Abstract
The intraspecific variation of Clarias gariepinus across four geographically distinct populations in Sudan was evaluated using meristic counts and morphometric measurements. Fish samples were obtained from Sinnar Dam area, Blue Nile (Site 1), Khor Abu Gassaba, White Nile (Site 2), Tordat Al Rahad an inland lake, mid Kordofan (Site 3) and Khashm El Girba, Atbara River (Site 4). Analysis of variance revealed highly significant differences (p<0.01) among populations in four of five meristic traits and 26 of 27 morphometric measurements. Post hoc comparisons (LSD test) showed that the Site 1 population exhibited the highest mean values (p<0.05) for all meristic and morphometric characters. In contrast, Site 3 population showed the lowest means in three of five meristic counts, while Site 2 individuals recorded the lowest morphometric means, except for orbital frontal width. Twelve morphometric variables and several meristic traits contributed substantially to total phenotypic variation, indicating their diagnostic utility in population differentiation. Head measurements were strongly and positively correlated with head length. Most morphometric traits exhibited high positive correlations (r>0.65), whereas others showed moderate to weak (r<0.40) associations. Standard length and body weight demonstrated strong positive correlations with most morphometric variables. The length–weight relationship was highly significant (p<0.0001), with correlation coefficients ranging from 0.766 t0.979. Principal Component Analysis grouped the populations into two clusters, with partial segregation of Site 1 population, suggesting relatively greater intra-population variability. The findings indicate a close phylogenetic affinity among the four populations, likely derived from a common clade, while identifying key morphological traits relevant for taxonomic discrimination and selective breeding programmes.
Keywords:Clarias gariepinus ; Meristics; Morphometrics; Sites; Sudan
Introduction
The African catfish, Clarias gariepinus, is widely distributed across Africa, Asia, and Southeast Asia [1] and is particularly common in the Nile and its tributaries [2,3]. In Sudan, studies on freshwater fish taxonomy initially focused on descriptive morphological characteristics [4,5]. This approach later expanded to include meristic counts and morphometric measurements [6-8]. More recently, molecular techniques have been applied to refine the classification and identification of freshwater fish species [7,9,10], although traditional morphometric and meristic methods remain central tools in fish taxonomic studies.
Phenotypic diversity in C. gariepinus were investigated in Nigeria [11,12] and in Turkey [13,14], revealing significant variation among populations. These differences have been attributed to genetic variability, environmental influences, or both.
The freshwater fish fauna in Sudan is similarly diverse, highlighting the importance of assessing meristic and morphometric traits in natural populations to determine patterns of homogeneity or heterogeneity among populations.
The objective of this work in C. gariepinus is to compare homogeneity and heterogeneity of meristic counts and morphometric characters three populations from three tributaries from the Nile and one from an inland lake.
Material and Methods
Source of specimens and identification
One hundred twenty-three specimens of C. gariepinus fish were obtained from 4 sites. These were Site 1 (Sinnar Dam area, Blue Nile, n=30); Site 2 (Khor Abu Gassaba, White Nile, n=30); Site 3 Tordat Al-Rahad, N. Kordofan State, n=33) and Site 4 (Khashm Al-Girba, Atbara River, n=30). Fish identification followed Abu Gideiri [4] and Bailey [5].

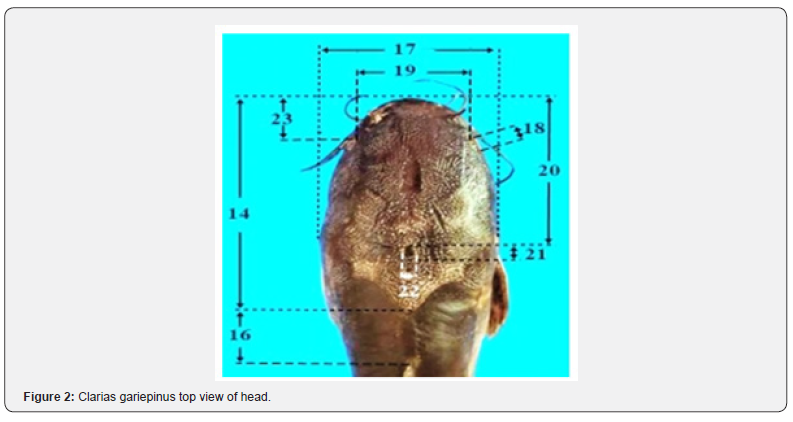
Meristic counts and morphometric measurements
Five meristic counts (The number of rays in the: dorsal fin (DFR); anal fin (AFR); pectoral fin (PFR); pelvic fin (PEFR) and caudal fin (CFR). Twenty-six morphometric measurements (Figures 1 & 2, Table 1) were measured following Ebraheem [7].
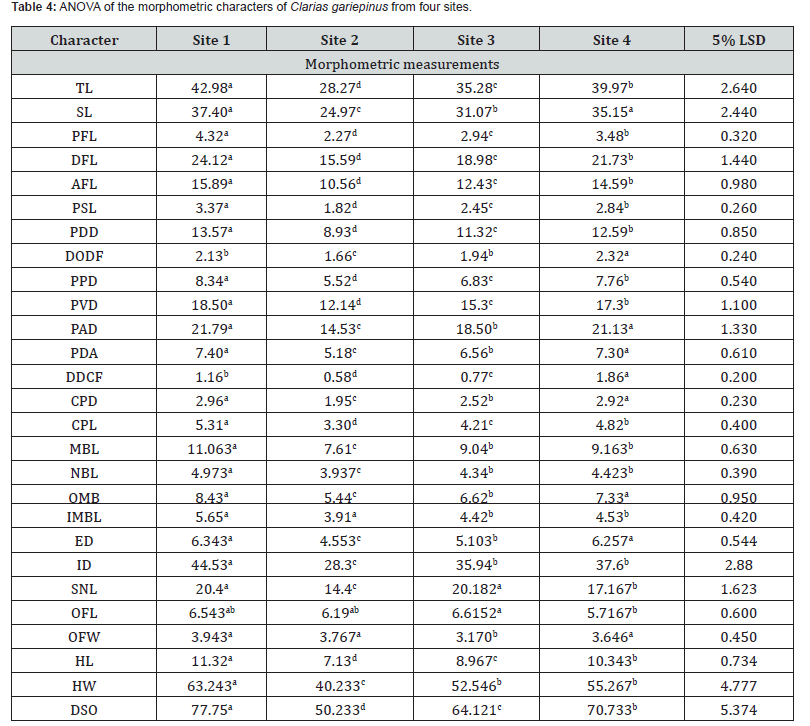
*Significant difference (p<0.05), **highly significance (p<0.01), different superscript in a raw indicates significant differences.
Different superscripts in a raw are statistically different.
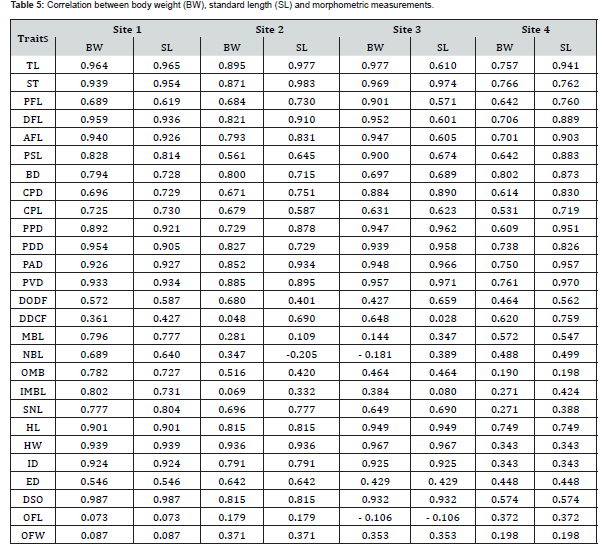
r>0.65 highly, r>0.40 to r<0.65 moderately and r<0.40 weakly correlated.
Morphometric measurements were taken from the left side of the 123 C. gariepinus fish, using a measuring board, a tape and a Vernier calliper to the nearest 0.01 cm accuracy.
Length-Weight Relationship
The Body weight (BW) was recorded in grams using a Docebel Braun balance. The relationship between Standard Length (SL) and Body Weight (BW) was obtained by linear regression analysis following the equation: SL = a + b BW ; Where: a = the intercept and b = growth mode.
Statistical Analysis
Data were analyzed using analysis of variance (ANOVA) in Microsoft Excel after normality testing. Means were compared by the Least Significant Difference test (p<0.05). Correlation coefficients were estimated for traits. Regression of BW and SL was performed in GraphPad QuickCalcs.
Results
Meristic Counts
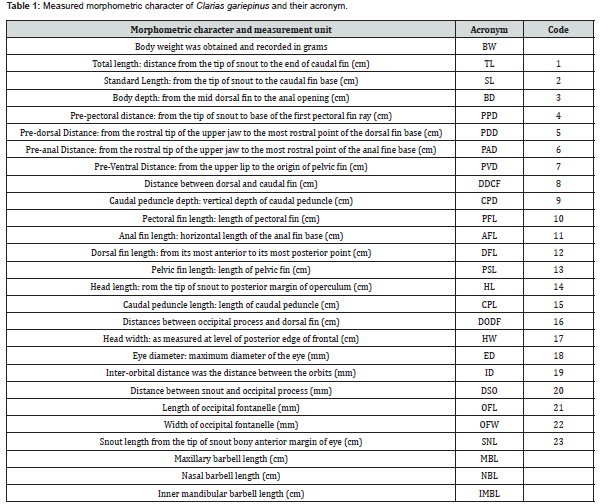
ANOVA results indicated that the meristic counts DFR, AFR, PFR and CFR four of (Table 2) were highly significantly different (p<0.01) among the populations of the 4 sites. The PEFR was just significantly different (p<0.05) among the 4 populations. The highest means of the meristic counts were found among Site 1 population followed by Site 2 samples with no significant difference (p>0.05) between them in AFR, PEFR, CFR and PEFR. The DFR means in Site 1 was significantly different (p<0.05) from the other three sites.
Morphometric measurements
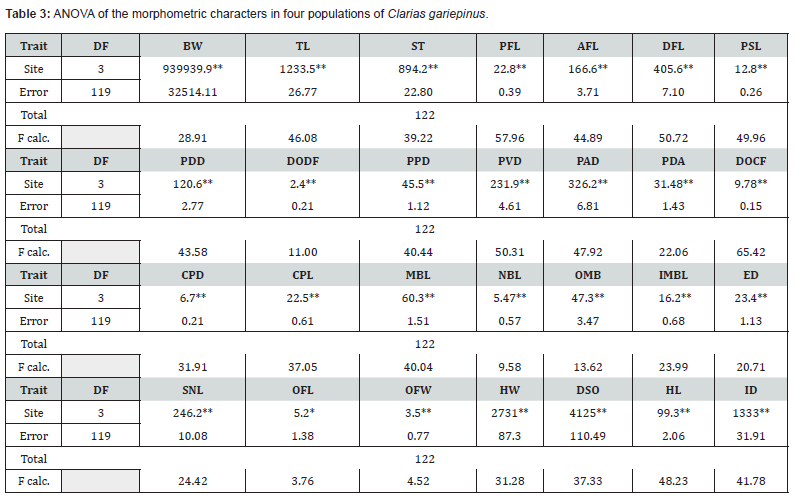
The ANOVA (Table 3) shows that:
a) The Sites significantly affected all measured traits. Most
traits are highly significant at p< 0.01, while OFL is significant at
p<0.05.
b) The high F-calculated values show strong betweenpopulation
variability relative to within-population variability.
This indicates morphological heterogeneity among the four
populations.
c) ANOVA of TL, SL and BW suggest significant differential
growth performance among sites.
d) All fin measurements (PFL, AFL, DFL, PSL) show highly
significant differences (p<0.01) suggesting adaptation to different
water regimes.
e) All PDD, DODF, PPD, PVD, PAD, PDA, DOC traits show
significant site effects related to body proportions.
There is strong statistical evidence of morphometric variation among the four populations of C. gariepinus. High F-values in structural traits imply possible adaptive divergence rather than random variation.
The morphometric characters measured (Table 3), indicated highly significant differences (p<0.01) in all 26 characters among the fish populations in the 4 sites. OFL was significant (p< 0.05) among the 4 populations. Site 1 C. gariepinus population scored the highest mean values with the exception of two characters, DODF and DDCF.
Table 4 shows that:
a) The 5% LSD suggests significant differences (p<0.05)
and morphological heterogeneity among the four populations.
b) Site 1 consistently recorded the highest mean values for
most morphometric characters and all fin-related parameters,
while Site 2 had the lowest values across almost all characters.
The environmental suitability for the populations followed the
order: Site 1> Site 4> Site 3> Site 2.
c) TL, SL and HL were significantly highest at Site 1.
d) Site 1 and Site 4 populations exhibited more robust body
structures, whilst Site 2 individuals were comparatively slender
and smaller.
e) Cranial and sensory structures ED, ID, SNL, OFL, OFW
were significantly larger in Site 1.
f) DODF, DDCF were highest in Site 4 suggesting differences
in microhabitat and/or water flow.
Correlations between BW, SL and morphometric measurements
The correlation coefficients (Table 5) show that:
a) Strong positive relationships (r>0.65) between BW and
most morphometric measurements in all sites. The strength of
association varies by trait and location.
b) Site 4 tends to display comparatively lower (r-value)
for many traits, though the relationships remain positive and
moderate to strong for key measurements such as BD. head HL,
and PVD.
c) DODF, DDCF, MBL, NBL, OFL, and OFW show low
correlations (r<0.40), and in some cases negative values (e.g., NBL
and OFL at Site 3). These weak associations suggest that these
measurements contribute less to variations in body weight.
d) TL, PDD, PAD), PVD, AFL and DFL showed very high
correlations (r ≥ 0.90) with SL across most sites. This proportional
growth reflects coordinated somatic development.
e) BD, CPD, CPL, PPD, SNL and PSL exhibited moderate
to strong positive correlations (r=0.60–0.89). Suggesting sizedependent
flexible growth.
f) Measurements such as DODF, DDCF, MBL, NBL, OMB and
IMBL showed weak, inconsistent, or even negative correlations
(e.g., NBL at Site 3), r<0.60).
g) The regression of SL and BW of C. gariepinus from the
four populations were:
Site 1; SL=48.15BW-1228, r=0.965; Site 2; SL=19.44BW-321,
r=0.871.
Site 3; SL=32.39BW-561.1, r=0.979.; Site 4; SL=22.31BW-347.1,
r=0.766 site 4.
The relation is very highly significantly correlated (p<0.0001).
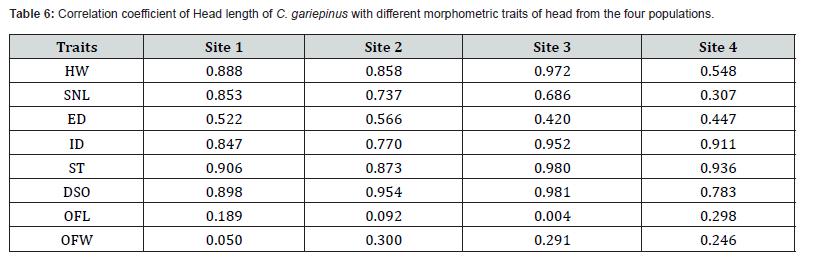
The majority of the measurements of the head, in the 4 populations, were highly and positively associated with head length (Table 6). In sites 3 and 4 the correlation is moderate in ED. OFL from all sites and OFW from site 1, showed weak positive correlation.
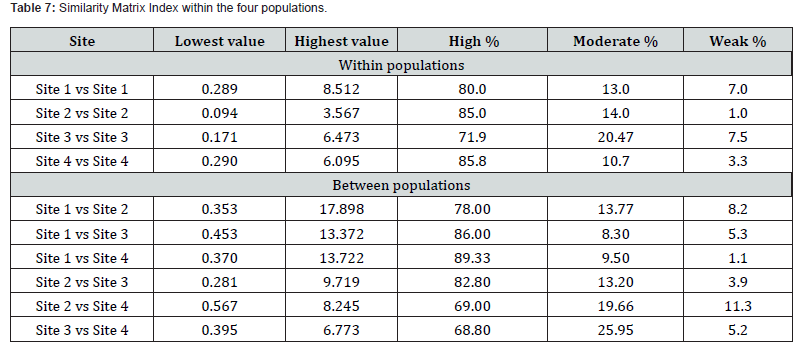
Similarity Matrix Index (SMI)
The SMI was used to quantify the structural resemblance within and between the four populations (Table 7). The SMI within-population displayed high values (71.9–85.8%); with highest homogeneity in Site 4 (85.8%) and Site 2 (85.0%). Site 3 shows comparatively lower SMI of 71.9% indicating heterogeneity. The SMI between-population comparisons show high readings (68.8–89.33%). The strongest resemblance occurs between Site 1 and Site 4 (89.33%), suggesting similar environmental conditions. In contrast, Site 3 vs Site 4 (68.8%) and Site 2 vs Site 4 (69.0%) display a relatively lower similarity percentages and higher moderate/weak components, implying greater compositional divergence.
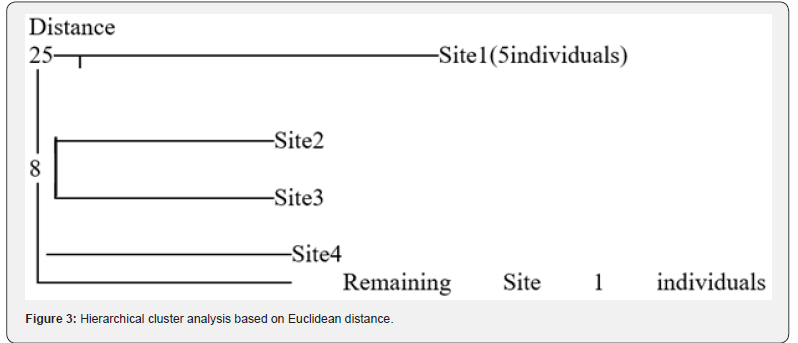
Principal Component Analysis
PCA of morphometric traits showed 80.7% accuracy across
populations, ranging from 71% at Site 3 to 85.8% at Site 4, with
variation at Site 1 and least at Site 2. The PCA (Figure 3) revealed
that:
a) Sites, with lower values along the Y-axis indicate higher
similarity.
b) At a distance of 25, Site 1 comprised five individuals
forming a distinct cluster, with four individuals separating from
the fifth, indicating intra-site variation.
c) At a distance of 8, Sites 2 and 3 clustered together,
reflecting their high similarity.
d) Site 4, joining at a distance slightly below 8, exhibited
moderate similarity to Sites 2 and 3.
e) The remaining individuals from Site 1 clustered
separately, highlighting their marked dissimilarity from the other
sites, indicating internal variation within Site 1.
Discussion
Clarias gariepinus, an ecologically significant and economically valuable freshwater fish, is widely distributed throughout Africa and extending into parts of Asia and Southeast Asia [1]. It is abundant in the Nile [2,3]. Its geographic range, high tolerance, and importance in capture fisheries and aquaculture have made it a focal species in studies of taxonomy, population structure, and phenotypic plasticity.
In Sudan, descriptions made by [4,5] provided base knowledge of freshwater fishes, but they were often limited in resolving intraspecific variation. Subsequent investigations incorporated quantitative meristic counts and morphometric measurements to enhance taxonomy and population comparisons [6,7,9]. These methods allowed researchers to statistically evaluate differences in meristic and morphometric traits. More recently, molecular techniques have been introduced to complement morphological analyses and refine species identification and phylogenetic relationships [7,9,10].
The ANOVA demonstrated highly significantly (p<0.01), spatial variation in meristic traits among the four studied populations in DFR, AFR, PFR, and CFR, while PEFR showed a lower but still significant difference (p<0.05). Site 1 exhibited the highest mean meristic counts, followed by Site 2, with no significant differences (p>0.05) in AFR, PEFR, and CFR between them. However, DFR in Site 1 differed significantly (p<0.05) from the other sites, indicating localized divergence. Evidence on usefulness of meristic counts variation in morphological characterization, and population differentiation in C. gariepinus align with previous studies [15-21] and supporting the taxonomic relevance of meristic traits. Using PCA, Umoh et al. [22] revealed that hybrids C. gariepinus from selected fish farms differed meristically in pectoral fin-ray, caudal fin-ray, anal fin-ray, dorsal fin-ray and pelvic fin-ray.
The present investigation indicated that the fish population in Site 1 recorded the highest mean value in all morphometric measurements (26 out of 27), and Site 2 the lowest means value (24 out of 28 traits, 88.9%). DDCF, OFL and OFW characters showed no correlation with SL and BW. The means, of TL, PFL, AFL, DFL, PSL, PDD, PPD, PVD, DDCF, HL and DSO as well as BW are most likely to be the characters that greatly contributed to the total variation in the four populations. Studies on C. gariepinus from the African freshwater bodies showed significant morphometric variations among populations from different environments [16,19,11,21,23]. Similarly, studies conducted in Turkey by Ihsan [13], Turan et al. [14] reported measurable differences in morphometric traits among geographically disconnected populations. These variations are attributable to environmental influences such as water flow [24], food availability [25], and habitat [26,27]. To this genetic divergence may accumulate due to geographic isolation or restricted gene flow [28] and consequently impact morphometrics.
The regression coefficient of the SL and the BW relationship of C. gariepinus from Site 1, Site 2, Site 3 and Site 4 populations indicates that the incremental change in (b) of the body weight is a consequence of change in SL. The highest b slope was among Site 1 (48.2) and the lowest (19.4) was recorded from Site 2. The result suggests that the SL has a high positive correlation with the BW and that the two variables can be used as indicators for selecting a high body weight candidate for selective breeding. Anderson and Gutreuter (1983) stated that it is of paramount importance to determine this relation to establish the production and the biomass estimations of the species. Site 1 represented the highest mean BW value, and it has the highest and positive SL relationships. This finding indicated that the heavier the fish are the better its environmental conditions as reported by Idris and Mahmoud [6] for Labeo niloticus. The present study showed a positive length-weight relationship in the four populations, between the individuals of the four populations and between the sexes among the four populations. It seems that the conditions existing in the freshwater habitat, in the Sudan, are in favour for the propagation and the growth of this species. Another important morphological character is the measurements of the head. Most measurements of the head in the four populations are highly and positively associated with the head length are important in the characterization and identification of the species. This supports the findings of Turan et al. [14], in Turkey, in six populations of C. gariepinus. However, the ED reveals moderate correlation in the four population. On the other hand, OFW and OFL, in the four populations and the SNL in Site 4 population, showed weak positive correlation. It is concluded that the measurements of the head are important in the characterization of the fish.
This study demonstrated that PCA of morphometric traits in four C. gariepinus populations yielded a correct classification rate (80.7%), indicating significant phenotypic structuring among sampling sites. Multivariate approaches such as PCA have been widely applied in Clarias systematics and stock discrimination to detect subtle morphological divergence attributable to environmental and/or genetic factors. Studies of Clarias species across West African basins emphasized the importance of morphometric differentiation and attributed this to geographic variation.
The highest correct assignment observed in Site 4 (85.8%) suggests greater morphological distinctiveness of this population, possibly reflecting ecological specialism. Morphological divergence in C. gariepinus populations has been associated with habitat heterogeneity, and local adaptation [29]. Conversely, the lowest classification rate in Site 3 (71%) may indicate greater overlap in morphometric traits with adjacent populations, suggesting phenotypic plasticity and/or environmental pressures shaping body form. Such overlap has been reported in populations of C. gariepinus from Nigeria [11].
The minimal variation detected in Site 2 suggests relative genetic homogeneity or environmental stability. This reduced morphometric variability within populations of Clarias is in line with Fagbuaro et al. [30] and is likely to be linked to ecological effects. In contrast, Site 1 exhibited the greatest morphological variability indicating mixed stock origin and/or ecological heterogeneity. Turan et al. [14] related within-population to fluctuating environmental conditions promoting phenotypic flexibility.
LSD analyses revealed strong affinities among Sites 1, 3, and 4, suggesting comparable ecological regimes as suggested by Teugels (1992). However, the comparatively lower similarity between Abu Gassba and Site 4 (69%), as well as between Site 3 and Site 4 (68.8%), indicates measurable divergence. The causes of such differentiation in Clarias populations are likely related to hydrological isolation, anthropogenic influences, or differential.
References
- Teugels GG, Adriaens D (2003) Taxonomy and phylogeny of Clariidae: An overview. In: Arratia G, Kapoor BG, Chardon M, Diogo R, Catfishes, (eds.), Publisher: Science Publishers, Inc. pp. 465-487.
- Neumann D, Obermaier H, Morti Z (2016) Annotated checklist for fishes of the Main Nile Basin in the Sudan and Egypt based on recent specimen record (2006-2015). Cybium 40: 287-317.
- Mahmoud ZN, Hagar EA, Abdalla MYM (2024) Fish diversity in the Nile system and ephemeral water bodies in Sudan: Records and a review. International Journal of Fisheries and Aquatic Studies 12(4): 1-8.
- Abu Gideiri YB (1984) Fishes of the Sudan. Khartoum University Press, pp. 166.
- Bailey RG (1994) A guide to the fishes of the river Nile in the Republic of Sudan. Jorn Nat Hist 28: 937-970.
- Idris MA, Mahamoud Z (2001) Studies on Morphometric Measurements and Meristic Counts on Labeo niloticus. Sudan Journal of Natural Science 1: 90-108.
- Ebraheem HA (2012) Morphometrics, Meristics and Molecular Characterization of Oreochromis niloticus, Sarotherodon galilaeus and Tilapia zillii (Cichlidae) from Kosti, Khashm El Girba and Al Sabloga. M. Sc. Thesis, Department of Zoology, Faculty of Science, University of Khartoum, Sudan.
- Abd Alrasoul EAA (2023) Meristic Morphological and Molecular identification of Electric. Catfish (Malapterurus electricus) from Sudan, with a description of a Subspecies. M. Sc Thesis, Department of Zoology, Faculty of Science, University of Khartoum.
- Mohammed ATM, Mahmoud ZN, Abushama HA (2019) Morphometric Measurements, Meristic Counts, and Molecular Identification of Alestes Dentex, Alestes Baremoze, Brycinus Nurse, and Brycinus macrolepidotus from the River Nile at Kreima. The Open Biology Journal 7: 25-38.
- Omer OM, Abdalla HA, Mahmoud ZN (2020) Genetic Diversity of Two Tilapia Species (Oreochromis niloticus and Sarthrodon galilaeus) Using Random Amplified Polymorphic DNA. The Open Biology Journal 7: 3-9.
- Ola Oladimeji FA, Oso JA, Oladimeji TE, Idowu EO, Adeleke K, et al. (2017) Phenotypic diversities of four populations of Clarias gariepinus (Siluriformes, Clariidae) obtained from Ogun and Ondo State waterbodies in South-Western Nigeria. Vestnik Zoologii 51(4): 285-294.
- Aawodi JC, Kawo AH, Abdullahi BA, Yola IA (2025) Comparative Assessment of Morphometric and Growth Characteristics of African catfish (Clarias gariepinus) in Cultured and Wild Habitats in Zaria, Nigeria. Fudma Journal of Sciences 9: 28-33.
- Ihsan A (2005) Morphometric comparisons of African catfish, Clarias gariepinus, population in Turkey. Folia Zoology 54(1-2): 165-172.
- Turan C, Yalçin Ş, Turan F, Akyurt I (2005) Morphometric comparisons of African catfish, Clarias gariepinus, populations in Turkey. Folia Zool 54(1-2): 165-172.
- Teugels GG (1982) Preliminary results of a morphological study of five African species of the subgenus Clarias (Clarias) (Pisces; Clariidae). Journal of Natural History 16(3): 439-464.
- Rognon X, Teugels GG, Guyomard R, Galbusera P, Adriamanga M, et al. (1998) Morphometric and allozyme variation in the African catfishes Clarias gariepinus and C. anguillaris. Journal of Fish Biology 53(1): 192-207.
- Naeem M, Salam A (2005) Morphometric study of fresh water bighead Carp Aristichthys nobilis from Pakistan in relation to the body size. Pakistan Journ Biol Sci 8(5): 759-762.
- Eyo JE (2008) Differences in meristic counts of the genus Clarias (Pisces: Clariidae) in Anambra River, Nigeria. Animal Research International 1(1): 31-35.
- Solomon SG, Okomoda VT, Ogbenyikwu AI (2015) Intraspecific morphological variation between cultured and wild Clarias gariepinus (Burchell) (Clariidae, Siluriformes). Fisheries and Aquatic Life 23(1): 53-61.
- Ola Oladimeji FA, Awodiran MO, Fagbuaro O, Akomolafe AO (2016) Morphological characterization of wild and cultured Clarias gariepinus (Burchell 1822) using principal component and cluster analyses. Notulae Scientia Biologicae 8(4): 428–436.
- Suleiman IO, Kabir M, Bolorunduro PI, Akinola OM (2019) Morphological characteristics and meristic count of Clarias gariepinus and Heterobranchus longifilis strains from Kano, Nigeria. Nig J Anim Sci Tech 2(1): 45 -53.
- Umoh I, Nlewadim A, Obuba L, Oguntade O (2015) Morphometric and Meristic Characteristics of Hybrid Catfish from Selected Fish Farms in Southern Nigeria. International Journal of Computer Trends and Technology 5(1): 14-19.
- Popoola OM, Olagunju OO (2021) Morphological differentiation in African catfish (Clarias gariepinus) from three different populations in Nigeria. Nigerian Journal of Technology Research 16(1): 13-20.
- Kelley JL, Davies PM, Collin SP, Grierson PF (2017) Morphological plasticity in a native freshwater fish from semiarid Australia in response to variable water flows. Ecol Evol 7(16): 6595-6605.
- Wimberger PH (1992) Plasticity of fish body shape: the effects of diet, development, family and age in two species of Geophagus (Pisces: Cichlidae). Biological Journal of the Linnean Society 45(3): 197-218.
- Brinsmead J, Fox MG (2005) Morphological variation between lake- and stream-dwelling rock bass and pumpkinseed populations. Journal of Fish Biology 61(6): 1619-1638.
- Al Jubaili RH, Taher MM, Al Mudhaffar RA (2025) Comparison of Morphometric Characteristics Between Cultivated and Wild Common Carp Cyprinus carpio Populations. Egyptian Journal of Aquatic Biology & Fisheries 29(2): 1281-1293.
- Pavlova A, Beheregaray LB, Coleman R, Gilligan D, Harrison KA, et al. (2017) Severe consequences of habitat fragmentation on genetic diversity of an endangered Australian freshwater fish: A call for assisted gene flow. Evol Appl 10(6): 531-550.
- Teugels GG (1986) A Systematic Revision of the African Species of the Genus Clarias (Pisces; Clariidae). Annales Musee Royal de l’Afrique Centrale 247: 1-199.
- Fagbuaro O, Oso J, Olurotimi M, Akinyemi O (2015) Morphometric and Meristic Characteristics of Clarias gariepinus from Controlled and Uncontrolled Population from Southwestern Nigeria. Journal of Agriculture and Ecology Research International 2(1): 39-45.






























