Hepatic Regeneration index and Completeness of Regeneration in Remnant Liver after Partial Hepatectomy for Hepatocellular Carcinoma and Cirrhosis
Xuesong Li1, Wan Yee Lau 1,2, Deyu Guo3, Shuguo Zheng1, Geng Chen4, Yujun Ji1, Shu Chen1 and Feng Xia1*
1Institute of Hepatobiliary Surgery and Southwest Cancer Center, Army Medical University, China
2Faculty of Medicine, The Chinese University of Hong Kong, China
3Institute of Pathology and Southwest Cancer Center, Army Medical University, China
4Institute of Hepatobiliary Surgery, Army Medical University, China
Submission:December 07, 2021; Published:December 13, 2021
*Corresponding author: Feng Xia, Institute of Hepatobiliary Surgery and Southwest Cancer Center, Army Medical University, China
How to cite this article: Xuesong L, Wan Yee L, Deyu G, Shuguo Z, Geng C, et al. Hepatic Regeneration index and Completeness of Regeneration in Remnant Liver after Partial Hepatectomy for Hepatocellular Carcinoma and Cirrhosis. Open Access J Surg. 2021; 13(4): 555868 DOI: 10.19080/OAJS.2021.13.555868.
Abstract
Background and objectives
This study aimed to evaluate whether the regeneration index (RIx) and completeness of regeneration of remnant liver volume (RLV) are related to liver fibrotic stages, resected liver parenchymal volumes (RLPV) and long-term survival outcomes in patients with hepatocellular carcinoma (HCC) and cirrhosis.
Methods
Sixty-two HCC patients were included in regular CT volumetry assessments for up to 17 months after partial hepatectomy. The RIx of remnant liver was calculated by the formula at the different time points as: [regenerated RLV- original RLV] / original RLV ×100. The patients were divided into subgroups according to the fibrotic stages and the RLPV as a percentage of total liver volume (TLV). The mean RIx and completeness of regeneration at 17 months of RLV were compared between each of the subgroups.
Results
The mean RIx declined as the fibrotic stages increased. It increased with increasing percentages of RLPV to TLV. RIx was high as measured by CT volumetry around 1 month after hepatectomy (t1) and then reached a plateau, especially in patients with severe liver fibrosis or with a minor hepatic volume resection. There were no significant differences in long-term disease-free survival and overall survival outcomes between the high RIx (≥50 percent) and low RIx (<50 percent) subgroups.
Conclusions
Liver regeneration had basically been completed (94.1%±14.1) within 4 months in cirrhotic livers after hepatectomy. The RIx showed no correlation with long-term disease-free and overall survival outcomes for HCC patients with cirrhosis.
Keywords: Liver Regeneration; Cirrhotic Liver; Partial Hepatectomy; Liver Volumetry
Abbreviations: Computed Tomography (CT); Hepatocellular Carcinoma (HCC); Partial Hepatectomy (PH); Remnant Liver Volume (RLV); Original Remnant Liver Volume (ORLV); Regenerative Index (RIx); Resected Liver Parenchymal Volume (RLPV); Total Liver Volume (TLV); Time Intervals for CT Volumetry After Liver Resection: Mean 2 months (t1); 6 months (t2); 10 months (t3); 17 months (t4)
Introduction
According to the annual projections, the World Health Organization estimated that more than 1 million patients would die from hepatocellular carcinoma (HCC) in 2030. The main etiologies of HCC are hepatitis B or C virus infection and alcohol abuse. Partial hepatectomy (PH) is still the first-line treatment for HCC [1-3]. However, the rule governing regeneration of the remnant liver after hepatectomy for these patients remains largely unclear [4]. The future Remnant Liver Volume (future RLV) and the restoration of the Remnant Liver Volume (RLV) by liver regeneration have been accurately measured and monitored by CT volumetry before and after surgery [5-7]. Many researchers reported that the remnant liver regenerates immediately after PH in non-cirrhotic livers. The early Regeneration Index (RIx) on day 7 ranges from 20% to 60%, and the RLV reaches 85% of its initial size at 6 months after right hepatectomy. The most relevant element related to RIx is the size of the original RLV [8- 10]. Few studies have been conducted on RIx and restoration of RLV by regeneration after PH in cirrhotic liver. These studies just revealed that the regenerative capacity of cirrhotic liver is lower and is more complicated than non-cirrhotic livers. Little is known on the impact of fibrotic stages of the liver and resected liver parenchymal volumes on liver regeneration of the remnant liver after hepatectomy. The study aimed to clarify the RIx and the completeness of regeneration of liver volume in the remnant liver in patients with cirrhotic liver after hepatectomy. The patients were divided into subgroups based on the fibrotic stages and percentages of Resected Liver Parenchymal Volume (RLPV). The regeneration of the remnant liver after surgery was assessed by CT volumetry for more than one year. The relationship between liver regeneration and long-term survival outcomes were also analyzed.
Material and Methods
Patient’s background
Consecutive patients who underwent partial hepatectomy for Hepatocellular Carcinoma (HCC) from October 2010 to April 2014 at the Department of Hepatobiliary Surgery, Southwest Hospital were retrospectively studied. The demographic characteristics and operative findings of these patients are summarized in (Table 1). No patients received TACE or chemotherapy, and no portal vein invasion was detected before surgery. The resected specimens were all verified to be HCC on histopathological examination. The study was approved by the Ethics Committee of the Southwest Hospital of the Army Medical University, and it was censored on April 30, 2016.
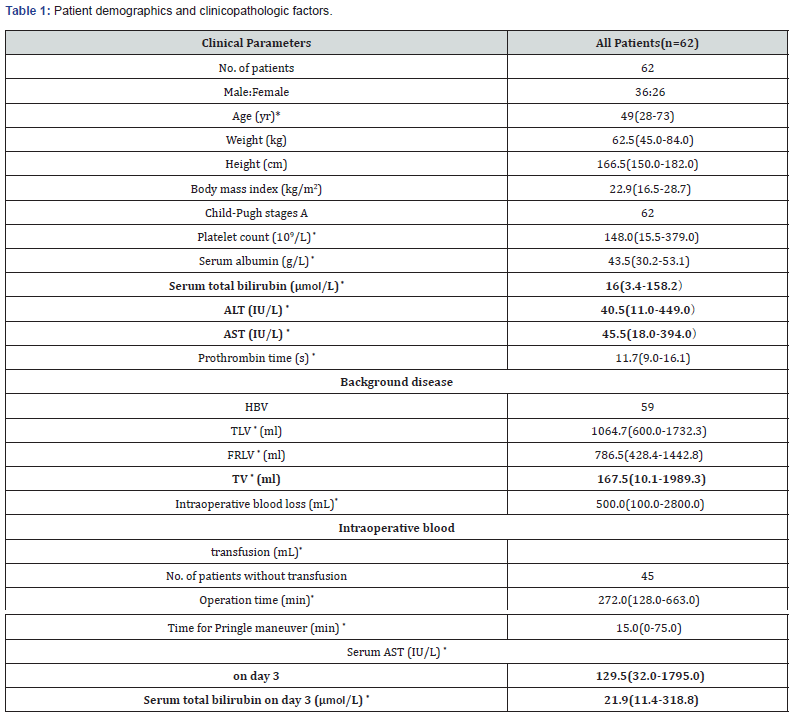
* Value expressed in median (interquartile range) and variance with range in parentheses.
CT technique
All patients underwent a pre-operatively and four postoperative CT volumetry assessments. The mean interval between the preoperative CT volumetry and operation was 7 days. The mean time of the first postoperative CT volumetry was 58.3±39.2 days (range 5 to 132 days), the second CT volumetry 188.9±52.7 days (range 77 to 275 days), the third CT volumetry 318.7±61.8 days (range 203 to 436 days), and the fourth CT volumetry 506.1±139.4 days (range 326 to 894 days). These time points were defined as t1, t2, t3, t4. The CT scanning included the entire upper abdomen, and all acquisitions started from the top of the liver in a craniocaudal direction to cover the whole liver during a single breath holding in maximal inspiration. The pre-contrast and then the hepatic arterial, portal venous, and delayed phases were obtained. Intravenous contrast enhanced imaging was obtained using a nonionic, iodinated contrast material (370 mg I/mL iopromide, Ultravist, Bayer Shering Pharma, Berlin, Germany) at a dose of 1.8 mL/kg of body weight. This contrast was administered with a power injector at a flow rate of 5 mL/second through an 18-gauge intravenous catheter placed in an antecubital vein. The imaging slice thickness was 5 mm.
Types and percentages of liver resection
Anatomical liver resections were based on the Couinaud’s liver segments. Non-anatomical hepatectomy followed the resection planes as planned in a preoperative simulator. The patients were divided into 3 subgroups according to the percentages of the total non-tumorous liver volume resected. The R1 group (n=22), 0-19%. The R2 group (n=32), 20-39%. The R3 group (n=8), ≥40%.
Fibrotic stages of liver specimens
All the surgically resected specimens were studied histopathologically. Using the metavir scoring system to assess the liver fibrosis stage by an experienced pathologist based on predefined criteria [11]. The patients were classified into 4 subgroups: the F1 subgroup with stellate enlargement of portal tracts but without septa formation (n=2); the F2 subgroup with enlargement of portal tracts with rare septa formation (n=9); the F3 subgroup with numerous septa formation (n=22); the F4 subgroup with cirrhosis (n=29). No patients in this study were in the F0 stage of the metavir score.
Measurement of liver volumetry
The liver parenchymal and tumorous contours of each of the histopathological slices were delineated manually. The volumes were calculated by summation of the areas on each axial section. Major vessels (e.g., inferior vena cava, extrahepatic portal vein) and fissures (e.g., fissure for ligamentum teres) were excluded from the volume summation. Volumetric data was calculated manually by the software of the manufacturer workstation (Amira 4.1.2) to include the Total Liver Volume (TLV), Tumor Volume (TV), resected liver parenchymal volume (RLPV) before surgery, and Remnant Liver Volume (RLV) at each CT volumetric study time points. The original remnant liver volume (original RLV) was calculated by [TLV (without TV)-RLPV (without TV)] before surgery. RLV was calculated at each CT volumetric study time points. The remnant liver RIx was calculated as: Remnant liver RIx (%) = [(regenerated RLV at each follow-up time point – original RLV)/original RLV] ×100.
Statistical analyses
All statistical analyses were performed by the SAS9.3 software. Quantitative data with normal distribution were expressed as median (interquartile range). Comparisons between groups of quantitative data were performed with the Kruskal-Walli’s test or the Wilcoxon test. A P value of less than 0.05 was considered as statistically significant. The association between patients’ parameters and regeneration of original RLV was analyzed by the linear regression analysis. The relationship between the liver RIx and tumor recurrence were assessed by the logrank test.
Results
For the 62 patients in this study, the types of liver resection included right hepatectomy (n=4), central hepatectomy (n=3), left hepatectomy (n=6), extended left hepatectomy (n=4), and minor liver resection (including segmentectomy and non-anatomical resection (n=45). There were no patients who developed liver failure. The 90-days postoperative mortality rate was 0%. The median of TLV (volume tumor excluded) was 1064.7 ml, the median TV was 167.5 ml, and median original RLV was 786.5 ml. The patient’s clinicopathologic factors, conditions during operation and operation methods are summarized in (Table 1). The median of RIx at CT volumetric assessments at the postoperative time points t1, t2, t3 and t4 were 25.8(0.5-91.2), 27.0(1.0-107.0), 26.5(2.2-139.5), and 33.9(0.3-127.2) respectively.
Liver volume and RIx according to the metavir system
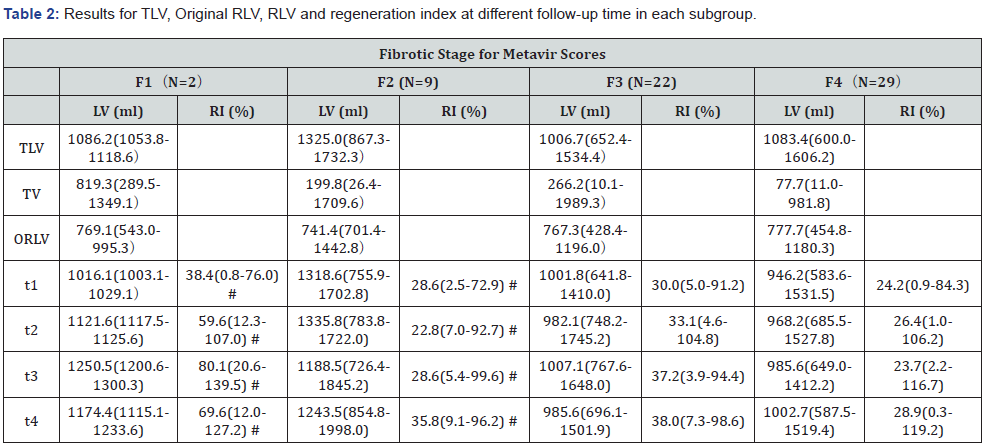
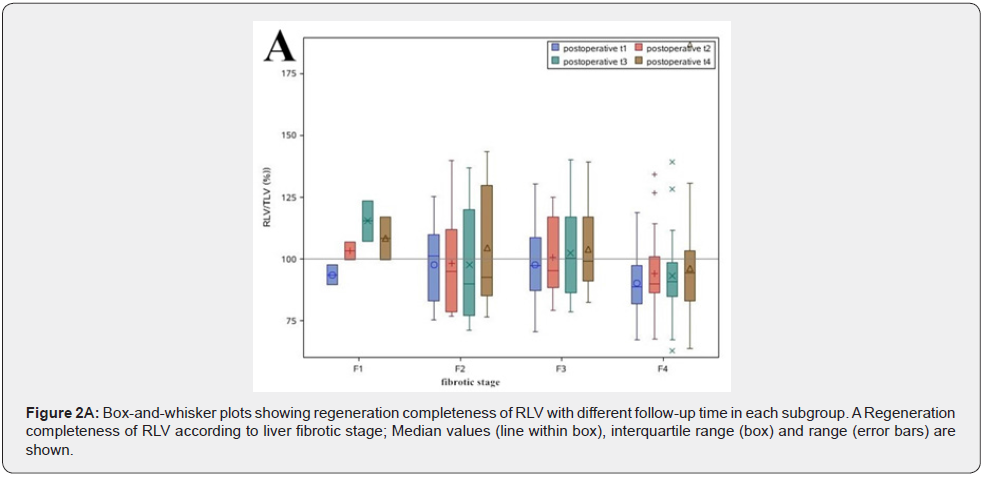
The liver volumes and RIx at different fibrotic stages are summarized in (Table 2). Among the F1 to F4 subgroups, the pre-operative TLV and original RLV showed no significant differences. At t1, the median RLV for the F1 to F4 subgroups increased to 1086.2(1053.8-1118.6) ml, 1325.0(867.3-1732.3) ml, 1006.7(652.4-1534.4) ml and 1083.4(600.0-1606.2) ml respectively. The corresponding RIx reached to 38.4(0.8-76.0), 28.6(2.5-72.9), 30.0(5.0-91.2) and 24.2(0.9-84.3) percent respectively. Then the RIx increased further only in the subgroup F1 from t2 to t4, but it slowed down in the subgroups of F2 to F4. The median RIx of each subgroup increased fastest from liver resection to t1. Using the metavir system, the median RIx were compared among the subgroups at a single follow-up time point. The median RIx was 80.1(20.6-139.5) in the F1 subgroup, and 23.7(2.2-116.7) in the F4 subgroup at t3, although there were no significant differences among the subgroups, but it seemed that there is a downward trend for RIx accompanied by fibrotic stage increased. The mean RIx in relation to the different metavir scores at each follow-up time points are shown as a box plot in (Figure 1A). The restoration of volume in the remnant livers after surgery in each subgroup is shown in (Figure 2A). The median RLV were restored to 93.7±5.7, 97.8±17.2, 97.7±14.1 and 90.2±13.0 percent of the TLV at t1 in the subgroups F1 to F4 respectively, then the increase of median RLV all reached a plateau from t2 to t4.
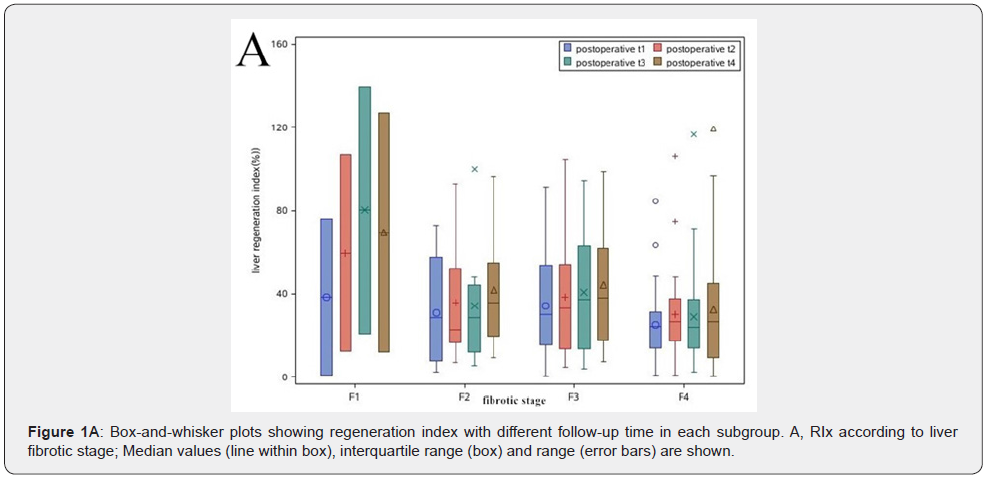
Note: F1 subgroup (n=2), Stellate enlargement of portal tract but without septa formation; F2 subgroup (n=9), Enlargement of portal tract with rare septa formation; F3 subgroup (n=22), Numerous septa formation; F4 subgroup (n=29), Cirrhosis. The data are presented as median (interquartile range).
# Mark located in the right side of RIx in F1 subgroup means no significant difference between F1 and F4 subgroupsat t1 (P=0.936), and t2 (P= 0.629), and at t3 (P=0.376), and t4 (P= 0.469).
# Mark located in the right side of RIx in F2 subgroup means no significant difference between F2 and F4 subgroups at t1 (P= 0.810), and t2 (P=0.877), and t3 (P= 0.668), and t4 (P= 0.295).
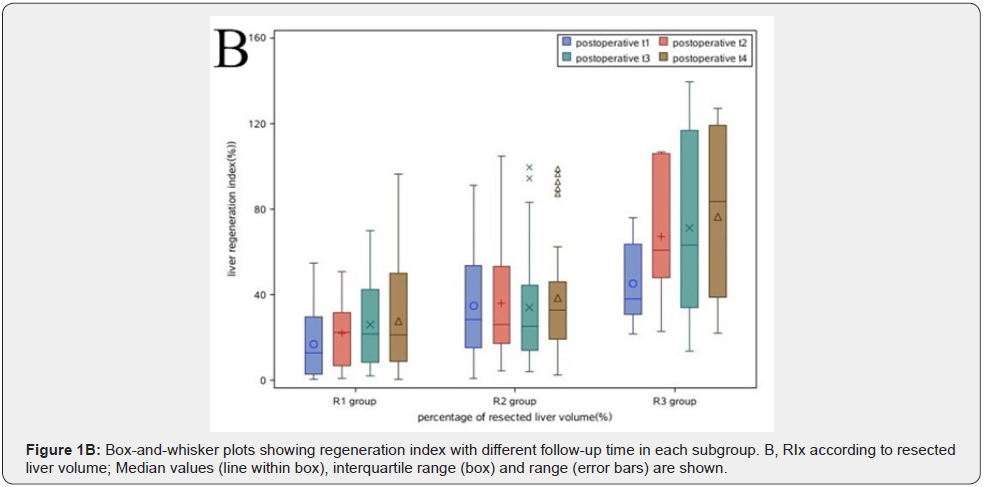
Liver volume and RIx according to percentages of resected liver volume
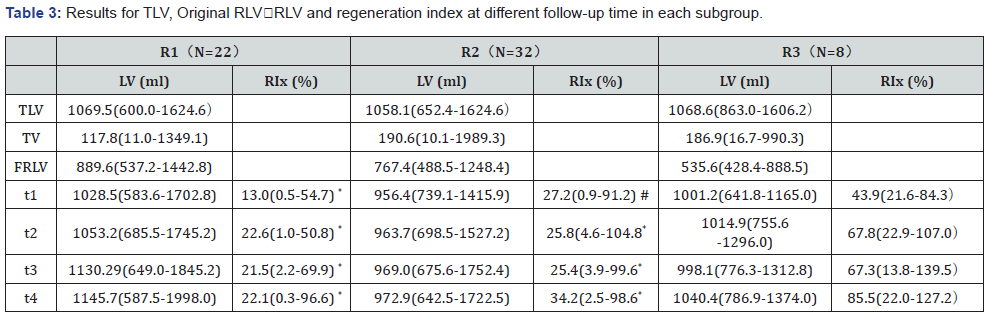
Note: all patients were classified into 3 subgroups according to percentage of removal of total non-tumorous liver volume, R1 group (n=22), 0-19 percent; R2 group (n=332), 20-39 percent (n=7); R3 group(n=8), more than and equal to 40 percent. Comparisons between groups of quantitative data were performed with Kruskal-Walli’s test and Wilcoxon test, a P value of less than .05 was considered significant.
*Mark located in the right side of RIx in R1 subgroup means significant difference between R1 and R3 subgroups at t1 (P= 0.00035), and t2 (P= 0.0006035), and t3 (P= 0.00003), and t4 (P= 0.00032).
*Mark located in the right side of RIx in R2 subgroup means significant difference between R2 and R3 subgroups at t2 (P= 0.01070.006), and t3 (P= 0.00470.010), and t4 (P= 0.0042008).
#Mark located in the right side of RIx in R2 subgroup means no significant difference between R2 and R3 subgroups at t1 (P= 0.2858054).
The patients were classified into three subgroups of R1, R2 and R3 according to the percentages of resected liver volume. There were no significant differences in age or sex distribution among them. The increase in RLV and RIx in relation to the percentages of liver resection are summarized in (Table 3). The mean preoperative TLV were 1069.5(600.0-1624.6) ml, 1058.1(652.4- 1624.6) ml, 1068.6(863.0-1606.2) ml, respectively. The mean TV were 117.8(11.0-1349.1) ml, 190.6(10.1-1989.3) ml, 186.9(16.7- 990.3) ml, respectively with no significant differences among the groups. The original RLV were 889.6(537.2-1442.8) ml, 767.4(488.5-1248.4) ml, and 535.6(428.4-888.5) ml, respectively. For the subgroups R1, R2 and R3 at t1, the RIx reached 13.0(0.5- 54.7), 27.2(0.9-91.2), 43.9(21.6-84.3) percent, respectively. The RIx only increased in R3 from t2 to t4, but it was less in R1 compared to R2. The median RIx of each subgroup all increased fastest within t1. The median RIx were compared among the subgroups at a single follow-up time point (Table 3). Shows that the RIx reached to 43.9(21.6-84.3), 67.8(22.9-107.0), 67.3(13.8- 139.5), 85.5(22.0-127.2) percent in the R3 subgroup from t1 to t4. However, the RIx only reached to 13.0(0.5-54.7), 22.6(1.0-50.8), 21.5(2.2-69.9), 22.1(0.3-96.6) percent in the R1 subgroup from t1 to t4. There were significant differences between the 2 groups (p<0.05). The same results were also found between the R2 and R3 subgroups. Thus, the more the liver volume was resected, the higher was the RIx acquired by the remnant liver from t1 to t4 [12]. The median RIx in relation to the different RLPV at each follow-up time point are shown as a box plot in (Figure 1B). When completeness of regeneration of RLV was studied, the median RLV of the R1 subgroup reached to its initial volume before surgery. However, the median RLV of the R3 subgroup was only restored to 91 percent of its original liver volume at t4. (Figure 2B) shows the median RLV as a percentage of TLV at different follow-up time points.

Multiple regression analysis
The analysis included the following factors which might impact on liver regeneration for the patients: BMI, original RLV, pre-surgical platelet count, prothrombin time (PT), tumor volume, serum total bilirubin, and follow-up time. Partial regression coefficients evidenced with stepwise regression revealed a significant association between the mean RIx and original RLV (p <0.0001).
Liver RIx and patient outcomes
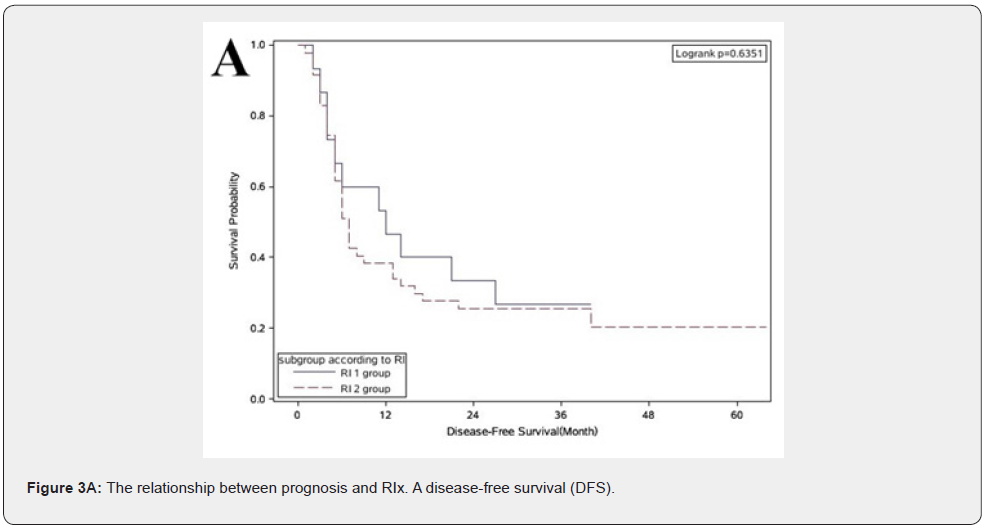
The recurrence rate for the patients was 75.8 percent (47/62), and the death rate was 38.7 percent (24/62). The patients were then divided into two subgroups by the percentages of RIx into the two subgroups: subgroup 1 (<50 percent); and subgroup 2, (≥50 percent). (Figure 3) shows that there was no significant difference between the two groups in median DFS and OS outcomes.

Discussion
Previous research reported that the RIx of non-cirrhotic liver after surgery rapidly reached to 28-64 percent in the 1st month, indicating that the early postoperative period was crucial for liver regeneration, and the RLV increased to nearly its original size at 6 month (13,14). However, the RIx in post-hepatectomy for cirrhotic liver is still unclear. Most patients in our study had a metavir score of F3 (n=22) or F4 (n=29). As our results showed that the mean RIx for all the patients was 25.8(0.5-91.2) percent at t1, then the increase in the RIx became slower from t2 to t4, liver regeneration occurs even in cirrhotic livers in the early postoperative period. In our study the RLV almost reached to its original volume within four months after hepatectomy, the major liver regeneration occurred within t1. The fibrotic stages and the resected liver parenchymal volume (RLPV) had remarkable effects on the completeness of regeneration of the liver remnant and the RIx in cirrhotic patients. These observations can be explained by the defective adaptation to surgical stress, the decreased regenerative capacity of the remnant liver parenchymal cells [15], the decreased portal blood flow to the remnant liver than in healthy people, and the excessive expression of IL-6 and CPR which are harmful to patient’s recovery and liver regeneration [16]. What are the mechanisms for cirrhosis to affect liver regeneration? In our study, the RIx was obviously higher in F1 than in F4 at all the different follow-up time points (Table 2), especially at t3 (80.1 vs 23.7 percent). Although the RIx did not show a significant difference between them, but the median RIx in F4 was obviously lower than RIx in the healthy liver previously reported [8]. Thus, the regeneration capacity was inhibited with increasing fibrotic stages. In addition, the RIx only reached to 69.6(12.0-127.2) percent at t4 in the F1 subgroup, the decrease in the RIx and RLV may be explained by tumor recurrence in a limited number of patients in the F1 subgroup. Kele and his associates reported that the RIx was linearly related to the resected liver parenchymal volume (RLPV) at 6 months after operation in non-cirrhotic livers8. In our study, the RIx was also strongly correlated with the RLPV, showing an increasing tendency at each follow-up time point when the liver remnant was regenerating. Besides, the significant difference among the subgroups suggested that the RIx had a positive correlation with RLPV [17].
A previous study showed that some pre- and post-operative factors had the potential to predict liver regeneration [18]. To detect the factors which might play a role on the RIx in patients with cirrhosis, our multiple linear regression suggested that only BMI and original RLV were significant factors. The follow-up time did not influence RIx. Our results revealed a negative relationship between original RLV and RIx (data not shown), which indicated that the smaller the RLV the more the release of proinflammatory cytokines which promote liver growth. Thus, the rate of TLV is more important to liver regeneration than the type of liver resection. A high tumor recurrence rate exists in patients after partial hepatectomy for HCC. The recurrence and death rates in our study were 73.9% and 52.2%, respectively. Tumor inoculation in animal models showed that tumor grows faster after PH and tumor cells can be stimulated by factors associated with liver regeneration [19,20]. Based on the RIx in our study, all patients were categorized into two subgroups by fifty percentage of RIx. The long-term DFS and OS outcomes showed no significant difference between the two subgroups. Our results indicated that liver regeneration might not directly promote liver tumor recurrence in patients with cirrhosis. There are limitations of this study. First, the number of patients in our study was small. Second, there is a potential of patient selection bias. Third, increase in liver volume by CT volumetry may not entirely reflect the increase in volume were all due to functional regeneration of liver parenchymal cells, but rather the increase could also be due to oedema or congestion of the remnant liver.
Conclusion
Liver regeneration in cirrhotic patients after partial hepatectomy was almost complete within 2 months after partial hepatectomy. The RIx and RLV increased very little after 4 months post-operation. The RIx declined with increasing fibrotic stages. The RIx was positively correlated with the resected liver parenchymal volume (RLPV). The completeness of regeneration of liver remnant volume was impaired with increasing fibrotic stages and RLPV, especially in the F4 and R3 subgroups when the remnant liver volume failed often to reach their original volumes before surgery. The RIx had no direct impact on long-term survival outcomes in our study.
Acknowledgements
Authors’ contributions: Xuesong Li contributed to the conception/design of the research. All authors contributed to acquisition, analysis, or interpretation of data as well as drafting the article. All authors agree to be fully accountable for ensuring the integrity and accuracy of the work and approved the final article.
References
- Court FG, Laws PE, Morrison CP, et al. (2004) Subtotal hepatectomy a porcine model for the study of liver regeneration. J Surg Res 116: 181-186.
- Chen MF, Hwang TL, Hung CF (1991) Human liver regeneration after major hepatectomy A study of liver volume by computed tomography. Ann Surg 213: 227-229.
- Glantzounis GK, Paliouras A, Stylianidi MC, et al. (2018) The role of liver resection in the management of intermediate and advanced stage hepatocellular carcinoma A systematic review. Eur J Surg Oncol 44: 195-208.
- Lin H, Vanden Esschert J, Liu C, Van Gulik TM (2011) Systematic review of hepatocellular adenoma in China and other regions. J Gastroenterol Hepatol 26: 28-35.
- D'Onofrio M, De Robertis R, Demozzi E, et al. (2014) Liver volumetry Is imaging reliable? Personal experience and review of the literature. World J Radiol 6: 62-71.
- Lim MC, Tan CH, Cai J, et al. (2014) CT volumetry of the liver where does it stand in clinical practice. Clin Radiol 69: 887-895.
- Mokry T, Bellemann N, Muller D, et al. (2014) Accuracy of estimation of graft size for living-related liver transplantation first results of a semi-automated interactive software for CT volumetry. PloS One 9: 1-7.
- Kele PG, De Boer M, Vander Jagt EJ, et al. (2012) Early hepatic regeneration index and completeness of regeneration at 6 months after partial hepatectomy. Br J Surg 99: 1113-1119.
- Pagano D, Gruttadauria S (2014) Impact of future remnant liver volume on post-hepatectomy regeneration in non-cirrhotic livers. Front Surg 1: 10.
- Zappa M, Dondero F, Sibert A, et al. (2009) Liver regeneration at day 7 after right hepatectomy global and segmental volumetric analysis by using CT. Radiol 252: 426-432.
- Mohamadnejad M, Tavangar SM, Sotoudeh M, et al. (2010) Histopathological Study of Chronic Hepatitis B A Comparative Study of Ishak and METAVIR Scoring Systems. Int J Organ Transplant Med 1: 171-176.
- Preziosi ME, Monga SP (2017) Update on the Mechanisms of Liver Regeneration. Semin Liver Dis 37: 141-151.
- Klink T, Simon P, Knopp C, et al. (2014) Liver remnant regeneration in donors after living donor liver transplantation long-term follow-up using CT and MR imaging. RoFo 186: 598-605.
- Yamanaka N, Okamoto E, Kawamura E, et al. (1993) Dynamics of normal and injured human liver regeneration after hepatectomy as assessed on the basis of computed tomography and liver function. Hepatology 18: 79-85.
- Ju MK, Choi GH, Park JS, et al. (2012) Difference of regeneration potential between healthy and diseased liver. Transplant Proc 44: 338-340.
- Lan AK, Luk HN, Goto S, et al. (2003) Stress response to hepatectomy in patients with a healthy or a diseased liver. World J Surg 27: 761-764.
- Machado MV, Diehl AM (2014) Liver renewal: detecting mis repair and optimizing regeneration. Mayo Clin Proc 89: 120-130.
- Matot I, Nachmansson N, Duev O, et al. (2017) Impaired liver regeneration after hepatectomy and bleeding is associated with a shift from hepatocyte proliferation to hypertrophy. FASEB J 31: 5283-5295.
- Harun N, Nikfarjam M, Muralidharan V, Christophi C (2007) Liver regeneration stimulates tumor metastases. J Surg Res 138: 284-290.
- Paschos KA, Bird NC (2010) Liver regeneration and its impact on post-hepatectomy metastatic tumour recurrence. Anticancer Res 30: 2161-2170.






























