembrolizumab in Biomarker-Unselected Patients with Recurrent and/or Metastatic Head and Neck Squamous Cell Carcinoma: Results from the Phase Ib KEYNOTE-012 Expansion Cohort. J Clin Oncol 34(32): 3838-3845.
- Machiels JP, Haddad RI, Fayette J, Licitra LF, Tahara M, et al. (2015) Afatinib versus methotrexate as second-line treatment in patients with recurrent or metastatic squamous-cell carcinoma of the head and neck progressing on or after platinum-based therapy (LUX-Head & Neck 1): an open-label, randomised phase 3 trial. Lancet Oncol 16(5): 583-594.
- Corry J, Peters LJ, Costa ID, Milner AD, Fawns H, et al. (2005) 'QUAD SHOT'--a phase II study of palliative radiotherapy for incurable head and neck cancer. Radiother Oncol 77(2): 137-142.
- Fortin B, Khaouam N, Filion E, Nguyen-Tan PF, Bujold A, et al. (2016) Palliative Radiation Therapy for Advanced Head and Neck Carcinomas: A Phase 2 Study. Int J Radiat Oncol Biol Phys 95(2): 647-653.
- Porceddu SV, Rosser B, Burmeister BH, Jones M, Hickey B, et al. (2007) Hypofractionated radiotherapy for the palliation of advanced head and neck cancer in patients unsuitable for curative treatment--"Hypo Trial". Radiother Oncol 85(3): 456-462.
- Paris KJ, Spanos WJ, Lindberg RD, Jose B, Albrink F, et al. (1993) Phase I-II study of multiple daily fractions for palliation of advanced head and neck malignancies. Int J Radiat Oncol Biol Phys 25(4): 657-660.
- Agarwal JP, Nemade B, Murthy V, Ghosh-Laskar S, Budrukkar A, et al. (2008) Hypofractionated, palliative radiotherapy for advanced head and neck cancer. Radiother Oncol 89(1): 51-56.
- Stevens CM, Huang SH, Fung S, Bayley AJ, Cho JB, et al. (2011) Retrospective study of palliative radiotherapy in newly diagnosed head and neck carcinoma. Int J Radiat Oncol Biol Phys 81(4): 958-963.
- Mohanti BK, Umapathy H, Bahadur S, Thakar A, Pathy S, et al. (2004) short course palliative radiotherapy of 20 Gy in 5 fractions for advanced and incurable head and neck cancer: AIIMS study. Radiother Oncol 71(3): 275-280.
- Chen AM, Vaughan A, Narayan S, Vijayakumar S (2008) Palliative radiation therapy for head and neck cancer: toward an optimal fractionation scheme. Head Neck 30(12): 1586-1591.
- Al-mamgani A, Tans L, Rooij VPH, Noever I, Baatenburg DjRJ, et al. (2009) Hypofractionated radiotherapy denoted as the "Christie scheme": an effective means of palliating patients with head and neck cancers not suitable for curative treatment. Acta Oncol 48(4): 562-570.
- Kancherla KN, Oksuz DC, Prestwich RJ, Fosker C, Dyker KE, et al. (2011) The role of split-course hypofractionated palliative radiotherapy in head and neck cancer. Clin Oncol (R Coll Radiol) 23(2): 141-148.
- Lok BH, Jiang G, Gutiontov S, Lanning RM, Sridhara S, et al. (2015) Palliative head and neck radiotherapy with the RTOG 8502 regimen for incurable primary or metastatic cancers. Oral Oncol 51(10): 957-962.
- Spanos W, Guse C, Perez C, Grigsby P, Doggett RL, et al. (1989) Phase II study of multiple daily fractionations in the palliation of advanced pelvic malignancies: preliminary report of RTOG 8502. Int J Radiat Oncol Biol Phys 17(3): 659-661.
- Spanos WJ, Perez CA, Marcus S, Poulter CA, Doggett RL, et al. (1993) Effect of rest interval on tumor and normal tissue response--a report of phase III study of accelerated split course palliative radiation for advanced pelvic malignancies (RTOG-8502). Int J Radiat Oncol Biol Phys 25(3): 399-403.
- Spanos WJ, Clery M, Perez CA, Grigsby PW, Doggett RL, et al. (1994) Late effect of multiple daily fraction palliation schedule for advanced pelvic malignancies (RTOG 8502). Int J Radiat Oncol Biol Phys 29(5): 961-967.
- Zumsteg ZS, Luu M, Yoshida EJ, Kim S, Tighiouart M, et al. (2017) Combined high-intensity local treatment and systemic therapy in metastatic head and neck squamous cell carcinoma: An analysis of the National Cancer Data Base. Cancer 123(23): 4583-4593.
- Florescu C, Thariat J (2014) Local ablative treatments of oligometastases from head and neck carcinomas. Crit Rev Oncol Hematol 91(1): 47-63.
- Young ER, Diakos E, Khalid-Raja M, Mehanna H (2015) Resection of subsequent pulmonary metastases from treated head and neck squamous cell carcinoma: systematic review and meta-analysis. Clin Otolaryngol 40(3): 208-218.
- Sun XS, Michel C, Babin E, Raucourt DD, Péchery A, et al. (2018) Approach to oligometastatic disease in head and neck cancer, on behalf of the GORTEC. Future Oncol 14(9): 877-889.






























