Will Climate Change Impacts Aggravate Malnutrition in Concealed Ways? The Effects of Temperature Elevation on Nutritive Value of the Edible Seaweed Ulva fasciata Delile
David Simiyu1, Amelia S Buriyo2*
1Department of Biology, Mwenge Catholic University, Tanzania
2Botany Department, University of Dares Salaam, Tanzania
Submission: February 05, 2018; Published: April 06, 2018
*Corresponding author: Amelia S Buriyo, Botany Department, University of Dar es Salaam, P.O. Box 35060, Dar es Salaam, Tanzania, Email: aburiyo@gmail.com
How to cite this article: Simiyu D, Buriyo AS. Will Climate Change Impacts Aggravate Malnutrition in Concealed Ways? The Effects of Temperature Elevation on Nutritive Value of the Edible Seaweed Ulva fasciata Delile. Oceanogr Fish Open Access J. 2018; 6(5): 555698. DOI: 10.19080/OFOAJ.2018.06.555698
Abstract
The edible green seaweed Ulva fasciata which is abundant in Tanzania, is highly nutritious and can therefore help in curbing severe malnutrition currently reported in the country. Seaweeds on the other hand, are reported to be affected by temperature. This study was therefore aimed at investigating the effect of rising temperature on the nutritive value of Ulva fasciata. Samples were collected at the Oysterbay shoreline in Dar es Salaam and grown under controlled conditions at the University of Dar es Salaam. Growth rate and selected nutrients (proteins, vitamin A, carbohydrates, iron, phosphorus, calcium and potassium) were determined using standard analytical methods. Data analysis was done using Paleontological statistics (PAST) version 1.27, 1999-2012 package. The results show that temperature elevation caused a decline in growth rate of Ulva fasciata. Furthermore, its protein content declined sharply from 19.8% dry weight to only 5.8% dry weight when temperature increased above the range of 28-29 °C. With the exception of Phosphorus which increased progressively with temperatures rise, other nutrients including minerals showed a significant decrease when Ulva fasciata was grown in higher temperature ranges. The results suggest that the ongoing global warming will decrease nutritive value of Ulva fasciata.
Keywords: Ulva fasciata; Nutritive value, Malnutrition, Global warming
Introduction
Various seaweeds are of nutritional interest in many parts of the world due to their richness in nutrients [1,2]. Seaweeds are also reported to have higher nutritive value than many terrestrial crops [3,4]. The edible seaweed Ulva fasciata Delile for instance, is rich in nutrients such as vitamins, protein, carbohydrates, lipids, fibres, iodine, calcium, potassium and iron [5,6]. For this reason, only a small amount of this species can fulfill the recommended daily intake (RDI) of several nutrients such as iodine, protein and vitamin A [7,8]. For this reason edible seaweeds have a potential of fighting malnutrition especially in countries where seaweeds flourish. Despite the fact that Tanzania is one of the countries most affected by malnutrition [9,10], and despite being endowed by variety of seaweeds [11], information about utilization as human food is still scanty.
Although there is a big potential of using edible seaweeds to eradicate malnutrition in communities highly affected like those in Tanzania, seaweeds' growth and distribution is highly influenced by environmental factors such as light, temperature, salinity, water movement and nutrients availability. Although studies that directly link U. fasciata's nutrients with predicted global temperature increase are scanty, there are studies which have reported that temperature of 25 °C brings maximum accumulation of amino acids and sugar accumulation in U. fasciata [12]. A study by Juneja et al. [13], revealed fluctuations in protein, carotenoids, lipid and starch in U. pertusa and other algal cells when temperature levels were altered. Although studies about how temperature influences growth and nutrients availability in various seaweeds have been done, information about how the nutritive value of Ulva fasciata will be affected by the predicted temperature increase as a result of global warming is hardly available.
The intergovernmental panel on climate change predicts that the global temperatures will increase by 5.8 °C by the year 2100 [14]. In Tanzania, seawater temperature rise is predicted to range between 3-4 °C by the end of the 21st century from the current average seawater temperature of between 27-28 °C [15]. The effects of global warming will cause a significant decline in food production especially in tropical Africa where farmers depend largely on rainfall [15,16]. As a result of this, edible seaweeds such as U. fasciata which is abundant in East Africa shores and highly nutritious could be effectively used as food supplements to help curb malnutrition. This study therefore aims at finding out how effects of temperature rise as a result of global warming will affect growth rate and nutritive value of U. fasciata so as to help farmers and decision makers to make informed decisions on possibilities of sustainable farming this seaweed for food purposes.
Materials and Methods
Samples of U. fasciata for this study, were collected from the shores of Oysterbay in Dar es Salaam Tanzania, located between 39°16-39°17.5'E and 6°46.5'S. From this area, the collected samples were transported to the University of Dar es Salaam for culturing. Seawater was also collected from the same area, transported and stored in a tank to be used in the experiment. The experiment was conducted in the growth chamber of the Botany Department at the University of Dar es Salaam. The seawater was filtered using a clean double-folded nylon cloth, then checked for salinity and pH which were maintained at 35±0.55%o and 8.2, respectively. Prepared seawater was then placed in 15 aquaria, 10 litres in each aquarium.
The aquaria were then grouped into groups of three to make five groups labled A to E. Each group was set at a different temperature range, from the range of 24-25 °C to the range of 32-33 °C as indicated in Table 1. Aquarium heaters were used to provide the desired temperature in each treatment, and the growth chamber was air conditioned at 18 °C.
Some of the collected U. fasciata samples were prepared for culturing. The young and healthy parts of the thalli were cut and cleansed, then they were gently blotted to remove excess water and weighed using an analytical balance. In each of the aquaria containing 10 litres of seawater, 10g of U. fasciata sample was inoculated. An aerator was immersed in each aquarium to provide ambient air to the culture media (Plate 1).

Throughout the experiments, seaweeds were exposed to 12:12 hours light: dark at 67.47μmol m-2 s-1 intensity with cool fluorescent tubes as source of light. This light intensity was lower compared to the intensity of light at the natural environment (study site) which has been recorded to range between 1500μmol m-2 s-1 and 1700μmol m-2 s-1 in the months of May to September [17]. But all the samples were subjected to the same level of light intensity thus any variation observed afterwards was due to the experimental temperature which was varied.
Seawater in the aquaria was replaced after every seven days to prevent nutrients depletion and growth of epiphytes. The experiment was conducted twice, with each experiment taking 28 days, this was done for the purpose of obtaining more data to ascertain reliable results and also because the growth chamber had no enough space to accommodate more than 15 aquaria at the same time.
Data collection
Determination of growth rate of U. fasciata
Fresh weight of the seaweeds growing in the aquaria was measured using analytical balance at three days interval to monitor their rate of growth. The seaweed was temporarily removed from their aquarium, gently blotted and measured. The seaweed was then returned into their aquarium. From these weights, growth rate was calculated using a formula K = [(Wt/W0)1/t- 1] x 100 %, described by Yong et al. [18] as the most accurate. Where K= specific growth rate per day, W0= the initial weight of the seaweed, and Wt = the weight of the seaweed after t number of days.
Determination of nutrients
In determining nutrients content in U. fasciata, analysis was done in two settings. The first was analyzing nutrients in the wild samples, and the second was analyzing nutrients in samples subjected to experimental conditions. In both settings, some nutrients were analysed in dry samples, and some in fresh samples. Vitamin A, was analysed by the analytical tests following standard methods described by Allen [19] using 1g of blotted fresh seaweeds in three replicates.
The seaweeds subjected to analytical tests for determination of protein, soluble carbohydrate, potassium, iron, phosphorus and calcium content were air-dried and then oven-dried at 60 °C for 48 hours to constant weight. Carbohydrates were extracted by hot water and calorimetrically determined by using procedures described by Allen [19]. In details, 1g of dry sample was weighed into 100ml conical flask, and 30ml of distilled water added. The mixture was boiled for 2 hours and then filtered. The filtrates were used to determine soluble carbohydrate contents of the sample using anthrone reagent.
Total nitrogen was determined by using a semi-micro Kjeldahl digestion [19] and colorimetric determination of the resultant ammonium by colour reaction (Endo-phenol blue method). In this method, 0.2g of dry sample was weighed into Kjedahl flask. To the sample, 0.2g of copper metal, 0.1g selenium (Kjeldahl tables), and 15ml of sulphuric acid were also added. In order to oxidise the organic matter, 2ml of hydrogen peroxide was added and then heated to boiling for 5 minutes. In the mixture 4g of K2SO4 was added and the mixture digested at 430 °C using a thermal Kjeldahl apparatus. The nitrogen present in the sample was thus converted to the ammonium form and the ammonium was determined calorimetrically by using a spectrophotometer. The amount of total nitrogen in the sample was obtained from the calibrated curve of standard NH4. To get the amount of protein, the obtained values of total nitrogen were each multiplied by a factor of 6.25 as described in Allen [19].
To determine the amount of selected minerals in the samples, a Kjeldahl thermo was used for digestion. A sample of 0.1 grams of each treatment seaweed material was weighed and put in Kjeldahl flasks. To the flasks 5ml conc. HNO3 and 1ml of perchloric acid were added and the resulting mixture was digested at 120 °C. The resulted solutions after digestions were subjected to analysis of the selected mineral nutrients. While Potassium, Calcium and Iron were determined using the Atomic Absorbance Spectroscopy (AAS), Perking Elmer 3100. The filtrate obtained above were retained for determination of Phosphorus. Ortho-phosphate was determined by colorimetry using a spectrophotometer according to ascorbic acid method of Allen [19]. The amount of Phosphorus in the sample was obtained from the calibration curve of standard PO4.
In determination of ß-carotene, a method developed by Nagata & Yamashita [20] was used. In its procedure, the dried extract (100mg) was vigorously shaken with 10ml of acetone- hexane mixture (4:6) for 1 minute and filtered through Whatman No.4 filter paper [21]. The absorbance of the filtrate was measured at 453, 505 and 663nm and contents of ß-carotene were calculated according to the following equation,
ß-carotene (mg/100mg) = 0.216A663 - 0.304A505 + 0.452A453. [21].
Data analysis
All statistical computations were done using Paleontological statistics (PAST) version 2.17, 1999-2012 package [22], at the confidence limit of 0.05. A single-factor Analysis of Variance followed by Tukey multiple comparison test (HSD) [23], were used to analyse the significant difference in the mean content of each selected nutrient in the samples of U. fasciata grown at different temperature set-ups and also to test whether the difference in growth rates at different temperatures was significant. Linear regression analysis was used to analyse the relationship between growth rate of U. fasciata and temperature variation. It was also used in statistical analysis of the relationship between temperature elevation and accumulation of the selected nutrients.
Results
Effect of temperature rise on growth rate of U. fasciata
Specific growth rate of U. fasciata was recorded to decrease with the increase in temperature ranges (Figure 1). The lowest specific growth rate of U. fasciata was recorded at the temperature range between 32 and 33 °C where growth rate averaged -0.94±0.2% per day. Ulva fasciata's specific growth rate was highest at the temperature range between 24 and 25 °C where the average specific growth rate was 4.67±0.1% per day. As indicated in Figure 1, U. fasciata's specific growth rate beyond the temperature range between 30-31 °C, was recorded to be negative. Thalli of U. fasciata were bleaching at the temperature above the range of 28-29 °C, and the seaweeds growing in the temperature range of 30-31 °C and 32-33 °C were disintegrating into fragments reducing their mass.
Using a linear regression model, it was observed that there was a strong relationship between temperature increase and decrease in growth rate (r= -0.97). It was also determined that 95% of the variations of growth rate values were explained by temperature variation, the coefficient of determination R2 was 0.95. According to ANOVA there were significant differences in growth rates of U.fasciata between different temperature ranges (P<0.05, F=303.7, DF=29). Results from Tukey's Multiple- comparison (HSD) test show that growth rate of U.fasciata was not significant between 24-25 °C vs 26-27 °C temperature ranges (Q=1.938, P>0.05), but growth rates compared between all other temperature ranges were significant.
Effect of temperature rise on selected nutrients in Ulva fasciata
Vitamin A
The average amount of ß-carotene determined in U.fasciata wild samples was 0.158±0.007mg/100mg. Results after experimentation shows that there was an increase in the amount of ß-carotene as temperature was increased reaching maximum accumulation at the range of 28-29 °C. At this range the amount of ß-carotene was measured to be 0.179±0.001mg/100mg wet weight (Figure 1). At the temperature exceeding 28-29 °C range, amount of ß-carotene began to decline reaching the minimum level at the temperature range of 32-33 °C where ß-carotene amount was just 0.055±0.002mg/100mg wet weight. Furthermore, it was observed that ß-carotene accumulation in wild samples nearly equals the amount measured in the samples growing at the temperature range of 26-27 °C which was 0.155±0.003mg/100mg wet weight.
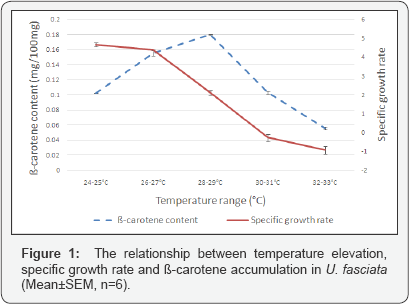
Using one-way ANOVA it was determined that there is a significant difference between ß-carotene contents accumulated by samples of U.fasciata grown in different temperature ranges (P<0.05, F=591.2, DF=29). The Tukey's multiple comparison test revealed that the accumulated ß-carotene contents were significantly different in the seaweeds grown in all compared temperature ranges except the difference in ß-carotene amounts accumulated between temperature range of 24-25 °C vs 30-31 °C (Q=0.1654, P>0.05) which was not significant. Furthermore, regression analysis (Polynomial fit, order 2), indicated a strong regression relationship between temperature elevation and ß-carotene content in U. fasciata, r = -0.95 and the coefficient of determination R2 was 0.903, indicating that 90.3% of the variations in ß-carotene accumulation was explained by the changes in temperature.

Protein
The average protein accumulated by this seaweed determined before experimentation was found to be 21.97±0.15% of its dry weight. Moreover, the protein content measured in U. fasciata after experimentation indicated that it increased slightly from 15.4±0.3% of the sample's dry weight to 16.3±0.1% when temperature was increased from the range between 24-25 °C to the temperature range between 26-27 °C respectively (Figure 2). The increase of temperature from the range of 26-27 °C to the range of 28-29 °C caused a sharp increase of protein (16.3±0.1% to 19.7±0.2%). Then protein content declined to 13.9.0±0.2% when temperature increased to the range of 30-31°C and continued to decline sharply to 5.6±1% when temperature reached the range of 32-33 °C.
Analysis by one way ANOVA revealed that there was a significant difference between protein contents in Ulva fasciata growing in different temperature ranges (F=112.8, DF=29, P<0.05). Turkey’s multiple comparison test revealed that the significant difference was between protein accumulation in U. fasciata growing at all temperature ranges except in the temperature ranges of 24-25 °C vs 26-27 °C and between 2425 °C vs 30-31 °C which revealed a difference that was not significant.
Linear regression analysis (polynomial regression fit, order 2) revealed a strong negative linear relationship between protein content and the determinant variable temperature, r = -0.96. The coefficient of determination R2 was 0.93 showing that 93% of the variations in protein content was explained by the changes in temperature.
Soluble Carbohydrates
The soluble carbohydrate content in the analysed samples decreased proportionally with the increase in temperature. The lowest content of soluble carbohydrate was observed in the temperature range of between 32-33 °C (6.5±1.2%), while the highest levels of soluble carbohydrates was in the temperature range of 24-25 °C (24.9±0.01%) as shown in Figure 2. Moreover, accumulation of soluble carbohydrate in the wild seaweeds determined soon after harvest was higher than in any of the seaweeds subjected to experimentation (25.6±0.18%).
Analysis by one way ANOVA revealed that there was a significant difference between the soluble carbohydrate contents of U. fasciata growing in different temperature ranges (F=187.8, DF=29, P<0.05). It was however found out that the compared mean difference in percentage soluble carbohydrates accumulated between temperature ranges of 24-25 °C vs 26-27 °C was not significant. This was revealed after doing a Tukey's multiple comparison test. The test also revealed that the difference in soluble carbohydrates accumulated by the seaweed sample between all other tested temperatures were significant. When regression analysis was performed, it was determined that there is a strong relationship between temperature rise and soluble carbohydrates content, r = -0.99 and R2=0.97.
Iron Content
As the temperature ranges were increased, it was observed that the levels of Iron in the U. fasciata samples also changed. Iron content increased from about 71.9±0.5mg/10g dry weight to 82.7±0.3mg/10mg when temperatures were increased from the range of 24-25 °C to the range of 26-27 °C (Figure 3). The content of Iron in the U. fasciata samples then decreased progressively as temperatures increased from the range of 2627 °C to 32-33 °C, the range at which the Iron content was the lowest (52.3±2.2mg/10g).
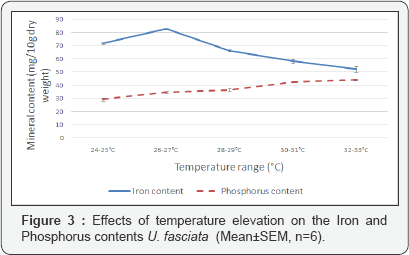
On the other hand, the amount of Iron determined in the seaweeds that were not subjected to experimentation was 85.72±1.2mg/10g. This amount nearly equals the amount accumulated by U. fasciata growing at the temperature range of 26-27 °C which was 82.65±0.35. Furthermore, analysis by One-Way ANOVA showed a significant relationship between temperature rise and iron content (F=83.12, DF=29, P<0.05). Tukey's multiple comparison test revealed that the difference in iron accumulation in the samples grown in all temperature ranges were significant. Moreover, the regression analysis which was performed to check the relationship between Iron accumulation and increasing temperature revealed a strong regression relationship (r = -0.85) and that 72% of the variation in Iron content were caused by temperature increase.
Phosphorus Content
Contrary to other nutrients, the increase in temperature from the lower range of 24-25 °C to the higher range of 32-33 °C resulted in a proportional increase in the content of phosphorus in U. fasciata samples (Figure 3). The highest accumulation of Phosphorus was averaged to be 44.1mg/10g at the temperature range of 32-33 °C and the lowest was 30.6mg/10g which was recorded at the range of 24-25 °C.
The amount of Phosphorus accumulated by the seaweeds growing at the temperature range of 28-29 °C which was 36.45±0.75mg/10g nearly equals the amount measured in samples that were not subjected to the experiments which was 38.40±1.75mg/10g (Table 1). One-way ANOVA showed that there is a significant relationship between increase in temperature range and change in phosphorus content in U.fasciata (F=75.11, DF=11.88, P<0.05).

According to Tukey’s multiple comparison test, the only difference in phosphorus content that was not significant was between samples from temperature ranges of 26-27 °C vs 28-29 °C and 30-31 °C vs 32-33 °C. Samples from all other temperature ranges had significant differences in their phosphorus contents. Linear regression analysis indicated a strong relationship between phosphorus accumulation and temperature increase (r=0.98). The coefficient of determination R2 was 0.97.
Calcium Content
Calcium content in U. fasciata increased slightly from 383.5±5.9mg/10g at the temperature range of 24-25 °C to 408.1±1.8mg/10g at the range of 26-27 °C. Calcium content reached highest levels at the range of28-29 °C (482.5±0.4mg/10g) and began to decrease as temperatures were increased to higher ranges of 30-31 °C and 32-33 °C where Calcium content was 435.7±5.7mg/10g and 324.5±3.3mg/10g respectively (Figure 4).
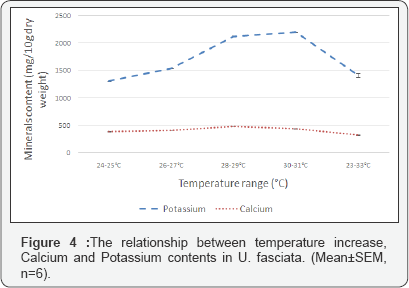
Calcium content was measured to be 426.37±9.11mg/10g in the seaweeds growing in the wild (before experimentation). This nearly equaled the amount accumulated in seaweeds growing at the range of 30-31 °C. The changes in calcium levels as temperature ranges increased were small but significant (F=212, DF=29, P<0.05; one-way ANOVA). Calcium content in U. fasciata samples from all temperature ranges showed significant difference. This was according to Tukey's multiple comparison test. Regression analysis was able to determine a strong relationship between variations in Calcium content and elevation of temperature, r=0.92 and R2=0.85.
Potassium Content
As temperature ranges were increased from 24-25 °C to the range of 30-31°C, potassium content also increased from 1305.8±3.3mg/10g to 2203.95±5.9mg/10g respectively. As temperature was further increased to 32-33 °C range, the levels of potassium in U.fasciata dropped sharply to 1413.1±39.4mg/10g (Figure 4). Levels of Potassium measured in the wild seaweeds was found to be 1912.81±28.20mg/10g. This amount was found to be between the amounts accumulated by seaweeds growing at temperature ranges of 26-27 °C and 28-29 °C.
The relationship between the changes in potassium content and rise in temperature ranges was determined to be statistically significant through one-way ANOVA (F=481, DF=29, P<0.05). Significant changes were determined by Tukey’s multiple comparison test to be between Potassium content accumulated by U. fasciata grown at all compared temperatures. When regression analysis was performed, it was determined that 78% of the variations in potassium content resulted from the change in temperature (R2-0.78), and the coefficient r was 0.88.
Discussion of the Results
Effect of temperature elevation on growth rate of Ulva fasciata
As one of an important aspect of nutrition, growth rate was one of the parameters studied. It was revealed in this study that, Ulva fasciata can grow at a fast rate (4.7% per day) when growing in the temperature range between 24-25 °C. This was also observed in other studies done in other species of Ulva. For example, in a study done by Ruangchuay et al. [24], it was found that U. intestinalis had the highest growth rate at a temperature of 25 °C.
Having recorded a growth rate of below 0 % per day when temperatures were elevated to 30 °C, it is evident that when temperature reaches this level U. fasciata species dies off. This was also reported in a study by Mohsen et al. [12], who observed that temperatures of 30 °C in the littoral region of the Mediterranean shore bordering Alexandria are lethal to U. fasciata.
The decrease in growth rate of this species with increase in temperature can be attributed to the disruptive stress that causes cellular and subcellular damage in seaweeds as explained in several studies such as those done by Davison & Pearson [25] and Eggert [26]. In the aforementioned studies, it was reported that such damages leads to relocation of resources for protection and repair thus causing growth rate to slow, development to delay and even mortality.
Growth of U. fasciata will subsequently continue to decrease affecting its yield if global temperatures continue to rise as predicted by the IPCC and other scholars such as Seitz & Nyangena [15] who used various simulation models to show that temperatures will continue to rise by up to 4 °C especially on the East African shores of the Indian Ocean by the end of the century. According to the data from the Tanzania meteorological agency (TMA), air temperatures in Dar es Salaam for the months of December to March have been averaged at 29 °C in the period between the year 1991 and 2007 [27]. The high temperatures in the above mentioned months have also been reported in other studies [15,28]. This means that in these months cultivation of U. fasciata in natural environment could be difficult. As reported in this study, when growing in a temperature range between 28 -29 °C, U. fasciata grow at a rate of only 2.1% per day as compared to the rate of 4.7% per day when temperatures are between 2425 °C.
Effects of temperature elevation on selected nutrients content in Ulva fasciata
It was observed in this study that, there is a significant increase in Vitamin A content when temperatures are increased. It was however also noted that Vitamin A content decreases dramatically when temperatures go beyond a range of 28-29 °C. This may be because at this range of temperature, U. fasciata began to disintegrate. The increase in ß-carotene content with increase in temperature was also observed in a study on secondary catorenoids formation by a green alga Chlorococcum sp. done by Liu and Lee (2000) in which it was shown that increase in temperature increases accumulation of carotenoids in algal species.
This increase in carotenoids can be attributed to cellular response to increased active free oxygen radicals formed at elevated temperatures [29] and increased biosynthetic enzyme activity in high temperatures [30]. As reported by Asensi-Fabado & Munne-Bosch [31], lipid-soluble vitamins such as Vitamin A play a role in both plants and animals metabolism because they are strong antioxidants and they also function as enzymatic cofactors. The predicted increase in temperature as a result of global warming will therefore bring about significant decline in the amount of Vitamin A in U.fasciata. To be used as a supplement food for Vitamin A, this edible seaweed will have to be grown in months cooler than 30 °C which according to Mtolera & Buriyo [17], are the months of March to September in Tanzania.
The increase in protein levels with the increase in temperature from the range of 24-25 °C to 28 -29 °C may be a result of increased production of heat shock proteins to protect the seaweed from the effects of heat stress. As explained by Collen et al. [32] in a study on red seaweed Chondrus crispus, high temperature stress drastically alter transcription of more than 15% of the said species' genes. Furthermore, the said study reported that high temperature treatments lead to an increase in expression of stress genes such as those that lead to antioxidants and heat shock proteins (HSP). This indicates that high temperatures stimulates production of more proteins hence supporting the results found in this study.
In addition to that, the amount of protein accumulated by U. fasciata below the temperature of 30°C (between 15 and 19.8% dry weight), correlates with the amount of proteins found in various species of the genus Ulva which is between 15 and 21% of their dry weight as reported by various studies [1,33]. High accumulation of Proteins below the temperature of 30 °C, indicates that this seaweed can indeed help curb malnutrition when cultivated below that range of temperature.
It was however discovered that further increase in temperature beyond the range of 28-29 °C caused a sharp decline in protein. This decline can be explained by considering the observed decline in biomass, disintegration and death of this species of seaweeds when temperature reaches 30 °C as reported in this study and others [12]. The low content of protein accumulated by U. fasciata growing at temperatures above the range of 28-29 °C, (below 14% of its dry weight) compromises the species potential use as a supplement food to increase protein in human diets. According to the National Academies [34], recommended daily amount of protein for nursing mothers for example, is about 71g per day, this means that, if global temperatures continue to rise and warm sea water temperatures to 30 °C and above, the amount of protein in U. fasciata will be lowered to the point that the amount of the daily consumption of this seaweed taken as food supplement will need to be very big to meet the recommended daily intake.
Nevertheless, protein content in the wild samples was found to be higher than in the samples grown at different temperature ranges. The experimental settings in this study tried to imitate the natural settings by providing appropriate salinity, pH and aeration to the seawater in the aquaria. These however are not the only parameters found in natural ocean environment which is vast and faced with fluctuations in temperature, salinity, pH and other extraneous parameters. These could therefore be reasons for higher accumulation of protein in the wild samples than in the seaweeds grown under different experimental temperatures. The higher content in protein accumulated in natural environment gives an even better prospect of cultivating this edible seaweed as long as it is within the months with temperature below the range of 28-29 °C.
With reference to the carbohydrate content in U. fasciata at different temperature ranges, this study has shown that temperature increase, profoundly decreases the amount of soluble carbohydrates in this edible seaweed. As indicated in the results, U. fasciata accumulated soluble carbohydrates by more than 24% of its dry weight when temperature was between 2425 °C. This indicates that, when temperatures are favorable, this edible seaweed can produce large amount of soluble carbohydrates making it even more nutritious. High amount of carbohydrates in U. fasciata growing in favorable conditions, has also been reported in other research works. A study by Padua et al. [5] on the chemical composition of three species of Chlorophyta for example, reported that U. fasciata contains carbohydrate to over 50% of their dry weight. In addition to that, the effect of temperature increase in soluble carbohydrate has also been reported in a study done by Nakamura & Miyachi [35] on the effect of temperature on starch degradation in an algae species Chlorella vulgaris, where it was revealed that increase in temperature decreases starch content.
Furthermore, it was discovered that, amounts of the mineral nutrients were affected differently by temperature elevation. The continuous increase in Phosphorus as temperatures were increased, might be a result of the seaweed's effort to curb the decline in growth as a result of temperature increase. This is because Phosphorus has been identified by various studies to probably be the main control of macro-algal growth in tropical coastal areas [36-38]. Therefore more of this nutrient was accumulated in the efforts to increase growth. The increase in percentage dry weight content of Phosphorus could also probably be due to decline in its utilization because there was a decline in growth, this lead to its percent in total dry weight of seaweed to increase as total biomass continued to decrease.
The decline in contents of other minerals in U. fasciata when temperature went beyond the tolerance level of the specific mineral was very sharp. Iron content for example, dropped by about 37% from its maximum accumulation (which occurred at a temperature range between 26-27 °C) when temperatures reached the range of 32-33 °C. Calcium and potassium content dropped by 33% and 36%, respectively, from their maximum levels when temperature levels reached 32 °C. The results suggest that if these plants are cultivated or naturally growing at higher temperature their nutritive value is compromised. This therefore calls for more efforts to be done to halt the trends of global temperature increase so that these seaweed species and others with similar attributes continue to be highly nutritious.
Although the accumulated minerals in U. fasciata as observed in this study declined at different temperature levels (Ca declined at temperature of 32 °C, K at 32 °C, and Fe at 28 °C) but their decline is an indication that temperature increase decreases the amount of most of the mineral nutrients in U. fasciata. It is therefore important to understand that, although U. fasciata contains enough amounts of mineral elements to make it a potential weapon against mineral deficiencies in humans, most of these minerals in this seaweed will be lowered significantly if seawater temperatures will continue rising.
This study has clearly shown that, if temperature will continue to increase at a predicted rate, then both growth rate and nutritive value of Ulva fasciata will be significantly lowered. Unless global temperature increase is halted, cultivation of this seaweed will be difficult in many months of the year, and its nutritive value will be less.
References
- Burtin P (2003) Nutritional Value of Seaweeds. Electronic Journal of Environment, Agriculture and Food Chemistry 2: 498-503.
- Devi G, Thirumaran G, Manivannan K, Anantharaman P (2009) Element Composition of Certain Seaweeds from Gulf of Mannar Marine Biosphere Reserve; Southeast Coast of India. World Journal of Dairy and Food sciences 4(1): 46-55.
- MacArtain P, Gill C, Brooks M, Campbell R, Rowland I (2007) Nutritional Value of Edible Seaweeds. Nutr Rev 65(12 Pt 1): 535-543.
- Kumar M, Gupta V, Kumari P, Reddy C (2011) Assessment of Caulerpa species for nutritional fatty acids and antioxidant potentials. Journal of food composition and analysis 24: 270-278.
- Padua M, Fontoura P, Mathias A (2004) Chemical composition of Ulvaria oxysperma (Kutzing) Bliding, Ulva lactuca (Linnaeus) and Ulva fasciata (Delile). Brazilian Archives of Biology and Technology 47(1): 49-55.
- Moustafa Y, Saeed S (2014) Nutritional evaluation of green macroalgae, Ulva sp. and related water nutrients in the Southern Mediterranean Sea coast, Alexandria shore, Egypt. Egyptian Academic Journal of Biological Sciences 5(1): 1-19.
- Kokilam G, Vasuki S (2013) Biochemical and phytochemical analysis on Ulva fasciata and Caulerpa taxifolia. International Journal of pharmacy and pharmaceutical science research 4(1): 7-11.
- Kailas A, Nair S (2015) Comparison of nutrient compositions and calorific values of eight tropical seaweeds. Phycological Society, India 45: 62-74.
- UNICEF Tanzania 2015 Nutrition Overview.
- FAO, IFAD, WFP (2015) The State of Food Insecurity in the World 2015: Meeting the 2015 international hunger targets, taking stock of progress. FAO, Rome, Italy
- Oliveira E, Osterlund K, Mtolera M (2005) Marine plants of Tanzania: A field guide to the seaweeds and seagrasses of Tanzania. Sida/ Department for Research Cooperation SAREC, The Hague.
- Mohsen AF, Nasr AH, Metwalli AM (1973) Effect of temperature variations on growth, reproduction, amino acid synthesis, fat and sugar content in Ulva fasciata delile plants. Hydrobiologia 42(4): 451-460.
- Juneja A, Ceballos RM, Murthy GS (2013) Effects if environmental factors and nutrient availability on biochemical composition of algae for biofuels production: a review. Energies 6(9): 4607-4638.
- Intergovernmental Panel on Climate Change (2007) Climate change 2007: Synthesis Report. IPCC, Valencia, Spain.
- Seitz J, Nyangena W (2009) Economic Impact of Climate Change in the East African Community (EAC), Final report. Global21 consulting, Toulouse, France.
- Kabasa JD, Sage I (2009) Climate Change and Food Security in Africa. In Besada H, Sewankambo N (Eds.), CIGI Special Report Climate Change in Africa; Adaptation, Mitigation and Governance Challenges CIGI, Ontario, USA, pp. 21-25.
- Mtolera M, Buriyo A (2004) Studies on Tanzanian Hypneaceae: Seasonal Variation in content and quality of Kappa-Carrageenan from Hypnea musciformis (Gigartinales: Rhodophyta). Western Indian Ocean Journal of Marine Science 3(1): 43-49.
- Yong YS, Yong WTL, Anton A (2013) Analysis of formulae for determination of seaweed growth rate. Journal of Applied Phycology 25(6): 1831-1834.
- Allen SE (1989) Chemical Analysis of Ecological Materials. (2nd edn), Blackwell Scientific Publications, Oxford, USA.
- Nagata M, Yamashita I (1992) Simple method for simultaneous determination of chlorophyll and carotenoids in tomato fruits. Nippon Shokuhin Kogyo Gakkaishi 39(10): 925-928.
- Pal J (2010) In vitro free radical scavenging activity of wild edible mushroom, Pleurotus squarrosulus (Mont.) Singer. Indian J Exp Biol 48(12): 1210-1218.
- Hammer O (1999) Paleontological statistics (PAST), Version 2.17. Natural History Museum, University of Oslo, Oslo, Norway
- Zar JH (2010) Biostastical Analysis (5th edn), Pearson, New Jersey, USA.
- Ruangchuay R, Dahamat S, Chirapat A, Notoya M (2012) Effects of culture on the growth and reproduction of Gut Weed, Ulva intestinalis Linnaeus (Ulvales, Chlorophyta). Sonklanakarin J Sci Technol 34(5): 501-507.
- Davison IR, Pearson GA (1996) Stress tolerance in intertidal seaweeds. Journal of phycology 32(2): 197-211.
- Eggert A (2012) Seaweed responses to temperature. In: Wiencke C, Bischof K (Eds.), Seaweed Biology, Springer-Verlag, Berlin.
- Tanzania Meteorological Agency 2008 Monthly temperature readings for Dar es Salaam in the period of 16 years (1991-2007). TMA, Dar es Salaam, Tanzania.
- McClanahan RT, Ateweberhan M, Muhando AC, Maina J, Mohammed SM (2007) Effects of climate and seawater temperature variation on coral bleaching and mortality. Ecological Monographs 77(4): 503-525.
- Tjahjono AE, Kakizono T, Hayama Y, Nishio N, Nagai S (1994) Isolation of resistant mutants for a green alga Haematococcus pluvialis, and their hybrid formation by protoplast fusion for breeding of higher astaxanthin producers. Journal of Fermentation and Bioengineering 77(4): 352-357.
- Liu BH, Lee YK (2000) Secondary carotenoids formation by the green alga Chlorococcum sp. J Appl Phycol 12(3-5): 301-307.
- Asensi-Fabado AM, Munne-Bosch S (2010) Vitamins in Plants: occurrence, biosynthesis and antioxidant function. Trends Plant Sci 15(10): 582-592.
- Collen J, Guisle-Marsollier I, Leger J, Boyen C (2007) Response of the transcriptome of the intertidal red seaweed Chondrus cripus to controlled and natural stresses. New Phytol 176(1): 45-55.
- Ratana-arporn P, Chirapart A (2006) Nutritional evaluation of traopical green seaweeds Caulerpa lentillifera and Ulva reticulate. Kasetsart Journal of Natural Sciences 40: 75-83.
- National Academies (2015) Dietary Reference Intakes (RDIs).
- Nakamura Y, Miyachi S (1982) Effect of temperature on starch degradation in Chlorella vulgaris cells. Plant Cell Physiol 23(2): 333341.
- Nixon SW, Pilson MEQ (1993) Nitrogen in estuarine and coastal marine exosystems. In: Carpenter EJ, Capone DG (Eds.), Nitrogen in marine environment Academic, New York, USA.
- Oviatt C, Doering P, Nowicki B, Reed L, Cole J, et al. (1995) An ecosystem level experiment on nutrient limitation in temperate coastal marine environments. Marine Ecology Prog Series 116: 171-179.
- Howarth RW, Anderson D, Cloern J, Elfring C, Hopkinson C, et al. (2000) Nutrient pollution of coastal rivers, bays and seas. Issues in Ecology 7: 1-15.






























