Abstract
The ayu, Plecoglossus altivelis, migrates between streams and the ocean across the Japanese Archipelago. In their southern distributional range, larvae often favor estuarine waters over marine waters as their initial habitats. The egg size of ayu increases with decreasing latitude. The aim of the present study was to determine the significance of egg size variation in ayu. The survival of newly hatched larvae was observed in freshwater, brackish water, and seawater using 12 clutches of various egg sizes. A total of 100 hatchlings from each sibling group were left unfed at 22°C. Positive correlations between egg size and survival were detected in larvae placed in freshwater and brackish water, whereas those placed in seawater were short-lived regardless of egg size. In conclusion, larger eggs are more adaptive for enhancing early survival at lower latitudes with reduced ocean productivity, synergizing with selecting estuarine waters as an initial habitat.
Keywords:Amphidromy; Egg size; Latitudinal cline; Brackish water
Introduction
Natural selection often favors aquatic organisms living in harsh environments that produce as many offspring as possible in order to maximize their chances of survival with limited energy reserves [1]. Although smaller eggs appear to be more suitable for achieving higher fecundity, egg size is variable rather than fixed to a constant quantity for any given species. This is because the effects of egg size on early survival can be mediated by the environmental conditions experienced by the offspring [2,3]. Theoretical studies predict that optimal egg size is determined by a trade-off between size and number of offspring in order to maximize the fitness of the female parent [4,5]. Thus, egg size varies among populations according to the local habitat traits responsible for the relationship between egg size and offspring survival [6,7]. For example, coho salmon, Oncorhynchus kisutch, migrate to the ocean after at least one year of rearing in freshwater and are believed to be the least dependent on estuaries among the juveniles of Pacific salmon species [8]. This anadromous fish species exhibits geographical variation in egg size, producing larger eggs at lower latitudes along the West Coast of North America, from Baja California to Alaska. The conversion efficiency of the energy stored in the yolk into larvae may decrease in warmer waters at lower latitudes [9]. Therefore, the latitudinal cline of coho salmon can simply be explained by the geographical variation in water temperature during its residence period in riverine waters.
Ayu Plecoglossus altivelis, a finned fish, is widely distributed across the Japanese Archipelago and along the Continental East Coast, migrating amphidromously between freshwater streams and the oceans [10,11]. After hatching in the riverbeds of rapids, the larvae drift downstream immediately to spend the winter in coastal areas. Ayu egg size varies geographically, with larger eggs observed at lower latitudes [12]. Newly hatched larvae that reach the ocean are inevitably forced to expend energy stored in the yolk to maintain homeostasis under osmotic pressure [13]. In the southern distributional area of the species, larvae often appear in the river mouth using estuarine waters as their initial habitat, in which the osmotic pressure is lower than that in marine waters (Takahashi et al. [14] for the Shimanto River in the Kochi Prefecture; Murase and Iguchi [15] for the Yakugachi River on Amami-Oshima Island; Tran et al. [16] for the Kalong and Tien Yen Rivers in Vietnam). The cost of early osmoregulation increas es as water temperatures rise; however, at comparable water temperatures, the energy expenditure for osmoregulation in marine waters exceeds that in estuarine waters [17]. Therefore, unlike in coho salmon, where water temperature is regarded as the only factor affecting egg size, in ayu, both water temperature and osmotic pressure are potential factors influencing egg size.
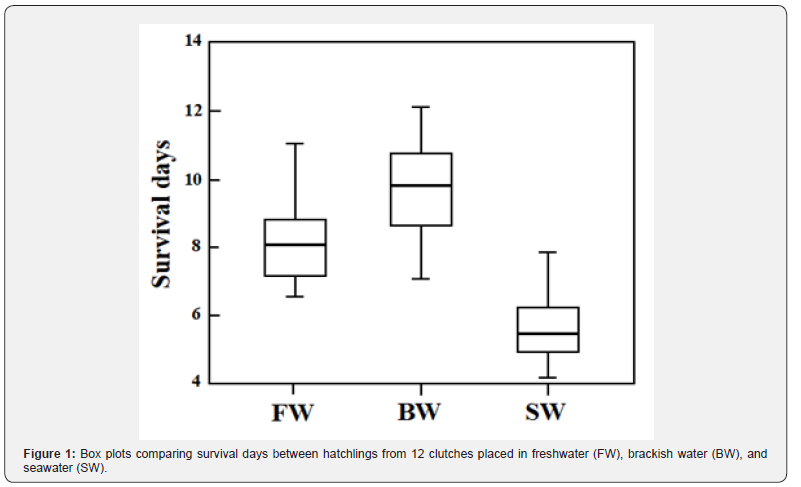
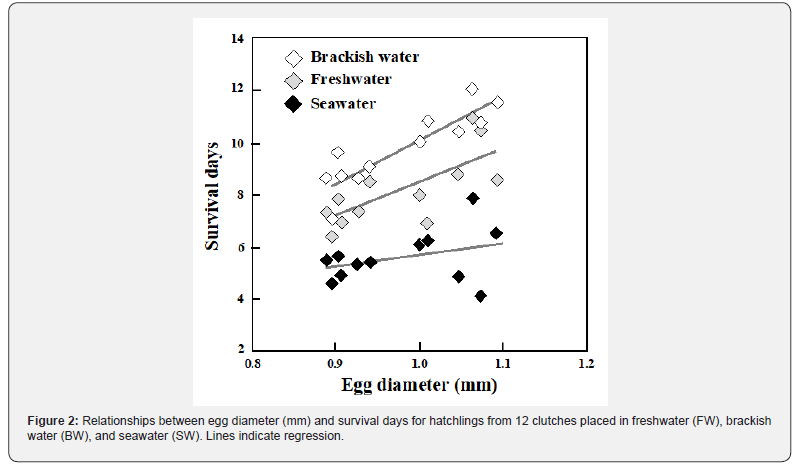
The present study aimed to estimate the adaptive significance of egg size variation in ayu and explain why larger eggs are favored at lower latitudes. The slow death of larvae after starvation was observed based on the endogenous energy reserves allocated for metabolic consumption and synthesis under different osmotic conditions. Osmoregulation is costly and requires energy from the yolk during the hatchling phase, which enhances the risk of starvation in marine environments with elevated osmotic stress. Simultaneously, it should be noted that osmoregulation capacity is not influenced by egg size, meaning that even larvae derived from larger eggs do not necessarily reduce the expenditure required for osmoregulation in seawater. On the other hand, in brackish water lacking significant osmotic pressure, early survival during the period of starvation may depend on egg size. To ascertain the distinctive characteristics of brackish water devoid of osmotic pressure, experiments were conducted involving hatchlings from clutches of varying egg sizes under unfed conditions. Subsequently, their survival was assessed in diverse aquatic environments, including seawater, brackish water, and freshwater.
Materials and Methods
Collection of larvae
Amphidromous ayu, approximately 6 months old, were collected from the lower course of the Shinano River (37.8° N; 136.3° E) that flows into the Sea of Japan in May 2005. They were reared in outdoor ponds (1 × 5 × 0.5 m) of the National Research Institute of Fisheries Science (36.2°N, 138.2°E) and fed assorted feed until maturation. In October, when the spawning season approached, pebbles were placed on the bottom of the upstream side of each pond, and a water flow of 100 cm/s was applied to simulate the spawning bed [18]. The spawning schedules of the females were determined by gently pressing their abdomens every few days to detect ovulation. The diameters of the ovulated eggs were measured using a profile projector (V-12B, Nikon Instech, Japan) with a resolution of 0.001 mm. For each ovulated female, egg size was calculated as the average diameter of 100 eggs chosen randomly from a clutch. Eggs within a clutch were fertilized with milt from a single live male and incubated at 20°C in flowing and aerated freshwater with a natural photoperiod. These embryos required more than 10 d to hatch.
Experimental procedure
The survival of newly hatched larvae was observed in freshwater (FW), brackish water (BW, 33% artificial seawater), and seawater [SW, 100% artificial seawater, see Iguchi and Takeshima [19]] using 12 clutches. A total of 100 hatchlings from each sibling group were left unfed in 2-L receptacles at 22°C. The survival status of the larvae was assessed based on their clarity; larvae that lost their transparency and turned white were considered dead. The dead larvae were recorded on a daily basis until the final larva perished. The average survival period was used as an index of the osmoregulation capacity of FW, BW, and SW.
Statistical analyses were performed using SPSS software version 25 (IBM Corporation, Tokyo, Japan).
Results
Newly hatched larvae placed in FW, BW, and SW demonstrated different survival processes, reflecting their osmoregulatory capacities (Figure 1: ANOVA, F2,33 = 32.017, P < 0.001), with those in BW living the longest, followed by those in FW and those in SW living the shortest (Games–Howell test, BW versus FW, P = 0.028; FW versus SW, P < 0.001), respectively. Survival spans, which decreased in the order of BW, FW, and SW, were also observed among siblings from the same clutch (repeated-measures ANOVA, F2,21 = 71.245, P < 0.001). Egg size varied between clutches, ranging from 0.890 mm to 1.093 mm in diameter. Positive correlations between egg size and survival days were detected in larvae placed in BW and FW, whereas those placed in SW were short-lived regardless of egg size (Figure 2: BW, r = 0.881, N = 12, P < 0.001; FW, r = 0.736, N = 12, P = 0.006; SW, r = 0.334, N = 12, P = 0.273). No interaction was found between BW and FW on the regression lines for egg size and survival days (BW, y = 16.38x-6.26; FW, y = 13.29x -4.84 where x and y are survival days and egg diameter, respectively; ANCOVA, F1,20 = 0.221, P = 0.644).
Discussion
Estuarine waters, in which BW reduces the osmotic cost to a negligible level, are the most energy-efficient habitats for ayu larvae, whereas SW requires more energy than FW for osmoregulation. Rivers and streams with running water do not meet the requirements for the initial habitat of larvae because of their inability to acquire sustenance by swimming (Tsukamoto et al., 1975). Therefore, drifting larvae that swiftly pass through riverine areas are more favorable. Some larvae that have reached the river mouth are allowed to remain in estuarine waters of varying sizes, depending on the river. Those that continue to disperse are inevitably exposed to salinity outside the estuary. In BW, larvae free from osmotic pressure are able to survive for a period that corresponds linearly to egg size, even when left unfed. However, those involved in SW, which forces them to pay high costs for osmoregulation, are not guaranteed survival rates that are proportionate to the extra investment in each individual offspring. This is most likely because the performance of ionocytes, which are responsible for osmoregulation during the larval stage [20] varies among clutches, irrespective of egg size.
In terms of the relative availability of aquatic food resources, oceans are more productive than inland waters from mid-to higher latitudes, whereas inland productivity exceeds that of the ocean at lower latitudes [21]. Latitudinal variations in relative productivity in the ocean lead to intraspecific variations in food availability among marine organisms. In the spectrum of the r-K strategy [1], residents at higher latitudes are favored for rapid reproduction, and a trait of higher fecundity with smaller eggs is selected to maximize the intrinsic rate of natural increase. In contrast, at lower latitudes, where intraspecific competition for food is intense, offspring derived from larger eggs can acquire a survival advantage in exchange for reduced fecundity due to their enhanced competitive ability. Latitudinal egg size variation in ayu can be explained by the r-K strategy. Furthermore, at lower latitudes, higher water temperatures cause larvae to expend more energy on osmoregulation [17]. Where ayu larvae can use estuarine waters as an alternative habitat by minimizing early dispersal, they can conserve the energy required for osmoregulation and run up the natal stream [22]. Larger larval sizes derived from larger eggs are essential to minimize early dispersal and remain in estuarine waters. In conclusion, larger eggs are more adaptive for promoting early survival at lower latitudes with lower productivity, synergizing with selecting estuarine waters as an initial habitat [23].
Acknowledgment
I am grateful to Taeko Hazama, Mariko Yokoyama, and Yukari Katagiri for their assistance with the experiments. The experimental procedures and housing conditions used in this study were in accordance with the ethical guidelines of the Ichthyological Society of Japan.
References
- Pianka ER (1970) On r- and K-selection. Am Nat 104(940): 592-597.
- Parker GA, Begon M (1986) Optimal egg size and clutch size: effects of environment and maternal phenotype. Am Nat 128: 573-592.
- Allen RM, Marshall D (2014) Egg size effects across multiple life-history stages in the marine annelid Hydroides diramphus. PLoS ONE e102253.
- Smith CC, Fretwell SD (1974) The optimal balance between size and number of offspring. Am Nat 108(962): 499-506.
- Kishi Y (1979) A graphical model of disruptive selection on offspring size and a possible case of speciation in freshwater gobies characterized by egg-size difference. Res Popul Ecol 20: 211-215.
- Nishino M (1980) Geographical variations in body size, brood size and egg size of a freshwater shrimp, Palaemon pauchdens DE HAAN, with some discussion on brood habit. Jap J Limnol 41: 185-202.
- Beacham TD, Murray CB (1993) Fecundity and egg size variation in North American Pacific salmon (Oncorhynchus). J Fish Biol 42(4): 485-508.
- Duffy EJ, Beauchamp DA, Buckley RM (2005) Early marine life history of juvenile Pacific salmon in two regions of Puget Sound. Estuar Coast Shelf Sci 64(1): 94-107.
- Fleming IA, Gross MR (1990) Latitudinal clines: a trade-off between egg number and size in Pacific salmon. Ecology 71: 1-11.
- McDowall RM (1992) Diadromy: origins and definition of terminology. Copeia 1992: 248-251.
- McDowall RM (1997) The evolution of diadromy in fishes (revised) and its place in phylogenetic analysis. Rev Fish Biol Fish 7: 443-462.
- Iguchi K (1993) Latitudinal variation in ayu egg size. Nippon Suisan Gakk 59: 2087.
- Iguchi K, Sakano H, Takeshima H (2010) Starving process of ayu hatchlings under different saline water conditions. Aquacult Sci 58: 459-463.
- Takahashi I, Kinoshita I, Azuma K, Fujita S, Tanaka M (1990) Larval ayu Plecoglossus altivelis occurring in the Shimanto Estuary. Nippon Suisan Gakk 56: 871-878.
- Murase I, Iguchi K (2019) Facultative amphidromy involving estuaries in ba abbyak amphidromous fish from a subtropical marginal range. J Fish Biol 95: 1391-1398.
- Tran HD, Kinoshita I, Ta TT, Azuma K (2012) Occurrence of Ayu (Plecoglossus altivelis) larvae in northern Vietnam. Ichthyol Res 59: 169-178.
- Iguchi K, Takeshima H (2011) Effects of saline water on early success of amphidromous fish. Ichthyol Res 58: 33-37.
- Iguchi K, Yamaguchi M (1994) Adaptive significance of inter- and intrapopulational egg size variation in ayu Plecoglossus altivelis (Osmeridai). Copeia 1994: 184-190.
- Iguchi K, Takeshima H (2007) Proposal of novel test for evaluating salinity tolerance of yolk-sac larvae in ayu. Aquaculture Sci 5: 417-421.
- Kaneko T, Shiraishi K, Katoh F, Hasegawa S, Hiroi J (2002) Chloride cells during early life stages of fish and their functional differentiation. Fish Sci 68: 1-9.
- Gross MR, Coleman RM, McDowall RM (1988) Aquatic productivity and the evolutions of diadromous fish migration. Science 239(4845): 1291-1293.
- Iguchi K, Konishi M, Takeshima H (2006) Early dispersal of ayu during marine stages as inferred from geographic variation in the number of vertebrae. Fish Sci 72: 737-741.
- Shiraishi Y, Suzuki N (1962) The spawning activity of ayu-fish, Plecoglossus altivelis. Bull Freshwat Fish Res Lab 12: 83-107.






























