Adrenal Pathology: A Historical-Etiological Overview from “Organum Succenturiatum” To Minimally Invasive Surgery
M Vassallo1, A Parra2, J Morao3* and C Zamora4
1General Surgeon, Full Professor at the Central University of Venezuela, University Hospital of Caracas. Chair of Surgical Clinic and Therapeutics “B”, Venezuela
25th year Medical Student, Luis Razetti School, Central University of Venezuela
3Fourth year Resident in General Surgery, University Hospital of Caracas. Chair of Surgical Clinic and Therapeutics “B”, Venezuela
45th year Medical Student, Luis Razetti School, Central University of Venezuela
Submission:February 23, 2026;Published:March 16, 2026
*Corresponding author:José Leonardo Morao Pompili, 4th Resident in General Surgery, University Hospital of Caracas. Chair of Surgical Clinic and Therapeutics “B”, Venezuela
How to cite this article:OM Vassallo, A Parra, J M, C Zamora. Adrenal Pathology: A Historical-Etiological Overview from “Organum Succenturiatum” To Minimally Invasive Surgery. Open Access J Surg. 2026; 17(2): 555959. DOIDOI: 10.19080/OAJS.2026.17.555959.
Summary
The adrenal gland, the nerve center of the stress response, presents a unique diagnostic and therapeutic challenge due to its great endocrine complexity and its challenging anatomical location.
Objective: To conduct a historical review of adrenal pathology, tracing its evolution from the initial anatomical description during the Renaissance to its consolidation as a cornerstone of minimally invasive endocrine surgery.
Methodology: A literature review of classic and contemporary sources was conducted to propose an original visual algorithm that, through a
timeline, integrates the historical emergence of each pathological entity with its specific etiologies, thus providing a clear view of scientific and
technological progress.
Discussion: Knowledge evolved from Eustachius’s anatomical description and Brown- Séquard ‘s validation of adrenal vitality to the clinical
definition of Addison’s, Cushing’s, and Conn’s syndromes. In the therapeutic field, the transition from Mayo’s first open adrenalectomy to
Gagner’s standardization of the laparoscopic approach is described.
Conclusion: Current surgical excellence rests on the integration of these historical discoveries. Mastery of anatomy, physiology, and technical
advancements is the fundamental academic pillar for the precise management of adrenal pathology in the modern era.
Keywords:Adrenal Gland, History of Medicine, Pheochromocytoma, Cushing’s Syndrome, Conn ‘S Syndrome, Adrenalectomy, Laparoscopic Surgery
Abbreviations:CAH: Congenital Adrenal Hyperplasia; HPA: Hypothalamic-Pituitary-Adrenal; CT: Computed Tomography; MRI: Magnetic Resonance Imaging; PRA: Posterior Retroperitoneoscopic Adrenalectomy; NOTES: Natural Orifice Transluminal Endoscopic Surgery
Introduction
The adrenal gland, located at the upper pole of the kidneys, is a crucial endocrine organ that functions as a dual entity with two functionally separate regions: the cortex and the medulla [1]. The adrenal cortex is responsible for the production of steroid hormones, including glucocorticoids, mineralocorticoids, and androgens. The medulla, on the other hand, synthesizes and secretes catecholamines, primarily adrenaline and noradrenaline [1]. Pathology of these glands, although infrequent, has profound systemic implications, affecting the metabolic, cardiovascular, and nervous systems.
In the field of medicine, knowledge of the history behind each pathology is fundamental, since these major discoveries create a solid foundation for future research to refine past knowledge and discover new entities, as well as new forms of treatment [2]. The study of the history of adrenal pathology presents a fascinating narrative of advances and enigmas, particularly because the organ’s function was primarily defined by pathology and the clinical observation of its deficiency, rather than by early physiological experimentation. Understanding this evolution, from anatomical ignorance to the ability to perform minimally invasive adrenalectomy, is crucial. This technical breakthrough, culminating in laparoscopic surgery, establishes the current standard of practice. However, technical competence must necessarily be integrated with the development of non-technical skills, and it is this foundational knowledge that is essential for patient safety and optimizing decision-making in the operating room [3].
The aim of this article is to provide a clear, practical, and comprehensive overview of the origins of each adrenal gland pathology, highlighting the key figures who discovered them and how their findings form the basis of current diagnostic and therapeutic approaches. Using a visual algorithm presented as a timeline, the article seeks to facilitate a clear yet precise understanding of the evolution of knowledge in this fundamental field of endocrine medicine.
Methodology
A narrative and critical review of the biomedical literature was conducted, focusing on the historical evolution and surgical management of adrenal pathology. Searches were performed in databases such as PubMed, Google Scholar, and Scopus using a combination of keywords. Specific search terms included: “ history of adrenal gland “, “ Eustachius “, “ Addison’s disease “, “ pheochromocytoma “ and “ laparoscopic adrenalectomy and “. The selection criteria prioritized primary sources to ensure the historical accuracy of the described events, supplemented by contemporary literature for the section on technological advancements. The collected information was systematized using a dual chronological (timeline) and etiological (separation by cortical and spinal cord pathology) approach, allowing for the construction of the proposed visual algorithm.
Discussion
Anatomical description (1564)
The first documented record that rescued the adrenal gland from anonymity is attributed to the Italian anatomist Bartolomeo Eustachi in 1564 [4]. Although in his Tabulae Anatomicae accurately described these structures located above the kidneys, their alternative designation being “ organum The term “ succenturiatum “ (spare organ) reflected the ignorance that prevailed at the time. For almost three hundred years, the lack of an evident organic function led the scientific community to consider them vestiges or accessory structures, without vital relevance. This concept of “uselessness” was the main obstacle to overcome [5].

First clinically described adrenal syndrome (1855)
The true importance of the adrenal gland was revealed through fatal pathology, when Thomas Addison published his classic monograph on in 1855 the constitutional and local effects of disease of the supra-renal capsules [6]. In this work he described the clinical symptoms associated with the destruction of these capsules, establishing the existence of chronic adrenal insufficiency, now known as Addison’s Disease [7]. Addison’s description was twofoldly significant. First, it demonstrated that the adrenal glands are essential for life, and second, it established that their function was critical for homeostasis. Thus, knowledge of adrenal function was driven by clinical observation of its deficiency, rather than by physiological understanding (Figure 1).
Experimental demonstration of vitality (1856)
The complete dismissal of the vestigial organ concept was provided by physiological experimentation. A year after Addison’s publication, the French physiologist Charles-Édouard Brown- Séquard demonstrated that bilateral removal of the adrenal glands in experimental animals caused rapid death. This finding was crucial, as it provided experimental validation confirming the organ’s absolute vitality, laying the groundwork for investigating the “essential substances” produced by these structures.
The physiological differentiation of the cortex and the medulla (1856 to 1895)
In 1856, the French physician Edmé Félix Alfred Vulpian detected a reactive substance (chromaffin) in the adrenal medulla that was released into the bloodstream. This finding provided the first biochemical evidence of the medullary component [8]. Subsequently, in 1895, George Oliver and Edward A. Schäfer experimentally confirmed the action of the adrenal medulla [9]. By injecting adrenal extracts, they demonstrated a potent vasopressor effect, effectively isolating the functional impact of adrenaline. The distinction between a cortex essential for life and a medulla with hemodynamic function laid the foundation for the modern understanding of adrenal syndromes.
The recognition of congenital adrenal hyperplasia (1865)
The understanding of congenital adrenal hyperplasia (CAH), a group of genetic disorders affecting cortisol and aldosterone synthesis, has its roots in the mid-19th century. Although the enzymatic etiology would not be discovered until much later, the Italian pathologist Luigi De Crecchio provided the first clear description of a case corresponding to this disease in 1865. He documented the finding of hyperplastic adrenal glands in the autopsy of an adult patient with ambiguous external genitalia and female internal anatomy. This early clinicopathological case demonstrated the profound connection between adrenal anatomy and function with sexual and hormonal development, opening an initial window into the study of congenital errors of adrenal metabolism [10].
Isolation of the active medullary principle (1901)
Later, the Japanese chemist Jokichi Takamine was able to isolate and characterize the active principle of the adrenal medulla, identifying epinephrine (adrenaline) as the compound responsible for the vasopressor effects [11]. This discovery marked the beginning of the era of modern endocrinology, allowing the biochemical understanding of adrenal physiology.
Description of myelolipoma (1905)
At the beginning of the 20th century, histological characterization made it possible to identify less frequent but pathologically distinct entities. In 1905, the pathologist Edgar von Gierke first described lesions formed by adipose and hematopoietic tissue in the adrenal gland, an entity that would later be named myelolipoma by Charles Oberling in 1929. Although benign and generally asymptomatic, their discovery marked an advance in tissue differentiation [12].
Identification of Waterhouse-Friderichsen Syndrome (1911)
A few years later, the British physician Rupert Waterhouse first described a syndrome characterized by massive bilateral hemorrhage of the adrenal glands occurring during fulminant meningococcal sepsis. Then, in 1917, the Danish pediatrician Carl Friderichsen expanded on these observations, identifying a major cause of acute adrenal insufficiency and irreversible shock [13].
Pheochromocytoma as a bone marrow challenge (1886 to 1927)
The medullary component gave rise to a fearsome neoplasm. The identification of pheochromocytoma was a process that extended from Felix ‘s pathological description. Fränkel in 1886, who associated it with medullary tumors in patients with hypertension, [14] until the term was coined by Ludwig Pick in 1912 [15]. This catecholamine-producing tumor presented a surgical challenge with very high mortality due to intraoperative hypertensive crises. The therapeutic shift occurred in 1927, when Dr. Charles H. Mayo performed the first successful transabdominal adrenalectomy to cure a pheochromocytoma [16]. This act demonstrated the feasibility of the intervention in a context of high physiological risk, ushering in the era of adrenal surgery (Figure 2).
The description of Cushing’s Syndrome (1932)
The discovery of the pathology caused by cortical hyperfunction was led by Harvey Cushing. In 1912, he initially reported what he called a “ polyglandular syndrome “ caused by pituitary dysfunction. However, in 1932 he published a series of cases describing the complete clinical picture, consolidating Cushing’s syndrome and establishing the complex interrelationship of the hypothalamic-pituitary-adrenal (HPA) axis [17]. Identifying the etiology, often pituitary or adrenal adenomas, was crucial for guiding therapeutic management, whether surgical or medical (Figure 3).
Isolation and characterization of cortical steroids (1930 to 1950)
Tadeus Reichstein and Edward Kendall carried out extensive research on the chemical structure and properties of steroids secreted by the adrenal gland [18]. Their work culminated in the isolation and identification of cortisol as the most important glucocorticoid. These researchers discovered that the adrenal gland secreted steroids with different physiological effects: glucocorticoids (regulators of carbohydrate metabolism) and mineralocorticoids (regulators of electrolyte balance). Then, Philip Hench discovered that cortisone injections caused a dramatic improvement in patients with severe rheumatoid arthritis [19]. In 1950 the joint work of Reichstein, Kendall and Hench was recognized with the Nobel Prize in Physiology or Medicine for their discoveries relating to the hormones of the adrenal cortex, their structure and biological effects [19].


Conn’s Syndrome (1955)
The spectrum of cortical hyperfunction was completed in 1955 with the description of primary hyperaldosteronism by Jerome W. Conn [20]. Based on the study of a 34-year-old patient with hypertension, tetany, and severe episodic muscle weakness, he proposed that an excess of mineralocorticoids was the underlying cause. Successful surgical resection of the adrenal adenoma confirmed his hypothesis, achieving complete remission of the hypertension and hypokalemia. This finding was groundbreaking, as it identified for the first time an etiology of arterial hypertension potentially curable by surgery, differentiating it from essential hypertension and broadening the therapeutic scope of adrenalectomy [21] (Figure 4).


Technological advances in diagnosis (1970 to 1980)
The development of adrenal pathology entered an era of technological advancements thanks to imaging technology and new surgical techniques. The introduction of Computed Tomography (CT) in 1970 marked the beginning of the diagnostic revolution, allowing non-invasive visualization of the adrenal glands with unprecedented resolution. This ability to detect masses and characterize them using densitometry surpassed all previous invasive methods [22]. Diagnostic imaging was further refined in the 1980s with Magnetic Resonance Imaging (MRI), which offered superior advantages for soft tissue characterization, crucial for differentiating benign cortical adenomas from adrenocortical carcinomas [23].
The incidentaloma phenomenon (1982)
The widespread use of computed tomography gave rise to a new clinical challenge described in 1982 by Glenn W. Geelhoed and Edward M. Druy: the incidentaloma. This term defined adrenal masses discovered incidentally in radiological studies performed for other non-endocrine reasons, forcing the development of new algorithms to distinguish between functional, malignant, and benign non-secretory lesions [24].
Staging of adrenocortical carcinoma (1984)
Adrenocortical carcinoma was recognized as a pathological entity from the late 19th century, based on descriptions of cases in autopsies around 1884. However, systematic and reproducible differentiation between benign adenomas and adrenocortical carcinomas was only achieved a century later. In 1984, Leonard M. Weiss published a histological scoring system based on nine morphological criteria that revolutionized diagnosis, allowing tumors to be classified as adenomas or carcinomas [25].
Macronodular hyperplasia (1964)
The improvement in pathological characterization also allowed for the identification of complex bilateral pathologies. In 1964, Marvin A. Kirschner described ACTH-independent bilateral macronodular adrenal hyperplasia. This finding challenged the traditional surgical paradigm, raising the need for simultaneous bilateral or cortical-sparing approaches in patients with subclinical Cushing’s syndrome [26].
The Surgical Revolution (1992)
Open adrenal surgery required large incisions, resulting in significant pain and prolonged hospital stays. This situation was revolutionized by Michel Gagner and his team, who in 1992 described the first laparoscopic adrenalectomy. Gagner reported the successful performance of this procedure in patients with Cushing’s syndrome and pheochromocytoma, and the demonstration of its technical feasibility in managing such complex functional tumors was a key factor in the global adoption of the technique [27].
In the following decades, multiple sources confirmed the advantages of laparoscopic adrenalectomy. The benefits of the laparoscopic approach over the open approach included less bleeding, less postoperative pain, and a significant reduction in hospital stay [28]. The effectiveness of the technique and the low conversion rate allowed laparoscopic adrenalectomy to become firmly established as the gold standard for most surgical adrenal pathologies, both functional and non-functional. This surgical success is the modern and efficient response to the indications established by the endocrinological discoveries of the 20th century (Figure 5).
Retroperitoneoscopic adrenalectomy (1996)
Following the standardization of transabdominal laparoscopy, the need arose for a more direct approach. Although Selçuk Mercan described the lateral approach in 1995, it was Martin K. Walz who popularized posterior retroperitoneoscopic adrenalectomy (PRA) in 1996 [29]. This technique allowed access to the gland without entering the peritoneal cavity and without the need to mobilize neighboring organs, establishing itself as the technique of choice for patients with a history of abdominal surgery or the need for bilateral adrenalectomy [30].
Robotic adrenalectomy (1999)
The incorporation of computerized assistance came in 1999, when Luigi Piazza reported the first successful robotic adrenalectomy using the ZEUS system. Subsequently, in 2001, Santiago Horgan and Daniel Vanuno consolidated the technique with the Da Vinci system. The three-dimensional vision and the articulation of the instruments allowed for more precise dissections of complex tumors, overcoming the ergonomic limitations of conventional laparoscopy [31].
Partial adrenalectomy with cortical preservation (2000)
In patients with high-risk hereditary syndromes, complete resection of both adrenal glands resulted in permanent adrenal insufficiency. To avoid this, groups such as Michael K. Walz’s proposed partial adrenalectomy with preservation of functioning cortex from the early 2000s, particularly in bilateral pheochromocytomas. This technique aimed to balance oncological control with the preservation of endocrine function, reducing dependence on exogenous glucocorticoids and mineralocorticoids, at the cost of a controlled risk of local recurrence [32].
Single-port adrenalectomy (2008)
In the quest to further reduce the surgical footprint, Sean A. Castellucci performed the first single-port adrenalectomy (LESS) in 2008. This technique concentrated all surgical trauma into a single umbilical incision, offering a superior aesthetic result, although with greater technical demands on the surgeon due to limited triangulation [33].
Transluminal endoscopic surgery (2008)
The landscape of scarless surgery was explored that same year when Silvana Perretta and Jacques Marescaux described the adrenal approach using natural orifice transluminal endoscopic surgery (NOTES) via a transvaginal approach. Although its routine clinical application remains limited, this milestone demonstrated the feasibility of operating on the adrenal gland without visible skin incisions, marking the current extreme of minimally invasive surgery [34].
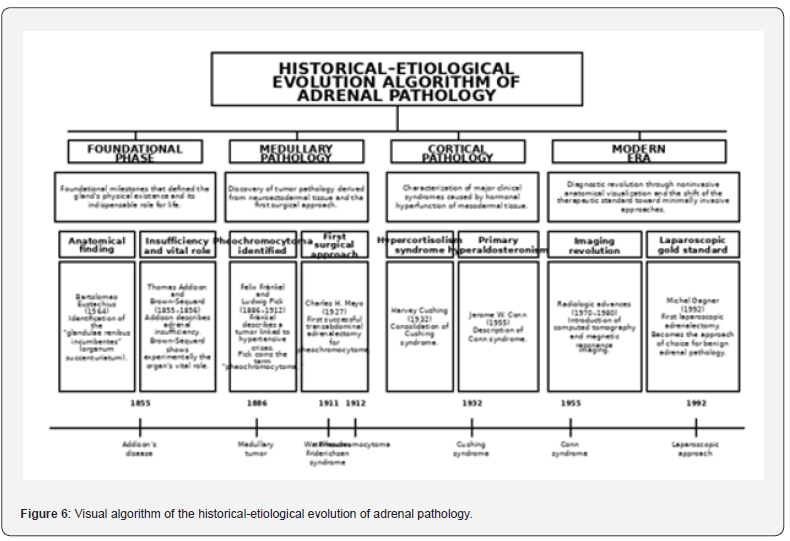
Description Of the Algorithmic Scheme
This review proposes a visual algorithm using a timeline that integrates three fundamental aspects: historical chronology, key figures in the discovery, and the etiological classification of pathological entities. This framework allows for a clear and didactic approach to understanding the most significant events in adrenal pathology. The algorithm’s structure is based on a horizontal timeline extending from 1564 to the present. A key aspect is that the main pathologies or syndromes are highlighted on this timeline precisely on the date they were discovered, facilitating an understanding of the sequence of findings.
The algorithm structures the historical evolution into four fundamental branches that classify milestones according to their etiological and technological nature. The first branch, the foundational phase, groups the discoveries that rescued the organ from anatomical invisibility and defined its vitality (Eustachius, Addison, Brown - Séquard). The second, spinal cord pathology, addresses the identification of neuroectodermal tumors and their first surgical approach (Fränkel, Pick, Mayo). The third, cortical pathology, details the clinical characterization of the major syndromes of steroid hypersecretion (Cushing and Conn). Finally, the modern era branch integrates the non-invasive diagnostic revolution (CT and MRI) with the therapeutic transformation toward the laparoscopic gold standard (Gagner). At the bottom, a timeline chronologically orders the sequential appearance of the most important described pathological entities, from Addison’s disease to laparoscopic resolution (Figure 6).
Conclusion
The history of adrenal pathology is undoubtedly a validation of the scientific method and interdisciplinary collaboration, traversing an extraordinary path from anatomical invisibility to prominence in precision medicine [1,7]. This journey was characterized by a fundamental dissociation in discovery: while knowledge of the cortex was driven by clinical observation (Addison, Cushing), understanding of the medulla resulted from physiological and biochemical experimentation [6,9]. At the center, Brown -Séquard ‘s findings were essential, connecting the mystery of the “ organum “ succenturiatum “ with clinical pathology in establishing the vitality of the organ [8].
For its part, the surgical intervention was shaped by a dual therapeutic approach, as the findings of Addison, Cushing, and Conn defined the indication, while the work of Mayo and Gagner’s transformative laparoscopic solution defined the feasibility and safety of the procedure [14,19]. The universal adoption of the minimally invasive approach not only reduced the morbidity of adrenalectomy but also consolidated the specialty [24,25]. Each of these advances has been driven by curiosity, rigor, and innovation [1,7], and understanding this historical-etiological journey is not just a tribute, it is the academic pillar that guides contemporary practice [1,7,24] . It is a demonstration that current surgical excellence is undoubtedly based on the mastery of each of these fundamental discoveries [24,25].
References
- Williams GH, Dluhy RG (1997) The history of adrenal gland research. Endocrinol Metab Clin North Am 26(2): 1-15.
- Vassallo M, Vandres MV, Documet Z (2010) Thyroiditis: a historical review to the present. Medicine in Time 43.
- Vassallo-Palermo Miguel, Morao-Pompili José Leonardo, Linares-Medina Argelis Sofía (2025) Non-technical skills in surgery: A literature review. rev. colomb. cir. [Internet] 40 (2): 226-233.
- Eustachius B Tabulae anatomicae. Rome 1563-1564.
- Bartholin C (1611) Anatomicae Institutions Corporis Humani.
- Addison T (1855) On the constitutional and local effects of disease of the adrenal capsules. London: Samuel Highley.
- Trousseau A (1868) Lectures on Clinical Medicine.
- Brown- Séquard CE (1856) Recherches expérimentales sur la physiologie et la pathologie des capsules surré Arch Physiol Norm Pathol 1: 385-403.
- Oliver G, Schäfer EA (1895) On the physiological action of extract of the adrenal capsules. J Physiol 18(3): 230-276.
- De Crecchio L (1865) On a case of monstrosity of the external genitalia, determined by a hypertrophy of the adrenal capsules. Morgagni 7: 433–464.
- Takamine J (1901) The isolation of the active principle of the supra-renal gland. J Physiol 27.
- Gierke E (1905) Über knowmarksgewebe in der nebenniere. Beitr Pathol Anat 7: 311–325.
- Waterhouse R (1911) A case of adrenal apoplexy. Lancet 1: 577-578.
- Fränkel F (1886) Ein Fall von doppelseitigem, völlig latent veraufenen Nebennierentumor und gleichzeitiger Nephritis mit Veränderungen am Circulationsapparat und Retinitis. Virchows Arch Pathol Anat 103: 244–63.
- Pick L (1912) Das Ganglioma embryonale sympathicum (Sympathoma embryonal) nebst einer Bemerkung über die neurogene Entstehung der echten Nebennierengeschwü Berl Klin Wochenschr 49: 16–22.
- May CH (1927) Paroxysmal hypertension with tumor of the retroperitoneal nerve. Report of a case. JAMA 89(13): 1047-1050.
- Cushing H (1932) The basophil adenomas of the pituitary body and their clinical manifestations (pituitary basophilism). Bull Johns Hopkins Hosp 2(5): 486-508.
- Kendall EC (1950) The development of cortisone as a therapeutic agent. Science 112: 352-358.
- Hench PS, Kendall EC, Reichstein T (1950) The effects of cortisone acetate and pituitary ACTH on rheumatoid arthritis, rheumatic fever and other conditions. In: Nobel Lecture Amsterdam: Elsevier 85(4): 545-666.
- Conn JW (1955) Primary aldosteronism, a new clinical syndrome. J Lab Clin Med 45(1): 3.
- Miguel V Palermo, Argelis S Linares, Hermógenes Malavé, Daniela Nieto, José LM Pompili (2024) Conn Syndrome Origins. About A Case [Internet]. Auctores.
- Hounsfield GN (1973) Computerized transverse axial scanning (tomography): Part 1. Description of system. Br J Radiol 46(552): 1016-1022.
- Korobkin M, Brodeur FJ, Francis IR, Quint LE, Dunnick NR (1998) CT time-attenuation washout curves of adrenal adenomas and nonadenomas. AJR Am J Roentgenol 170(3): 747-752.
- Geelhoed GW, Druy EM (1982) Management of the adrenal "incidentaloma". Surgery. 92(4): 866-874.
- Weiss LM (1984) Comparative histological study of adrenal cortical tumors. Am J Surg Pathol 8(3): 163-169.
- Kirschner MA, Powell RD, Lipsett MB (1964) Cushing's syndrome: nodular cortical hyperplasia of adrenal glands with clinical and pathological features suggesting adrenocortical tumor. J Clin Endocrinol Metab 24: 947-955.
- Gagner M, Lacroix A, Bolté E (1992) Laparoscopic adrenalectomy in Cushing's syndrome and pheochromocytoma. N Engl J Med 327(14): 1033.
- Kercher KW, Heniford BT, Matthews BD, Sing RF, Esquivel RF, et al. (2002) Laparoscopic vs. open adrenalectomy. Surg Endosc 16: 100-102.
- Mercan S, Seven R, Ozarmagan S, Tezelman S (1995) Endoscopic retroperitoneal adrenalectomy. Surgery 118(6): 1071-1075.
- Walz MK, Peitgen K, Hoermann R, Giebler RM, Mann K, et al. (1996) Posterior retroperitoneoscopy as a new minimally invasive approach for adrenalectomy: results of 30 adrenalectomies in 27 patients. World J Surg 20(7): 769-774.
- Piazza L, Caragliano P, Scardigno M, Sgroi AV, Marino G, et al. (1999) Laparoscopic robot-assisted right adrenalectomy and left ovariectomy (case reports). Chir Ital 51(6): 465-466.
- Horgan S, Vanuno D (2001) Robots in laparoscopic surgery. J Laparoendosc Adv Surg Tech A 11(6): 415-419.
- Castellucci SA, Curcillo PG, Ginsberg PC, Saba SC, Jaffe JS, et al. (2008) Single port access adrenalectomy. J Endourol 22(8): 1573-1576.
- Perretta S, Allemann P, Asakuma M, Dallemagne B, Marescaux J (2008) Adrenalectomy using natural orifice translumenal endoscopic surgery (NOTES): a transvaginal retroperitoneal approach. Surg Endosc 22: 1090.






























