Abstract
Assessing population dynamics is essential for the effective management of fish stocks. In this context, the current study examined key parameters: growth, mortality, and recruitment of Stephanolepis diaspros in the Gulf of Gabès, utilizing the FiSAT II (FAO-ICLARM Stock Assessment Tools) software (version 1.2.2) for analysis. The specimens were collected from monthly sampling of S. diaspros along the continental shelf of the Gulf of Gabès, between the parallel 35oN and the Libyan border, mainly at depths between 0 and 40 m and by collecting monthly samples from Sfax fishing harbour. A total of 1116 individuals were collected from August 2015 to February 2017. Most were small, ranging in size from 3.7 to 23.7 cm, and weighing 1.05 to 235.3 g respectively. Von Bertalanffy growth was deployed to determine the growth parameters of this alien fish. The asymptotic length is L∞ = 37 cm , with a growth performance φ ' = 2.628 , justifying the moderate growth rate K = 0.31 yr−1 . Theoretical age (t0 = −1.976 yr−1) and longevity or maximum age (tmax = 9.664 yr−1) were determined. Battacharya’s method incorporated in the FISAT II software was applied to identify the different age groups (cohorts) and the mean length of each group. Age and Growth Plotting the decomposition of the length frequency distribution clarified four age groups with a dominance of the group II.
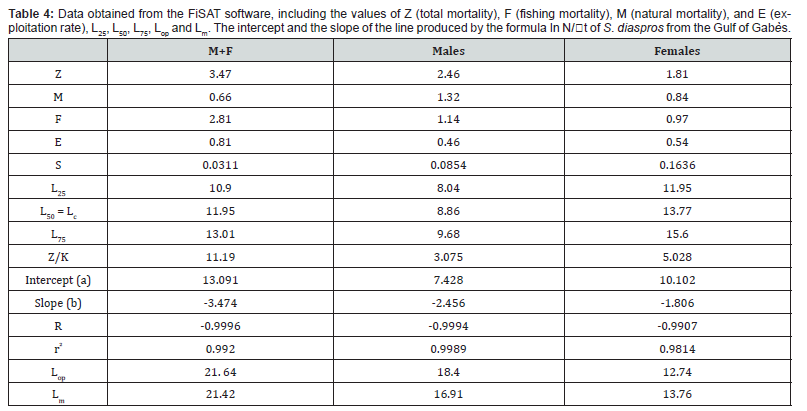
Natural mortality for this alien species (M = 0.66 yr−1) is lower than fishing mortality (F = 2.81 yr−1) . The exploitation rate (E = 0.81) is higher than the critical value (E = 0.5) . These results suggest that this species is overexploited in the Gulf of Gabès and the size of first capture (Lc = 11.95 cm) is lower than the size of first maturity (Lm = 21.4 cm) also, a condition that can disturb the stock.
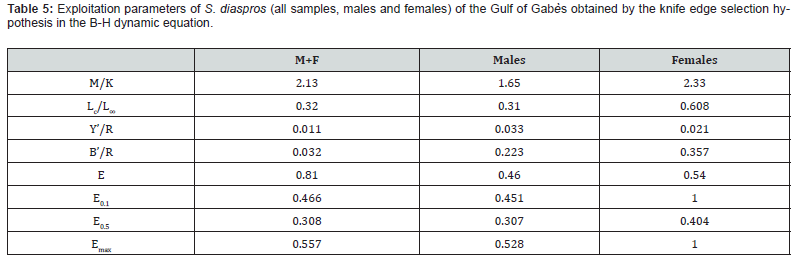
Analysis of relative yield per recruit (Y’/R) indicates that the exploitation rate exceeds Emax (0.557) and E0.5 (0.308) , thereby confirming overfishing. To ensure sustainable and rational management of this species, it is advised to decrease the exploitation rate from 0.81 to 0.308, representing a 49.8% reduction. The current research on the population dynamics of S. diaspros and emphasizes the importance of proactive measures to protect pelagic fish populations and maintain sustainable fishery resources. All parameters in the study of stock assessment of this species (Z, M, F, L∞, E, Emax, E0.5, Lc, Lm) shows significant differences between males and females.
No prior studies or assessments were available concerning fish stock evaluation, making these results the first data on the overall status of this species in the Mediterranean Sea.
Keywords:Stephanolepis diaspros; Eastern Mediterranean Sea; Growth; Mortality; Exploitation; Yield per recruit
Introduction
The Mediterranean Sea is a globally recognized biodiversity hotspot, hosting approximately 17000 marine species, many of which are endemic. However, this semi-enclosed basin is undergoing significant ecological transformations due to various anthropogenic pressures, including habitat degradation, overfishing, pollution, and climate change. Among these challenges, the introduction of alien species through Lessepsian migration has emerged as a key driver of change, particularly in the eastern Mediterranean Sea. Lessepsian migration refers to the movement of marine organisms from the Red Sea into the Mediterranean via the Suez Canal, which has acted as a corridor for bioinvasions since its opening in 1869. This phenomenon has introduced nearly 1000 alien species to the Mediterranean, with many establishing themselves as dominant components of local ecosystems [1,2].
The Monacanthidae family, commonly known as filefishes, is closely related to the Balistidae family, and the two are often grouped together. Monacanthidae includes around 95 species, most of which are found in tropical regions. Of these, only one species, the reticulated leatherjacket (Stephanolepis diaspros), originally from the Red Sea, has been reported in the Mediterranean Sea [3].
The Reticulated leatherjacket, Stephanolepis diaspros Fraser Brunner, 1940 is among the earliest Lessepsian settlers in the Mediterranean [4,5]. This specie is characterized by its compressed lateral body and rough skin. It inhabits inshore in sandy and rocky habitats in vegetation usually at depths not exceeding 20 meters [3,5]. Common sizes of S. diaspros range from 7 to 15 cm, with the maximum as 30.5 cm [6]. They feed on phytobenthos and small rock-dwelling organisms, especially small benthic invertebrates, mainly crustaceans, foraminifers, gastropods, ostracods, sea urchins [3,7,8]. Young individuals feed also in open water areas with sandy and muddy substrates [9].
S. diaspros is distributed in Arabian and in Red Sea (Randall 1995) [9], and entered the Mediterranean Sea through Suez Canal, such as other lessepsian migrant species [10]. Since the first record off Palestine [11], this demersal fish has now become common in several Mediterranean regions, where it appears to have successfully adapted to local conditions [9]. Thus, a sustainable population was successfully established in Turkish seas, for instance [12,13]. Also, it reached Gulf of Palermo, Sicily [14], the Adriatic Sea [15], and Maltese waters [16].
In the Tunisian coast, characterized by a warm and oligotrophic waters, S. diaspros was first recorded by Chakroun [17] in the Gulf of Gabès, furtherly, reported by Bradaï [18], Bradaï et al. [19] and Zouari-Ktari et al. [7,20,21] in the area. Northward, this species was recorded by Bdioui et al. [22] in a brackish area, the Bizerte Lagoon and by Ben Amor and Capapé [23] in Tabarka (northern Tunisia) and constituted the northernmost record of S. diaspros in the region and the westernmost in the Mediterranean Sea.
The presence and the establishment of a population of S. diaspros in the Gulf of Gabès raise both ecological and economic concerns. Ecologically, it competes with native species for resources and may alter local food webs. Economically, it has become an incidental catch in regional fisheries and represents an additional resource for some coastal communities [2,6].
This rapid expansion of S. diaspros in the Gulf of Gabès, raises important questions about its potential ecological impact and its population dynamics within this vulnerable ecosystem. Its increasing commercial exploitation calls for a better understanding of its biology, particularly its growth and population structure, in order to support sustainable management. Despite its growing significance, there is limited information on its biological traits and population dynamics in Tunisian waters except the both works of Zouari-Ktari et al. [7,21].
The present study aims to address this gap by estimating growth parameters and analyzing the population structure of S. diaspros in the Gulf of Gabès based on length-frequency data using the tools provided by the FISAT II software. Understanding these aspects is crucial for assessing its ecological impact and for informing management strategies in this rapidly changing ecosystem and potential fisheries implications. Such information is very important in Tunisian fisheries management strategies.
Despite the increasing presence of S. diaspros in the Mediterranean Sea, research has predominantly focused on its biological aspects, such as reproduction and diet. Studies have documented its reproductive biology in regions like the Gulf of Gabès [21] and the Gulf of Suez [24], as well as its feeding habits [7]. The study by El-Ganainy and Sabra [25] is the only work that addresses age, growth, mortality, and yield per recruit in the Gulf of Suez. Thus, comprehensive stock assessments of S. diaspros populations are notably lacking in both the Mediterranean and its native range in the Red Sea and Indo-pacific Ocean. This gap in knowledge hinders effective management and conservation strategies for the species, underscoring the need for further research into its population dynamics and stock status.
The present study represents the first comprehensive investigation into the growth, population dynamics, and stock assessment of S. diaspros in the Mediterranean region. By addressing this critical knowledge gap, the research aims to provide essential data for the sustainable management and conservation of this species, which has become increasingly significant in local fisheries.
Materials and Methods
Sampling Site study area
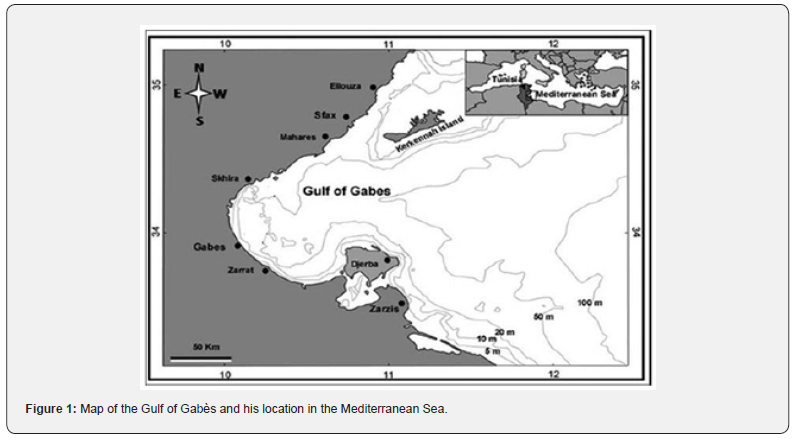
The Gulf of Gabès spans approximately from 33° to 35.5°N in latitude and from 9.5° to 12.5°E in longitude, known as the Lesser Syrtis, is located in the south-eastern part of Tunisia, along the eastern Mediterranean coast (Figure 1). It is characterized by its wide continental shelf, one of the most extensive in the Mediterranean Sea, which can reach up to 80 kilometers in width and is bounded by the Kerkennah Islands to the northeast and Djerba Island to the southeast. This shallow marine area is influenced by semi-diurnal tides. Thus, it is one of the few areas in the Mediterranean with a significant tidal range and presents a high level of ecological productivity, making it an important fishing ground for both artisanal and industrial fisheries. It has a high primary productivity due to nutrient inputs from sediment resuspension (driven by tidal currents), freshwater discharges (e.g., from the Wadi Akarit and Wadi Gabès) and upwelling events along the Kerkenah Islands [26,27].
The Gulf is known for its diverse habitats, including seagrass beds (Posidonia oceanica), sandy and muddy bottoms, and coastal lagoons, which support a rich marine biodiversity. The Gulf also acts as a nursery and spawning ground for migratory species such as cephalopods, turtles, and sharks, many of which are listed as endangered by the IUCN [27-32]. However, the region is also subjected to various anthropogenic pressures, such as overfishing, pollution, and habitat degradation, which threaten its ecological balance.
Due to its unique environmental conditions and strategic geographical position, the Gulf of Gabès has become a favourable site for the establishment and expansion of Lessepsian species, including S. diaspros, whose presence may influence local biodiversity and fisheries dynamics. The present study focuses on this area to evaluate the growth patterns and population structure of this invasive filefish species.
Specimens sampling and measurements
Monthly sampling of S. diaspros was carried out in pelagic trawlers, along the continental shelf of the Gulf of Gabès, mainly at depths ranged 0 and 40 m as well as through monthly collecting samples from Sfax fishing harbour. Fishes were taken from the net, identified to species, segregated by sex and then frozen for laboratory examination. In the laboratory, the total length (TL) of each specimen collected was measured to the nearest 1 mm using an ichtyometer and the total weight was also recorded to the nearest 0.01 g using an electronic balance.
Growth and Exploitation Parameters Growth parameters
The analysis of population parameters was conducted based on length frequency distribution using the FAO-ICLARM Stock Assessment Tools (FISAT II) software package (version 1.2.2) [33]. It is one of the most widely used models for growth studies in fisheries biology. Bhattacharya’s method [34] incorporated in FISAT II software was applied to identify age classes (cohorts) within the population and determine the mean length of each class. The Asymptotic length (L∞) and the growth coefficient (K) of the Von Bertalanffy Growth Formula (VBGF) were estimated using the ELEFAN-Ι according to Pauly and Morgan [35]. Then, the VBGF was fitted to estimates of length-at-age expressed by the following equation:
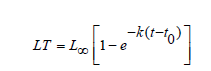
Where: TL: mean fish length at age t (cm), L∞ : asymptotic length (cm) at which growth is zero, K: growth rate (yr−1) , t0 : the theoretical age at which the fish has zero size [33].
The theoretical age (t0) was calculated according to the following relationship elaborated by [36]:
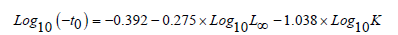
The von Bertalanffy Growth Function (VBGF) was fitted to the length frequency data to estimate the asymptotic length (L∞) and the growth coefficient (K).
The estimated values of (L∞) and k were used to determine the growth performance index (φ') and growth inflection point age (ttp) using the following formulae of Pauly [37]:
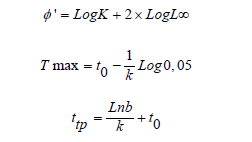
The growth performance index (φ') was computed to compare with other population.
Age and growth
Age estimation was performed for S. diaspros in the Gulf of Gabès using an age-length key. The age-length key was developed based on the length-frequency distribution of the sampled individuals and applied to the entire population as well as separately for males and females. To construct this key, we utilized the FISAT software, which is a widely used tool in fisheries biology for analyzing fish population dynamics. FISAT allows for the efficient estimation of growth parameters and age structures based on length-frequency data. By assigning individuals to age groups according to their total length, we were able to determine the age composition of the population. This method provided valuable insights into the growth dynamics, reproductive potential, and age structure of this invasive species, essential for its management and understanding of its ecological impact in the Gulf of Gabès.
Mortality, Exploitation, and Survival
The total instantaneous mortality rate (Z ) was calculated using a length-converted catch curve method incorporated in FiSAT II software as described by Pauly [37] and expressed as:
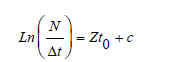
Where N is the number of samples in different length groups; Δt is the time required for the corresponding body length group to go from the lower limit to the upper limit; Ln = (N/Δt) is the number of deaths in the population N at time t and c is the intercept.
Natural mortality (M) was estimated according to Pauly [38] as:
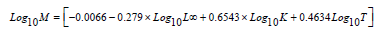
Where: (L∞) and (K) are Von Bertalanffy parameters and (T ) is average annual surface temperature. According to data obtained from relevant local authorities, T is 19.5 oC.
A number of relationships exist between the total (Z ) , natural (M) and fishing (F) mortality coefficients, as well as with the exploitation rate (E) : Z = M + F ; When two of the mortality parameters are known, the third is automatically deduced. Fishing mortality (F) = Z − M and The exploitation rate (E) = F / Z [39].
Following Gulland work on the exploitation rate (E) of a fishery resource, when E < 0.5, the stock is said to be underexploited, when E > 0.5 the stock is overexploited, and when E = 0.5 or F = M, the stock is optimally exploited [39].
The survival rate (S ) are represented by the formula [40]
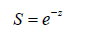
where e is Euler’s number.
Recruitment and First-Capture Size
Recruitment refers to the addition of new individuals to a harvestable stock and plays a crucial role in shaping fish population dynamics [41]. Thus, recruitment is the arrival of the youngest age group of individuals, called recruits, into the fishery each year for the first time [42].
These individuals are characterized by a size known as the recruitment size in the fishery (Lc or L50), corresponding to the size at which the fish becomes exploitable, that is, likely to be encountered in fishing gear. Several stocks show considerable interannual variations in the number of recruits. It is therefore important to take this into account in stock dynamics assessments. Monthly recruitment patterns were calculated from the length frequency data series. For this purpose, the program incorporated in FISAT II [43] was used. The monthly recruitment estimates were generated following the procedure described by Dadzie et al. [44].
The ogive selection method proves that the probability of a fish being caught is correlated with its length. On this basis, the length at which 50% of the catch retains by the fishing (L50) (length at first capture) was estimated from the catch curve analysis (probability of capture by length) incorporated in the FISAT II, according to Pauly [45,46]. In addition, the lengths associated with the 25th and 75th percentiles of catches were established at cumulative probabilities of 25% and 75%, respectively. Length at first sexual maturity (Lm) was obtained according to Froese and Binohlan [47] as follow:
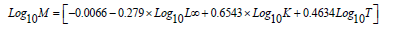
The optimal length of capture (Lopt ) is the length at optimum yield. It is determined for a given cohort according to the equation of Beverton [48]:
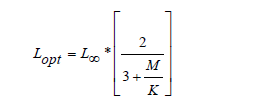
L∞ and K are function of Von Bertalanffy growth, while the value of M depends on the natural mortality rate. The recruitment period for S. diaspros in the Gulf of Gabès was determined using FiSAT II (recruitment patterns method), based on individual size frequency data.
Relative yield-per-recruit (Y’/R) and relative biomass-perrecruit (B’/R): Beverton and Holt Y’/R analysis
Y’/R (Relative Yield per Recruit) estimates the expected catch (in weight) per recruit, considering growth and mortality rates. Lower values suggest that each recruit contributes less to the fishery, potentially due to high mortality or early capture. B’/R (Relative Biomass per Recruit) reflects the average biomass each recruit contributes to the stock over its lifetime. Values below 1 indicate that the biomass per recruit is reduced compared to an unfished population, often due to fishing pressure.
The knife edge approach implemented in the FiSAT II software was upplied for the Beverton and Holt analysis to generate plots of the relative yield per recruit (Y’/R) and relative biomass per recruit (B’/R) against as functions of the exploitation rate [49,50]. The analysis determined the maximum allowable exploitation rate (Emax) that yields the highest relative yield-per-recruit (MSY =Maximum Sustainable Yield).
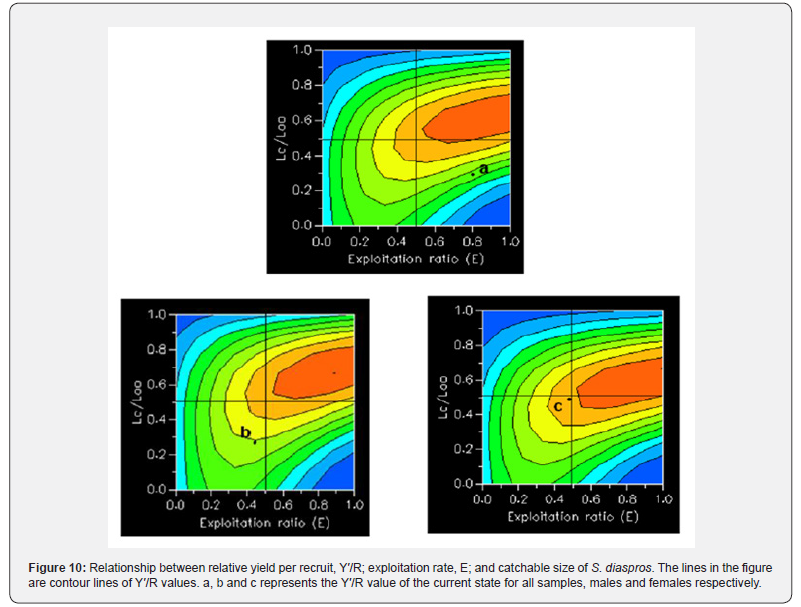
Additionally, E0.1 , the exploitation rate at which the marginal increase in relative yield-per-recruit reaches 10% of its virgin stock, and E0.5 , the exploitation rate corresponding to 50% of the unexploited relative biomass per recruit (B/R) (TRP = target reference point), were calculated. Yield contours were created to illustrate the yield isopleths, enabling an assessment of the impacts of changes in exploitation rate Emax and the critical length ratio (Lc = L50 / L∞) on yield.
Virtual population analysis (VPA)
The previously determined growth and mortality parameters (L∞, K, to, M, F, a, and b) were used as input data for the Virtual Population Analysis (VPA) performed following the methodology outlined by Gayanilo et al. [33].
Results
Length-Frequency Distribution
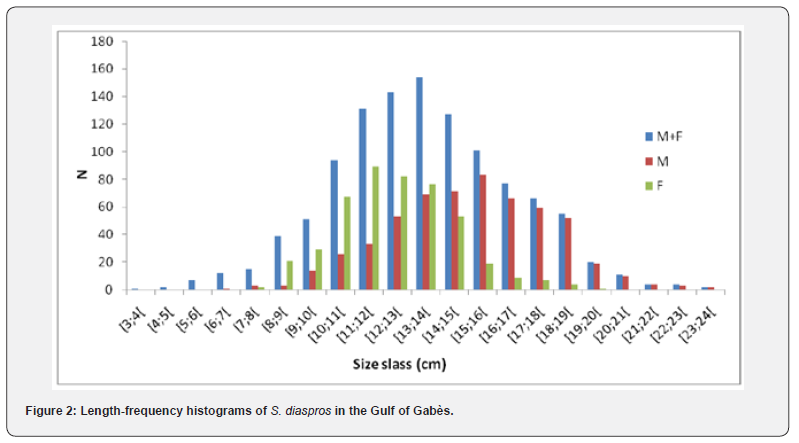
A total of 1116 specimens, with total lengths (TL) varying between 3.7 and 23.7 cm, were examined. The greatest number of specimens caught was between 11 and 14.9 cm (Figure 2), and the most common size is 12.5 for all specimens, 15.5 cm for males and 13.6 cm for females. The characteristics of this population were resumed in Table 1.
Growth and Exploitation Parameters Growth parameters
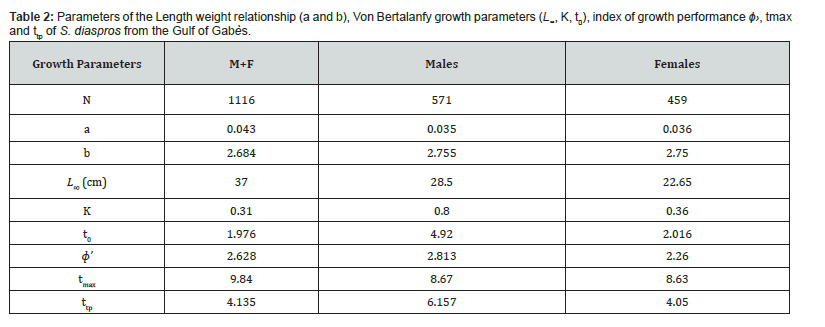
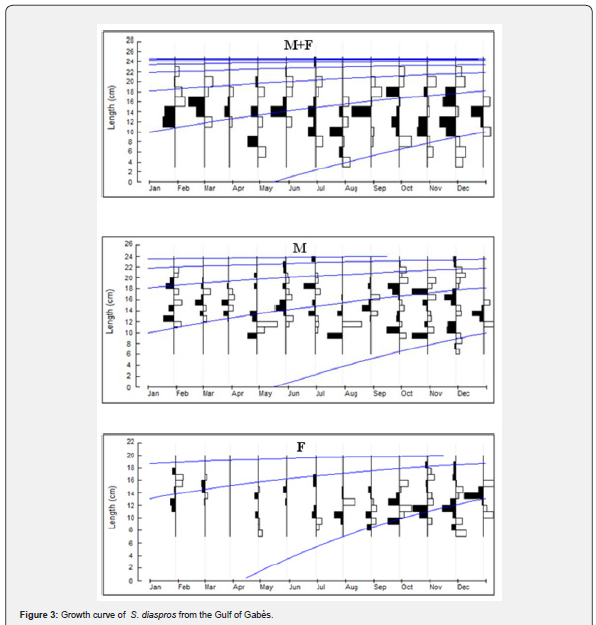
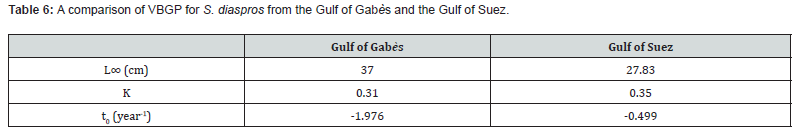
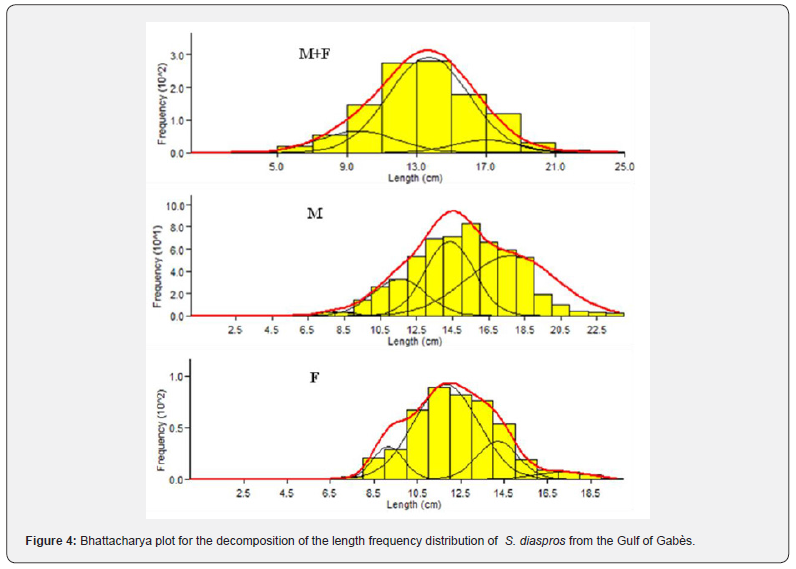
The asymptotic length is the theoretical maximum length that individuals of this exotic species can reach in this fraction of Mediterranean Sea, in relation to the factors influencing their environment. The value of K corresponds to the speed at which these fish grow to reach this greatest length locally. The asymptotic length obtained is 37 cm. The growth rate obtained K for this species is 0.31 yr-1, length-based index of growth performance (ɸ› = 2.628), theoretical age (t0 = 1.976 yr−1) and longevity or maximum age in the Gulf of Gabès (tmax = 9.84 yr−1). The largest growth inflection point age (ttp ) was detected for males (6.157) because they have an important value of growth rate (Table 2). The von Bertalanffy growth curve used to determine these parameters is shown in Figure 3. The comparison of the growth parameters between all sample, males and females are presented in Table 2. The representation of growth curve is provided in Figure 3. The blue growth curves are superimposed on black size frequency distributions of the samples. Each blue curve illustrates the progression of a cohort or age class that appeared successively (on the same date).
Age and Growth
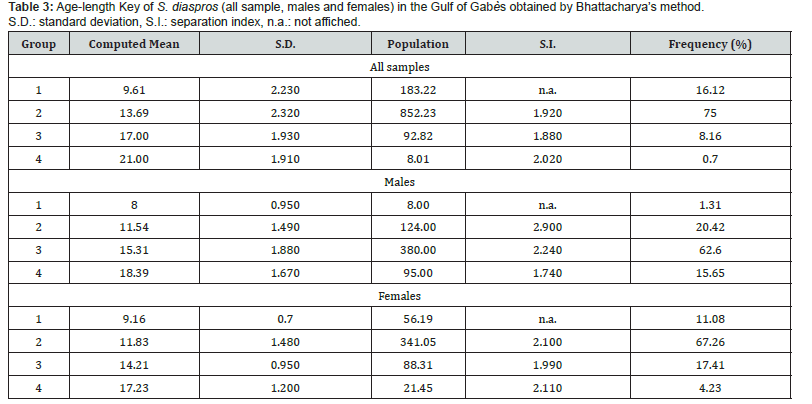
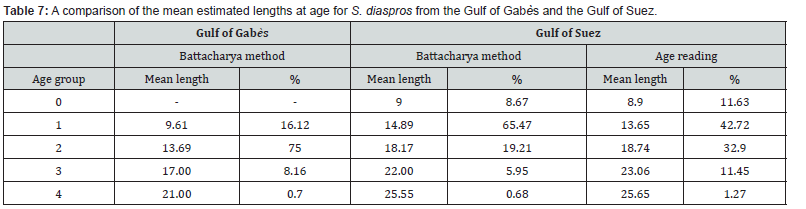
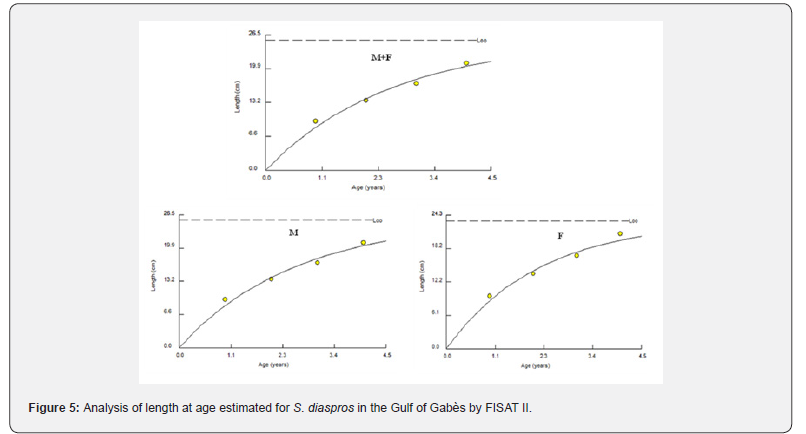
Plotting the decomposition of the length frequency distribution (Figure 4) clarified four age groups; I, II, III and IV; for all sample, males and females of the lessepsian specie S. diaspros in the Gulf of Gabès. Group III was predominantly represented among males (62.6 %), whereas Group II was more prevalent among combined sexes and females (67.26 %) (Table 3).
The estimated mean length at age allowed us to plot lengthat- age curves for the entire sample and for males and females separately (Figure 5).
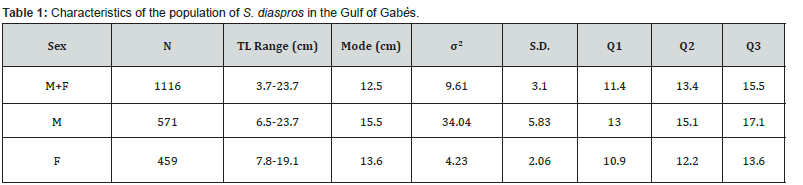
σ²: variance; S.D.: standard deviation, Q1 (First Quartile): the 25th percentile. It marks the value below which 25% of the data fall; Q2 (Second Quartile or Median): This is the 50th percentile. It divides the data set into two equal halves; Q3 (Third Quartile): This is the 75th percentile. It marks the value below which 75% of the data fall.
Mortality, Exploitation, and Survival
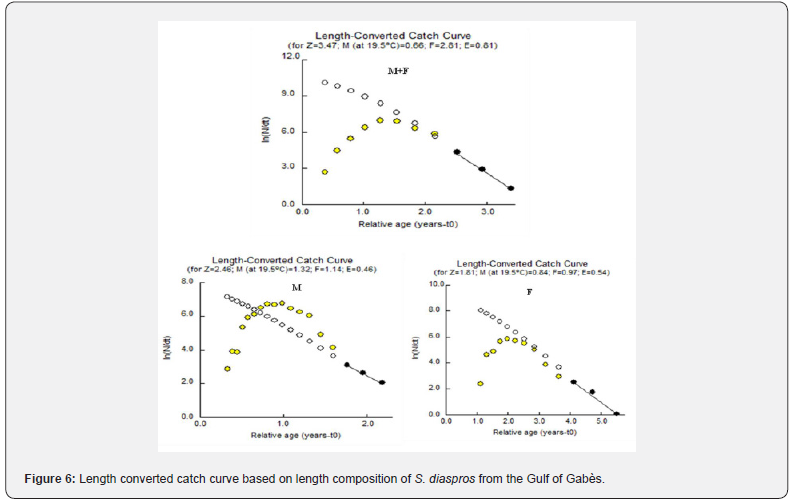
Three instantaneous mortality rates for S. diaspros populations can be calculate: The total instantaneous mortality (Z ) can be estimated using the L-converted catch curve method in FiSAT II software. Length-based catches with extrapolated data points are represented in Figure 6. The points represent the natural logarithm of the numbers per age class. The white points represent the theoretical values that should be considered. The yellow points are not considered because they are either too close to or too far from the asymptotic length. The black points are those used for the least squares calculation of the linear regression. These black points are used for the estimation of total mortality.
The slope of the line produced by the formula LnN / Δt = −3.47t +13.091 for all sample, LnN / Δt = −2.46t + 7.428 for males and In N / Δt = −1.81t +10.102 for females represents the estimated values of Z (Table 4). The values obtained with a mean surface temperature of 19.5°C are total mortality (Z ) , natural mortality (M) , fishing mortality (F) and exploitation rate (E) in the Gulf of Gabès marine waters. The exploitation rate (E) for all sample, males and females are respectively 0.81, 0.46 and 0.54) (Figure 6, Table 4).
Recruitment and First-Capture Size
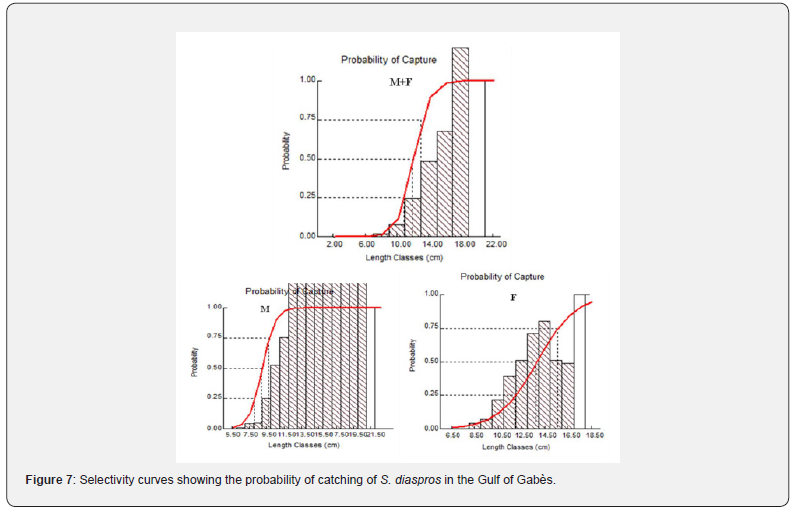
The potential length of 50% of the population being captured was estimated as 11.95 cm for all sample, 8.86 cm for males and 13.77 cm for females (Figure 7). The length at first sexual maturity (Lm) was found to be 21.4 cm for all sample, 16.91 cm for males and 13.76 cm for females, in addition to 21.64 cm for the optimum length (Lop ) for all sample, 18.4 cm for males and 12.74 cm for females, which generate the maximum sustainable yield (Figure 7, Table 4).
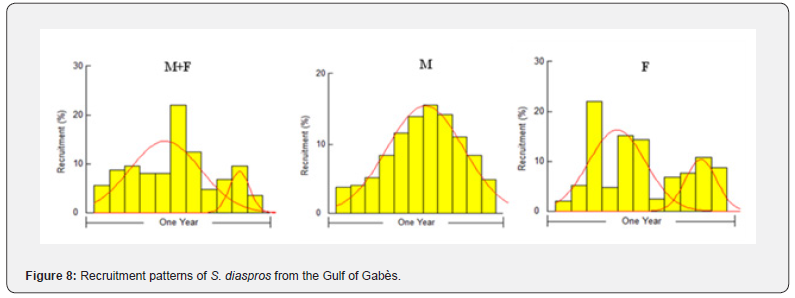
The recruitment histogram for S. diaspros shows continuous recruitment throughout the year (Figure 8), with a significant peak in the months of June-July for all sample and males but females show 2 peaks in June-July and another in October.
Relative yield-per-recruit (Y’/R) and relative biomass-perrecruit (B’/R): Beverton and Holt Y’/R analysis
These exploitation rate of S. diaspros were obtained by the knife edge selection hypothesis in the B-H dynamic equation. The Y’/R and B’/R values were evaluated according to Lc / L∞ and M / K . The analytical model of relative yield per recruit was applied to clarify the current and optimal status of S. diaspros stock in the Gulf of Gabès (Table 5). The value of relative yield per recruit (Y’/R) was 0.011 for all samples, 0.033 for males and 0.021 for females and the value of relative biomass-per-recruit (B’/R) was 0.032 for both sexes, 0.223 for males and 0.357 for females.
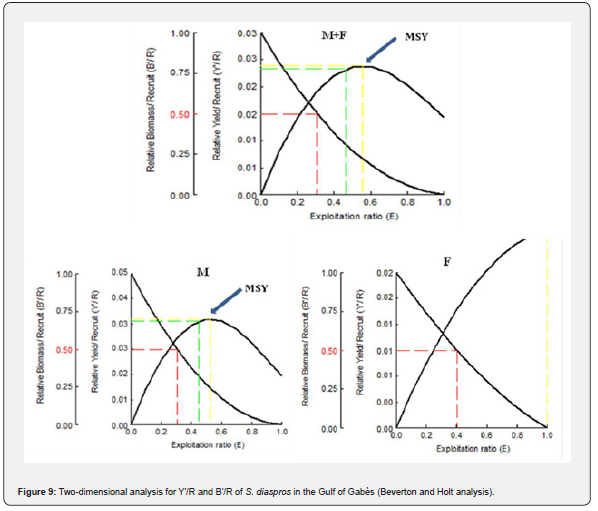
The values of E0.1 , E0.5 and Emax were resumed in Table 5. The peak relative yield per recruit (Y’/R) occurred at Emax, signifying the maximum sustainable yield (MSY). The findings demonstrate that the exploitation rate (E = 0.81) surpasses the threshold that would yield the maximum relative yield per recruit (Emax or MSY = 0.557) and E0.5 for all samples (Figures 9 & 10).
Virtual population analysis
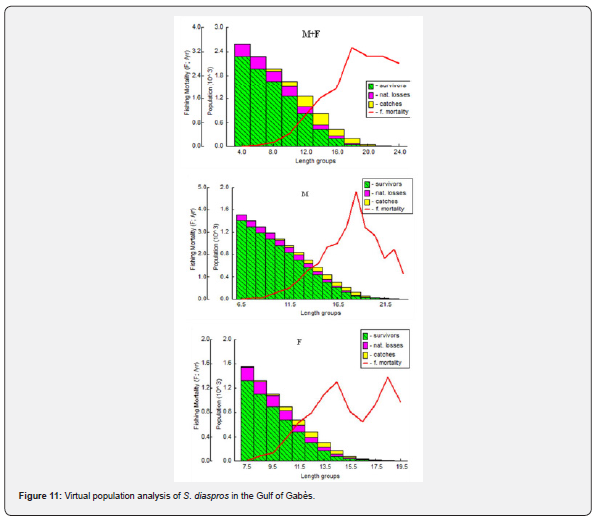
Virtual population analysis (VPA) provides valuable information on the proportion of survivors and the losses attributed to both natural mortality and fishing activities which are assessed by plotting various length groups against fishing mortality. In the present study, survival rates and natural mortality decreased as body length increased were observed for both males and females (Figure 11). The highest catch rates were recorded in the length groups of 10 to 18 cm for combined sexes, between 11.5 and 18.5 cm for males and between lengths classes 10.5 to 14.5 cm for females.
Fishing mortality also increased after a length of 8 cm, 9.5 cm and 7.5 cm for combined sexes, males and females respectively and attained a peak at a mid-length of 18 cm for all samples and 18.5 for males. For females we find two peaks in mid length 14.5 and 18.5 cm.
Discussion
The growth pattern, exploitation level, recruitment capacity, and mortality are among the key factors influencing fish population dynamics. Aside from the study by El-Ganainy and Sabra [25], which remains the only comprehensive research to date on the population dynamics of this species in the Gulf of Suez, no prior studies or assessments have been conducted on this parameter for the alien fish S. diaspros in the Red Sea or in the Mediterranean Sea. As such, the present findings represent the first available data on the overall status of this species in the study area.
S. diaspros, commonly referred to as filefish, is a pelagic species abundant along the coastline of the Gulf de Gabès. It is harvested year-round but no holds considerable financial significance in Tunisian’s markets. Despite not being widely recognized as a food fish, it shows significant potential as an alien species. This makes the study of its population dynamics and related characteristics tend to be imperative, forming the basis for the current research focus.
Utilizing the length-frequency data, the current study provides inferences on the population of S. diaspros in the Gulf of Gabès. We have examined the fundamental elements necessary to formulate a rational and sustainable management strategy for this species stock. Among 1116 sampled individuals, with size frequency distribution from 3.7 to 23.7 cm, the overall sex ratio (SR = 0.87) indicated a significant disparity, favoring males. The proportion of females is 46.53% and the proportion of males is 53.47%. Kartas and Quignard [51] suggest that the physiological condition of fish and fluctuations in the quantity and quality of sex-specific food may account for variations in the sex ratio and the differential susceptibility of each sex to the employed fishing gear.
S. diaspros exhibits clear sexual dimorphism, with noticeable differences between males and females. In adult males, the second spine of the second dorsal fin is elongated, and the caudal peduncle often features several rows of horny patches. Additionally, males tend to be larger in body size compared to females. In this study, the average length of males was 15.03 m with the predominance of 15.5 cm, while that for females, the average length was 12.32 cm and predominance of 13.6 cm. The largest sizes were 23.7 cm and 19.1 cm for males and females respectively. El-Ganainy and Sabrah [25] found that the total length of S. diaspros ranged from 7 to 26.1 cm with an average of 14.59±3.61 cm. In their biological studies of the filefish S. diaspros from the Gulf of Suez (Egypt), El- Ganainy and Sabrah [24] reported that females were represented in small sizes (8.0 - 16.0 cm), while males largely outnumber females up to the length interval of 16.0 - 26.0 cm. They also found a monthly sex ratio of S. diaspros that showed a predominance of males over females nearly during all the fishing season, and in sizes larger than 16 cm.
Variations in sex composition among fish populations can be influenced by several factors, including differences in natural and fishing mortality rates between sexes, or seasonal segregation of males and females. This segregation may also result from differences in age and size at sexual maturity [52]. In the case of the filefish, the notably high proportion of males is likely due to the disappearance of females following spawning [53], as well as selective fishing practices that may disproportionately affect one sex.
The growth performance index (φ ') for the combined sexes was 2.62, with an asymptotic length (L∞) of 37 cm and a growth coefficient (K) of 0.31. This value reflects a moderate growth rate, typical of an invasive species adapting to a new environment. When considering the sexes separately, males exhibited a higher growth performance index of 2.813, with a smaller L∞ of 28.5 cm and a significantly higher K value of 0.8, indicating a faster growth rate compared to females. In contrast, females showed a lower growth performance index of 2.26, with a smaller L∞ of 22.65 cm and a K value of 0.36, indicating slower growth. The higher growth performance index for males suggests that they have a greater capacity for rapid growth, which may enhance their ability to adapt and proliferate in the new ecosystem. These differences in growth rates and performance indices between sexes are crucial for understanding the invasive potential of this species, highlighting its ability to thrive and potentially impact local biodiversity in the Gulf of Gabès.
The asymptotic length happens to be a vital factor for demarcating the mesh size limits of fishing gears [54]. The asymptotic length is considerably higher in the combined population compared to males and females separately, which can be attributed to the presence of numerous small-sized individuals of undetermined sex. Furthermore, this value is higher in males than in females, as previously mentioned, due to the generally greater length of males. This observation also explain the notably higher growth rate recorded in males (K=0.80) compared to females (K=0.33) and the combined population (K=0.31) .
The size distribution of a fish population can be influenced by several external factors, with fishing methods and water temperature playing particularly important roles [55]. These factors, along with the type of data models used in analysis, can ultimately affect estimates of growth rates and the maximum size that fish in the population are expected to reach [56].
The L∞ value estimated in our study is considerably higher than that reported by El-Ganainy and Sabrah [25] (L∞ = 27.8) , while the K values in both studies remain relatively similar. A comparative summary of the Von Bertalanffy Growth Function (VBGP) parameters for the Gulf of Gabès and the Gulf of Suez is presented in Table 6.
The identification of four age groups (I to IV) in the lengthfrequency distribution of S. diaspros indicates a stable and continuous recruitment within the population in the Gulf of Gabès. This structure suggests successful establishment and adaptation of this Lessepsian migrant in the Mediterranean environment. The presence of multiple age classes reflects ongoing reproduction and survival across different cohorts, which is essential for maintaining population resilience. The dominance of Group II among males and Group III among females may reflect differences in growth rates, mortality, or maturation between sexes. Such age distribution patterns are crucial for understanding the population dynamics and for developing effective management strategies.
In the study conducted on S. diaspros from the Gulf of Suez, five age classes were identified using two complementary methods: the examination of growth rings on thin sections of the dorsal fin spine and the Bhattacharya method. The results indicated rapid growth during the first year of life, with individuals reaching nearly 50% of their estimated maximum length. In subsequent years, growth rate declined considerably. A comparison of the mean estimated lengths at age for S. diaspros from the Gulf of Gabès and the Gulf of Suez is presented in Table 7.
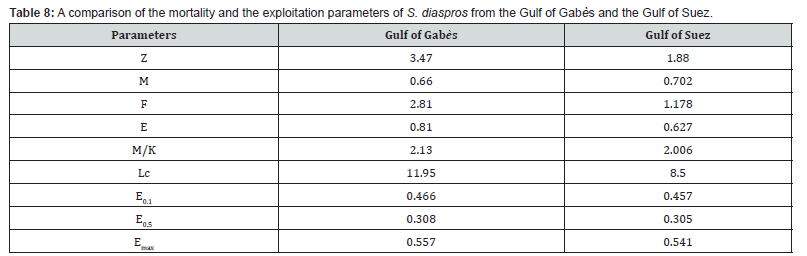
The total (Z ) , natural (M) , and fishing (F) mortalities for S. diaspros along the coast of the Gulf of Gabès waters were estimated at 3.47, 0.66, and 2.81 yr-1, respectively. In this case, the value of fishing mortality is very higher than natural mortality, for all samples but closed for males and females. Also, the exploitation rate (E) of our species is higher than the reference exploitation rate (E = 0.5) which symbolize overexploitation and an anthropogenic action. This implies that the mortality of most of these fish is linked to fishing and not to predation or habitat and physicochemical characteristics. Human harvesting is therefore responsible for stock fluctuation in this ecosystem. According to Al-Nahdi et al. [57], the maximum level of exploitation of a resource is reached, when the exploitation rate is greater than or equal to 0.5, or when fishing mortality (F) is equal to or greater than natural mortality (M) . It is therefore possible to affirm that this fish species, considered as by-catch is overexploited in the Gulf of Gabès. In addition, the Z/K ratio (11.19 for all sample, 3.075 for males and 5.028 for females) greater than 1 again confirms high fishing-related mortality. A Z/K ratio greater than 1 suggests that mortality surpasses growth, potentially indicating overexploitation. Conversely, a ratio less than 1 implies that growth outpaces mortality, which is characteristic of a healthy, growing population [58].
The opening catch, L50 , of S. diaspros was 11.95 cm. Specimens ≤ 11.95 cm can be considered a supplementary population. It’s considered one of the most important premises for analyzing the relationship between the relative yield per recruit (Y’/R) and E. This is bound to lead to very intense exploitation of this resource, as many of the young individuals have no chance of reaching optimum growth size. As the size of first capture ( Lc or L50 ) is lower than the size of first maturity Lm , we can say that this is a case of biological overexploitation. A fishery focused on the spawning stock generally leads to such overexploitation.
The recruitment histogram for S. diaspros indicates yearround recruitment, with a marked peak in June–July for the overall population and males, while females display two distinct peaks: one in June–July and another in October. These months, included in the major upwelling, are thought to be favorable for young fish feeding, thanks to the planktonic proliferation during this period, whereas recruitment tends to be lowest during the winter months, likely due to the drop in water temperature [59]. The availability of food and favorable environmental conditions are among the key factors that can significantly influence fish recruitment [60-64].
According to Ktari-Zouari et al. [21], the gonadosomatic index (GSI) of female S. diaspros shows significant variation during the spawning season, which extends from July to December, with two noticeable peaks in July and October. The overall pattern of females GSI suggests a prolonged breeding period lasting from April to December, with fluctuations throughout. The development of ovaries and the growth of oocytes in fish may be influenced by changes in water temperature. Spawning season of S. diaspros typically begins in July, coinciding with rising summer temperatures, suggesting that temperature shifts may play a key role in triggering ovarian development in this alien species. Additionally, there appears to be a strong link between photoperiod and GSI, as both reach their highest levels at the end of spring and the start of summer [65,66]. Similar to females, the GSI values of males reached a peak in July [21].
The current Y’/R and B’/R values for S. diaspros in the Gulf of Gabès (0.011 and 0.032 for both sexes, 0.033 and 0.223 for males and 0.021 and 0.357 for females) indicate that the population, particularly males, is under significant fishing pressure, leading to reduced yield and biomass per recruit, potentially compromising the stock’s sustainability. Implementing targeted management strategies is essential to ensure the long-term viability of the species and the fishery.
Also, the examination of Y’/R and B’/R indicates that the current exploitation level (E = 0.81) substantially exceeds both Emax and E0.5 (0.557 and 0.308, respectively). The reduction of exploitation rate should be occurred from 0.81 to 0.308 with a rate of 49.8% to ensure effective management and to sustain the spawning stock biomass. Similarly, El-Ganainy and Sabrah [25] reported an exploitation ratio that exceeded the level associated with the maximum relative yield per recruit (Emax=0.541) by approximately 16%, indicating that fishing pressure in the Gulf of Suez had surpassed the critical threshold. Furthermore, they found that the estimated length at first capture (Lc = 8.5 cm) was very close to the length at first sexual maturity (9.0 cm) [24], suggesting that juvenile individuals are heavily targeted by the fishery. This early exploitation poses a serious threat to the stock dynamics of the species, as the removal of immature individuals can significantly reduce future yields. The high vulnerability of juvenile fish to trawl fishing highlights the urgent need for protective measures. Safeguarding juvenile fish, particularly through the periodic spatial closure of spawning and nursery grounds, is likely a key factor for ensuring the longterm sustainability of the resource. This could be effectively implemented by establishing marine reserves within the Gulf of Suez aimed at protecting the spawning stock biomass, followed by continuous monitoring to evaluate the effectiveness of such management strategies.
A comparison of the mortality and the exploitation parameters of S. diaspros from the Gulf of Gabès and the Gulf of Suez were resumed in Table 8.
The yield isopleths categorize the S. diaspros fishery in quadrant D for all population and in quadrant C for males and females separately [50], signifying the inclusion of juvenile fish in the landings throughout the study period (growth and recruitment overfishing).
Hard work must be done to reduce fishing effort in general and tighten control over landing sites and fishing ports to ensure compliance with the minimum allowable size of the catch. Thus, strengthen the monitoring and regulation of shallow fishing sites in order to better preserve our fishery resources and ensure the sustainability of marine ecosystems [67].
This finding is consistent with the results obtained from the virtual population analysis (VPA). Thus, Virtual population analysis provides valuable insights into the proportion of survivor and the losses resulting from both natural causes and fishing pressure. This is achieved by comparing different length groups against levels of fishing mortality. In the present study, the number of survivors and natural losses decreased as fish size increased. However, fishing-related mortality rose sharply beyond a length of 18 cm for the overall sample, 18.5 cm for both males and females, and showed an additional peak at 14.5 cm specifically for females. The highest catch rates were recorded in fish measuring between 10 and 18 cm overall, 11.5 to 18.5 cm for males, and 10.5 to 14.5 cm for females (Figure 11).
Therefore, we can note that the stock of S. diaspros, especially the females, caught off the Gulf of Gabès is overexploited and cannot achieve a sustainable fishing rate. This reflects a tendency to catch immature or maturing fish. Raising awareness among fishers about the negative impact of this practice could gradually change their behaviour and motivations.
References
- Coll M, Piroddi C, Steenbeek J, Kaschner K, Lasram BRF, et al. (2010) The Biodiversity of the Mediterranean Sea: Estimates, Patterns, and Threats. PLOS ONE 5(8): e11842.
- Katsanevakis S, Nikolaou A, Tsirintanis K, Rilov G (2025) Lessepsian migration in the Mediterranean Sea in an era of climate change: Plague or boon? Science Talks, 13.
- Golani D, Öztürk B, Baflusta N (2006) Fishes of the Eastern Mediterranean. In: Istanbul: Turkish Marine Research Foundation pp. 265.
- Whitehead PJP, Bauchot ML, Hureau JC, Nielsen J, Tortonese E (1986) In: (Eds.) Fishes of the North-eastern Atlantic and the Mediterranean. Paris: UNESCO. 2.
- Froese R, Pauly D (2019) FishBase. World Wide Web electronic publication. www.fishbase.org, version.
- Metin G, Akyol O (2021) Maximum size of Stephanolepis diaspros (Tetraodontiformes: Monacanthidae). Mar Sci Technol Bull 10(1): 23-27.
- Zouari KR, Bradaï MN, Bouain A (2008) The feeding habits of the Lessepsian fish Stephanolepis diaspros (Fraser-Brunner, 1940) in the Gulf of Gabès (eastern Mediterranean Sea). Cah Biol Mar 49(4): 329-335.
- Froese R, Pauly D (2014) FishBase. In: World Wide Web electronic publication. Retrieved from www.fishbase.org.
- Golani D, Relini OL, Massutí E, Quignard JP (2002) CIESM atlas of exotic species in the Mediterranean. Fishes. CIESM Publishers, Monaco 1.
- Por FD (1978) Lessepsian Migration: The Influx of Red Sea Biota into the Mediterranean by Way of the Suez Canal. In: Ecological Studies, Springer-Verlag, Berlin-Heidelberg. 23.
- Steinitz W (1927) The migration of Indo-Pacific species into the Mediterranean since the beginning of the Quaternary period Int Rev Gesamten Hydrobiol 22(1): 1-90.
- Taskavak E, Bilecenoglu M (2001) Length–weight relationships for 18 Lessepsian (Red Sea) immigrant fish species from the eastern Mediterranean coast of Turkey. J Mar Biol Assoc U K 81(5): 895-896.
- Sangun L, Akamca E, Akar M (2007) Weight–Length Relationships for 39 Fish Species from the North-Eastern Mediterranean Coast of Turkey. Turk J Fish Aquat Sci 7(1): 37-40.
- Catalano E, Zava B (1993) On the presence of Stephanolepis diaspros Fr. Brunn in Italian waters (Osteichthyes, Monacanthidae). Suppl Ric Biol Selvaggina 21: 379-382.
- Dulčić J, Pallaoro A (2003) First record of the filefish, Stephanolepis diaspros (Monacantidae), in the Adriatic Sea. Cybium 27(4): 321-322.
- Deidun A, Castriota L, Andaloro F, Maraventano G, Prazzi E, et al. (2015) Documenting the occurrence of the Lessepsian fish Stephanolepis diaspros within the Strait of Sicily (central Mediterranean). BioInv Rec 4(2): 103-107.
- Chakroun F (1966) Capture of rare animals in Tunisia. Bull Inst Natn Scient Tech Oceanogr Pêche Salammbo NS 1(2): 75-79.
- Bradaï M N (2000) Diversity of the fish population and contribution to the knowledge of sparids in the Gulf of Gabè PhD Thesis, University of Sfax, Tunisia.
- Bradaï MN, Quignard JP, Bouain A, Jarboui O, Ouannes GA, et al. (2004) Native and exotic ichthyofauna of the Tunisian coasts: inventory and biogeography. Cybium 28 (4): 315-328.
- Zouari KR, Ghorbel M, Bradaï MN, Ghorbel OA, Jarboui O, et al. (2003) Analysis of two benthic trawling campaigns carried out in the Gulf of Gabès region. Bulletin of the National Institute of Marine Sciences and Technologies. Proceedings of the 6th Conference of the Tunisian Association of Marine Sciences. Tunis (Tunisie) pp. 99-102.
- Zouari KR, Bradaï MN (2011) Reproductive biology of the Lessepsian filefish Stephanolepis diaspros (Fraser-Brunner, 1940) in the Gulf of Gabès (southern Tunisia). Rev Fish Biol Fish 21(3): 641-648.
- Bdioui M, Ben Amor MM, Capapé C (2004) Occurrence of Stephanolepis diaspros (Osteichthyes: Monacanthidae) in the Lagoon of Bizerte (northern Tunisia). Cah Bio Mar 45(4): 373-375.
- Ben Amor MM, Capapé C (2008) Occurrence of a filefish closely related to Stephanolepis diaspros (Osteichthyes: Monacanthidae) off northern Tunisian coast (south-western Mediterranean Sea). Cah Bio Mar 49: 323-328.
- El-Ganainy AA, Sabrah MM (2013) Biological studies on the filefish Stephanolepis diaspros, Fraser-Brunner, 1940 (Monacanthidae) from the Gulf of Suez, Egypt. Egypt J Aquat Biol Fish 17(1): 113-121.
- El-Ganainy AA, Sabrah MM (2008) Age, Growth, Mortality and Yield per Recruit of the Filefish Stephanolepis diaspros (Fraser-Brunner, 1940) (Pisces: Monacanthidae), in the Gulf of Suez, Egypt J Fish Aquat Sci 3(4): 252-260.
- Hattour MJ, Sammari C, Nassrallah BS (2010) Hydrodynamics of the Gulf of Gabès deduced from observations of currents and levels. Revue Paralia 3: 1-16.
- UNEP/MAP (2012) State of the Mediterranean Marine and Coastal Environment 2012. United Nations Environment Programme / Mediterranean Action Plan.
- Enajjar S, Bradai MN, Bouain A (2009) The Gulf of Gabès: a nursery area for sharks and rays. In Proceedings of the 13th EEA Conference, Palma de Mallorca, Spain. Program and Abstracts, p. 76.
- UNEP-MAP RAC/SPA (2010) The Mediterranean Sea Biodiversity. Initiative-PIM.
- El Zrelli R, Rabaoui L, Ureta RRH, Gallai N, Castet S (2020) Economic impact of human-induced shrinkage of Posidonia oceanica meadows on coastal fisheries in the Gabès Gulf (Tunisia, Southern Mediterranean Sea). Mar Pollut Bull 155: 111124.
- Dauvin JC, Fersi A, Pezy JP, Bakalem A, Neifar L (2021) Macrobenthic communities in the tidal channels around the Gulf of Gabès, Tunisia. Mar Pollut Bull 162: 111846.
- IUCN (2024) Mediterranean and Black Seas. IUCN Portals. IUCN Portals+1RAC-SPA+1.
- Gayanilo FC, Sparre P, Pauly P (2005) The FAO-ICLARM Stock Assessment Tools (FiSAT II). Revised version. User’s manual. FAO Computerized Information Series (Fisheries), Rome: World fish Center, FAO. 7: 168.
- Bhattacharya CG (1967) A simple method of resolution of a distribution into Gaussian components. Biometrics 23: 115-135.
- Pauly D, Morgan GR (1987) Length-based methods in fisheries research. In: (Eds.) Proceedings de la conference ICLARM No. 13. International Center for Living Aquatic Resources Management (ICLARM), Manille, Philippines.
- Von Bertalanffy LV (1938) A quantitative theory of organic growth (inquiries on growth laws II). Hum Biol 10(2): 181-213.
- Pauly D (1983) Length-converted catch curves: A powerful tool for fisheries research in the tropics (part I). Fishbyte 1(2): 9-13.
- Pauly D (1980) A selection of simple methods for the assessment of tropical fish stocks. FAO Fisheries Circular, No. 729. FAO.
- Gulland J A (1971) Fish Resources of the Ocean. Fishing New Books, London. pp. 255.
- Zhao C, Cong YF, Wang L, Song D, Du X, et al. (2022) Growth characteristics and stock assessment of perch Perca fluviatilis in Lianhuan Lake. Chin J Fish 35(6): 66-74.
- Camp E, Collins AB, Ahrens RN, Lorenzen K (2020) Fish Population Recruitment: What Recruitment Means and Why It Matters. EDIS 2: 1-6.
- Moreau J, Palomares MLD, Torres FSB, Pauly D (1995) Demographic Atlas of Freshwater Fish Populations in Africa. ICLARM Rapport Technique, 45: 140.
- Gayanilo FC, Sparre P, Pauly D (1995) FAO-ICLARM Stock Assessment Tools (FiSAT) User’s Guide. FAO Computerised Information Series (Fisheries), Rome 8: 126.
- Dadzie S, Seedo AF, Moreau J (2007) Population dynamics of Parastromateus niger in Kuwaiti waters as assessed using length–frequency analysis. J Appl Ichthyol 23(5): 592-597.
- Pauly D (1984a) Fish Population Dynamics in Tropical Waters: A Manual for Use with Programmable Calculators. ICLARM Studies and Reviews 8: 325.
- Pauly D (1984b) Length-converted catch curves. A powerful tool for fisheries research in the tropics. (Ill: conclusion). ICLARM Fishbyte 2(3): 9-10.
- Froese R, Binohlan C (2005) Empirical relationships to estimate asymptotic length, length at first maturity, and length at maximum yield per recruit in fishes, with a simple method to evaluate length frequency data. J Fish Biol 56(4): 758-773.
- Beverton RJH (1992) Patterns of reproductive strategy parameters in some marine teleost fishes. J Fish Bio 41: 137-160.
- Beverton RJH, Holt S J (1966) Manual of methods for fish stock assessment part II tables of yield function. FAO Fish Biology Technical Paper 38(4): 10.
- Pauly D, Soriano ML (1986) Some practical extensions to Beverton and Holt’s relative yield-per-recruit model. In: Maclean JL, Dizon LB, Hosillos LV (eds.), The First Asian Fisheries Forum. Asian Fisheries Society, pp: 491-496.
- Kartas F, Quignard JP (1984) The Fertility of Teleost Fish. Paris : Masson. Collection de Biologie des Milieux Marins, 5.
- Smith SH (1956) Life history of lake herring of Green Bay, Lake Michigan. Fish Bull 109: 87-138. U.S. Fish and Wildlife Service.
- Nakazono A, Kawase H (1993) Spawning and biparental egg-care in a temperate filefish, Paramonacanthus japonicus (Monacanthidae). Environ Biol Fishes 37(3): 245–256.
- Gebremedhin S, Bruneel S, Getahun A, Anteneh W, Goethals P (2021) Scientific methods to understand fish population dynamics and support sustainable fisheries management. Water 13: 574.
- Tu CY, Chen KT, Hsieh CH (2018) Fishing and temperature effects on the size structure of exploited fish stocks. Scientific Reports 8: 7132.
- Etim L, Sankare Y, Brey T, Arntz W (1998) The dynamics of unexploited population of Corbula trigona (Bivalvia: Corbulidae) in a brackish-water lagoon, Côte d'Ivoire. Arch Fish Mar Res 46(3): 253-262.
- Al-Nahdi A, Al-Marzouqi E, Al-Rasadi JC, Groeneveld J (2009) Size composition, reproductive biology, age and growth of largehead cutlassfish (Trichiurus lepturus) Linnaeus from the Arabian Sea coast of Oman. Ind J Fisheries 56(2): 73-79.
- Allen MS, Hightower JE (2010) Chapter 2: Fish Population Dynamics: Mortality, Growth, and Recruitment. In: American Fisheries Society p. 43-79.
- Kindong R, Gao C, Dai X, Tian S, Wu F (2018) Population dynamic parameters for Cyprinus carpio in Dianshan Lake. Thalassas: Int J Mar Scien 34: 279-288.
- Shoji J, Toshito SI, Mizuno KI, Kamimura Y, Hori M, et al. (2011) Possible effects of global warming on fish recruitment: Shifts in spawning season and latitudinal distribution can alter growth of fish early life stages through changes in daylength. ICES J Mar Sci 68(6): 1165-1169.
- Okamoto DK, Schmitt RJ, Holbrook SJ, Reed DC (2012) Fluctuations in food supply drive recruitment variation in a marine fish. Proceedings of the Royal Society B: Biological Sciences 279 (1747): 4542-4550.
- Tableau A, Brind'Amour A, Woillez M, Le Bris H (2016) Influence of food availability on the spatial distribution of juvenile fish within soft sediment nursery habitats. J Sea Res 111: 76-87.
- Gebrekiros ST (2016) Factors affecting stream fish community composition and habitat suitability. J Aquacult Mar Biol 4(2): 00076.
- Kripa V (2017) Role of environmental variables on spawning and recruitment of small pelagics in an upwelling system. In: course manual summer school on advanced methods for fish stock assessment and fisheries management. CMFRI, Kochi pp: 289-295.
- Brewer GD (1978) Reproduction and spawning of the northern anchovy, Engraulis mordax, in San Pedro Bay, California. Calif. Fish Game 64: 175-184.
- Chen CT, Ma MC, Liu KM (1993) Ovarian cycling of Japanese barracuda Sphyraena japonica in northeastern Taiwan waters. J Fish Soc Taiwan 20: 301-311.
- Pauly D, Munro JL (1984) Once more on the comparison of growth in fish and invertebrates. Fishbyte, The WorldFish Center 2(1): 1-21.






























