Surgical Site Infection in Abdominal Procedures: An Update on Prevention and Clinical Approach
Aishwarya Yannamani1, Thulasi Ram Gudi2, Blanca Estefanie Avalos Quijano3, Coralvia Yaroslangna Villanueva Pérez4, Miguel Eduardo Rodriguez Rodriguez5, Jhon Nicol Navarro Gonzalez6, Pushan Aggarwal1, Karla Banuelos Gonzalez7, Oluwatomisin Olapoju8, Nengimote Prince Ala-Binte8, Daniel Arias9, María Fernanda Artavia Pineda9 and Maria Isabel Gomez10*
1Kasturba Medical College, Manipal, Karnataka, India
2Merit Health River Region, Vicksburg, USA
3Universidad Evangelica de El Salvador, El Salvador
4Universidad Nacional Experimental Francisco de Miranda, Venezuela
5Universidad de Oriente, Venezuela. Larkin Community Hospital, USA
6Universidad del Zulia. Venezuela
7Centro de Estudios Universitarios Xochicalco, Mexico
8Richmond Gabriel University, Saint Vincent, and the Grenadines
9Universidad de Ciencias Médicas (UCIMED), Costa Rica
10Universidad del Valle de México, México
Submission:April 21, 2023; Published:May 01, 2023
*Corresponding author: Maria Isabel Gomez Coral, Universidad Del Valle de México, México, USA
How to cite this article: Aishwarya Yannamani1, Thulasi Ram Gudi, Blanca Estefanie Avalos Quijan, Coralvia Yaroslangna Villanueva Pérez, Miguel Eduardo Rodriguez Rodriguez, et al. Surgical Site Infection in Abdominal Procedures: An Update on Prevention and Clinical Approach. Open Access J Surg. 2023; 14(2): 555885 DOI: 10.19080/OAJS.2023.14.555885.
Abstract
A surgical site infection is an infection occurring within 30 days of the procedure. They are common postoperative complications that can cause significant patient morbidity and mortality. SSIs occur in 2% to 4% of all patients undergoing inpatient surgical procedures. They constitute the second most common type of healthcare-associated infection after urinary tract infections. The etiology of the infection is multifactorial, with both patient-related and surgical-related factors playing a role. Patient-related factors include advanced age, immunocompromised status, obesity, diabetes mellitus, smoking, and poor nutritional status. Surgical-related factors include prolonged operative time, contaminated surgical instruments or equipment, poor surgical technique, and inadequate wound care. Common symptoms of SSI involve surgical site erythema, delayed healing, fever, pain, tenderness, or swelling. The diagnosis is commonly based on evidenced purulent discharge from superficial and deep wounds, organisms isolated from cultures. Overall, the management of surgical site infections includes wound care, antimicrobial therapy, wound exploration, and debridement. In addition, effective infection prevention strategies, including appropriate antimicrobial prophylaxis and adherence to infection prevention practices, are crucial in reducing the incidence of SSIs in surgical patients. This article provides a broad overview of the clinical approach to this day-to-day condition.
Keywords: Surgical site infection; Abdominal surgery; Prevention; Treatment
Keywords: SSI: Surgical site infection; CT: Computerized tomography; MRI: Magnetic resonance imaging; HAI: Healthcare-associated infection; UTI: Urinary tract infection; CDC: Centers for Disease Control and Prevention; MSSA: Methicillin-susceptible Staphylococcus aureus; MRSA: Methicillin-resistant Staphylococci; NPWT: Negative pressure wound therapy
Introduction
A surgical site infection (SSI) is an infection that occurs after surgery in the part of the body within 30 days of the procedure [1]. There are 3 types of surgical site infections: superficial incisional (occurs just in the area of the skin where the incision was made), deep incisional (occurs beneath the incision area in muscle and the tissues surrounding the muscles), and organ or space surgical site infection (can occurs in any area of the body other than skin, muscles, and surrounding tissue that was involved in the surgery) [2]. SSI can be classified based on how clean or contaminated they are: clean wound, clean/contaminated wound, contaminated wound, and dirty/infected wound [3]. SSIs occur in 2% to 4% of all patients undergoing inpatient surgical procedures [4]. Risk factors for SSI include patient factors (age, tobacco use, diabetes, and malnutrition) and procedure-specific risk factors (including emergency surgery and the degree of bacterial contamination of the surgical wound at the time of the procedure).
Microbial contamination of the surgical wound is the first step in the development of an SSI, which may come from either endogenous (patient’s skin, mucous membranes, and hollow viscera) or exogenous sources (air, instruments, materials, and staff members) [5]. To prevent SSI, healthcare providers should practice good hand hygiene before and during surgery, wear sterile protective equipment, and thoroughly clean the skin at the surgery site. The most common symptoms of SSI are redness, delayed healing, fever, pain, tenderness, or swelling. The diagnosis can be made by clinical examination and microbiological swabs to identify causative organisms. If there is suspicion of deep-seated infection, ultrasound or CT/MRI imaging might be used [5]. SSI can be treated with antibiotics, depending on the causative organism. However, additional surgery may be needed to treat the infection. This narrative review aims to update patients’ prevention and clinical approach to SSIs in abdominal procedures.
Epidemiology
Surgical site infections (SSIs) are common postoperative complications that can cause significant patient morbidity and mortality. Epidemiological studies have shown that SSIs affect approximately 2-5% of all surgical procedures and are the second most common type of healthcare-associated infection (HAI) after urinary tract infections (UTIs) [6,7]. However, SSI prevalence can vary widely depending on the type of surgery, patient population, and healthcare setting. A study conducted by the Centers for Disease Control and Prevention (CDC) found that the overall SSI rate in the United States was 1.9% in 2015, with higher rates reported for certain types of surgeries such as colon surgery (3.8%) and hysterectomy (2.4%) [8]. Moreover, SSI incidence rates also vary depending on the type of surgery and patient population. A meta-analysis of 25 studies found that the overall incidence rate of SSIs was 5.6% in patients undergoing surgery [6,7]. SSIs were higher in patients undergoing abdominal surgeries (9.2%) and lower in orthopedic surgeries (2.4%).
Lastly, mortality rates associated with SSIs can be significant, particularly in vulnerable populations such as the elderly or immunocompromised patients. A recent study found that patients who developed SSIs had a 2.5-fold higher risk of mortality than patients who did not [8]. Risk factors for SSIs include patientrelated factors such as age, comorbidities, and immune status, as well as surgical-related factors such as duration of surgery, surgical technique, and type of surgery. A systematic literature review identified several risk factors for SSIs, including advanced age, obesity, smoking, diabetes mellitus, and prolonged surgical time [9]. SSIs are a common postoperative complication resulting in significant patient morbidity and mortality. Prevalence and incidence rates of SSIs vary depending on the type of surgery and patient population, and risk factors for SSIs include patientrelated and surgical-related factors. Therefore, effective infection prevention strategies, including appropriate antimicrobial prophylaxis and adherence to infection prevention practices, are crucial in reducing the incidence of SSIs in surgical patients.
Etiology & Pathogenesis
Surgical site infections (SSIs) are caused by the invasion of microorganisms into a surgical site, resulting in inflammation and tissue damage. The etiology of SSIs is multifactorial, with both patient-related and surgical-related factors playing a role [10,11]. Patient-related factors contributing to developing SSIs include advanced age, immunocompromised status, obesity, diabetes mellitus, smoking, and poor nutritional status. These factors impair the body’s immune response and increase the risk of infection [12]. Surgical-related factors that increase the risk of SSIs include prolonged operative time, contaminated surgical instruments or equipment, poor surgical technique, and inadequate wound care. This type of surgery also influences the risk of SSIs, with procedures involving the implantation of prosthetic materials or foreign bodies having a higher risk of infection [13,14].
The pathogenesis of SSIs involves a complex interplay between the host immune response and the invading microorganisms. For example, microorganisms can enter the surgical site through direct contact with contaminated surgical instruments or equipment or through hematogenous spread from a distant infection site [14]. Once microorganisms enter the surgical site, they adhere to the surrounding tissue and form a biofilm, which provides a protective environment for bacterial growth and resistance to antimicrobial agents. The bacteria then secrete toxins and enzymes that cause tissue damage and trigger an inflammatory response [13]. The pathophysiology of SSIs involves a cascade of events that lead to tissue destruction, impaired wound healing, and systemic complications. The inflammatory response to infection can cause fever, leukocytosis, and other signs of systemic infection. Tissue destruction can lead to impaired wound healing, dehiscence, and the formation of abscesses or fistulas. In some cases, SSIs can lead to sepsis, septic shock, and other life-threatening complications. The severity of SSIs depends on the virulence of the infecting organism, the host immune response, and the timely initiation of appropriate antimicrobial therapy [12,13]. In summary, SSIs result from invading microorganisms into a surgical site. The etiology of SSIs is multifactorial, with both patient-related and surgical-related factors contributing to the risk of infection. SSIs’ pathogenesis involves forming a biofilm by invading microorganisms, which triggers an inflammatory response and tissue damage. The pathophysiology of SSIs involves a cascade of events that lead to tissue destruction, impaired wound healing, and systemic complications.
Clinical Presentation
Surgical site infections (SSIs) are a common complication following abdominal procedures, with varying clinical presentations, symptoms, and signs depending on the type of infection. The three SSI types are superficial, deep incisional, and organ/space infections. First, superficial SSIs are limited to the skin and subcutaneous tissues, with symptoms appearing within 30 days of surgery. Clinical presentation includes erythema, warmth, tenderness, and induration around the surgical incision site. Drainage may also be present, and wound cultures may reveal bacterial growth. Risk factors for superficial SSIs include obesity, smoking, and prolonged hospitalization [15,16]. The second type of SSI, deep incisional SSI, involves deeper tissues beneath the skin and can present within 30 to 90 days after surgery [17]. Clinical presentation includes fever, wound dehiscence, and purulent drainage. There may also be erythema and tenderness around the incision site, with possible abscess formation. Diagnosis is confirmed through imaging, wound cultures, and histopathology [16,18]. Lastly, organ/space SSIs occur in any body area other than the skin or subcutaneous tissues, such as the abdominal cavity [19,20]. Clinical presentation includes fever, leukocytosis, and abdominal pain. Patients may also experience nausea, vomiting, and diarrhea. In addition, imaging may reveal fluid collections, abscesses, or other signs of infection. Diagnosis is confirmed through imaging, culture, and histopathology [21].
Prevention Strategies
FWhile the preventative measures for surgical site infections differ depending on the wound class, various approaches are taken to prevent SSI. One of these includes the avoidance of elective surgeries in patients with active infections. In addition, the use of prophylactic antibiotics is also implemented to prevent SSI. For example, intravenous antibiotics, such as second-generation cephalosporins, are recommended against aerobic and anaerobic bacteria at least 30 minutes before a skin incision for elective colorectal surgeries [22]. More recently, adding a single dose of an oral antibiotic, such as 1 g of ornidazole, to the intravenous antibiotic has been demonstrated to effectively reduce SSI in deep and organ space [22]. While proper skin preparation is routinely implemented for preventing SSI, some pre-operative measures significantly impact managing the risk of infection. For example, the implementation of showers with chlorhexidine is statistically significant for preventing SSI [23]. Abdominal wall antisepsis with alcoholic chlorhexidine solution is also essential for skin preparation before surgery [23]. Surgical techniques, like pre-operative preparation, is also essential for preventing SSI due to the importance of skin closure for colonizing bacteria on the surgical wound. Suturing techniques, continuous sutures, or interrupted stitches do not have a significant impact on preventing SSI. However, different techniques to close the surgical incision impact the wound dehiscence rate. Wound dehiscence occurs at a higher rate in interrupted stitches when compared to continuous sutures [24]. Moreover, the materials used to close the incision are necessary to prevent infections. Although there are limited clinical studies, using sutures with antimicrobial properties, such as triclosan-coated sutures, is recommended in efforts to reduce bacterial colonization and biofilm formation [24,25].
Treatment & Prognosis
The general management of surgical site infections includes non-surgical measures such as wound care, antimicrobial therapy, and even invasive intervention in wound exploration and debridement. The basics of wound management apply to all surgical wounds as well- ensure good vascularity of the site, maintain cleanliness around the wound, control comorbidities to acceptable levels, and use topical agents where necessary. In the event of SSIs, it is important to ensure good wound care- frequent wound packing, good dressing, good drainage of secretions, and timely and meticulous clearance of slough, pus, and debris from the site [28,29].
Antibiotics and antimicrobial drugs may be warranted if infections set in around the surgical site or disseminate to distal sites. Occurrence of localized cellulitis, presence of patient risk factors such as comorbidities (Diabetes Mellitus, etc.) or chronic steroid use, or overt signs of infection (fever, significantly elevated white cell count) is some signs that warrant prompt antimicrobial drug administration [28,29]. The approach to selecting antimicrobial drugs can be tailored based on knowledge of causative pathogens. Therefore, initiating Empiric Antibiotic Therapy while awaiting culture and sensitivity testing is frequently pertinent. It has been observed in recent times that while the most common causative organism for surgical site infections remains to be MSSA / Staphylococcus aureus, Coagulase-negative Staphylococci, Streptococcus sp., and Enterococci, there has also been a sharp/steep ascent in the cases of Methicillin-Resistant Staphylococci (MRSA) [30]. Hence, culture and sensitivityguided antimicrobial treatment act as a precise tool, with a higher clearance rate of such infections. The duration of the course of antimicrobial therapy can also be determined based on the extent of the infection- superficial infections requiring shorter regimens, as compared to more prolonged and intensive regimens for widespread and extensive infections that may even involve other organs (systems) [31-35]. Therefore, it is essential to pay attention to the evolution of the infection and adjust drug therapy accordingly- to detect and manage resistant pathogens/ infections early on and avoid dire consequences that may lead to amputations or even mortality.
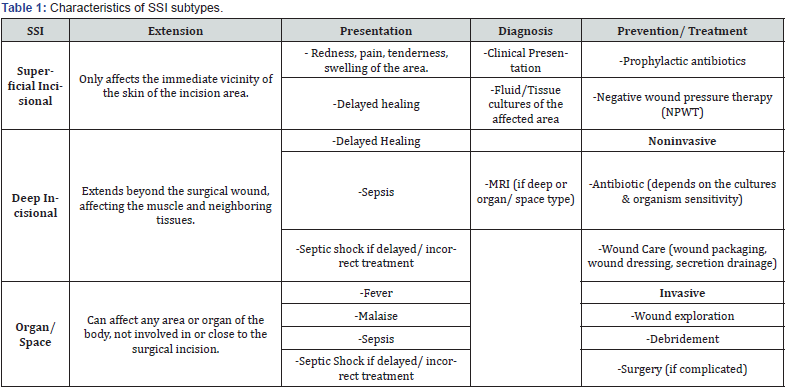
Invasive management in wound exploration and debridement may be warranted when SSIs become complicated with the undrained fluid collection, large amounts of necrotic debris, or the spread of infection into deeper skin layers (significantly beyond the fascia). Superficial wound exploration can be done bedside or in outpatient settings by practicing sterile precautions, carefully excising devitalized tissue, and draining accumulated pus/fluid with clear and healthy margins. Deeper exploration may require reopening wounds and invasive exploration that may have to be done in Operating Rooms. While this may seem invasive, it is explicable, given the potential risks of untreated SSIs. Regarding post-surgical care, Negative pressure wound therapy (NPWT) has been associated with lower infection rates and earlier healing of wounds compared to comparable wounds without similar care [36,37]. A summary of the definition, diagnosis, clinical manifestations, prevention strategies, and management of each type of SSI is illustrated in Table 1.
Conclusion
As discussed, SSIs are a common complication following abdominal surgical procedures that occur within 30 days of surgery and involve the surgical incision, deep tissue, or organ/space. They can result in significant morbidity and mortality. Various pathogens can be involved, including bacteria, viruses, and fungi. The etiology of SSIs is multifactorial, with both patient and surgical factors playing a role. The prevalence of SSIs varies depending on the type of surgery performed, but it is estimated to be between 2-5% of all surgical procedures, with a much higher incidence for certain types of surgery, such as colorectal surgery. SSIs’ pathogenesis involves colonizing the surgical site by pathogenic microorganisms, followed by their invasion and proliferation. The immune response to these pathogens can also contribute to tissue damage and inflammation. Clinical presentation of SSIs can vary depending on the type and severity of infection. Symptoms include fever, wound drainage, redness and swelling at the surgical site, and increased pain or tenderness. The diagnosis is typically made based on clinical signs and symptoms and laboratory tests such as wound cultures and blood tests to identify the causative organism. Prevention of SSIs involves a combination of measures, including appropriate use of antibiotics, sterile technique during surgery, and patient optimization before and after surgery. Treatment strategies typically involve a combination of antibiotics and surgical intervention, such as debridement or drainage of the infected site. In severe cases, hospitalization and intensive care may be required. Future research studies are needed to improve the outcomes for patients with SSIs. Areas of research include the development of more effective prevention strategies, the identification of novel therapeutic targets, and a better understanding of the pathogenesis of these infections.
References
- Lardière-Deguelte S, Ragot E, Amroun K, Piardi T, Dokmak S et.al. (2015) Hepatic abscess: Diagnosis and management. J Visc Surg 152(4): 231-243.
- Khim G, Em S, Mo S, Townell N (2019) Liver abscess: diagnostic and management issues found in the low resource setting. Br Med Bull. 11;132(1): 45-52.
- Akhondi H, Sabih DE (2022) Liver Abscess. In: StatPearls [Internet]. Stat Pearls Publishing.
- Nepal P, Ojili V, Kumar S, Kaur N, Nagar A (2020) Beyond pyogenic liver abscess: a comprehensive review of liver infections in emergency settings. Emerg Radiol. 27(3): 307-320.
- Chattopadhyay B (1983) Pyogenic liver abscess. J Infect 6(1): 5-12.
- Siu LK, Yeh KM, Lin JC, Fung CP, Chang FY (2012) Klebsiella pneumoniae liver abscess: a new invasive syndrome. Lancet Infect Dis 12(11): 881-887.
- Stanley SL Jr (2003) Amoebiasis. Lancet 361(9362): 1025-1034.
- Shi SH, Zhai ZL, Zheng SS (2018) Pyogenic Liver Abscess of Biliary Origin: The Existing Problems and Their Strategies. Semin Liver Dis 38(3): 270-283.
- Moinuddin M (1987) Pyogenic liver abscess. Compr Ther 13(3): 26-31.
- Reid-Lombardo KM, Khan S, Sclabas G (2010) Hepatic cysts and liver abscess. Surg Clin North Am 90(4): 679-697.
- Rustgi AK, Richter JM (1989) Pyogenic and amebic liver abscess. Med Clin North Am 73(4): 847-58.
- Sifri CD, Madoff LC (2015) Infections of the liver and biliary system (liver abscess, cholangitis, cholecystitis). In: Bennett JE, Dolin R, Blaser MJ (eds.). Principles and Practice of Infectious Diseases, 8th Philadelphia: Elsevier Saunders.
- Herbinger KH, Fleischmann E, Weber C, Perona P, Löscher T, et al. (2011) Epidemiological, clinical, and diagnostic data on intestinal infections with Entamoeba histolytica and Entamoeba dispar among returning travelers. Infection 39(6): 527-535.
- Wuerz T, Kane JB, Boggild AK, Krajden S, Keystone JS, et al. (2012) A review of amoebic liver abscess for clinicians in a nonendemic setting. Can J Gastroenterol 26(10): 729-733
- Andrade-Filho Jde S (2012) Analogies in medicine: anchovy paste in the liver. Rev Inst Med Trop Sao Paulo 54(4): 234.
- Longworth S, Han J (2015) Pyogenic liver abscess. Clin Liver Dis (Hoboken) 6(2): 51-54.
- Khim G, Em S, Mo S, Townell N (2019) Liver abscess: diagnostic and management issues found in the low resource setting. Br Med Bull 132(1): 45-52.
- Mohsen AH, Green ST, Read RC (2002) Liver abscess in adults: ten years experience in a UK centre. QJM 95(12):797-802.
- Rahimian J, Wilson T, Oram V, et al. Pyogenic liver abscess: recent trends in etiology and mortality. Clin Infect Dis 39: 1654-1659.
- Kaplan GG, Gregson DB, Laupland KB. (2004) Population-based study of the epidemiology of and the risk factors for pyogenic liver abscess. Clin Gastroenterol Hepatol2: 1032-1038.
- Webb GJ,Chapman TP, Cadman PJ, Gorard DA (2014) Pyogenic liver abscess. Frontline Gastroenterol 5(1): 60-67.
- González JJ, Baldonedo RF, J A Alvarez Pérez L Sanz, G Carreño, A Junco, et al. (2001) Clinical course, treatment, and multivariate analysis of risk factors for pyogenic liver abscess. Am J Surg 181(2):177-186.
- Reid-Lombardo KM, Khan S, Sclabas G (2010) Hepatic and liver abscess. Surg Clin North Am 90(4): 679-697.
- Halvorsen, R. A (1984) The variable CT appearance of hepatic abscesses." American journal of roentgenology 1425(1984): 941-946.
- Amroun K, Piardi T, Dokmak S, Bruno O, Appere F, et al. (2015) Hepatic abscess: Diagnosis and management. J Visc Surg 152(4): 231-243.
- Sarah Longworth, Han J. Pyogenic liver abscess. Clin Liver Dis (Hoboken) 6(2): 51-54.
- Fontanilla T, Noblejas A, Cortes C, Minaya J, Mendez S, et al. (2013) Contrast-enhanced ultrasound of liver lesions related to arterial thrombosis in adult liver transplantation. J Clin Ultrasound 41 (8): 493-500.
- Cavailloles FA, Mure A, Nasser H, Lecapitaine AL, Granier F (2014) Multiple liver amoebic abscesses detected on FDG PET/CT. Clin Nucl Med 39 (1): 79-80.
- Jindal A, Pandey A, Sharma MK, Mukund A, Vijayaraghavan R, et al. (2021) Management Practices and Predictors of Outcome of Liver Abscess in Adults: A Series of 1630 Patients from a Liver Unit. J Clin Exp Hepatol 11(3): 312-320.
- Nyabera A, Shaban L, Hijazin K, Tumarinson T (2021) Community-Acquired Methicillin-Resistant Staphylococcus Aureus Hepatic Liver Abscess. Cureus13(1): e12560.
- Cherian J, Singh R, Varma M, Vidyasagar S, Mukhopadhyay C (2016) Community-Acquired Methicillin-Resistant Pyogenic Liver Abscess: A Case Report. J Investig Med High Impact Case Rep 4(3): 2324709616660576.
- Hasan S, Fearn R (2018) Fungal liver abscess in an immunocompetent patient who underwent repeated ERCPs and subtotal cholecystectomy. BMJ Case Rep 2018: bcr2017222013.
- Hernandez-Calleros J, Tellez-Avila FI, Torre A, Uribe M (2009) Image-guided percutaneous procedure plus metronidazole versus metronidazole alone for uncomplicated amoebic liver abscess. Cochrane Database Syst Rev (1): CD004886.
- Babikir TB, Alnehmi FS, Abdalrahman HH, Abdelmalik MAA, Ali AM, Fadlalmola HA, et al. (2022)Percutaneous needle aspiration versus catheter drainage in the management of liver abscess: an updated systematic review and meta-analysis. ANZ J Surg.
- Harclerode TP, Gnugnoli DM. Percutaneous Abscess Drainage. In: StatPearls [Internet].
- Men S, Akhan O, Köroğlu M (2002) Percutaneous drainage of abdominal abcess. Eur J Radiol. 43(3): 204-218.
- Hayashi Y, Uchiyama M, Inokuma T, Torisu M (1989) Gas-containing pyogenic liver abscess--a case report and review of the literature. Jpn J Surg 19 (1): 74-77.
- Zerem E, Hadzic A (2007) Sonographically guided percutaneous catheter drainage versus needle aspiration in the management of pyogenic liver abscess. AJR Am J Roentgenol 189(3): W138-142.
- Mortelé KJ, Segatto E, Ros PR (2004) The infected liver: radiologic-pathologic correlation. Radiographics. 24(4): 937-955.
- vanSonnenberg E, Wittich GR, Goodacre BW, Casola G, D'Agostino HB (2001) Percutaneous abscess drainage: update. World J Surg 25(3): 362-369.
- Ndong, Abdourahmane, et al. “Efficacy of Laparoscopic Surgery in the Treatment of Hepatic Abscess: A Systematic Review and Meta-Analysis.” Annals of Medicine and Surgery 75: 103308.
- Cemalettin Aydin, Turgut Piskin, Fatih Sumer, Bora Barut, Cuneyt Kayaalp (2010_) Laparoscopic Drainage of Pyogenic Liver Abscess. JSLS: Journal of the Society of Laparoendoscopic Surgeons 14(3): 418-420.
- Tak, Nikhil, Rajendra Bagree, Gaurav Jalendra, Hetish M. Reddy (2021) Laparoscopic Management of Liver Abscess: A Viable Alternative in USG Guided Percutaneous Drainage Failure Cases.” International Surgery Journal (8): 637-640.
- Tan L, Zhou HJ, Hartman M, Ganpathi IS, Madhavan K, et al. (2013) Laparoscopic drainage of cryptogenic liver abscess. Surg Endosc 27(9): 3308-3314.
- Reid-Lombardo KM, Khan S, Sclabas G (2010) Hepatic cysts and liver abscess. Surg Clin North Am. 90(4): 679-97.
- Frey CF, Zhu Y, Suzuki M, Isaji S (1989) Liver abscesses. Surg Clin North Am 69(2): 259-271.
- Liew KV, Lau TC, Ho CH, Cheng TK, Ong YS, (2000) Pyogenic liver abscess--a tropical centre's experience in management with review of current literature. Singapore Med J 41(10): 489-492.
- Romano G, Agrusa A, Frazzetta G, De Vita G, Chianetta D et al. (2013) Laparoscopic drainage of liver abscess: case report and literature review. G Chir 34(5-6): 180-182.
- Leggieri N, Marques-Vidal P, Cerwenka H, Denys A, Dorta G, et al. (2010) Migrated foreign body liver abscess: illustrative case report, systematic review, and proposed diagnostic algorithm. Medicine (Baltimore) 89(2): 85-95.
- Ghosh JK, Goyal SK, Behera MK, Tripathi MK, Dixit VK, et al. (3015) Efficacy of aspiration in amebic liver abscess. Trop Gastroenterol 36(4): 251-255.
- Garcea G, Rajesh A, Dennison AR (2012) Surgical management of cystic lesions in the liver. ANZ J Surg 83(7-8): E3-E20.
- Ch Yu S, Hg Lo R, Kan PS, Metreweli C (1997) Pyogenic liver abscess: treatment with needle aspiration. Clin Radiol 52(12): 912-916.
- Kodama T, Yokoyama T (1995) [Liver abscess (solitary liver abscess, multiple liver abscess, subcapsular liver abscess]. Ryoikibetsu Shokogun Shirizu 7: 100-103.






























