Abstract
For the first time, the concentration and distribution patterns of polyaromatic hydrocarbons were evaluated in Iranian parts of the Gulf of Oman. The sampling area included 10 transects facing the coastlines of Hormozgan and Sistan and Baluchestan Provinces in the South of Iran. Collection of sediment and water samples was performed in the Spring and Fall of 2007 and 2009, respectively. Polyaromatic hydrocarbons ranged from 9.06 to 972.77 ng/l in water samples. In sediments, polyaromatic concentrations varied from 10.2 to 157.1 ng/g dry weight. The determined values were lower than sediment quality guidelines values. Results of isomeric ratios confirmed that petrogenic dominated over pyrogenic origins for polyaromatic hydrocarbons pollution, in water and sediments from the Gulf of Oman. Monitoring of pollutants is recommended, regarding the high extension of oily activities in the Gulf of Oman.
Keywords:Pollution Monitoring; Sediment Pollution; Petroleum Hydrocarbons; Poly Aromatic hydrocarbons; Gulf of Oman; Hormozgan Province; Sistan and Baluchestan province
Introduction
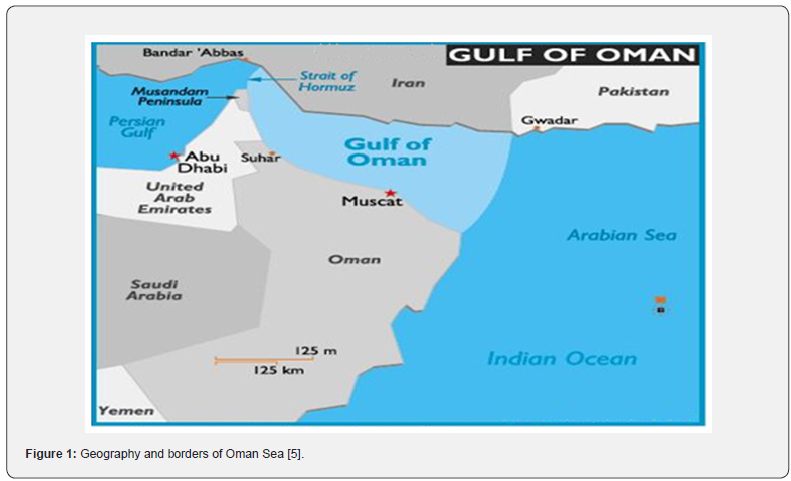
The Gulf of Oman is situated in the Middle East and known as the Western expansion of the Arabian Sea into the Persian Gulf (Figure 1) with length and width of 545 and 370 km, respectively. The Gulf of Oman is surrounded by Iran, United Arab Emirates and Oman. In Iran, the two southern provinces including Hormozgan and Sistan and Baluchestan share borders with the Gulf of Oman. Hormoz Strait confers a vital importance to the Gulf of Oman and makes it a shipping path for oil-producing countries neighboring the Persian Gulf. Due to the high rate of oil production and high volume of marine transportations, the Gulf of Oman is critically threatened by petroleum pollution [1,2]. It is noticeable that over half of the world’s oil exports are transferred through the Gulf of Oman. It is estimated that the Persian Gulf receives about 120186 t of petroleum per year [3]. This strategic position and the activities related to the petroleum industry cause chemical pollution including PAHs and TPHs. Furthermore, following the Persian Gulf, the Gulf of Oman is at risk from various kinds of pollution sources, including municipal waste, agricultural and industrial discharges, hot and saline waste waters from desalinization plants, contamination sources related to petroleum and discharge of ballast water of the ships [1].
Furthermore, to its strategic importance for oil exporting, fisheries and related industries are among the most important sources of income for the populace and the livelihood of residents in coastal cities depends on industrial fisheries from the Gulf of Oman. Thus, maintaining the health of the marine environment of the Gulf of Oman is important for marine biodiversity and consumer health.
In spite of the socio-economic aspects of the Gulf of Oman, information on organic contaminants is rare and limited to restricted studies [1] and investigations with the main concentrations of the Persian Gulf area [2,4]. In the current study, for the first time focus was on the Iranian parts of the Gulf of Oman, to study the poly aromatic hydrocarbons (PAHs) [5].
Material and Methods
Area study and sample collection
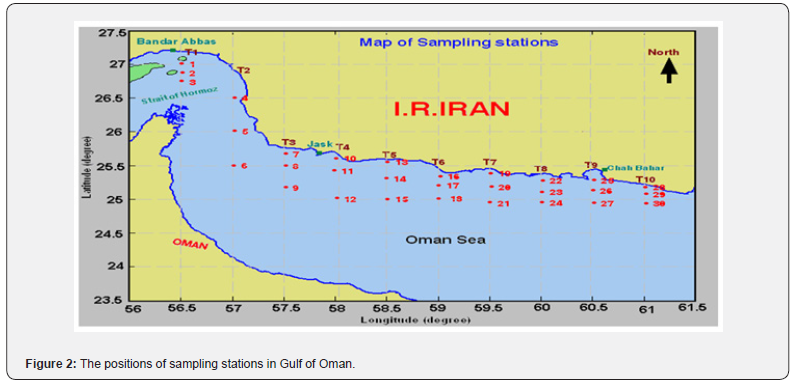
The sampling stations were selected along the Hormozgan and Sistan and Baluchestan Provinces coastline, as they cover the Iranian part of the Oman Sea from Hormoz Strait to Gowatr Gulf (Figure 2). The sampling area included 30 stations in ten transacts, represented respectively by indices from T1 to T10, while each transact comprised three stations. Sediment samplings were performed using GPS co-ordinates to record the geographical location of stations in the Spring and Fall of 2007 and 2009, respectively.
Sediment sampling was performed using a Van Veen Sampler (made of stainless steel, with overall dimensions of approximately 20×30×60 cm and 5 kg weight) on a 250 cm2 surface. All samples were collected in three duplications. After collection, the sediments were transferred into a pre-cleaned container, mixed and homogenized. Sub samples were removed and freezed. In the laboratory, the freeze-dried and sieved fraction of sediment with sizes fewer than 2 mm was kept at -20°C temperature until analysis. Water samples were collected by Niskin, Multi- Capasity Water Sampler coupled with pressure meter to determine the sampling depth. Separatory funnel method by n-Hexan was used to extract the poly aromatic hydrocarbons from water samples and the extract was transferred to the laboratory for PAHs analysis [6].
Chemical analysis of petroleum hydrocarbons
The determined sixteen EPA polyaromatic hydrocarbons included naphthalene (Na) , acenaphthylene (ACEY), acenaphthene (ACE), fluorine(FL), phenanthrene (PHEN), anthracene (AN), fluoranthene (FLU), pyrene (PY), benz[a] anthracene (B(a)A), chrysene (CHR), benzo[b]fluoranthene (B(b) FLU), benzo[a]pyrene (B(a)PY), benzo[k]fluoranthene (B(k)FLU), dibenz[a,h]anthracene, benzo[ghi]perylene and indeno[1,2,3-cd] pyrene . At the preliminary step, internal standards (pyrene-d10, phenanthrene-d10, naphthalene d8, perylene d12) were added to about 10 g of sediments. A mixture of 50:50 hexane and dichloromethane (40 ml) was exposed to microwave radiation under specific conditions as follows: microwave power of 1200 watts, temperature increase to 115°C in 10 min, extraction time of 20 min at 115°C, and cooling to ambient temperature within 1 h [2,6,7].
Active copper was used to remove sulfur interference [8]. Active copper was prepared in the laboratory through the reaction of CuSO4·5Hsub>2O with Zn in acidic condition [6].
The silica-alumina column was used for cleaning up, purification, and fractionation of petroleum hydrocarbons. The chromatography column was prepared using a simple 50 mL burette with 1 cm internal diameter. The column was filled with 10 mL of silica and 10 ml of alumina and 1 g sodium sulfate, respectively. After transferring the extract to the top of the column, the first fraction containing the aliphatic hydrocarbons was eluted with 20 mL hexane. The second and third fractions were eluted with 30 mL hexane/dichloromethane mixture (90:10) and 10 mL dichloromethane, respectively. The latter fractions contained polyaromatic hydrocarbons. The volumes of extract were reduced to 15 mL via rotary evaporation and to lower volumes for instrumental analysis, by high-purity nitrogen gas [4,6,9-11].
Instrumental analysis of extracts for PAHs was performed
using a gas chromatography system (Agilent 6890N) employing
an auto-injection system equipped with a mass selective
detector (Agilent 5973N). Instrumental analysis was based on
guidance provided in EPA-method 8000B and 8275A [3,4,12-
14]. Instrumental analysis was performed based on single ion
monitoring (SIM) mode at selected quantitation ions.: 128, 152,
154, 166, 178, 202, 228, 252, 276, and 278. Other instrumental
conditions included the following:
Column type: capillary column (0.25 mm × 30 m × 0.5 μm) of
DB-5ms (Agilent 122-5536) Injector temperature: 290oC
Column temperature program: 60oC (1 min hold) to 100oC
(with 10o/min ramp) and finally to 300oC (with 4oC /min ramp
and 15 min hold)
Carrier gas: Helium, 1.5 ml / min
Injection volume: 1 μL, in splitless mode
Ionization mode: electron impact
Electron energy: 69.9 EV
Quality control
The standard mixture of PAH components was prepared by Chiron AS. Additionally, the internal standards were purchased from Chiron AS, including pyrene-d10, phenanthrene-d10, naphthalene d8, and perylene d12. All used solvents including n-hexane, dichloromethane, methanol and acetone were prepared as chromatography grade from Sigma-Aldrich. Silica gel 60 and aluminium oxide neutral 90 active were purchased from Merck. Anhydrous sodium sulphate, copper sulphate and zinc powder with 99% purity were obtained from Sigma-Aldrich. Components were quantitated based on the external standard method. With each analytical batch (nine samples), the established calibration curves were checked at the midpoint. The retention time of each individual component was compared to reliable standards. Several techniques were employed to show the validity of the analytical manuals and the quality of obtained data. The accuracy and precision of the applied methods were validated using these techniques. With each set of samples, method blanks were performed to investigate the possibility of interferences or contamination peaks at monitoring ions. To assess the precision of the analytical methods, three duplications of all experiments were performed. Overall, the certified reference material was IAEA-417 for petroleum hydrocarbons present in the sediment samples. The achieved acceptable average deviations from the reference values were 10% for PAHs. The calculated method detection limits (MDLs) and the recovery methods for PAHs were 0.1- 0.3 (ng/g) and 92 – 120%, respectively.
Statistical analysis
Detected data were expressed as average ± SD (standard deviation). The significance level in all analyses was 0.05. Parametric test (one-way ANOVA) and non-parametric tests (Kruskall–Wallis) were used to assess the significant differences of polyaromatic hydrocarbons concentrations among the sampling sites. Whereas the T-test and Mann–Whitney U test were performed to assess the significant differences between sampling seasons.
Results and Discussion
PAHs in water and sediments
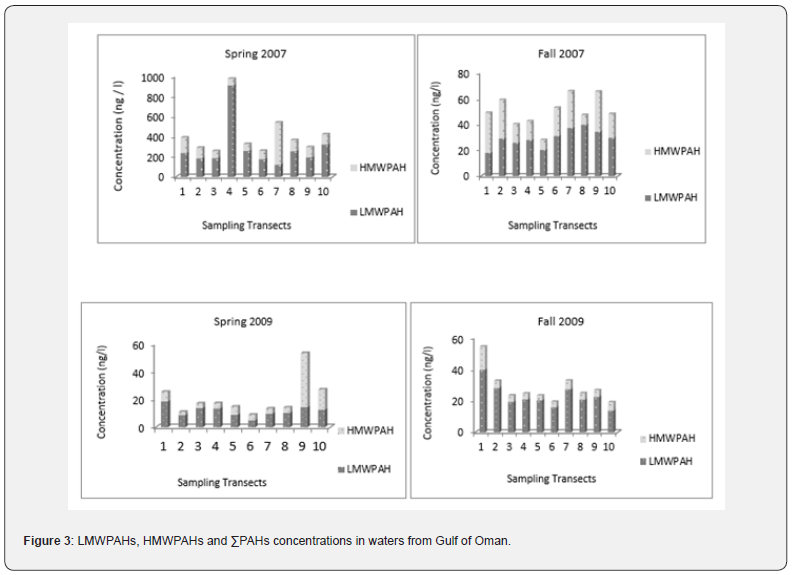
The concentrations of ΣPAHs, low molecular weight PAHs (LMWPAHs) and high molecular weight PAHs (HMWPAHs) in water from the Gulf of Oman during 2007 and 2009, are presented in Figure 3.
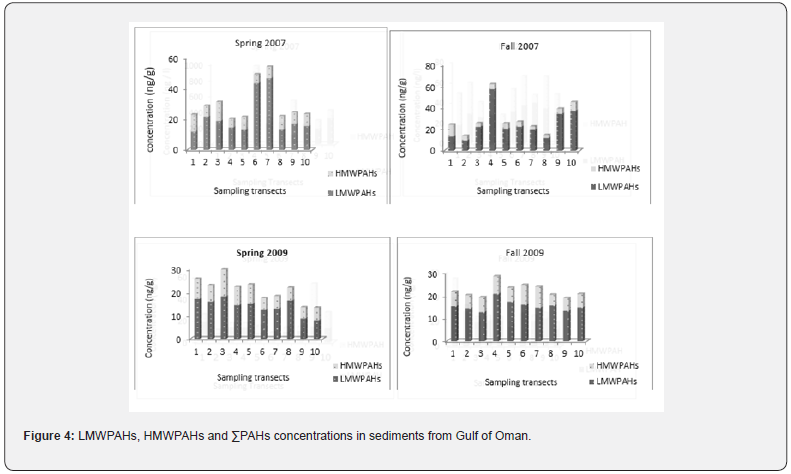
Wide ranges of ΣPAHs among the sampling stations were detected. The range of variation was from 9.06 to 972.77 ng/l despite the high values observed in the 2007 spring, the other detected concentrations were below 70 ng/l. The hotspots for ΣPAHs detected in the Iranian waters of Oman Sea were related to transect 4 in the 2007 spring. Statistical surveys did not confirm the significant differences between ΣPAHs among the sampling transects (p<0.05). Figure 4 represents the determined values for sediments from the Gulf of Oman.
ΣPAHs ranged from 10.2 to 157.1 ng/g in sediment samples. The high detected values were related to Transects 3 and 4 in the Fall and Spring of 2009. However, no identical distribution pattern was followed in ΣPAHs among different seasons and transects. Except fall 2007, no significant differences were shown in PAHs distribution among sampling stations (p<0.05).
Distribution patterns of PAHs in water and sediments
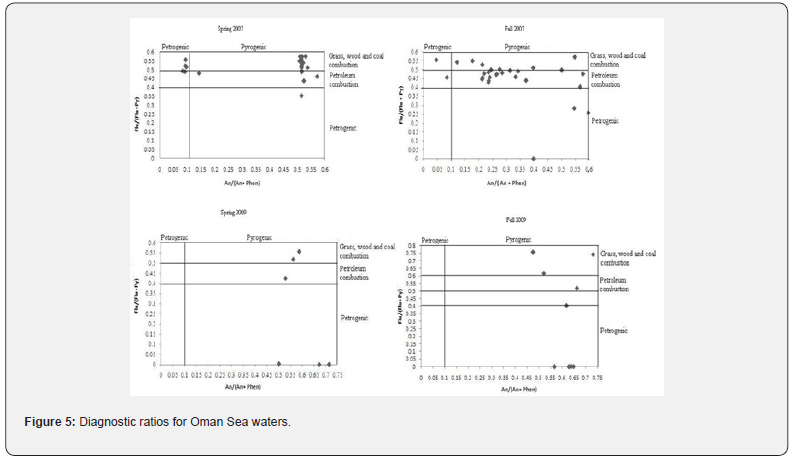
Except Transect 1 in the 2007 Fall, in all studied area and sampling times, the ratio of LMPAHs/HMPAHs were higher than 1 for water samples, indicating the input of petroleum from release of crude oil. LMPAHs/HMPAHs values increased in the Fall of 2007. Variation of molecular ratios An/Phen and Flur/Py are shown in Figure 5. Flur/Py values lower than 1 and An/Phen lower than 10 support the petrogenic sources [15]. The maximum and minimum values of An/Phen ratio in the 2007 Spring were 8.01 and 0.91, respectively. In the 2007 Fall, An/Phen ratio showed a range from 0.64 to 2.9. Similar results were observed in 2009, and all the calculated ratios were lower than 10, confirming the pyrogenic sources for poly aromatic hydrocarbons in water samples from the Gulf of Oman. Whereas, Flur/Py ratios showed values more than 1 and reached 2.9, supporting pyrogenic origins. Totally, it has been shown that PAHs are of petrohenic and pyrogenic sources in the Gulf of Oman; however, the petrogenic sources dominated over pyrogenic sources.
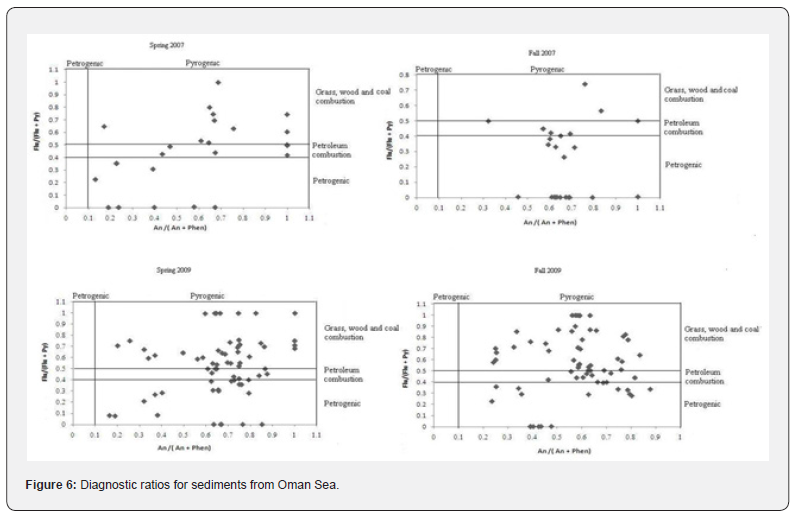
All detected values of LMPAHs/HMPAHs for sediment samples varied from 1.03 to 44.3, confirming the petrogenic source of poly aromatic hydrocarbons. Diagnostic ratios for sediments in the Gulf of Oman are shown in Figure 6. Identical pattern for An/Phen and Flur/Py variation were detected in sediments and water samples. Whereas all detected An/Phen ratios were lower than 10, the Flur/Py included both values lower and higher than 1, showing the presence of petrogenic and pyrogenic origins in the marine ecosystem of the Gulf of Oman. This observation is attributed to the high intensity of activities related to the oil industry in the Gulf of Oman and Persian Sea, connected by the Hormoz Strait.
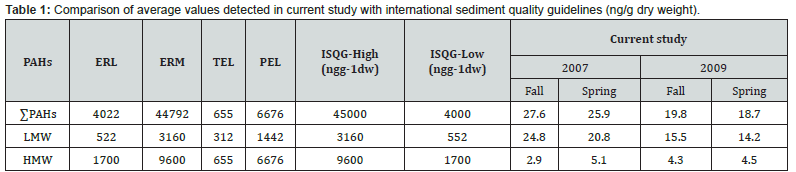
The values detected in the current study were compared with international standards (Table 1) including the National Oceanographic and Atmospheric Administration [16], Interim marine sediment quality guidelines [17] and Florida Department of Environmental Protection [18]. All determined levels in sediments from the Gulf of Oman were much lower than the international sediment quality guidelines. It means that in spite of hotspot presence in the Gulf of Oman, totally there was no risk from polyaromatic hydrocarbon pollutants for the marine ecosystem of the Gulf of Oman.
Comparison with other results
Table 2 contains the results obtained for waters from areas worldwide. In comparison with high polluted areas such as China and the Persian Gulf, the study areas are classified as unpolluted. However, the presence of hotspots in Spring 2007 is noticeable and reached pollution level in the North of the Persian Gulf [19].
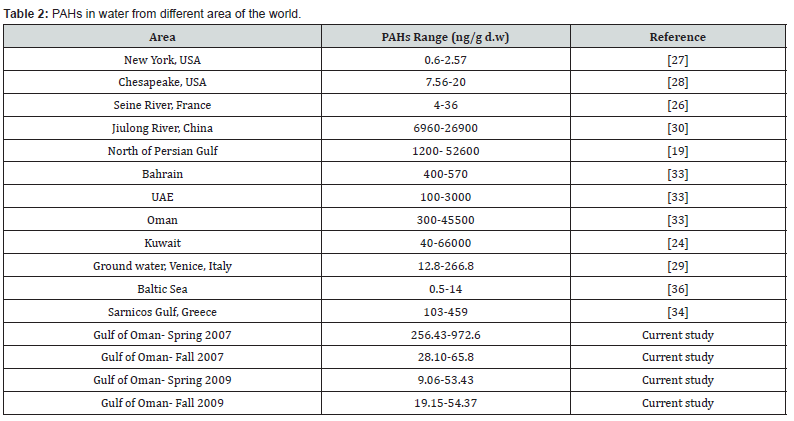
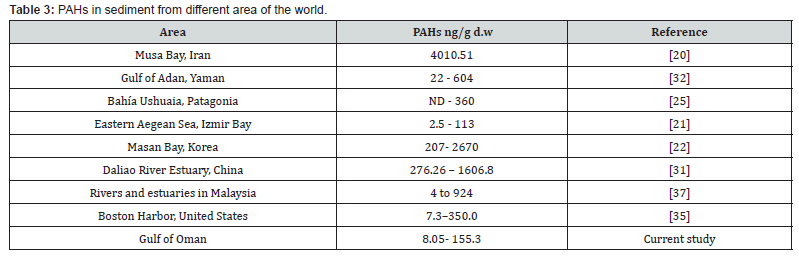
Table 3 indicates the PAH distribution in sediment samples from different parts of the world. The results of this study were much lower than ΣPAHs reported for sediments of Musa Bay in Iran [20] with high activity in oil-related industry. The detected range is close to ΣPAHs concentration in Izmir Bay [21] and far from PAH pollution in sediments of Masan Bay and Daliao River Estuary in industrial countries Korea and China, respectively [22]. Harbours are known as the most oil-polluted areas. Sediments from the Gulf of Oman contained ΣPAHs lower than Boston Harbor in United States.
Marine sediments are classified as low, moderate, high and very highly polluted, corresponding to PAHs concentrations of <100, 100–1000, 1000–5000 and >5000 ng/g d.w, respectively [23]. Sediments from Gulf of Oman are classified in low to moderate polluted areas [24-37].
Conclusion
In spite of special position and existence of high rate of marine transportation, the Gulf of Oman is not at risk of pollution by polyaromatic hydrocarbons. In comparison with international sediment quality guidelines showed the low probability of adverse biological effects from polyaromatic hydrocarbons. However, professional management applications are needed to avoid the spreading of detected hotspots. Continuous monitoring of other groups of organic contaminants seems essential.
Acknowledgement
We would like to thank the Iranian Fisheries Research Institute for financial supporting of this research (project no. 2-029-200000-04-8502-8503).
References
- Awad H, Al Eissa S, Al Moharamy M (1990) Oil pollution in waters, fish and sediments in Gulf of Oman Environment. JKAU Marine Science 1: 9-16.
- De Mora S, Tolosa I, Fowler SW, Villeneuve JP, Cassi R, et al. (2010) Distribution of petroleum hydrocarbons and organochlorinated contaminants in marine biota and coastal sediments from the ROPME Sea Area during 2005. Marine Pollution Bulletin 60: 2323-2349.
- Araghi EP, Farzadnia S (2011) Petroleum inputs to the Persian Gulf. Research Journal of Environmental Sciences 5(2): 134-141.
- Tolosa I, De Mora SJ, Fowler SW, Villeneuve JP, Bartocci J, et al. (2005) Aliphatic and aromatic hydrocarbons in marine biota and coastal sediments from the Gulf and the Gulf of Oman. Marine Pollution Bulletin 50(12): 1619-1633.
- Worldatlas (2015) Gulf of Oman.
- MOOPAM (2005) Manual of Oceanographic Observation and Pollutant Analyses Methods. In: (4th), Regional Organization for the Protection of the Marine Environment, Kuwait.
- USEPA (2007) Method 3546, Microwave extraction. Washington, DC: US Environmental Protection Agency.
- USEPA (1996f) Method 3660B, Sulfur cleanup. Washington, DC: US Environmental Protection Agency.
- USEPA (1996b) Method 3600C, Cleanup. Washington, DC: US Environmental Protection Agency.
- USEPA (1996a) Method 3610B, Alumina cleanup. Washington, DC: US Environmental Protection Agency.
- USEPA (1996e) Method 3630C, Silica gel cleanup. Washington, DC: US Environmental Protection Agency.
- Rajendran R, Imagawa T, Tao H, Ramesh R (2005) Distribution of PCBs, HCHs and DDTs, and their ecotoxicological implications in Bay of Bengal, India. Environment International 31(4): 503-512.
- USEPA (1996c) Method 8000B, Determinative chromatographic separations. Washington, DC: US Environmental Protection Agency.
- USEPA (1996d) Method 8275A, Semivolatile organic compounds (PAHs and PCBs) in soils/sludges and solid wastes using thermal extraction/gas chromatography/mass spectrometry (TE/GC/MS). Washington, DC: US Environmental Protection Agency.
- Budzinski H, Jones I, Bellocq J, Pierard C, Garrigues P (1997) Evaluation of sediment contamination by polycyclic aromatic hydrocarbons in the Gironde estuary. Marine Chemistry 58(1-2): 85-97.
- Long E, MacDonald D, Smith S, Calder F (1995) Incidence of adverse biological effects within ranges of chemical concentrations in marine and estuarine sediments. Environmental Management 19: 81-97.
- Burton JGA (2002) Sediment quality criteria in use around the world. Limnology 3: 65-76.
- MacDonald D (1994) Approach to the assessment of sediment quality in Florida coastal waters. Florida Department of Environmental Protection Office of Water Policy, Florida.
- El Samra M, Emara H, Shunbo F (1986) Dissolved petroleum hydrocarbon in the northwestern Arabian Gulf. Marine Pollution Bulletin 17(2): 65-68.
- Tehrani GM, Hshim R, Sulaiman AH, Tavakoly SB, Sany RKJ, et al. (2012) Assessment of Contamination by Petroleum Hydrocarbons in Sediments of Musa Bay, Northwest of the Persian Gulf-Iran. International Conference on Environment, Energy and Biotechnology. IACSIT Press, Singapore, p. 75-80.
- Darilmaz E, Kucuksezgin F (2007) Distribution and origin of hydrocarbons in surficial sediments from the eastern Aegean Sea (Izmir Bay). Marine Pollution Bulletin 54(11): 1824-1830.
- Yim U, Hong S, Shim W, Oh J, Chang M (2005) Spatio-temporal distribution and characteristics of PAHs in sediments from Masan Bay, Korea. Marine Pollution Bulletin 50(3): 319-326.
- Baumard P, Budzinski H, Garrigues P, Sorbe J, Burgeot T, et al. (1998) Concentrations of PAHs (polycyclic aromatic hydrocarbons) in various marine organisms in relation to those in sediments and to trophic level. Marine Pollution Bulletin 36(12): 951-960.
- Bu-Olayan A, Thomas B (2001) Heavy Metal Accumulation in the Gastropod, Cetithium Scabridum , from the Kuwait Coast. Environmental monitoring and assessment 68: 187-195.
- Commendatore MG, Nievas ML, Amin O, Esteves JL (2012) Sources and distribution of aliphatic and polyaromatic hydrocarbons in coastal sediments from the Ushuaia Bay (Tierra del Fuego, Patagonia, Argentina). Marine environmental research 74: 20-31.
- Fernandes M, Sicre MA, Boireau A, Tronczynski J (1997) Polyaromatic hydrocarbon (PAH) distributions in the Seine River and its estuary. Marine Pollution Bulletin 34(11): 857-867.
- Gigliotti CL, Brunciak PA, Dachs J, Glenn IV TR, Nelson ED, et al. (2002) Air-water exchange of polycyclic aromatic hydrocarbons in the New York-New Jersey, USA, Harbor Estuary. Environmental Toxicology and Chemistry 21(2): 235-244.
- Gustafson KE, Dickhut RM (1997) Distribution of polycyclic aromatic hydrocarbons in southern Chesapeake Bay surface water: evaluation of three methods for determining freely dissolved water concentrations. Environmental Toxicology and Chemistry 16(3): 452-461.
- Manodori L, Gambaro A, Piazza R, Ferrari S, Stortini A, et al. (2006) PCBs and PAHs in sea-surface microlayer and sub-surface water samples of the Venice Lagoon (Italy). Marine Pollution Bulletin 52(2): 184-192.
- Maskaoui K, Zhou J, Hong H, Zhang Z (2002) Contamination by polycyclic aromatic hydrocarbons in the Jiulong River Estuary and Western Xiamen Sea, China. Environmental Pollution 118(1): 109-122.
- Men B, He M, Tan L, Lin C, Quan X (2009) Distributions of polycyclic aromatic hydrocarbons in the Daliao River Estuary of Liaodong Bay, Bohai Sea (China). Marine Pollution Bulletin 58: 818-826.
- Mostafa AR, Wade TL, Sweet ST, Al-Alimi AKA, Barakat AO (2009) Distribution and characteristics of polycyclic aromatic hydrocarbons (PAHs) in sediments of Hadhramout coastal area, Gulf of Aden, Yemen. Journal of Marine Systems 78(1): 1-8.
- ROPME (2002) Regional Report of the Marine Environment. Regional Organization for the Protection of the Marine Environment, Kuwait.
- Valavanidis A, Vlachogianni T, Triantafillaki S, Dassenakis M, Androutsos F, et al. (2008) Polycyclic aromatic hydrocarbons in surface seawater and in indigenous mussels (Mytilus galloprovincialis) from coastal areas of the Saronikos Gulf (Greece). Estuarine, Coastal and Shelf Science 79(4): 733-739.
- Wang XC, Zhang YX, Chen RF (2001) Distribution and partitioning of polycyclic aromatic hydrocarbons (PAHs) in different size fractions in sediments from Boston Harbor, United States. Marine Pollution Bulletin 42(11): 1139-1149.
- Witt G (1995) Polycyclic aromatic hydrocarbons in water and sediment of the Baltic Sea. Marine Pollution Bulletin 31(4-12): 237-248.
- Zakaria MP, Takada H, Tsutsumi S, Ohno K, Yamada J, et al. (2002) Distribution of polycyclic aromatic hydrocarbons (PAHs) in rivers and estuaries in Malaysia: a widespread input of petrogenic PAHs. Environmental Science & Technology 36(9): 1907-1918.






























