Abstract
Epiprotect is a biosynthetic cellulose dressing utilised as treatment for partial thickness burns. Aims of this study include Retrospectively evaluating the utilisation of EpiProtect® in paediatric patients of a wide age range with burn injuries of varying total body surface area percentage (TBSA %), depth, and anatomical distribution. Also, to retrospectively identify complications at first dressing change in order to assess potential for same-day discharge of all eligible patients with EpiProtect® application in prospective studies. A retrospective analysis of medical notes was conducted for 39 patients who underwent Epiprotect application between December 2020 and February 2022. At our institution, Epiprotect is applied within 24 hours of injury under oral sedation +/- oral analgesia on the unit. The first check is performed at 48 hours. Thirty-Nine (n=39) patients (mean, 3 years old) with a mean TBSA% of 5% underwent application of Epiprotect for burns sustained mainly as scalds (95% of injuries). Only two patients experienced complications at the first check in the form of displacement requiring reapplication. 62% of cases remained as inpatient till the 48-hour check was conducted, 13% were discharged the same day of application and the remaining 25% required admission for other reasons. Epiprotect® is well tolerated and can feasibly be applied with same day discharge.
Keywords:Burn Injury; Scald; Wound Dressings; Biosynthetic Dressing; Paediatric Burns
Abbreviations:TBSA%: Total Body Surface Area Percentage; SPT: Superficial Partial Thickness; DD: Deep Dermal; FT: Full Thickness; LDI: Laser Doppler Imaging
Introduction
Children account for approximately half of all burn injuries treated annually in the UK with 25000 emergency department attendances and almost 4000 admissions. Scalding is one of the most common mechanisms of burns in children accounting for more than half of cases in children aged less than 5 years old [1]. EpiProtect® (S2 Medical; Regen Medical, Swindon, UK) is a biosynthetic cellulose dressing utilised as a treatment for partialthickness burns and a temporary dressing for full-thickness burns [2]. The biotechnology incorporates a variant of cellulose Acetobacter Xylinium, a gram-negative bacterium, synthesised in microfibrillar bundles [3]. This mimics and is analogous, in its nano-structure, to the collagen matrix of human skin and is also gas-permeable. It contains no animal, plant or human cells, and has high water absorption capacity, good permeability, and resistance to degradation [4]. Epiprotect® provides an adherent, transparent dressing that reduces exudates from burn wounds [2]. Of note, our department’s catchment population is diverse, in terms of language, culture, religion and geography. For cultural/ religious reasons, other dressings of porcine origin may not be accepted by some families. Our department, a regional paediatric burns unit, commenced the use of EpiProtect® in November 2018.
The aims of our study were to:
i. Retrospectively evaluate the utilisation of EpiProtect®
in paediatric patients of a wide age range with burn injuries of
varying total body surface area percentage (TBSA %), depth, and
anatomical distribution.
ii. Retrospectively identify complications at first dressing
change to assess the potential for same-day discharge of all eligible
patients with EpiProtect® application in prospective studies.
Methods
Data collection
The research was performed after approval by a local hospital audit/research committee; approval from the ethics board was not required, as this was a non-interventional study. As this was a retrospective study, verbal consent had been documented in notes before the use of EpiProtect® dressing as part of the department’s protocols. Data was collected retrospectively from patients who underwent EpiProtect® application in our regional paediatric burns unit located in Yorkshire, United Kingdom between December 2020 – February 2022. Data points collected included demographics, medical history, burn characteristics; aetiology, mechanism of injury, date and time of EpiProtect® application and first change; complications with first dressing change, inpatient/outpatient status, duration of admission; need for acute/delayed surgery; and healing time. This was obtained by review of notes available through the hospital’s electronic record system.
Standard operating procedure (SOP) for epiProtect® application and aftercare
Our standard operating procedure was as follows; burn distribution, size and depth were first assessed by referrers and then on admission by the burns team. Suitability for EpiProtect® was then determined after review by the burn’s registrar or consultant. Parents/caregivers were counselled and consented to the use of EpiProtect® with verbal consent obtained before application. The criteria for application were >3% total body surface area (TBSA) confluent burn wounds sustained less than 24 hours before application without contamination or visible signs of infection. Burns had to be predominantly superficial partial thickness burns on initial assessment. The anatomical location of burns was not a confounding factor to the dressing’s application. Contraindications in our unit are predominantly deep burns, presentation after 48 hours of injury and any concerns of infection. EpiProtect® was applied under oral sedation and/ or oral analgesia in the unit’s dressings/procedure room. Oral sedation used was oral/buccal midazolam with oral morphine/ intranasal diamorphine. Application was not done under a general anaesthetic or in an operating theatre environment unless indicated for other reasons such as patients with larger burns or requiring surgery. All children were fasted for 2 hours before oral sedation administration and monitored until fully recovered from sedation. Using aseptic non-touch technique (ANTT) with a minimum 2-person team (1 sterile, 1 unsterile person). Thorough wound cleansing was performed, swabs were taken, and clinical photograph was obtained and uploaded to our electronic records system. EpiProtect® was applied to the clean wound smoothing out bubbles and ensuring one-to-two-centimetre overlap with normal skin. Standard secondary dressings were applied over the EpiProtect®, consisting of a non-adherent dressing layer, usually Bactigras®, followed by Kerlix® and a further securing outer dressing based on the location.
The EpiProtect® itself was either left unsecured or secured with steri-strips or Histoacryl®. Histoacryl® was used typically in areas with a high risk of slippage such as the thighs, in younger children who may be more non-compliant or at staff’s discretion. The First EpiProtect® check was carried out 48 hours after application. This was either as an inpatient or outpatient with no standardisation as to which patients were deemed suitable for discharge and first check as an outpatient. In most cases, the patients were admitted until the first dressing check in 48hrs EpiProtect® was then left in situ until wound healing by re-epithelialisation at which point it spontaneously would lose adherence and fall off. In some cases, trimming of small nonadherent segments on the edges of the wound was done to keep contact with unhealed areas. After a first dressing change, our patients are followed up in the burns dressing clinic as outpatients until the wounds are fully healed and if needed in the paediatric scar clinic for those with hypertrophic scarring developing. As part of our unit’s policy, only wounds that take longer than 14 days to heal are followed up in MDT scar clinics with surgeons, therapists, and psychologists. All children have access to burns physiotherapists, occupational therapists and psychologists.
Results
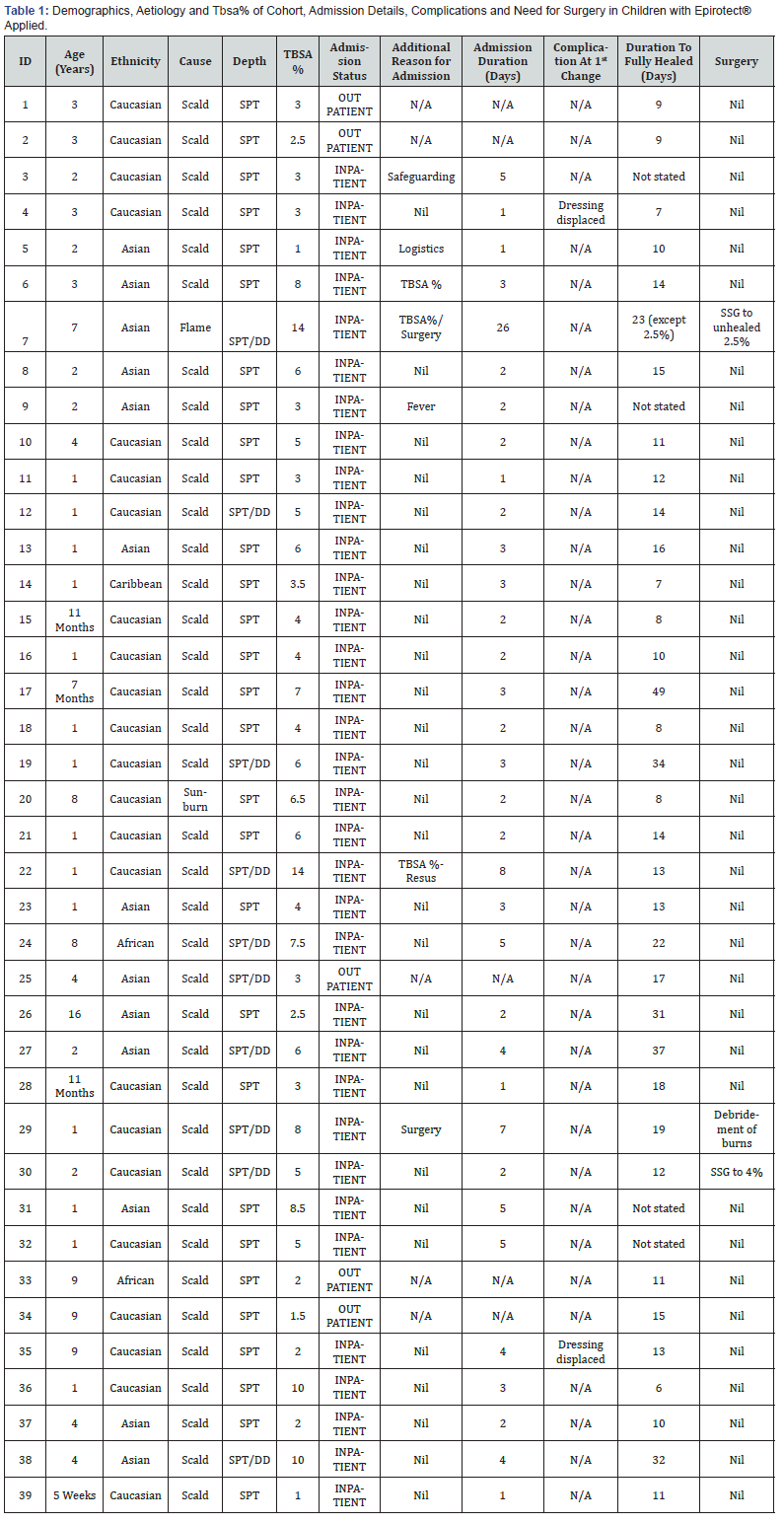
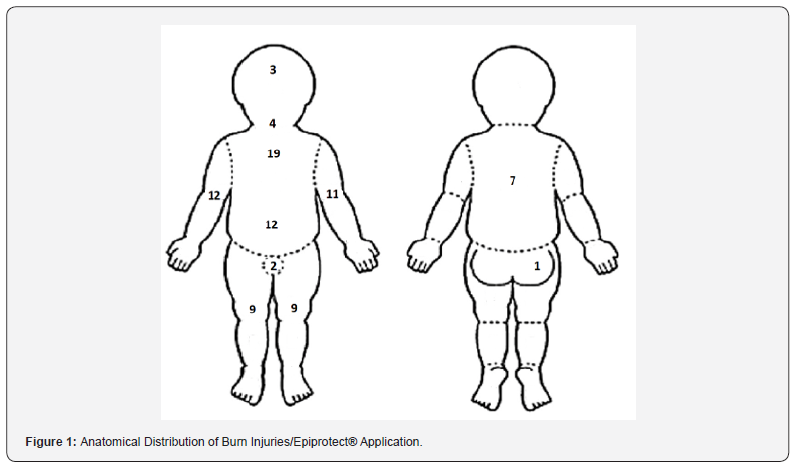
Over the two-year data collection period included in our study (December 2020 - February 2022), EpiProtect® was applied in 39 cases with a median age of 2 years and a median TBSA% of 4%. Further demographic data is presented in (Table 1). The prevailing burn wound depth was superficial partial thickness (SPT) representing 74.36%(n=29) whilst the most common mechanism was a scald injury representing 94.87% (n=37) of cases. Flame and sunburn were the implicated aetiology in one case each. The anatomical distribution of the burn wounds upon which EpiProtect® was applied is presented visually in (Figure 1) most burns affected the upper limbs, chest, and abdomen respectively. Most children (n=34) were inpatients until their first dressing change scheduled 48 hours postadmission. The range of admission stay was 1-26 days with a median duration of 2.5 days. The outlier in terms of admission duration was a child with a relatively larger and deeper burn (Resuscitation burn of 14% TBSA) who needed a general anaesthetic for the application of EpiProtect® which was done concomitantly with debridement of deeper burns in theatre, whilst all others in the group had a successful application with oral sedation on the unit. Six children had additional reasons for their inpatient stay other than the application of EpiProtect®. Some alternative reasons for staying in included the need for surgery, the size of the burn (n=4) relative to the need for resuscitation, safeguarding concerns, travel logistics, and fever.In terms of complications, the EpiProtect® displaced prematurely in 5.13% (n=2) cases which was picked up on the first check and reapplied. It was not documented if these were secured using Histoacryl® or steri-strips in the clinical records. All dressings were reinforced with further layers of dressings.
Superficial Partial Thickness, DD- Deep Dermal, FT - Full Thickness, TBSA - Total Body Surface Area.
Only one child was not brought to their first check appointment but on later follow-up had not experienced complications with the dressing or wound healing. There were no recorded intolerances to the dressing. One child developed a clinical burns wound infection confirmed on swabs taken. With regards to healing, this was determined by identifying in dressing clinic notes the date ‘fully healed’ was documented. This however was not reserved for only the superficial parts of burns and often referred to the time at which all burns had healed. Thus, was unreliable in children with mixed depths of burns. Furthermore, as wounds were reviewed weekly at dressing clinics; it would infer the actual healing duration for these burns was shorter than recorded. The median time to fully heal was 13 days, with a range of 6 to 49 days. In the child with the longest duration of healing; this was from a deep 0.25% patch that was treated non operatively thus ‘fully healed’ was only documented after this patch had healed. Two children required operative management of their burn wounds during admission as seen in (Table 1). The first child with TBSA 8% (SPT/ DD) required debridement of some burn wounds at the time of EpiProtect® application and the other, split skin grafting as an inpatient to 2.5% unhealed deep areas out of a total TBSA 14%. A third child underwent semi-elective split skin grafting for 4% unhealed TBSA after discharge. All other patients were followed up in the dressing clinic and were discharged from the service with advice and safety netting once fully healed.
Discussion
In this paper, we demonstrate our experience and utilisation procedures of EpiProtect®. Our study findings suggest that it is minimal dressing-related problems occurring in the first 48 hours after dressing application that would warrant an inpatient admission in this cohort. Although our policy does not encourage routine overnight admission of our patients, only 5 out of our cases examined were discharged on the day of application. Being a regional burns unit for children, many patient families may live considerably far distances from the hospital and would find it financially burdensome to return for a dressing change within 48 hours. We found that these patients we successfully discharged on the day of application were families where the logistics of travel were easier coupled with a child with no clinical indication for admission. In our study period, 38 out of a cohort of 39 children underwent EpiProtect® application on the burn unit with only oral sedation and analgesia.
This practice differs from most burn units and centres in the UK where similar biosynthetic cellulose dressings have been applied under general anaesthesia in an operating theatre environment. The advantages of oral sedation combined with potential same-day discharge are the return of the child to a familiar environment, prudent use of sparse inpatient burns beds and reducing the proposed risk of neurodevelopmental injury associated with general anaesthetics in children [5,6]. Our study findings suggest that it is feasible for application and same-day discharge except in conditions such as suspicion of non-accidental injury, late time of application, or on clinical grounds (size of burn usually). On reviewing individual patient notes, the description of burns was typically based on the clinical review by a doctor in the burn department on initial assessment. Some of these were found to be deeper in later reviews and thus took longer to heal. Our study experienced similar complications with dressing displacement as Quereshi et al [7]. This was usually mitigated with the use of Histoacryl® but was limited in our unit when specialist nurses/doctors were not present due to prescription restrictions. EpiProtect® dressing changes were generally seamless, fewer, requiring simple analgesia only and were minimally distressing for children and parents.
Limitations
Future work will focus on expanding the study population and prospectively assessing the outcomes of standardized same-day discharge protocols where appropriate. We also plan to incorporate routine use of Laser Doppler Imaging (LDI) for more precise burn depth assessment and systematically evaluate pain scores during dressing changes. Gathering feedback from patients, parents, and colleagues will be integral to refining these protocols and ensuring comprehensive care. Long-term follow-up of these patients will provide essential data on burn healing, the extent of scarring, and effective scar management strategies. Such information will deepen our understanding and improve patient care outcomes. Additionally, designing protocols for consistent and standardized documentation of healing times and burn depth, alongside excluding cases with incomplete data, will enhance data reliability and reduce bias.
Conclusion
EpiProtect® demonstrates excellent tolerability as an epidermal substitute dressing. Our findings highlight a safe method of application without the need for general anaesthesia, thereby eliminating routine hospital stays for recovery. This approach minimizes the risks associated with general anaesthesia and offers logistical and cost advantages by reducing inpatient stays and optimizing burns bed availability. The adoption of this standard operating procedure (SOP) can support same-day discharge for children treated with EpiProtect® with a review in 48hours in patients who have smaller TBSA and have no other contraindication for discharge. Finally, we aim to analyse a fiveyear retrospective database (2018–2023) from our unit to identify trends and insights that can further inform clinical practice and improve patient outcomes.
Lay Summary
i. We present our study
ii. Epiprotect® is well tolerated and poses no religious or
cultural considerations.
iii. Epiprotect® can be feasibly and safely applied with
same day discharge, and first dressing check done as outpatient.
References
- Kemp A, Jones S, Lawson Z, Maguire SA (2014) Patterns of burns and scalds in children. Arch Dis Child 99(4): 316–321.
- Regan Medical UK- Part of Joint operations family, https://www.regenmedical.co.uk/epiprotect/. Last accessed 26/9/2023.
- Gayathry G, Gopalaswamy G (2014) Production and Characterization of Microbial Cellulosic Fibre from Acetobacter Xylinum. Indian 39(1): 93-96.
- Matilda Karlsson, Pia Olofsson, Ingrid Steinvall, Folke Sjöberg, Johan Thorfinn, et al. (2019) Three Years' Experience of a Novel Biosynthetic Cellulose Dressing in Burns. Advances in Wound Care 8(2): 71- 76.
- Lee JH, Zhang J, Wei L, Yu SP (2015) Neurodevelopmental implications of the general anesthesia in neonate and infants. Exp Neurol 272: 50–60.
- Slikker W Jr, Zou X, Hotchkiss CE, Divine RL, Sadovova N, et al. (2007) Ketamineinduced neuronal cell death in the perinatal rhesus monkey. Toxicol Sci 98(1): 145–158.
- Qureshi MA, Lalwani P, Asad I, Khan-Assad N, Mohamedali S, et al. (2021) Single stage first layer biosynthetic cellulose dressing versus non-adherent gauze management in paediatric burns. Burns Open 5(4): 17-23.






























