Ameliorative Effects of Ginkgo Biloba on Haloperidol-Induced Vacuous Chewing Movement BDNF Plasm Levels in Rats: A Dose-Dependent Effect
Shuchang He*1, Hongxiao Jia2, Shuang Wu1 and Xiangyang Zhang*3
1Department of Psychology, Peking University, China
2Beijing Anding Hospital, Capital Medical University, China
3Beijing HuiLongGuan Hospital, China
Submission: March 27, 2017; Published: April 13, 2017
*Corresponding author: Shuchang He, School of Psychological and Cognitive Sciences, Peking University, Beijing 100871, China, Email: shuchangh@pku.edu.cn
Xiangyang Zhang, Beijing HuiLongGuan Hospital, Chang-Ping District, Beijing 100096, China, Email:zhangxy@gmail.com
How to cite this article: Shuchang H, Hongxiao J Shuang W Xiangyang Z. Ameliorative Effects of Ginkgo Biloba on Haloperidol-Induced Vacuous Chewing Movement and BDNF Plasm Levels in Rats: A Dose-Dependent Effect. Psychol Behav Sci Int J. 2017; 3(1): 555603. DOI: 007 10.19080/PBSIJ.2017.03.555603
Abstract
Background: The pathophysiology of tardive dyskinesia (TD) remains unknown. Recent clinical evidence shows that low brain-derived neurotrophic factor (BDNF) may play an important role in its pathophysiology. Ginkgo biloba (EGb-761), a potent antioxidant, may have neuroprotective effects through enhancing BDNF levels. This study aimed to assess the efficacy of EGb-761 for the treatment of TD and the effect on plasma BDNF levels in a rodent model.
Methods: Fifty rats were randomly assigned into five groups: control group [injected saline intraperitoneally (IP) for 9 weeks], TD model group and three treatment groups (injected haloperidol 2mg/kg/day for 4 weeks, and then saline, 50mg/kg, 100mg/kg or 200mg/kg EGb-761 daily for the following 5 weeks). We performed behavioral assessments shown as vacuous chewing movements (VCMs) weekly and analyzed BDNF levels in plasma at the end of treatment.
Results: Haloperidol administration led to a progressive increase in VCMs, but all three groups of daily doses of EGb-761 significantly decreased VCMs, with both 100mg and 200mg showing the greater decreased in VCMS. Haloperidol also decreased BDNF plasma level, but 100mg EGb-761 administration significantly increased BDNF levels with no significant difference from the control group.
Conclusion: BDNF may be a significant biomarker related to the pathophysiology of TD. EGb-761 reduces TD symptoms possibly via its effect on BDNF system, which is related to its dosing.
Keywords: Tardive dyskinesia; Extract of ginkgo biloba; EGb-761; BDNF; Haloperidol
Abbreviations: TD: Tardive Dyskinesia; BDNF: Brain-Derived Neurotrophic Factor; IP: Intra-Peritoneally; VCMs: Vacuous Chewing Movements; AIMS: Abnormal Involuntary Movement Scale; BPSD: Behavioral and Psychological Symptoms; AD: Alzheimer's Disease; PD: Parkinson's Disease; HAL: Haloperidol; NS: Normal Physiological Saline; EGb-761: Extract of Ginkgo Biloba Leaves; ELISA: Enzyme-Linked Immuno-Sorbent Assay; ANCOVA: Analyses of Co-Variance; LSD: Least Significant Difference
Introduction
Tardive dyskinesia (TD) is a serious, often disabling, movement disorder associated with long-term neuroleptic treatment, in which characteristic abnormal, involuntary,persistent movements of the tongue and lips and facial musculature are seen [1]. However, TD is a common side effect of antipsychotic drugs treatment in patients with schizophrenia, such as haloperidol, perphenazine or chlorpromazine treatment, and the incidence rate has been estimated that up to 32% [2]. As a consequence, the adverse events induced by these drugs make schizophrenia patients having to face more difficulty in daily life. As so far, the etiology and pathophysiology of TD remain unclear. A recent neurodegeneration hypothesis has been proposed to explain the development of TD, suggesting that long-term antipsychotic treatments can enhance neurotoxic free radical generation [3], which can induce potentially irreversible damage in motor regions of the brain such as the basal ganglia, resulting in TD [4,5]. Currently, no standard treatment approach is available for physicians confronted with such patients.
Brain-derived neurotrophic factor (BDNF), the most abundant of the neurotrophins in the brain, plays an important role in neuronal survival, development and maintenance [6,7]. BDNF increases dopamine uptake in mesencephalic, and GABA uptake in basal forebrain, preventing neurons damage of the basal forebrain and the striatum induced by cholinergic or glutamatergic mechanism [8,9], which was implicated in the pathogenesis of TD [10,11]. Moreover, BDNF had been found to enhance the survival of neurons, to confer protection against the neurotoxic effects, and to increase the activity of the protective enzyme [12]. Interestingly, we recently reported lower serum BDNF levels in patients with TD [13,14] and found that these levels were inversely correlated with their abnormal involuntary movement scale (AIMS) score, suggesting that BDNF may play an important role in TD pathophysiology [13].
Ginkgo biloba is an ancient Chinese tree, and extracts from its leaves have been used therapeutically in traditional medicine for centuries [15,16]. EGb-761, the standardized extract of Ginkgo biloba leaves, has been widely exploited to treat cerebrovascular and peripheral vascular insufficiency, cognitive and functional symptoms associated with "mild to moderate" dementia [17]. Studies have showed that it may improve the cognitive function and the social functioning, behavioral and psychological symptoms (BPSD), activities of daily living and quality of life in demented patients [18-20]. Beck et al. [21] reported that Ginkgo biloba extract EGb-761 improved cognitive flexibility and response inhibition in elderly adults with subjective memory impairment. Many clinical trials have demonstrated that EGb- 761 treatment may be effective in the treatment and prevention of neurodegenerative disorders such as Alzheimer's disease (AD) [20,22-24] and Parkinson's disease (PD) [23,25-27].
Recently, a systematic review also confirmed the efficacy of EGb-761on the treatment of patients suffering from Alzheimer's, vascular or dementia with BPSD [28]. The potent antioxidant and neuroprotective activity and free radical scavenging actions were proposed to underlie the beneficial effects of EGb-761 [29-31]. Moreover, our previous study showed that EGb-761 enhanced the effectiveness of antipsychotic drug haloperidol and reduced its extra pyramidal side effects [32]. We found that EGb-761 was an effective treatment for reducing the symptoms of TD in schizophrenia patients [33]. Several studies reported that EGb761 treatment in dosage strengths of 50 mg/kg/day could attenuate and prevent vacuous chewing movements in a rodent model of TD, though not compared the efficacy of different doses [34,35]. Interestingly, our recent study showed that EGb-761 treatment improved symptoms of TD and increased BDNF levels. Furthermore, the improvement of AIMS total score correlated with the increase in BDNF levels, indicating that EGb- 761 has neuroprotective and neuromodulatory effects on TD by enhancing BDNF [36,37].
However, to our knowledge, there is little study on whether EGb-761 may reduce TD symptoms and increase BDNF levels in an animal model of TD [35]. The purpose of the present study, therefore, was to examine: 1) Repeated the result that plasma BDNF levels lower in TD rats after drug administration than normal controls; 2) Whether EGb-761 may lead to a significant reduction in haloperidol-induced VCMs, and a significant increase in BDNF levels of TD rats? 3) Were there different curative effects after different doses of EGb-761 treatment? 4) Was improvement in TD symptoms associated with changes in plasma BDNF levels after EGb-761 treatment?
Materials and Methods
Animals
Science Academy, Beijing. They were housed under standard laboratory conditions and maintained on a normal light-dark cycle with free access to food and water. Experimental protocols were approved and supervised by the Institutional Animal Ethics Committee of the Peking University, and followed the guidelines of China National Science Academy for the care and use of experimental animals.
Drugs
Haloperidol (HAL) (Shanghai Jiufu Pharmaceutical Co., Ltd, China) was dissolved in normal physiological saline (NS) to the concentration of1mg/ml, and was administered intraperitoneally (IP) in a volume of 1 ml/kg. Extract of Ginkgo Biloba Leaves (EGb- 761) was obtained from Dr. Willmar Schwabe Pharmaceuticals (Karlsruhe, Germany), and was dissolved in NS to 25.2 mg/ 5 ml, which was administered IP in a constant volume of 5ml per 1000g of bodyweight. Animals were divided into 5 groups: 1) control group (n=13): rats were injected saline IP for 4 weeks, and then administered saline IP for the following 5 weeks; 2) TD model group: rats were injected with HAL (2 mg/kg/day) for 4 weeks, and then administered saline IP for the following 5 weeks; 3) 50 mg EGb-761 group: rats were injected with HAL (2 mg/kg/day) for 4 weeks, and then administered EGb-761 (50 mg/kg/day) IP for the following 5 weeks; 4) 100 mg EGb-761 group: rats were injected with HAL (2 mg/kg/day) for 4 weeks, and then administered Egb-761 (100 mg/kg/day) IP for the following 5 weeks; 5) 200mg EGb-761 group: rats were injected with HAL (2 mg/kg/day) for 4 weeks, and then administered EGb-761 (200 mg/kg/day) for the following 5 weeks.
VCMs Assessments
Rats treated chronically with neuroleptic drugs often develop spontaneous mouth movements which have been described as "vacuous chewing movements" (VCMs) [38]. VCMs were defined as single mouth openings in the vertical plane not directed toward physical material, and three types of oral behaviors were recorded [39]: vertical jaw movements (each vertical opening and closing of jaw was regarded as one VCM), bursts of jaw tremor and tongue protrusions. For calculation purposes, each burst of jaw tremor was regarded as two VCMs. A VCM consisted of a rapid movement of the jaw that resembled chewing, but did not appear to be directed at any particular stimulus. In addition, counting was stopped whenever the rat began grooming, and restarted when grooming stopped, because the periods of grooming were obscured.
On the test day, rats were placed individually in a small (20cmx30cmx30cm) Plexiglas cage. Animals were given 5 min to get acclimatized to the observation cage before behavioral assessments. The behavioral parameters of oral dyskinesia were measured continuously for a period of 5 min [40,41]. In all the experiments, the scorer was unaware of the treatment conditions given to the animals. VCMs were assessed in each individual weekly, with a total of nine times (test for 9 weeks in all). In order to ensure the effectiveness of TD model, we removed rats with low VCMs scores (<5) (n=10) from all rats injected with HAL (i.e. rats in Group 2~5) at the end of the fourth week.
Plasma BDNF Measurement
On week 9, rats were deeply anesthetized (nembutal, 80 mg/kg, intraperitoneal injection) and plasma samples were obtained from each rat's heart after the behavioral assessment, which were collected in tubes containing EDTA. The plasma was separated, aliquot and stored at-70oC before use. Plasma BDNF levels were measured within one month by sandwich enzyme- linked immunosorbent assay (ELISA) using a commercially available kit (R&D systems, Beijing, China), as described in the previous report [42]. All samples were assayed by a research assistant blind to the clinical situation. Inter- and intra-assay variation coefficients were 7% and 5%, respectively.
Statistical analysis
The principal outcome analysis consisted of repeated- measures analyses of covariance (ANCOVA) for the VCMs score, with a between-subject factor of drug [saline, haloperidol alone (TD model), 50 mg, 100 mg, 200 mg] and a within-subject factor of time (every week, total 9 time points), with VCM score at baseline as a covariate, followed by the post hoc test [Fisher's least significant difference (LSD) test)] to compare the differences between the dosage groups. BDNF plasma levels were analyzed by one-way ANOVA. Post hoc tests were conducted to determine the difference between groups, followed by LSD test. In all tests, the criterion for statistical significance was P<0.05. All statistical analyses were conducted in SPSS Statistics, version 20.0 (SPSS, Inc., Chicago).
Results
Haloperidol-induced VCMs
Figure 1 shows that VCMs increased gradually with haloperidol (2 mg/kg/day) IP injection, and reached a peak until the 5th week, then changed in different situation with different dose of EGb-761, but the general trend is down. However, VCMs of control group changed weekly in nine weeks, being fewer than 5 from beginning to end. Repeated-measures ANCOVA on VCMs over the withdrawal period showed a significant group effect (F=10.885, df=4, p<0.001), a significant time effect (F=3.365, df=7, p<0.01), and a non-significant group-by-time effect (F=.640, df=28, p>.05). In 2nd and 3rd week, there were no significant differences among all groups (2nd week: NC vs TD vs low-dose EGb-761 vs middle-dose EGb-761 vs high-dose EGb-761: 2.429 vs 12.714 vs 14.857 vs 12.714 vs 13.142, all p>.05) (3rd week: NC vs TD vs low-dose EGb-761 vs middle-dose EGb-761 vs highdose EGb-761: 1.857 vs 14.000 vs 14.429 vs13.857 vs 14.000, all p>.05). In 4th week, control group had significantly lower score on VCMs than three treatment groups (NC vs low-dose EGb-761/middle-dose EGb-761/high-dose EGb-761: 1.714 vs 18.000/16.000/19.429, all p<.05). In 5th week, control group had significantly lower score on VCMs than TD and three treatment groups (NC vs TD/low-dose EGb-761/middle-dose EGb-761/ high-dose EGb-761: 1.857 vs 20.286/21.429/19.714/20.286, all p<.001), but there were no significant differences among TD and three treatment groups. From 6th to 9th week, control group had significantly lower score on VCMs than TD model group (p<.001), low-dose EGb-761 group (p<.001), and high-dose EGb-761 group (p<.01), but not significantly lower VCMs than middle-dose EGb-761 group (p>.05).
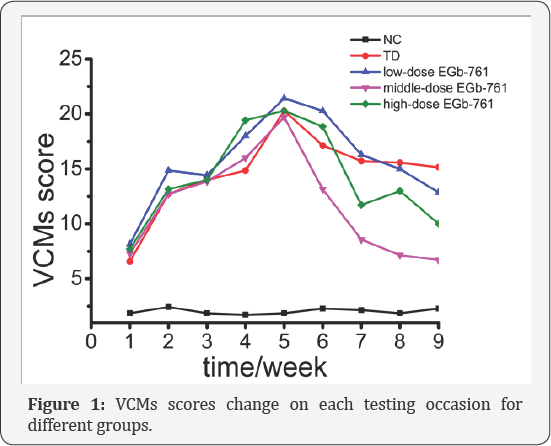
Plasma BDNF levels
As shown in Figure 2, differences of plasma BDNF levels among all of five groups were significant (F(4,45)=8.183, p<.001). Specifically, plasma BDNF levels were significantly higher in control group than TD model group (211.21±42.91 vs 163.63±29.98, p<.05), low-dose EGb-761 group (211.21±42.91 vs 150.05±15.98, p<.01), and high-dose EGb-761 group (211.21±42.91 vs 161.41±7.30, p<.05), but there was no significant difference in plasma BDNF between normal controls and rats in middle-dose EGb-761 group (211.21±42.91 vs 197.90±24.71, p>.05) (Figure 2a). Comparing plasma BDNF of TD model group with three treatment groups (Figure 2b), we got results that there were significant differences among the four groups (F(333)=6.5 9 3, p<.01), and plasma BDNF levels of middle- dose EGb-761 group were significantly higher than others' (middle vs TD: p<.01; middle vs low: p<.001; middle vs high: p<.01). No significant difference in plasma BDNF levels among TD model group, low- and high-dose EGb-761 group existed (p>.05) (Figures 1 & 2).
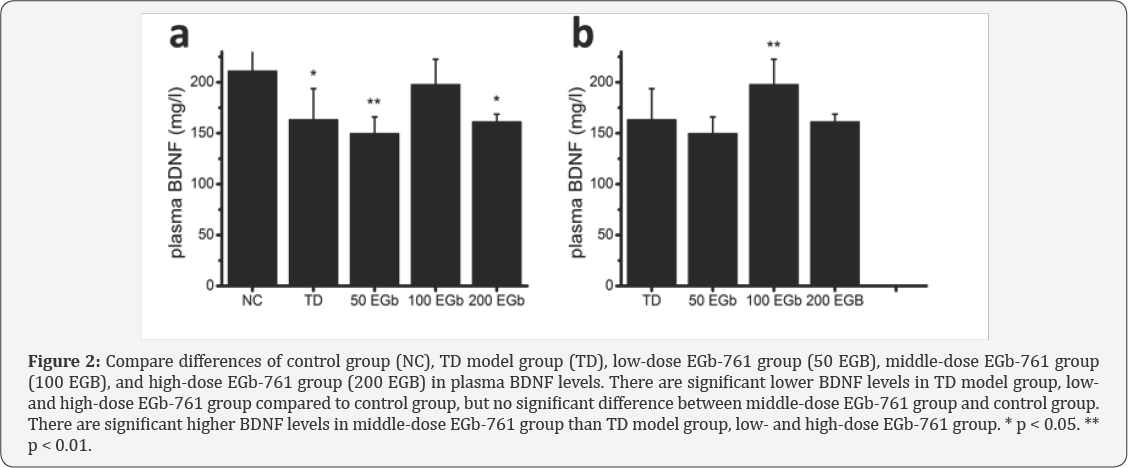
Discussion
The major findings of this study were that 1) rats in TD model group had lower plasma BDNF levels than normal controls; 2) different doses of EGb-761 treatment improved TD symptoms in varying degrees; 3) VCMs of rats in middle-dose EGb-761 group was closest to one of normal controls, and there was no significant difference between middle-dose EGb-761 group and control group in VCMs measurement; 4) only 100mg EGb-761 treatment increased significantly BDNF levels among three treatment groups, and there was no significant difference between middle-dose EGb-761 group and control group in plasma BDNF levels.
BDNF levels were significantly lower in TD patients than normal controls, which are consistent with the previous reports [13,36,37]. These decreased BDNF levels are inosculated with the neuronal degeneration hypothesis of tardive dyskinesia, because BDNF exerts various effects on neuronal survival and maintenance, protect against neuronal degeneration induced by ischemia, hypoglycemic insults, and glutamatergic mechanism, all hypothesized contributors to neuroleptic-induced TD [7,8,43]. Lower BDNF levels would be a high risk factor for TD development, conceivably because of that down-regulation of this neurotrophin could lead to further neurotoxicity/ neurodegeneration in organism with TD. However, the exact mechanism for lower BDNF levels in TD pathology is still undiscovered, and deserves further investigation, as well as the relationship between BDNF and other risk factors, such as oxidative stress, and antipsychotic treatment. Then solving these questions may be extremely rewarding for TD treatment, in which BDNF will play a role indubitably.
In the present study, TD symptoms of three treatment groups had been improved significantly, but in different levels. Low- and middle-dose EGb-761 group had significantly lower VCMs than TD model group. There was a marginally significant reduction in VCMs in high-dose group compared to TD model group. However, no significant difference in VCMs measurement between high-dose group and control group, further VCMs of rats in TD model group were significantly higher than normal controls, so it appeared that high-dose EGb-761 had a significant and favorable effect on TD symptoms. It was distinctive that there was no significant BDNF increase in low- and high-dose group compared to TD model group. Significantly enhanced BDNF levels only occurred in middle-dose group among three treatment groups. BDNF levels of rats in middle-dose group were closest approach to normal levels and their differences with normal controls were not statistically significant. Moreover, VCMs of animals in middle-dose group were the most proximate to normal controls.
EGb-761 has been shown in previous studies to improve mitochondrial function and increase energy metabolism, promotes hippocampal neurogenesis and neuroplasticity, such as long-term potentiation, spine density, neuritogenesis and neurogenesis [44]. Furthermore, one of possible mechanisms to explain how EGb-761 could reduce TD may relate to enhanced BDNF levels. A recent study found that EGb-761 and VE decreased dyskinetic jaw movements and increased BDNF expression levels in prefrontal cortex, striatum, substantia nigra, and globus pallidus in the rat model of TD [35]. Zhang et al. [36] reported that the increase in BDNF was correlated with the improvement in AIMS ratings among Chinese schizophrenic patients with TD who were treated with EGb-761 and this indirect evidence had led to the speculation that enhancing BDNF levels could decrease further neuronal injury and even reverse some existing TD symptoms. On the other hand, the efficacy of EGb-761 contains antioxidant properties and free radical scavenging actions, which have been therapeutic for other neurodegenerative disorders, such as AD, and also suggested for TD treatment [3,32,45,46]. Zhang et al. [33] demonstrated that EGb-761 appeared to be an effective treatment for reducing the symptoms of TD in schizophrenia patients, and improvement may be mediated through the well-known antioxidant activity of this extract.
Oxidative stress has a critical role in the pathophysiology of TD, and bauhinia forficata and crocin as the antioxidants showed a protective effect against TD [47,48]. BDNF levels increasing might be promoting antioxidant or free radical scavengers of EGb-761 to enhance its efficiency in treatment of TD symptoms. Zhang et al. [36] found that the better responders to EGb-761 treatment were these whose BDNF levels were closer to normal levels. However, the relationship between the two needs to be further elucidated. Other possible mechanisms through how EGb-761 could improve TD symptoms may relate to its efficacy as a symptomatic treatment for mild-to-moderate AD [49-51], and it may be due that EGb-761 has neuroprotective and neuromodulatory effects [14,24,29,52]. Zhang et al. [36] concluded that EGb-761 may stimulate other neuroprotective processes relevant to TD as well as AD, for instance activating the CREB signaling pathway more broadly as a neuronal protective effect [42], elevating brain levels of pCREB to lead to enhanced hippocampal synaptogenesis and neurogenesis in a rodent model of AD [53,54]. Form this point, both of low- and high- dose EGb-761 improved just TD symptoms, instead of curing TD radically.
It seemed that there was no significant difference in VCMs between high-dose group and control group, though BDNF levels of high-dose group were not enhanced. Therefore, improvement in TD symptoms did not mean improvement inside damage in TD rats, and TD symptoms were likely to be recurrent because of low BDNF levels. Lower BDNF levels were more associated with a high risk for TD development [13]. The mostly discovery of our study was that middle-dose EGb-761 improved TD symptoms and increased BDNF levels, which were nearly the same to normal, as well as the best curative effect. We can infer that more improvements are available when long-lasting middle-dose EGb- 761 treatment in TD.
The major limitation of the present study was that we didn’t test the correlation between increased BDNF levels and decreased VCMs, so we could not directly detect the relationship of enhancing BDNF levels and TD symptoms improvement in the treatment of EGb-761 [55-59]. Other limitations included the small sample size, the insufficient treatment grouping, the relatively short duration of treatment, lack of measuring BDNF levels and VCMs scores at baseline, and obtaining BDNF levels at only Week 9 rather than at multiple time points.
Conclusion
BDNF levels may be implicated in the pathophysiology of tardive dyskinesia and may promote or mediate its improvement with antioxidant treatment. Merely alleviating TD symptoms may work in the short term, but don’t fundamentally solve the problem. When BDNF levels are enhanced significantly, TD is improved indeed by EGb-761. In addition, only middle-dose EGb-761 can maximize the treatment effects, and too high or too low dose is not perfect.
Contributors
Zhang Xiangyang and Jia Hongxiao designed the study, wrote the manuscript. He Shuchang collected the data and Wu Shuang wrote first draft of the manuscript. All authors approved the final manuscript.
Funding
This research was financially supported by a research grant to He Shuchang (Natural Science Foundation of China, Grant NO 81571322).
References
- Cloud LJ, Zutshi D, Factor SA (2014) Tardive dyskinesia: therapeutic options for an increasingly common disorder. Neurotherapeutics 11(1): 166-176.
- Thelma B, Srivastava V, Tiwari AK (2008) Genetic underpinnings of tardive dyskinesia: passing the baton to pharmacogenetics. Pharmacogenomics 9(9): 1285-1306.
- Lohr JB, Kuczenski R, Niculescu AB (2003) Oxidative mechanisms and tardive dyskinesia. CNS Drugs, 17(1): 47-62.
- Cho CH, Lee HJ (2013) Oxidative stress and tardive dyskinesia: pharmacogenetic evidence. Prog Neuropsychopharmacol Biol Psychiatry 46: 207-213.
- Teo JT, Edwards MJ, Bhatia K (2012) Tardive dyskinesia is caused by maladaptive synaptic plasticity: a hypothesis. Movement Disord 27(10): 1205-1215.
- Altar CA, Cai N, Bliven T, Juhasz M, Conner JM, et al. (1997) Anterograde transport of brain-derived neurotrophic factor and its role in the brain. Nature 389: 856-860.
- Tsukinoki K, Saruta J, Muto N, Sasaguri K, Sato S, et al. (2007) Submandibular Glands Contribute to Increases in Plasma BDNF Levels. J Dent Res 86(3): 260-264.
- Cheng B, Mattson MP (1994) NT-3 and BDNF protect CNS neurons against metabolic/excitotoxic insults. Brain Res 640(1-2): 56-67.
- Knusel B, Winslow JW, Rosenthal A, Burton LE, Seid DP, et al. (1991) Promotion of central cholinergic and dopaminergic neuron differentiation by brain-derived neurotrophic factor but not neurotrophin 3. Proc Nati Acad Sci USA 88(3): 961-965.
- Gerlach J, Reisby N, Randrup A (1974) Dopaminergic hypersensitivity and cholinergic hypofunction in the pathophysiology of tardive dyskinesia. Psychopharmacology 34(1): 21-35.
- Tsai G, Goff DC, Chang RW, Flood J, Baer L, et al. (1998) Markers for glutamatergic neurotransmission and oxidative stress associated with tardive dyskinesia. Am J Psychiatry 155(9): 1207-1213.
- Spina MB, Squinto SP, Miller J, Lindsay RM, Hyman C (1992) Brain- Derived Neurotrophic Factor Protects Dopamine Neurons Against 6-Hydroxydopamine and N-Methyl-4-Phenylpyridinium Ion Toxicity: Involvement of the Glutathione System. J Neurochem 59(1): 99-106.
- Tan YL, Zhou DF, Zhang XY (2005) Decreased plasma brain-derived neurotrophic factor levels in schizophrenic patients with tardive dyskinesia: association with dyskinetic movements. Schizophr Res 74(2-3): 263-270.
- Luo Y (2001) Ginkgo biloba neuroprotection: Therapeutic implications in Alzheimer's disease. J Alzheimers Dis 3(4): 401-407.
- Jacobs BP, Browner WS (2000) Ginkgo biloba: A living fossil. Am J Med 108(4): 341-342.
- Kleijnen J, Knipschild P (1992) Ginkgo biloba. Lancet 340: 1136 -1139.
- Defeudis FV (1991) Ginkgo Biloba Extract (EGb): Pharmacological Activities and Clinical Appli-cations. Elsevier, Paris, pp. 61-66.
- R Hoerr (2003) Behavioural and Psychological Symptoms of Dementia (BPSD): Effects of EGb 761. Pharmacopsychiatry 36: 56-61.
- Ralf lhl (2012) Gingko biloba extract EGb-761: clinical data in dementia. Int Psychogeriatr 24: S35-S40.
- S Kanowski, R Hoerr (2003) Ginkgo biloba Extract EGb 761 in Dementia: Intent-to-treat Analyses of a 24-week, Multi-center, Doubleblind, Placebo-controlled, Randomized Trial. Pharmacopsychiatry 36(6): 297-303.
- Beck SM, Ruge H, Schindler C, Burkart M, Miller R, et al. (2016) Effects of Ginkgo biloba extract EGb 761® on cognitive control functions, mental activity of the prefrontal cortex and stress reactivity in elderly adults with subjective memory impairment-a randomized doubleblind placebo-controlled trial. Hum Psychopharmacol 31(3): 227-242.
- Dodge HH, Zitzelberger T, Oken BS, Howieson D, Kaye J (2008) A randomized placebo-controlled trial of Ginkgo biloba for the prevention of cognitive decline. Neurology 70: 1809-1817.
- Kim MS, Lee JI, Lee WY , Kim SE (2004) Neuroprotective Effect of Ginkgo biloba L. Extract in a Rat Model of Parkinson's Disease. Phytother Res 18(8): 663-666.
- Shi C, Liu J, Wu F, Yew DT (2010) Ginkgo biloba Extract in Alzheimer's Disease: From Action Mechanisms to Medical Practice. Int J Mol Sci 11(1): 107-123.
- Ahmad M, Saleem S, Ahmad AS, Yousuf S, Ansari MA, et al. (2005) Ginkgo biloba affords dose-dependent protection against 6-hydroxydopamine- induced parkinsonism in rats: neurobehav-ioural, neurochemical and immunohistochemical evidences. J Neurochem 93(1): 94-104.
- Rojas P, Serrano-Garci'a N, Mares-Samano JJ, Medina-Campos ON, Pedraza-Chaverri J, et al. (2008) EGb761 protects against nigrostriatal dopaminergic neurotoxicity in 1-methyl-4-phenyl-1,2,3,6- tetrahydropyridine- induced Parkinsonism in mice: role of oxidative stress. Eur J Neurosci 28(1): 41-50.
- Rojas P, Montes S, Serrano-Garci'a N, Rojas-Castaneda J (2009) Effect of EGb761 supplementation on the content of copper in mouse brain in an animal model of Parkinson's disease. Nutrition 25(4): 482-485.
- Von Gunten A, Schlaefke S, Uberla K (2016) Efficacy of Ginkgo biloba extract EGb 761® in dementia with behavioural and psychological symptoms: A systematic review. World J BiolPsychiatry 17(8): 622633.
- Luo Y, Smith JV, Paramasivam V, Burdick A, Curry KJ, et al. (2002) Inhibition of amyloid-beta aggregation and caspase-3 activation by the Ginkgo biloba extract EGb761. Proc Natl Acad Sc iUSA 99(19): 121971202.
- Maclennan KM, Darlington CL, Smith PF (2002) The CNS effects of Ginkgo biloba extracts and ginkgolide B. Prog Neurobiol 67(3): 235257.
- Tanaka K, Galduroz RFS, Gobbi LTB, Galduroz JCF (2013) Ginkgo Biloba Extract in an Animal Model of Parkinson's Disease: A Systematic Review. Curr Neuropharmacology 11(4): 430-435.
- Zhang XY, Zhou DF, Zhang PY, Wu GY, Su JM, et al. (2001a) A doubleblind, placebo-controlled trial of extract of Ginkgo biloba added to haloperidol in treatment-resistant patients with schizo-phrenia. J Clin Psychiatry 62(11): 878-883.
- Zhang WF, Tan YL, Zhang XY, Chan RC, Wu HR, et al. (2011) Extract of Ginkgo biloba treatment for tardive dyskinesia in schizophrenia: a randomized, double-blind, placebo-controlled trial. J Clin Psychiatry 72(5): 615-621.
- An HM, Tan YL, Shi J, Wang ZR, Li J, et al. (2013) Extract of Ginkgo biloba is equivalent to vitamin E in attenuating and preventing vacuous chewing movements in a rat model of tardive dyskinesia. Behav Pharmacol 24(7): 610-616.
- Shi J, Tan YL, Wang ZR, An HM, Li J, et al. (2016) Ginkgo biloba and vitamin E ameliorate haloperidol-induced vacuous chewingmovement and brain-derived neurotrophic factor expression in a rat tardive dyskinesia model. Pharmacol Biochem Behav 148: 53-58.
- Zhang XY, Zhang WF, Zhou DF, Chen DC, Xiu MH, et al. (2012) Brain- derived neurotrophic factor levels and its Val66Met gene polymorphism predict tardive dyskinesia treatment response to ginkgo biloba. Biol Psychiat 72(8): 700-706.
- Zhang XY, Zhou DF, Cao LY, Wu GY (2006) The effects of Ginkgo biloba extract added to haloperidol on peripheral T cell subsets in drug-free schizophrenia: a double-blind, placebo-controlled trial. Psychopharmacology 188(1): 12-17.
- Tamminga CA, Dale JM, Goodman L, Kaneda H, Kaneda N (1990) Neuroleptic-induced vacuous chewing movements as an animal model of tardive dyskinesia: a study in three rat strains. Psychopharmacology 102(4): 474-478.
- Naidu PS, Kulkarni SK (2001) Possible involvement of prostaglandins in haloperidol induced orofacial Dyskinesia in rats. Eur J Pharmacol 430(2-3): 295-298.
- Bishnoi M, Chopra K, Kulkarni SK (2008a) Protective effect of Curcumin, the active principle of turmeric (Curcuma longa) in haloperidol- induced orofacial dyskinesia and associated behavioural, biochemical and neurochemical changes in rat brain. Pharmacol Biochem Behav 88(4): 511-522.
- Bishnoi M, Chopra K, Kulkarni SK (2008b) Protective effect of L-type calcium channel blockers against haloperidol-induced orofacial dyskinesia: a behavioral, biochemical and neurochemical study. Neurochem Res 33(9): 1869-1880.
- Xu Y, Cui C, Pang C, Christen Y, Luo Y (2007) Restoration of impaired phosphorylation of cyclic AMP response element-binding protein (CREB) by EGb-761 and its constituents in Abeta-expressing neuroblastoma cells. Eur J Neurosci 26(10): 2931-2939.
- Lewin GR, Barde YA (2003) Physiology of the neurotrophins. Annu Rev Neurosci 19: 289-317.
- Muller WE, Heiser J, Leuner K (2012) Effects of the standardized Ginkgo biloba extract EGb 761® on neuroplasticity. Int Psychogeratr 24: S21-24.
- Zai CC, Tiwari AK, De Luca V, Muller DJ, Bulgin N, et al. (2009) Genetic study of BDNF, DRD3, and their interaction in tardive dyskinesia. Eur Neuropsychopharmacol 19: 317-328.
- Zhang XY, Zhou DF, Su JM, Zhang PY (2001b) The effect of extract of ginkgo biloba added to haloperidol on superoxide dismutase in inpatients with chronic schizophrenia. J Clin Psychopharmacol 21(1): 85-88.
- Kamyar M, Razavi B M, Hasani F V, Mehri S, Foroutanfar A, et al. (2016) Crocin prevents haloperidol-induced orofacial dyskinesia: possible an antioxidant mechanism. Iran J Basic Med Sci 19(10): 1070-1079.
- Peroza LR, Busanello A, Leal CQ, Ropke J, Boligon AA, et al. (2013) Bauhinia forficata prevents vacuous chewing movements induced by haloperidol in rats and has antioxidant potential in vitro. Neurochem Res 38(4): 789-796.
- Correll CU, Schenk EM (2008) Tardive dyskinesia and new antipsychot- ics. Curr Opin Psychiatry 21(2): 151-156.
- Mazza M, Capuano A, Bria P, Mazza S (2006) Ginkgo biloba and donepe-zil: A comparison in the treatment of Alzheimer's dementia in a random-ized placebo-controlled double-blind study. Eur J Neurol 13(9): 981-985.
- Yancheva S, Ihl R, Nikolova G, Panayotov P, Schlaefke S, et.al. (2009) Ginkgo biloba extract EGb 761(R), donepezil or both combined in the treatment of Alzheimer's disease with neuropsychiat-ric features: A randomised, double-blind, exploratory trial. Aging Ment Health 13(2): 183-190.
- Defeudis FV (2002) Bilobalide and neuroprotection. Pharmacol Res 46(6): 565-568.
- Tchantchou F, Lacor PN, Cao Z, Lao L, Hou Y, et.al. (2009) Stimulation of neurogenesis and synaptogenesis by bilobalide and quercetin via common final pathway in hippocampal neurons. J Alzheimers Dis 18(4): 787-798.
- Tchantchou F, Xu Y, Wu Y, Christen Y, Luo Y (2007) EGb-761 enhances adult hippocampal neurogenesis and phosphorylation of CREB in trans-genic mouse model of Alzheimer's disease. FASEB J 21(10): 2400-2408.
- An HM, Tan YL, Shi J, Wang ZR, Li J, et al. (2016) Beneficial effects of EGb761 and vitamin E on haloperidol-induced vacuous chewing movements in rats: Possible involvement of S100B mechanisms. Behav Brain Res 297: 124-130.
- Burt DR, Creese I, Snyder SH (1977) Antischizophrenic drugs: chronic treatment elevates dopamine receptor binding in brain. Science 196: 326-328.
- Elkashef AM, Wyatt RJ (1999) Tardive dyskinesia: possible involvement of free radicals and treatment with vitamin E. Schizophr Bull 25(4): 731-740.
- Le BP, Katz MM, Berman N, Itil TM, Freedman AM, et al. (1997) A Placebo-Controlled, Double-blind, Randomized Trial of an Extract of Ginkgo Biloba for Dementia. JAMA 278(16): 1327-1332.
- Rubinstein M, Muschietti JP, Gershanik O, Flawia MM, Stefano FJ (1990) Adaptive mechanisms of striatal D1 and D2 dopamine receptors in response to a prolonged reserpine treatment in mice. J Pharmacol Exp Ther 252(2): 810-816.






























