Abstract
The problem of implant failure is also a major issue in contemporary surgical practice because revision surgeries cause huge financial and patient costs. The use of self-healing polymers, which can repair mechanical or chemical damage on their own, is a paradigm shift from passive implantable devices to adaptive biomaterial systems. This mini review explores the clinical translation prospects of self-healing polymers in the field of surgery. We consider four fundamental healing processes (reversible covalent bond, supramolecular, microencapsulated systems, and vascular networks) and assess existing evidence in orthopedic, soft tissue, cardiovascular, and prosthetic bioproducts. Although early clinical results indicate positive mechanical recovery and biocompatibility, biological performance results are still behind the curve of engineering progress. The major obstacles to clinical adoption are the scalability of manufacturing, complicated regulatory routes of combination products, and the requirement for long-term safety data. The next frontier is artificial intelligence, predictive healing, and 4D bioprinting of personalized implants. Self-healing polymers are at the pivotal point between bench and bedside, and it is necessary to have a concerted effort among materials scientists, surgeons, and regulatory bodies to achieve the transformational potential of self-healing polymers in the surgical practice.
Keywords:Self-Healing Polymers; Biomedical Implantation; Regenerative Medicine; Biomaterials; Clinical Translation
Abbreviations:UPY: Ureidopyridinone; GMP: Good Manufacturing Practice; PMMA: Polymethylmethacrylate; ETO: Ethylene Oxide
Introduction
About 10-15 out of every surgical operation involves implant failure, and revision surgery costs healthcare systems more than 20 billion US Dollars every year [1]. The three major types of failure modes, namely mechanical fatigue, wear-induced damage, and biological encapsulation, have one thing in common: they are progressive and can often be undetected until clinical signs appear [2]. Conventional biomaterials, be it metallic, ceramic, or polymeric, are machined to act as mechanical components, yet they do not have adaptive designs to damage occurring [3]. Self-healing polymers constitute a radical break to this paradigm of no action. These systems are defined as those materials that can self-heal damage without any outside help and have the potential to restore mechanical integrity, inhibit crack growth, and improve useful life [4]. The difference between self-healing and biodegradable materials is vital to surgeons: the same is that both are related to the material transformation; whereas, in biodegradation, the structural integrity is deliberately removed, self-healing is focused on its maintenance [5].
Self-healing polymers are not only promising clinically because of their ability to repair damage. Such materials have the potential to decrease the inflammatory response related to wear debris, reduce fibrous encapsulation by use of surface renewal, and reduce the number of revision surgeries [6]. Nonetheless, the process of translation of engineered materials to clinical devices involves complex biological environments, sterilization procedures, and regulatory standards. The objective of this review is to: (1) outline self-healing responses that have been clinically translated with potential, (2) review the current evidence-based studies in all areas of surgery, and (3) determine the obstacles to clinical implementation that are critical. We are still interested in applications in which self-healing is used to provide a solution to particular unmet clinical requirements as opposed to engineering expertise.
Basic Mechanisms of Surgical Environment
The self-healing polymers proceed through two major mechanisms: intrinsic mechanisms that involve reversible chemical attachments in the polymer framework, and extrinsic mechanisms that involve the incorporation of distinct healing agents [7]. These mechanisms can be summarized in terms of surgical relevance as in Table 1. Reversible covalent kinetic bonds, especially the Diels-Alder reaction of maleimide and furan groups, have shown thermal reversibility, which can be used in the orthopedic practice [16,17]. These systems are capable of being subjected to several healing cycles; however, they need higher temperatures (usually 80-120°C), which might be impractical to achieve in vivo activation [18]. New developments in roomtemperature Diels-Alder elastomers have the potential to be translated into clinical practice [19]. Hydrogen bond, π-π stacking, and metal-ligand coordination are supramolecular interactions that provide quick healing at physiological temperatures [20]. The Ureidopyridinone (UPy) quadruple hydrogen bonding has demonstrated specific efficacy in soft tissue applications with a healing efficacy of greater than 90% in minutes [21]. However, such non-covalent interactions can be weakened under wet biophysical conditions [22].
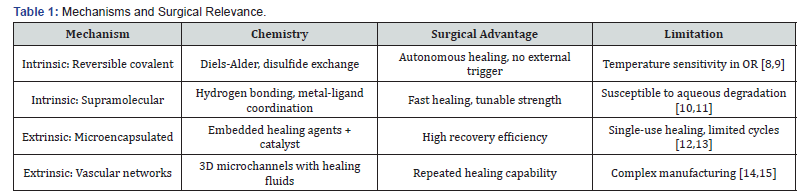
Microencapsulated Systems. A healing agent (monomer, catalyst, or hardener) is incorporated into microcapsules that are scattered through the polymer matrix [23]. When a crack propagates, capsules burst, and their content is released, causing polymerization that seals crack faces. The finite capsule supply is effective when dealing with one-damage events, but it restricts several rounding-offs of healing [24]. Biological circulation is imitated by the use of vascular networks that include empty channels that contain healing agents [25]. This solution allows healing and reuse of various agents but involves complex 3D printing that is difficult to comply with the existing Good Manufacturing Practice (GMP) regulations [26].
One of the most important things that surgeons should take into account is that the actual autonomy or healing without any external stimulus is not common. The majority of systems demand the specific stimuli (temperature, pH, light) that should be compatible with surgical processes and biological limitations [27].
Clinical Applications: Existing Evidence
Orthopaedic and dental surgery
The surgeon must identify the medical history of a patient before proceeding with the surgery or treatment.
The problem of self-healing bone cements is that polymethylmethacrylate (PMMA) has a severe drawback in cyclic loading, which leads to catastrophic failure due to the formation of microcracks [28]. Self-healing preparations which contain microencapsulated monomers have shown 70-80 per cent recovery of compressive strength in vitro [29]. In vivo experiments of rabbit femur defects demonstrated a decrease in radiolucent lines and enhanced bone integration against the traditional cements [30]. Their main issue is to strike a compromise between the healing performance and the mechanical modulus. Excessive and deficient content of healing agents reduces the first strength and the maximum repair capacity, respectively [31]. Existing formulae have compressive strengths of 60-80 MPa - sufficient for dental use but unsatisfactory in load-bearing orthopedic implants [32].
Soft tissue and wound management
The development of self-healing hydrogels has gone the farthest in the clinical translation of the gel in wound closure systems [33]. These substances blend tissue gluing properties with self-sealing properties and overcome dehiscence, which is a problem associated with 2-5% of surgical wounds [34]. The recent ones are injectable, self-healing hydrogel adhesives with rigid tissue adhesion and on-demand biodegradation [35]. These systems develop in seconds when injected, follow the topography of the wound, and adhere in wet conditions. Preclinical trials show fewer cases of wound dehiscence and faster wound healing than sutures or traditional fibrin sealants [36]. Self-healing hydrogels have excellent biocompatibility and minimize heat production in polymerization compared to cyanoacrylates [37]. They need not contain blood components and have tunable degradation rates comparable to tissue regeneration schedules, unlike fibrin glue [38].
Cardiovascular and vascular surgery
Vascular grafts have special problems: a mismatch in compliance with native vessels generates intimal hyperplasia and graft failure. Mechanically tunable self-healing polyurethaneelastomers have been demonstrated to be promising in restoring vessel compliance and avoiding the formation of aneurysms. Magnesium alloys with self-healing coatings exhibit pH/nearinfrared responsiveness, antibacterial, osteogenic, and angiogenic properties, which are applicable to the vascular stenting application. But the long-term patency data are still not available, and fears still exist about the cytotoxicity of healing agents in blood-contacting applications.
Prosthetic interfaces
A crucial setting where the self-healing material can be beneficial is the interface point between the residual limb and prosthetic socket area to minimize skin breakages and enhance comfort. Self-healing socket liners adjust to general daily changes in the volume of residual limbs, which alleviates shear forces and prevents pressure ulcers. Based on the recent systematic reviews of self-healing polymers in prosthetics and orthotics, the translational pathway is one that has been developed beyond external prosthetics to implantable devices. Osseointegrated implants with self-healing properties are potentially effective in the prevention of periprosthetic inflammation and enhanced fixation in the long term (Figure 1).
Biological Performance and Safety Issues
The biological performance of self-healing polymers has
not been described in detail compared with their mechanical
performance. The essential points to consider are:
Cytotoxicity of healing agents: Microencapsulated systems
expel reactive monomers and catalysts, which can drain into
adjacent tissues. The kinetics of release and systemic distribution
must be quantified in order to obtain regulatory approval.
Host inflammatory response: The inflammatory cascades
can be triggered by the very process of healing. Though acute
inflammation causes tissue integration, chronic inflammation
causes fibrous encapsulation and failure of the device. Biostability:
There is little research on the healing efficiency of implants after
12-24 months of implantation. Healing capacity may be impaired
due to polymer ageing, oxidation, and mechanical fatigue when
it is most required. Effects of sterilization: implantable devices
are sterilized by gamma and ethylene oxide (ETO) lighting; both
are capable of damaging healing processes. It can also be crosslinked
with gamma radiation to create permanent bonds between
reversible bonds, and the ETO residual can poison catalysts. These
deficits underline the fact that biological data on performance is
much behind mechanical characterization, and such a research
priority is severely vital.
Obstacles to Clinical Translation
Technical barriers: The fabrication of scalability and batch-tobatch reproducibility should be more problematic for the existing production approaches. The self-healing polymers usually demand a fine control over the molecular weight distributions and the interlocking densities that are challenging to control on large-scale levels. Complexity in regulation: Since self-healing polymers are combination products (device + drug/biologic when the healing agents are bioactive), they need two regulatory pathways. These materials are dynamic and hence, make it harder to perform traditional stability tests and determine shelf-life. Economic factors: Cost-benefit analysis should prove that lower revision surgery rates are worth increasing the cost of the first material. Evidence is needed in healthcare systems that selfhealing implants are better in terms of long-term outcomes to justify high premium prices.
Clinical adoption: There are practical barriers to surgeon education and the long-term follow-up. Implantation methods and the requirement of special monitoring protocols may impede adoption due to the learning curve. Ethical and liability issues: As long as the healing mechanism fails and leaves the patients with defective implants, the liability becomes a specific issue, unlike the failure of the conventional device.
Expert Opinion: What would make a surgeon adopt this technology? Evidence of reduced revision rates (more than 50% based on five years of follow-up data), demonstrable intraoperative evidence of the activation of healing, and simplified explantation procedures in case they are necessary.
Future Directions
AI-based materials: Machine learning can be used to generate patient-specific healing needs, depending on comorbidities, activity, and local biological conditions. This is in line with the fact that the author has expertise in artificial neural networks in biomedical applications. 4D bioprinting: It allows implants to be deployed minimally invasively as well as repair damage in situ through a combination of shape-memory polymers and selfhealing capabilities. Electrically manipulated microcapsules in hierarchical hydrogels have specific potential in the regeneration of the spinal cord. Bioelectronic integration: Neural interfaces and biosensors. Self-healing conductive polymers make it possible to maintain the activity of these devices even when they are deformed mechanically. Such materials may also track the health of the implants and cause therapeutic responses. Personalized Medicine: Healing kinetics that are patient-specific, like genetic profiles and local tissue conditions, are the final goal. Spatially varying healing scaffolds bioprinted can be used to match the regional mechanical challenges.
Conclusion
The self-healing polymers are at a crossroad, where they are no longer an engineering fantasy but are now a clinically viable biomaterial. Though the preclinical results reveal strong evidence backing the mechanical recovery and biocompatibility, the distance between material development and clinical proof is still enormous. The collaboration between materials scientists, surgeons, regulatory scientists, and healthcare economists must be long-term in order to succeed. The recommendation is simple: to determine whether self-healing polymers will serve their purpose to reduce the rate of implant failures and enhance patient outcomes, prospective clinical trials with rigorous followup need to be conducted. These adaptive materials will either be the standard care or a laboratory success in the next decade.
References
- Rumon M, Akib A, Sultana F, Moniruzzaman M, Niloy M, et al. (2022) Self-Healing Hydrogels: Development, Biomedical Applications, and Challenges. Polymers 14(21): 4539.
- Bertsch P, Diba M, Mooney D S (2022) Self-Healing Injectable Hydrogels for Tissue Regeneration. Chemical Reviews 123(2): 834–873.
- K A, Shyam R, Palaniappan A, Oh T, Nathanael A (2021) Self-Healing Hydrogels: Preparation, Mechanism, and Advancement in Biomedical Applications. Polymers 13(21): 3782.
- Yang P, Li Z, Fang B, Liu L (2023) Self-healing hydrogels based on biological macromolecules in wound healing: A review. International journal of biological macromolecules 253(Pt 8): 127612.
- Mo C, Xiang L, Chen Y (2021) Advances in Injectable and Self-healing Polysaccharide Hydrogel Based on the Schiff Base Reaction. Macromolecular Rapid Communications 42(10): e2100025.
- Xie J, Peng Y, Wang Z, Li J (2022) Recent Advances of Self-Healing Polymer Materials via Supramolecular Forces for Biomedical Applications. Biomacromolecules 23(3):641-660.
- Jiang C, Zhang L, Yang Q, Huang S, Shi H, et al. (2021) Self-healing polyurethane-elastomer with mechanical tunability for multiple biomedical applications in vivo. Nature Communications 12(1): 4395.
- Zhao Y, He P, Yao J, Li M, Han L, et al. (2023) pH/NIR-responsive and self-healing coatings with bacteria killing, osteogenesis, and angiogenesis performances on magnesium alloy. Biomaterials 301: 122237.
- Diba M, Spaans S, Ning K, Ippel B, Yang F, et al. (2018) Self‐Healing Biomaterials: From Molecular Concepts to Clinical Applications. Advanced Materials Interfaces 5(7): 1800118.
- Matini A, Naghib S (2025) Recent advances in self-healing hydrogel design for enhanced wound care: A comprehensive study on polysaccharides and proteins in comparison with synthetic polymers. International journal of biological macromolecules 323(Pt 2): 147075
- Anand K, Sharma R, Sharma N (2024) Recent advancements in natural polymer-based self-healing nanomaterials for wound dressing. Journal of Biomedical Materials Research. Part B, Applied biomaterials 112(6): e35435.
- Zhang H, Lin X, Cao X, Wang Y, Wang J, et al. (2023) Developing natural polymers for skin wound healing. Bioactive Materials 33: 355-376.
- Ren H, Zhang Z, Cheng X, Zou Z, Chen X, et al. (2023) Injectable, self-healing hydrogel adhesives with firm tissue adhesion and on-demand biodegradation for sutureless wound closure. Science Advances 9(33): eadh4327.
- Pourshahrestani S, Zeimaran E, Kadri N, Mutlu N, Boccaccini A (2020) Polymeric Hydrogel Systems as Emerging Biomaterial Platforms to Enable Hemostasis and Wound Healing. Advanced Healthcare Materials 9(20): e2000905.
- Nie L, Wang L, Hu S, Wei Z, Ding X, et al. (2025) Dopamine-conjugated hyaluronic acid hydrogel interpenetrated by genipin crosslinked quaternary ammonium chitosan for potential biomedical adhesives applications. Colloids and Surfaces. B, Biointerfaces 252: 114683.
- Côté-Cyr M, Bourgault S (2025) Design and applications of self-assembled polypeptide matrices in wound healing. Frontiers in Bioengineering and Biotechnology 13: 1646622.
- Jiang Y, Zhou Y, Tian Y, Nabavi N, Ashrafizadeh M, et al. (2025) Conductive polymers in smart wound healing: From bioelectric stimulation to regenerative therapies. Materials Today Bio 34: 102114.
- Menikheim S, Lavik E (2020) Self-healing biomaterials: The next generation is nano. Wiley Interdisciplinary Reviews. Nanomedicine and nanobiotechnology 12(6): e1641.
- Wu W (2025) Shaping the Future of Surgical Procedures: A Bibliometric Analysis of Advanced Polymeric Surgical Materials Research in the Last Decade. Surgical Innovation 32(6): 524-535.
- Tripathy D, Rout S, Saha I, Halder J, Rajwar T, et al. (2025) Synthetic Polymer-Based Interventions in Wound Healing: A Clinical Perspective on their Efficacy and Limitations. Current pharmaceutical design pp. 23.
- Ratwani CR, Kamali AR, Abdelkader AM (2023) Self-healing by Diels-Alder cycloaddition in advanced functional polymers: A review. Progress in Materials Science 131: 101001.
- Khan NI, Halder S, Gunjan SB, Prasad T (2018) A review on Diels-Alder-based self-healing polymer composites. IOP Conference Series: Materials Science and Engineering 377(1): 012007.
- Safaei A, Brancart J, Wang Z, Yazdani S, Vanderborght B, et al. (2023) Fast self-healing at room temperature in Diels–Alder elastomers. Polymers 15(17): 3527.
- Li B, Cao PF, Saito T, Sokolov AP (2022) Intrinsically self-healing polymers: From mechanistic insight to current challenges. Chemical Reviews 122(24): 17247–17313.
- Ren Y, Dong X (2024) Dynamic polymeric materials via hydrogen-bond cross-linking: Effect of multiple network topologies. Progress in Polymer Science 151: 101890.
- Ooya T, Cho IS (2021) Supramolecular self-healing gels. In Supramolecular Chemistry in corrosion and biofouling protection pp. 85–108.
- Agrawal N, Arora B (2022) Self-healing polymers and composites: Extrinsic routes. Mini-Reviews in Organic Chemistry 19(5): 420–433.
- Kuhl N, Bode S, Hager MD, Schubert US (2015) Self-healing polymers based on reversible covalent bonds. In Self-healing materials: Principles and applications pp. 1–45.
- Dallaev R (2024) Advances in materials with self-healing properties: A brief review. Materials 17(10): 2464.
- Chiang MY, Cheng HW, Lo YC, Wang WC, Chang SJ, et al. (2021) 4D spatiotemporal modulation of biomolecule distribution in anisotropic corrugated microwrinkles via electrically manipulated microcapsules within hierarchical hydrogel for spinal cord regeneration. Biomaterials 272: 120762.
- Menikheim SD, Lavik EB (2020) Self‐healing biomaterials: The next generation is nano. WIREs Nanomedicine and Nanobiotechnology 12(6): e1641.
- Del Prado-Audelo ML, Caballero-Florán IH, Mendoza-Muñoz N, Giraldo-Gomez D, Sharifi-Rad J, et al. (2021) Current progress of self-healing polymers for medical applications in tissue engineering. Iranian Polymer Journal 30(8): 587–603.
- Chen Y, Chai M, Xuan C, Lin J, Yang H, et al. (2024) Tuning the properties of surgical polymeric materials for improved soft-tissue wound closure and healing. Progress in Materials Science 141: 101249.
- Liu Y, Hsu SH (2018) Synthesis and biomedical applications of self-healing hydrogels. Frontiers in Chemistry 6: 449.
- Yang H, Yang Y, Li Y, Hope J, Choo W (2023) Extrinsic conditions for the occurrence and characterizations of self-healing polyurea coatings for improved medical device reliability: A mini review. ACS Omega 8(33): 30087–30098.
- Almutairi MD, Aria AI, Thakur VK, Khan MA (2020) Self-healing mechanisms for 3D-printed polymeric structures: From lab to reality. Polymers 12(7): 1534.
- Mashkoor F, Lee SJ, Yi H, Noh SM, Jeong C (2022) Self-healing materials for electronics applications. International Journal of Molecular Sciences 23(2): 622.
- Ganewatta MS, Wang Z, Tang C (2021) Chemical syntheses of bioinspired and biomimetic polymers toward biobased materials. Nature Reviews Chemistry 5(11): 789–806.






























