Burrow Construction Morphology of Ocypode Rotundata Miers 1882 (Ocypodidae: Brachyura) from the Sandy Coastal Areas of Karachi, Pakistan
Sahir Odhano1*, Noor Us Saher2, Michael S Rosenberg3, Mohammad Farooque Hassan4 and Hidayatullah Soomro5
1Department of Fisheries and Aquaculture, Shaheed Benazir Bhutto University of Veterinary and Animal Sciences (SBBUVAS), Sakrand, Pakistan
2Centre of Excellence in Marine Biology, University of Karachi, Pakistan
3Center for Biological Data Science, Virginia Commonwealth University, USA
4Department of Pathology, Shaheed Benazir Bhutto University of Veterinary and Animal Sciences (SBBUVAS), Sakrand, Pakistan
5Department of Poultry Science, Shaheed Benazir Bhutto University of Veterinary and Animal Sciences, Sakrand, Pakistan
Submission:November 12, 2022; Published:December 05, 2022
*Correspondence author: Sahir Odhano, Department of Fisheries and Aquaculture, Shaheed Benazir Bhutto University of Veterinary and Animal Sciences (SBBUVAS), Sakrand, Pakistan
How to cite this article: Sahir O, Noor Us S, Michael S R, Mohammad Farooque H, Hidayatullah S. Burrow Construction Morphology of Ocypode Rotundata Miers 1882 (Ocypodidae: Brachyura) from the Sandy Coastal Areas of Karachi, Pakistan.Oceanogr Fish Open Access J. 2022; 15(4): 555917. DOI: 10.19080/OFOAJ.2022.15.555917
Abstract
Burrow morphology of ghost crab Ocypode rotundata was studied at two stations of Sandspit beach, Karachi. A quadrate method Two-meter square (2m2) was used for burrow cast profiling. Burrow casts were shaped by pouring plaster of Paris (liquified 2:1 ratio) into the ghost crab burrows. The Burrow casts and sediment samples were collected monthly from the studied site from Jan 2012 to Dec 2013. Besides this effect of human trampling on the sandy beach was also studied. Total Six types of burrow structures were observed (I, Y, C, L, J and multiple branched (M-type)), with burrow depth between 460 mm to 1300 mm. The most common casts collected were straight I- shaped (36.8%), while M- type casts were the rarest (5.7%). All the burrow openings were found positively correlated with carapace length (r2 = 0.81). The fine grain found dominated during grain size analysis from both stations. During anthropogenic survey, the effect of human trampling was less due to private huts as compared to the control area where a smaller number of burrows were found. Current study revealed strong relationship of ghost crabs with environment, therefore, may be useful to study as ecological indicators for sandy beaches.
Keywords: Anthropogenic impact; Burrow morphology; Ghost crabs; Grain size analysis; Human trampling; Sandspit; Sediment analysis
Abbreviations: OD: Opening Burrow Diameter; TL: Total Length; TD: Total Depth; CL: Carapace Length; CW: Carapace Width; Ch L: Chela Length; TBL: Total Burrow Length
Introduction
Ghost crabs are semi-terrestrial Brachyura from the genus Ocypode Weber, 1795 (Family: Ocypodidae) [1-5]. These crabs are frequently found in tropical to sub-tropical areas along the sandy coasts of the world, including the Atlantic, Pacific, and Indian Oceans, as well as major seas such as the Mediterranean and Red Sea. [5-8]. O. rotundata Miers 1882 is abundant in the Indo-Pacific region, found in large quantities above the high tide mark on sandy shores [5,8-12]. This species is the most conspicuous macro scopic invertebrate inhabiting the sandy beaches found along the coasts of Pakistan [6].
Ghost crabs are relatively large and mostly observed during dawn and dusk, but juveniles may also be seen during the daytime because they cannot spend as much time inside their burrows as adults. Ghost crabs usually feed during the night and are considered omnivorous scavengers, feeding on macroscopic, microscopic, live or dead animal or plant materials [13].
Marine invertebrate organisms living in the soft sediments of marine coastal area have developed burrowing adaptability [14]. Burrow construction behaviour is predominantly species specific, but individuals of some species may alter burrow structure when encountering variation in sediment properties to better regulate their activities in the transformed environment [14-18]. Ghost crabs can construct deep and complex burrows up to two meters [11] which provide shelter against both climatic extremes and predators and serve as sanctuaries during moulting and while gravid [4,18]. Crabs in the family Ocypodidae show significant intraspecific variation during construction of burrows in response to different individual and environmental conditions, such as sediment structure, vegetation forms, coastal height, tidal difference, gender, and age of individual [4,19-21]. The burrow openings of ghost crabs are circular with neighbouring accumulated sand mounds and are generally surrounded by intense feeding lines [22]. There are clearly visible entrances to ghost crab burrows on the surface of sandy beaches maintained as territory [23] and counting these burrows serves as a measure of local ghost crab density [8,18,24-26].
Coastlines of the world are dominated by sandy beach ecosystems [27,28], but increasing human population is destroying natural sandy beach habitats at an accelerating rate [8,29]. Because ghost crabs are a key component of the sandy beach food chain, they can be used for the assessment of human impact on the beach environment [8,30]. In Pakistan, the significance of ghost crab burrow morphology and relative growth analysis on beaches has yet to be explored. In this study, we examined burrow structure morphology of O. rotundata from two stations of Sandspit coast of Karachi, Pakistan and the influence of human impact on burrow density.
Materials and Methods
Study area
The Pakistan coastal belt covers about 990km from Ketibunder to Jiwani Bay. The Karachi coast, located in Sindh Province, is approximately 100km in length [31]. The Karachi coast comprises of many sandy beaches and is well known for its picnic spots. Sandspit beach is one of the more popular beaches of Karachi, with wide areas of mangrove on its backshore. The backshore and foreshore of Sandspit beach are separated by a wide strip of road which connects the Hawksbay coastline with Manora beach island over a distance of about 20km [32]. The study was carried out at two stations of Sandspit: (S1) Hawksbay (24o51’ N 66o53’ E): located on Hawksbay drive opposite to WWF wetland centre regional office, and (S2) Manora (24o50’24” N 66o54’36” E): located at the southwest of Karachi on Manora drive near leftover of CEMB shore lab. Both sites contain large population of ghost crabs. The S1 site is also famous as a nesting site of the green turtle Chelonia mydas [32]. Each station was divided into an upper tidal zone and a lower tidal zone. The upper zone ranges from splash zone to surf zone (approx. 38 feet), while the lower zone ran from the surf zone to the lowest tide level (approx. 38 feet).
Sampling methodology
The present study was carried out over two years from Jan 2011 to Dec 2012. Data was collected through line transect methods in four months (March, April, August and September) of each year. Four transects were placed in each zone for burrow count and cast filling. The measurement was taken from high tide zone to low tide zone with a line transect method. Total two quadrats (2m2) were used in each tide level from high tide level to low tide level. Measurements included the diameter of burrow openings (OD), the distance of each burrow from the sea (DS) at low tide, and the total burrow count from each zone (TB). Burrow counts and cast filling were replicated during each visit, in order to search for any new form of burrow structure and examine human disturbance over time. An aqueous solution consisting of plaster of Paris and water with a 2:1 ratio was prepared in the field. This solution was emptied into the selected crab burrows until the burrows were filled completely with the solution and took 30 to 60 minutes to dry [4]. Few crabs emerged from burrows while filling with the solution; these crabs were tagged and placed into marked bags and brought to the laboratory for further analysis. On several occasions, crabs were trapped inside their burrows and could not emerge because of the depth, branching, or drying effect of the cast. The casts were excavated carefully as they become solidified and carefully taken to the laboratory for measurement of burrow proportions related with opening burrow diameter.
In the laboratory, casts were cleaned prior to measurement with a moderate size paint brush to remove dust. Measurements of each cast were recorded, including opening burrow diameter (OD), total length (TL), and total depth (TD), and associated captured crabs were measured for carapace length (CL), carapace width (CW) and enlarged chela length (Ch. L).
Sediment analysis
Sediment samples were collected near casting areas from a depth of 30cm for each quadrate replicate. Sediment samples were analyzed for sediment structure and morphology, including percent organic matter and grain size analysis [18]. For percent organic matter, 200g of sediment were placed into pre-weighted well labelled crucibles, placed into an oven at 70˚C for 5hrs, weighed again, and then placed into a furnace at 400˚C for 24hrs. After 24 hours, the crucibles were weighed a final time.
For grain size analysis, sediment was dried at room temperature and treated to have a permanently crumpled appearance. About 100g of sediment sample were taken from the dried sample and sieved through a sediment shaker machine with a standard mesh sized sieve for 15 minutes. Sediments retained on each sieve were collected, weighed, and analyzed to determine percent grain size.
Anthropogenic impact
A 20m2 area at a reference site and each station were chosen to estimate anthropogenic impact at each site. The reference site was a popular beach regularly visited for picnics. Ghost crab densities were calculated twice a month by counting the number of active burrows on the beach surface at each site. Burrows were counted by the line transect method. Four transects were positioned in each of the upper and lower shoe zones and burrow densities were observed as the number of burrows per transect. These counts were replicated during each visit, with counts carried out 8 times each year, twice a month in March, April, August, and September.
Statistical analysis
Linear regression was used to examine the relationship between the morphometric and burrow measures, with Student’s t-test used to compare the two sites. A one-way ANOVA was employed to test differences among the sampling locations and the control location.
Results
Burrow morphology
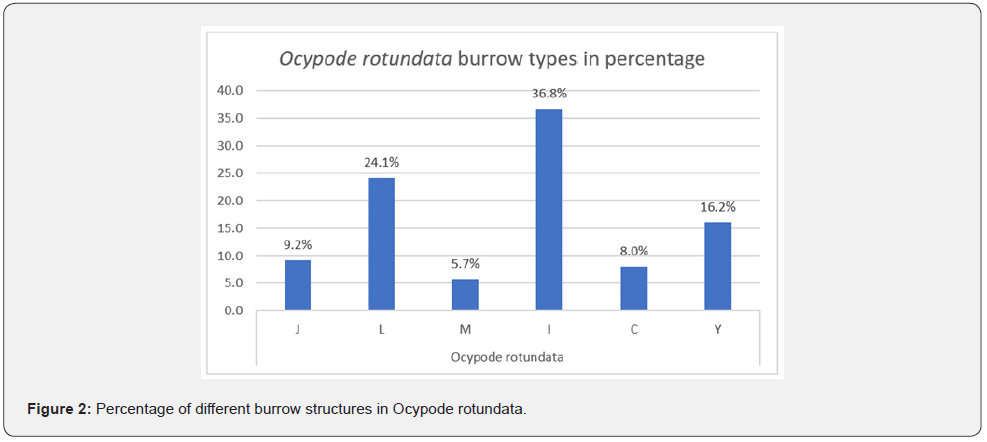
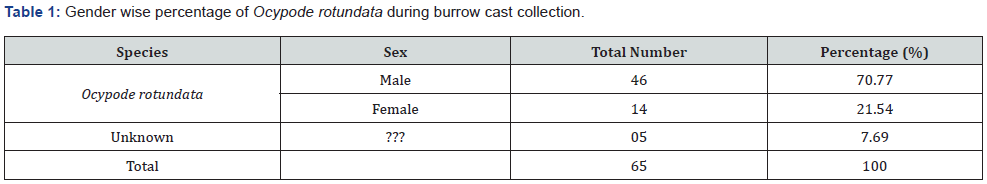
In total, 65 complete burrow casts of O. rotundata were collected. Male crabs were more common burrow occupants, with 46 males (70.77%) vs. 14 females (21.54%), with an additional five (7.69%) empty burrows (Table1). The casts included six types of burrow structures: J-, L-, C-, Y-, I-, and multiple branched (M-) (Figure 1). The most common casts collected were straight I- shaped (36.8%), while M- type casts were the rarest (5.7%) (Figure 2).
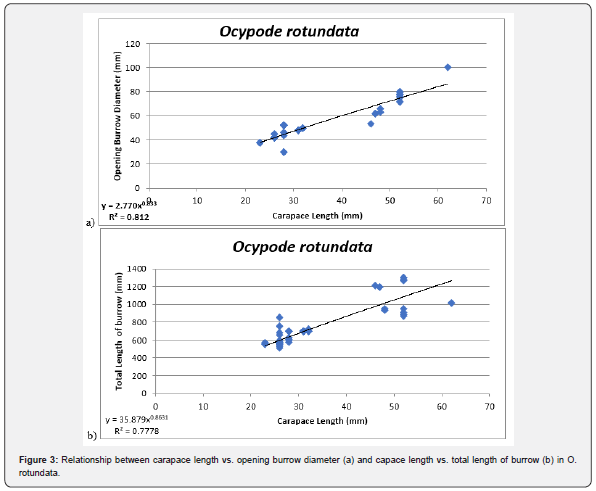
Linear regression analysis revealed positive allometric relationships between both burrows opening diameter (BOD) and total burrow length (TBL) with carapace length (CL) (R2= 81.2% and 80.7%) respectively (Figure 3).
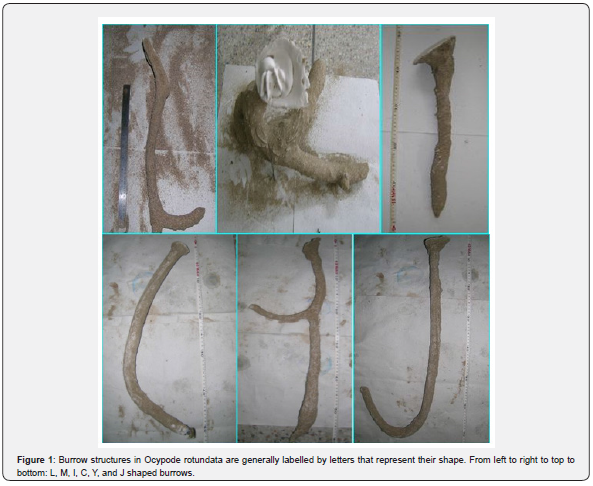
Using Student’s t-test, no significant differences were observed in crab size between the two study sites, whether measured by carapace length (p=0.58) or width (p=0.30). J-shaped burrows had the smallest volume with a mean opening diameter (OD) of 38.31mm (Table 2). The primary and secondary arms of Y shaped burrows joined together into a straight shaft and ended up in a chamber at the base (Figure 1). All the primary arms were found to be oriented towards seaside, with the secondary arms opposite the sea and not opened at the surface. The multiple branched burrow structure (M) had the largest OD (87.0 ± 67.7mm) despite containing the smallest size crabs (CL = 26.00mm (Table 2). The largest crabs tended to be found in I-shaped burrows (Table 2).
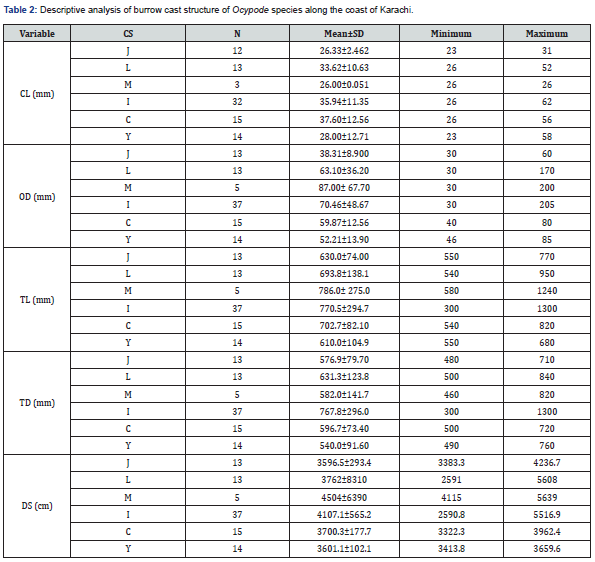
(OR: O. rotundata; CL: Carapace Length; OD: Opening Burrow Diameter; TL: Total Length of Burrow; TD: Total Depth of Burrow; CS: Cast Shape).
Sediment analysis
The yearly grain size analysis revealed that fine grain dominated the sediment composition from both stations (Table 3). The coarse silt sand was consistently less than 2% in any core, site, or year (Figures 4 & 5).
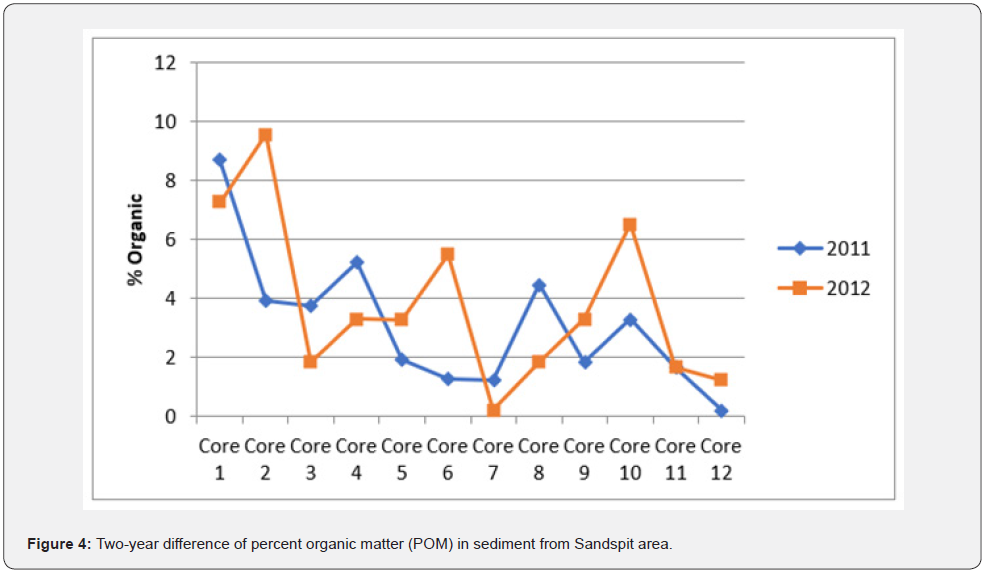
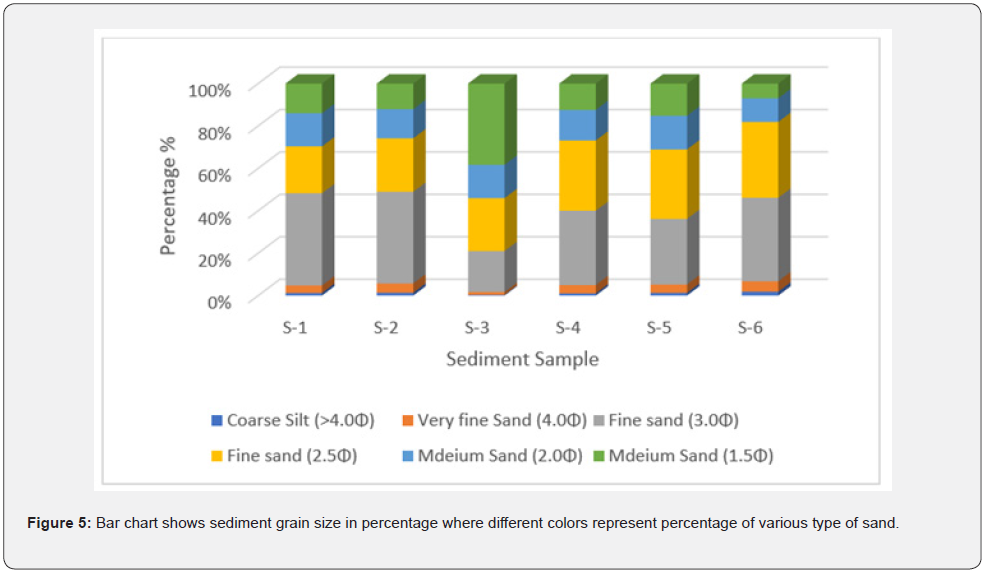
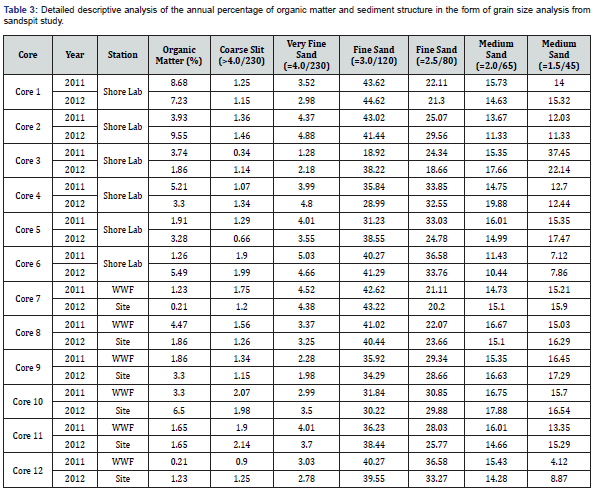
Anthropogenic impact
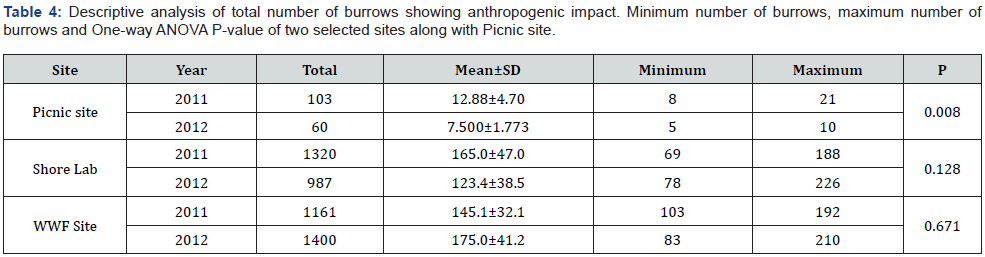
Both selected stations were observed to have less impact from human intervention than the control area. Both sites are usually used by a few people for bathing and picnicking due to access to private huts throughout the study area. Occasionally the number of people increased during weekends and holidays. Both stations are similar in topography; burrow density was compared using ANOVA (Table 4). Burrow density was substantially smaller at the control site than either of the two study sites, but also differed significantly at the control site between the two years, but not at either of the two study sites.
Discussion & Conclusion
Smaller crabs and juveniles construct smaller burrows closer to the sea than larger, adult crabs. Adult ghost crabs have a larger gill surface area which helps them maintain respiratory water for a longer period, allowing them to construct burrow further away from the sea, whereas juveniles with their smaller gill surface areas, cannot tolerate as long a period away from the water [4]. Chan et al. [4] stated that burrows which are shallow with a single tube structure are considered to be temporary burrows. Such behaviour was also observed in other species of crabs such as Ocypode ceratophthalmus (Pallas), Austruca iranica (Pretzman), Austruca annulipes (H Milne Edwards) and Austruca sindensis (Alcock), Leptuca pugilator (Bosc), Sesarma longipes (Krauss), Carisoma carnifex (Herbst), and Macrophthalmus parvimanus (Gurin) [4,31,33,34].
Increasing gill surface area and constructing deeper, more complex burrows help protect ghost crabs from predators, mainly shore birds and wandering animals (i.e., cats, dogs); dogs have been observed to dig up ghost crab burrows on Sandspit beach (S.O., personal observation). Crabs are cold blooded, and harsh environmental conditions may directly influence burrow density as extreme temperatures (below 15˚C or above 30˚C) induces them to hide inside burrows for protection [15,25,18,26,35-37].
The air temperature in Karachi rises to more than 40˚C during summer (April-June) and drops below 15˚C during winter (December and January); at these times burrow density increases, and crabs remain inside their burrows all day. The deeper parts of the burrows remain moist, preventing crabs from suffering from desiccation [13,18]. Similar patterns were observed by Takahashi [38] from Taiwan and India.
The present study revealed that the opening burrow direction was tilted towards the sea, regardless of the structure. When the burrow structure is complex with many branches, secondary arms face landward. Similar results of burrow opening direction were observed from Hong Kong and India [4,22], whereas Hayasaka [39] reported secondary shafts opened at the surface like primary shafts. The secondary arm provides additional protection from predators [22]. Larger crabs protect themselves by constructing deeper burrows with single tube [4], as was found in the current study.
Ghost crabs are considered ecological indicators of anthropogenic impact in urban areas; this impact can be evaluated through burrow counts [24,30,40-42]. Urbanization and human use can have a major impact on the ghost crab burrow density [30,43,44], as human use of beaches lowers ghost crab burrow density [26,40,45-62].
Acknowledgement
Authors thankful to acknowledge for the support of Centre of Excellence in Marine Biology, University of Karachi, and anonymous reviewers for their review.
References
- Jackson LF, Smale MJ, Berry PF (1991) Ghost crabs of the genus Ocypode (Decapoda, Brachyura, Ocypodidae) of the east coast of South Africa. Crustaceana 61: 280-286.
- Turkay M, Sakai K (1996) The Ocypode ghost crabs (Crustacea: Decapoda: Brachyura) of the Arabian Peninsula and adjacent regions. Fauna Saudi Arabia 15: 117.
- Strachan PH, Smith RC, Hamilton DAB, Taylor AC (1999) Studies on the ecology and behaviour of the ghost crab, Ocypode cursor (L.) in Northern Cyprus, Scientia Marina 63: 51-60.
- Chan BKK, Chan KKY, Leung PCM (2006) Burrow architecture of the ghost crab Ocypode ceratophthalma on a sandy shore in Hong Kong. Hydrobiologia 560: 43-49.
- Sakai K, Türkay M (2013) Revision of the genus Ocypode with the description of a new genus, Hoplocypode (Crustacea: Decapoda: Brachyura). Mem Queensl Museum 56: 665-794.
- Yousuf F, Ali F, Kazmi QB (2007) Some Ghost Crabs of the Genus Ocypode (Decapoda: Brachyura: Ocypodidae) from Pakistan’s Waters (Northern Arabian Sea). Turkish Journal of Zoology 31: 107-112.
- Defeo O, McLachlan A, Armitage D, Elliot M, Pittman J (2021) Sandy beach social–ecological systems at risk: regime shifts, collapses, and governance challenges. Front Ecol Environ 19(10): 564-573.
- Yong AY, Lim SS (2021) Plasticity of foraging strategies adopted by the painted ghost crab, Ocypode gaudichaudii, in response to in situ food resource manipulation experiments. Zoological Studies p. 37-60.
- Alcock A (1900) Materials for a carcinological fauna of India. The Brachyura Catometopa or Grapsoidea. The journal of the Asiatic Society of Bengal 69: 279-486.
- Chappgar BF (1956) One the marine crabs (Decapoda: Brachyura) of Bombay state part I. Journal of the Bombay Natural History Society 54: 399-439.
- George RW (1982) A revision of the fiddler crabs of Australia (Ocypodinae: Uca). Records & Supplements - Western Australian Museum p. 1-99.
- Seike K, Nara M (2008) Burrow morphology of ghost crabs Ocypode ceratophthalma and sinensis in foreshore, backshore and dune sub environment of sandy beach of Japan. The Journal of the Geological Society of Japan 114(11): 591-596.
- Weinstein RB (1995) Locomotor behavior of nocturnal ghost crabs on the beach: focal animal sampling and instantaneous velocity from three-dimensional motion analysis. Journal of Experimental Biology 198(4): 989-999.
- Trivedi JN, Vachhrajani KD (2016) On burrow morphology of the ghost crab, Ocypode ceratophthalmus (Decapoda: Brachyura: Ocypodidae) from sandy shore of Gujarat, India. International Journal of Marine Science 6(15): 1-10.
- Wolfrath B (1992) Burrowing of the Fiddler-Crab Uca-Tangeri in the Ria Formosa in Portugal and Its Influence on Sediment Structure. Marine Ecology Progress Series 85: 237-243.
- Griffis RB, Chavez FL (1988) Effects of sediment type on burrows of Callianassa californiensis Dana and gigas Dana. Journal of Experimental Marine Biology and Ecology 117: 239-253.
- Griffis RB, Suchanek TH (1991) A model of burrow architecture and trophic modes in Thalassinidean shrimp (Decapoda: Thalassinidea). Marine Ecology Progress Series 79: 171-183.
- Lucrezi S, Schlacher TA, Walker S (2009) Monitoring human impacts on sandy shore ecosystems: A test of ghost crabs (Ocypode spp.) as biological indicators on an urban beach. Environ Monit Assess 152: 413-424.
- Takeda S, Kurihara Y (1987) The distribution and abundance of Helice tridens (De Haan) burrows and substratum conditions in a North-eastern Japan salt marsh (Crustacea: Brachyura). Journal of Experimental Marine Biology and Ecology 107: 9-19.
- Morrisey DJ, DeWitt TH, Roper DS, Williamson RB (1999) Variation in the depth and morphology of burrows of the mud crab Helice crassa among different types of intertidal sediment in New Zealand. Marine Ecology Progress Series 182: 231-242.
- Lim SSL, Diong CH (2003) Burrow-morphological Characters of the Fiddler Crab, Uca annulipes (H. Milne Edwards, 1837) and Ecological Correlates in a Lagoonal Beach on Pulau Hantu, Singapore. Crustaceana 76(9): 1055-1069.
- Chakrabarti A (1981) Burrow patterns of Ocypode ceratophthalma (Pallas) and their environmental significance. Journal of Palaeontology 55: 431-441.
- Vannini M (1976) Researches on the coast of Somalia. The shore and the dune of Sar Uanle. 10. Sandy beach decapods. Monitore Zoologico Italiano. Supplemento 8(1): 255-286.
- Moss D, McPhee DP (2006) The impacts of recreational four-wheel driving on the abundance of the ghost crab (Ocypode cordimanus) on a subtropical sandy beach in SE Queensland. Coastal management 34: 133-140.
- Barboza CA, Mattos G, Soares GA, Zalmon IR, Costa LL (2021) Low densities of the ghost crab Ocypode quadrata related to large scale human modification of sandy shores. Frontiers in Marine Science 11(8): 589542.
- Costa LL, Soares GA, Zalmon IR (2021) Burrow occupation rates and spatial distribution within habitat of the ghost crab Ocypode quadrata (Fabricius, 1787): Implications for impact assessments. Regional Studies in Marine Science 44: 101699.
- Brown AC, McLachlan A (2006) The ecology of sandy beaches. Elsevier, Burlington, USA, pp. 373.
- Branco JO, Hillesheim JC, Fracasso HAA, Christoffersen ML, Evangelista CL (2010) Bioecology of the ghost crab Ocypode quadrata (Fabricius, 1787) (Crustacea: Brachyura) compared with other intertidal crabs in the Southwestern Atlantic. Journal of Shellfish Research 29: 503-512.
- Defoe O, McLachlan A, Schoeman DS (2009) Threat to sandy beach ecosystems: review. Estuarine, Coastal and Shelf Science 81: 1-12.
- Barros F (2001) Ghost crabs as a tool for rapid assessment of human impacts on exposed sandy beaches. Biological Conservation 97: 399-404.
- Qureshi NA, Saher NU (2012) Burrow morphology of three species of fiddler crab (Uca) along the coast of Pakistan. Belgian Journal of Zoology 142: 114-126.
- Khan MZ, Ghalib SA, Hussain B (2010) Status and new nesting sites of Sea Turtles in Pakistan. Chelonian Conservation and Biology 9: 119-123.
- Braithwaite CJR, Talbot MR (1972) Crustacean burrows in the Seychelles, Indian Ocean. Palaeogeography, Palaeoclimatology, Palaeoecology 11: 265-285.
- Christy JH (1982) Burrow structure and use in the sand fiddler crab, Uca pugilator (Bosc). Animal Behaviour 30: 687-694.
- Hughes DA (1966) Behavioural and ecological investigations of the crab Ocypode ceratophthalmus (Crustacea: Ocypodidae). Journal of Zoology 150: 129-143.
- Christoffers EWI (1986) Ecology of the ghost crab Ocypode quadrata (Fabricius) on Assateague Island, Maryland and the impacts of various human uses of the beach on their distribution and abundance. Michigan City, Michigan State University, East Lansing, Michigan, Doctoral dissertation.
- Weinstein RB (1998) Effects of Temperature and Water Loss on Terrestrial Locomotor Performance in Land Crabs: Integrating Laboratory and Field Studies. Integrative and Comparative Biology 38(3): 518-527.
- Takahashi S (1932) On the burrows of Ocypode ceratophthalma Kwagaku 2: 329-335.
- Hayasaka I (1935) The burrowing activities of certain crabs and their geological significance. The American Midland Naturalist Journal 16: 99-103.
- Neves FM, Bemvenuti CE (2006) The ghost crab Ocypode quadrata (Fabricius, 1787) as a potential indicator of anthropic impact along the Rio Grande do Sul coast, Brazil. Biological Conservation 133: 431-435.
- Schlacher TA, Dugan J, Schoeman DS, Lastra M, Jones A, et al. (2007) Sandy beaches at the brink. Diversity and Distributions 13: 556-560.
- Costa LL, Machado PM, Barboza CAM, Soares GA, Zalmon IR (2022) Recovery of ghost crabs metapopulations on urban beaches during the Covid-19 "anthropause". Mar Environ Res 180: 105733.
- Costa LL, Soares GA, Zalmon IR (2021) Burrow occupation rates and spatial distribution within habitat of the ghost crab Ocypode quadrata (Fabricius, 1787): Implications for impact assessments. Regional Studies in Marine Science 1(44): 101699.
- Yosef R, Spivak V, Edelman S, Kosicki JZ (2022) Anthropogenic beach-activities influences the endemic Red Sea Ghost Crab (Ocypode saratan). Marine Policy 138: 104986.
- Schlacher TA, Thompson L, Price S (2007) Vehicles versus conservation of invertebrates on sandy beaches: Mortalities inflicted by off-road vehicles on ghost crabs. Marine Ecology 28: 354-367.
- Moffett M, Mclachlan A, Winter P, Ruyck A (1998) Impact of trampling on sandy beach macrofauna. Journal of Coastal Conservation 4: 87-90.
- Barboza CA, Mattos G, Soares G A, Zalmon IR, Costa LL (2021) Low densities of the ghost crab Ocypode quadrata related to large scale human modification of sandy shores. Frontiers in Marine Science 11(8): 589542.
- Yosef R, Spivak V, Edelman S, Kosicki JZ (2022) Anthropogenic beach-activities influences the endemic Red Sea Ghost Crab (Ocypode saratan). Marine Policy 138: 104986.
- Buller AT, McManus J (1979) Sediment sampling and analysis in: Estuarine Hydrology and Sedimentation. pp. 87-103.
- Bulleri F, Underwood AJ, Benedetti CL (2007) The assessment and interpretation of ecological impacts in human-dominated environments. Environmental Conservation 34: 181-182.
- Costa LL, Machado PM, Barboza CAM, Soares GA, Zalmon IR (2022) Recovery of ghost crabs metapopulations on urban beaches during the Covid-19 "anthropause". Mar Environ Res 180: 105733.
- Crane J (1975) Fiddler Crabs of the World Ocypodidae: Genus Uca. Princeton University Press, New Jersey, United States of America, pp. 375.
- De C (2005) Biophysical model of intertidal beach crab burrowing: application and significance. Ichnos 12: 11-29.
- Defoe O, McLachlan A, Schoeman DS (2009) Threat to sandy beach ecosystems: review. Estuarine, Coastal and Shelf Science 81: 1-12.
- Defeo O, McLachlan A, Armitage D, Elliot M, Pittman J (2021) Sandy beach social–ecological systems at risk: regime shifts, collapses, and governance challenges. Front Ecol Environ 19(10): 564-573.
- Frey RW, Mayou TV (1971) Decapod burrows in Holocene barrier island beaches and washover fans, Georgia. Senckenbergiana maritima 3: 53-77.
- Hill GW, Hunter RE (1973) Burrow of the Ghost crab Ocypode quadrata (Fabricius) on the barrier islands, South-Central Texas Coast. Journal of Sedimentary Petrology 43: 24-30.
- Lim S, Tantichodok P (2011) Comparison of Burrow Morphology of Juvenile and Young Adult Ocypode ceratophthalmus from Sai Kaew, Thailand. Journal of Crustacean Biology 31: 59-65.
- Miers EJ (1882) On the species of Ocypode in the collections of the British Museum. Annals and Magazine of natural history 10(5): 376-388.
- Saher NU (2008) Population dynamics and biology of fiddler crabs in the mangrove areas of Karachi coast. Doctoral Dissertation, Karachi city, University of Karachi, Sindh, Pakistan, pp. 265.
- Schuchman E, Warburg MR (1978) Dispersal, population structure and burrow shape of Ocypode Marine Biology 49: 255-263.
- Weber F (1795) Entomological nomenclature according to systematic entomology ill. Fabricius added to the recently discovered species and varieties 1(8): 1-72.






























