The Changes of Hepatic Transporters of Gadolinium-Ethoxybenzyl-Diethylenetriamine Pentaacetic Acid on Signal Intensity of MRI during Hepatocarcinogenesis in Rats
Lin Wu1**, Piyun Zhang2**, Yongguo Liu1, Hongzhi Zhao1 and Feng Xia3*
1Department of Hepatobiliary, Chongqing University Central Hospital & Chongqing Emergency Medical Center, China
2Department of Gastroenterology, Chongqing University Central Hospital & Chongqing Emergency Medical Center, China
3Institute of Hepatobiliary Surgery, Army Medical University, China
Submission:December 08, 2021; Published:December 13, 2021
*Corresponding author: Feng Xia, Institute of Hepatobiliary Surgery, Southwest Hospital, Army Medical University, Chongqing, China
**Note: **These authors contributed equally to the study
How to cite this article: Lin W, Piyun Z, Yongguo L, Hongzhi Z, Feng X. The Changes of Hepatic Transporters of Gadolinium-Ethoxybenzyl- Diethylenetriamine Pentaacetic Acid on Signal Intensity of MRI during Hepatocarcinogenesis in Rats. Open Access J Surg. 2021; 13(4): 555867 DOI: 10.19080/OAJS.2021.13.555867.
Abstract
Objective: To investigate the further mechanism of enhancement of hepatocellular carcinoma (HCC) on gadolinium-ethoxybenzyl-diethylenetriamine pentaacetic acid (Gd-EOB-DTPA)-enhanced magnetic resonance imaging (MRI).
Methods: Fifty rats were divided into six experiment groups, a control group and three untreated groups. In addition, every four weeks the rats were applied for MRI and analyzed the expressions of oatps and mrps in livers. It was compared for signal intensity on Gd-EOB-DTPA-enhanced MRI with the transporter expressions to clarify the relationship between signal intensity and the consecutive changes of transporters. Pearson correlation analysis was used to assess the correlation between relative enhancement (RE) of intensity and the expression of transporters.
Results: The expressions of oatp1, oatp2 and oatp4 gradually decreased, while 24-Week-T group was significantly lower than 24-Week-NT group (p=0.01, p=0.025, p<0.01). Mrp1 and mrp2 gradually increased, while the 24-Week-T group were significantly higher than 24-Week-NT group (p=0.03, p=0.01). The signal intensity gradually decreased during hepatocarinogenesis (p<0.0001). The RE of 24-Week-T group was even lower than 24-Week-NT group significantly (p=0.006). Analysis showed the RE of intensity was correlated with the expression of oatp1, oatp4, mrp1 and mrp2 (p<0.05, respectively). There was no difference between untreated groups and control group in transporter expressions and RE.
Conclusion: The consequtive changes of oatps and mrps is correlated with the signal intensity of Gd-EOB-DTPA-enhanced MRI which suggested that the signal intensity was determined by the down-regulation of oatp1 and oatp4 and the up-regulation of mrp1 and mrp2 during the progress of DEN-induced hepatocarcinogenesis.
Keywords: Gd-EOB-DTPA, Hepatic transporter, MRI, Hepatocarcinogenesis.
Introduction
Hepatocellular carcinoma is the fifth common cancer and the third cause of cancer death worldwide [1,2]. The majority of HCC patients are derived from the hepatitis B or C viruses-related cirrhosis [3,4]. Therefore, conventional imaging including ultrasound, CT and MRI for the early screening of HCC is essential. Compared with other conventional imaging, previous studies showed Gd-EOB-DTPA-enhanced MRI had high diagnostic sensitivity and specificity for early detection of HCC, especially for those tumor nodules less than 20mm [5-12]. Gd-EOB-DTPA is a specific MRI contrast agent, which is used for the diagnosis of liver diseases, especially the HCC [13-20]. It is a newly developed contrast agent derived from Gd-DTPA, which combines properties of non-specific extracellular and hepatobiliary-specific contrast agent [21]. Gd-EOB-DTPA is specially taken up by the hepatocytes and excreted up to 50% by hepatobiliary pathway which is mediated by two different transport systems located at the sinusoidal (organic anion transporting polypeptide, oatp) and canalicular membranes (multidrug resistance protein, mrp) of hepatocyte [22-24].
The relationship between the signal enhancement on Gd-EOB-DTPA-enhanced MRI and uptake and excretion mechanism of Gd-EOB-DTPA with oatps and mrps remains unclear completely. It was reported that Gd-EOB-DTPA entered the hepatocytes though oatp1 and excreted into the bile via mrp2 [16,20,24,25]. However, these studies only focus on the effects of oatp1 and mrp2, and the significance of the other members of oatps and mrps located on hepatocyte remains unclear because no results have been published [26]. In addition, the other hepatic transporters might also be responsible for signal intensity of Gd-EOB-DTPA-enhanced MRI.
In a previous study, Tsuda N et al. reported that the signal enhancement of the liver behaved a low signal intensity on Gd- EOB-DTPA-enhanced MRI, which was correlated with the upregulation of mrp2 in the thioacetamide-induced liver cirrhosis in rat [24]. They also reported that the signal intensity on Gd- EOB-DTPA-enhanced MRI was correlated with the expression of oatp1 and mrp2 in the hepatocellular nodules during hepatocarcinogenesis in rat [25]. However, the expressions of other oatp and mrp family in liver during hepatocarcinogenesis remains unclear. As for our study, we had a more consecutive and overall analysis of the relationship between hepatic transporters and signal intensity of Gd-EOB-DTPA during hepatocarcinogenesis at different time points.
Since there is, no results reported about the consecutive changes of hepatic transporters during hepatocarcinogenesis correlated with Gd-EOB-DTPA, except oatp1 and mrp2. Therefore, the purpose of the present study was to clarify the impact of consecutive changes of overall hepatic transporter expressions of Gd-EOB-DTPA on signal intensity of MRI during the progress of diethylnitrosamine (DEN)-induced hepatocarcinogenesis.
Materials and Methods
Animal model
A total of fifty male Sprague-Dawley rats aged from 7 to 8 weeks, weighing about 180g-200g (The Animal Centre of the Third Military Medical University, Chongqing, China) were divided into six experiment groups, a control group and three untreated groups (5 rats/group). The time points separated six experiment groups as follows: 4-Week, 8-Week, 12-Week, 16-Week, 20-Week, 24-Week. All the experiment group of rats received weekly dose of DEN (Sigma–Aldrich, St. Louis, MO, USA, purity=0.95g/mL) in drinking water (0.01% v/v) corresponding to the estimated water consumption of 6 days. Once finishing the DEN water, they were given normal water the rest of the week, and DEN water was prepared freshly every week. The control group of rats was given normal water. Because this is a long term study and age is also one of the factors that probably modulates several parameters. Three untreated groups (8-Week-Untreated, 16-Week-Untreated, 24-Week-Untreated) of rats were fed normal diet to investigate whether the factor of age affect any parameters. At the setting time point, every four weeks an experiment group and every eight weeks an untreated group of rats were firstly applied for MRI, and sacrificed for global observation, histological analysis and analysis of gene expression of transporters later.
During the entire experiment, the rats had free access to drinking water and basal diet, which were maintained on a 12-h artificial light-dark cycle at 22±2℃ and 55±5% humidity in a specific pathogen free room with a filtered air supply. The Animal Care Ethics Committee of the Third Military Medical University approved all experimental procedures involving animals.
Preparation of liver tissues
Each group of rats was sacrificed with a lethal dose of pentobarbital sodium. Tissue samples of liver were cut into 15 blocks and then 10 blocks were rapidly removed into PCR tubes, snap frozen in liquid nitrogen and stored at 80℃ until RNA extraction. The other 5 blocks were fixed in 10% formalin, and then paraffin-embedded sections and eosin-stained preparations were microscopically observed to check the presence of HCC. The tissue of each rat in the 24-Week group were microscopically divided into two sub-groups (neoplastic tissue and paraneoplastic tissue) as follows: 24-Week-tumor (24-Week-T) group and 24-Week-non-tumor (24-Week-NT) group.
RNA extraction and quantitative reverse transcription PCR
Total RNA was extracted from frozen tissue of liver using TRIzol (Invitrogen, Carlsbad, CA) according to the manufacturer’s protocol. Extracted mRNA was reverse-transcribed, and cDNA was synthesized using a PrimeScript RT regent kit (Perfect Real Time, Takara Bio Inc.). In addition, glyceraldehyde-3- phosphate dehydrogenase (GAPDH) was used as an internal reference. Specific primers (Invitrogen, Cergy Pontoise, France) were designed to amplify the cDNA of different transporters according to their sequences available in GenBank as presented in supporting Table S1. After reverse transcription, mRNA expressions levels of oatp1, oatp2, oatp3, oatp4, mrp1,mrp2 and mrp3 were quantitatively examined by using the CFX96 real-time PCR (Bio-Rad Laboratories, Hercules, CA) and a kit (SYBR Premix Ex Taq II Kit, Perfect Real Time; Takara Bio).
Magnetic resonance imaging
Each group of rats were subjected to Gd-EOB-DTPA-enhanced MRI at the setting time points using a 3.0T clinical imager (TrioTim; Siemens Medical Solutions, Erlangen, Germany). The rats were firstly received intraperitoneal injection of 40 mg/ kg pentobarbital sodium for anesthesia. Then Gd-EOB-DTPA (Primovist, Bayer AG, Berlin, Germany) was tail vein-injected into rats at 0.025mmol Gd/kg. MRI was obtained with the sequence of T1_TSE_RR_TRA and T2_TSE_TRA_448_P2. T1 imaging parameters were as follows: repetition time (TR)/echo time (TE) = 550/9msec, a flip angle of 90°, 448 ×240 matrix, one excitation, 156×157mm field of view, 25slices of 2 mm in thickness. T2 imaging parameters were as follows: repetition time (TR)/echo time (TE) = 6000/92msec, a flip angle of 120°, 512×408 matrix, one excitation, 145×230mm field of view, 25 slices of 2mm in thickness. Gd-EOB-DTPA-enhanced MRI was taken before and 3 min after injection refer to the previous study [27].
Image analysis
The signal intensities (SI) of rats in each group were measured for each MR image, and relative enhancement of intensity was calculated with the following equation: RE (%)= ([SIpost- SIpre]/ SIpre)×100, where SIpre and SIpost are signal intensities of the liver before and 3 min after Gd-EOB-DTPA injection.
Statistical analysis
The difference in the gene expression levels of oatp1, oatp2, oatp3, oatp4 and mrp1, mrp2, mrp3 in each group were evaluated with one-way analysis of variance (ANOVA) and the Tukey posthoc test by the statistical analysis software SPSS 17 for Windows (sample size of each group: n = 5). The gene expression and RE between 24-Week-NT and 24-Week-T group were evaluated with paired t test. The difference in RE among each groups was also evaluated with ANOVA and Tukey post-hoc test. Pearson correlation analysis was used to assess the correlation between relative enhancement of intensity and the expression of transporters. P<0.05 was considered statistically significant.
Results
Detection of HCC in DEN-induced HCC model by MRI

To demonstrate whether MRI can be used to detect HCC in an animal model, firstly we established DEN-induced HCC in rats. We applied MRI at different time points after induction with DEN. Meanwhile, we sacrificed animals at different time points after induction with DEN for global observation of tumor development and HE staining for observation of liver histology. Our data showed that MRI could not detect tumor nodules in livers of animals until 24 week’s induction (Figure 1U). Histological examination showed that apparent pseudo lobules were observed in whole liver of rats at 16 week’s induction (Figure 1L). Severe cirrhosis was observed in whole liver of rats at 20 week’s induction (Figure 1M). Multiple HCC nodules were observed in each rats of 24-Week group (Figure 1G and N). This data indicated that liver cancer could be induced by carcinogen DEN and MRI could be used for detection of HCC.
The decreased signal intensity of Gd-EOB-DTPA-enhanced MRI during induction of HCC by DEN


To investigate the changes of signal intensity after Gd-EOBDTPA- enhanced MRI during hepatocarcinogenesis. The signal intensity in each group of rats were measured quantitatively as RE. These data showed RE gradually decreased during induction with DEN (Figure 2A, p<0.0001). It was also found that RE in 24-Week-T group was even more lower than 24-Week-NT group by paired t test (Figure 2B, p=0.006). Typical T1, T2 and T1- Enhanced images of Control group and 24-Week group before and 3 min after Gd-EOB-DTPA injection are shown in Figure 3. The signal intensity of 24-Week group significantly decreased in comparison with the Control group after Gd-EOB-DTPA injection, especially the HCC nodule (Figure 3F, arrow). The HCC nodule showed definite hypo intensity 3 min after Gd-EOB-DTPA injection in hepatobiliary phase. These data indicated that the signal intensity of Gd-EOB-DTPA-enhanced MRI gradually decreased during hepatocarcinogenesis, especially the 24-Week-T group.
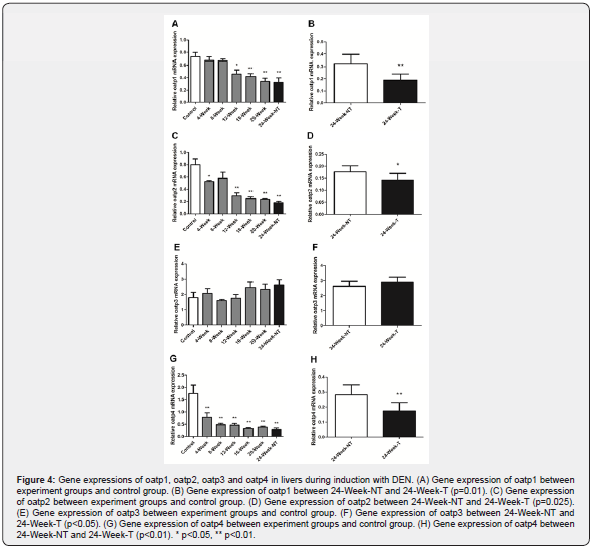
The altered expression of oatp family during induction of HCC by DEN
In order to know whether expression of oatp family members was changed during DEN induction, we obtained liver samples from normal animals and animals at different time points of DEN induction and detected gene expression by quantitative RT-PCR. Our data showed that expression levels of oatp1, oatp2 and oatp4 was gradually decreased during induction with DEN (Figure 4A, C & G). However, expression level of oatp3 was not changed significantly (Figure 4E). Particularly, expression levels of oatp1, oatp2 and oatp4 was further decreased in HCC, as compared with surrounding liver tissues (Figure 4B, D & H). This data indicated that oatp1, oatp2 and oatp4 were down regulated in HCC.
The altered expression of mrp family during induction of HCC by DEN
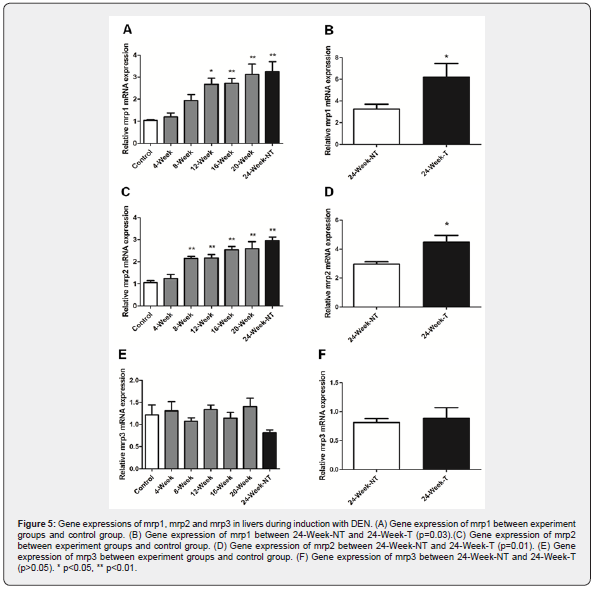
The gene expression of mrp family members in liver samples were also detected by quantitative RT-PCR. Our results showed that expression levels of mrp1 and mrp2 were gradually increased during induction with DEN (Figure 5A & 5C). Nevertheless, expression level of mrp3 was not changed significantly (Figure 5E). Particularly, expression levels of mrp1 and mrp2 were further increased in HCC, as compared with surrounding liver tissues (Figure 5B & D). This data indicated that mrp1 and mrp2 were up regulated in HCC.
Relationship between altered expression of oatp and mrp families with RE
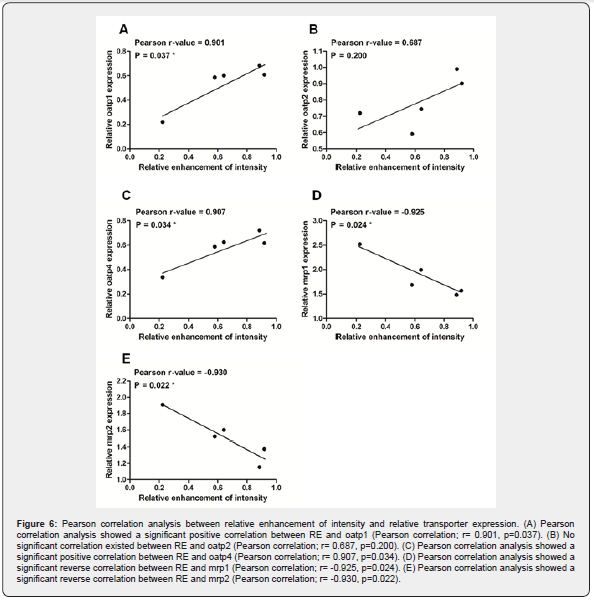
In order to know which oatp and mrp are responsible for enhanced signal intensity of MRI, we made Pearson correlation analysis to assess the correlation between relative enhancement of intensity and the altered expression of transporters. Our results showed that the relative enhancement of intensity was positively correlated with the expression of oatp1 and oatp4 (Figure 6A & C, p<0.05, respectively). In addition, the relative enhancement of intensity was reversely correlated with the expression of mrp1 and mrp2 (Figure 6D and E, p<0.05 respectively). However, although expression of oatp2 was down regulated during the hepatocarcinogenesis, the Pearson correlation analysis suggested there was no significant correlation between RE and oatp2 (Figure 6B). This data indicated that oatp1, oatp4, mrp1 and mrp2 might have effects on signal intensity of Gd-EOB-DTPA-enhanced MRI.
Effect of age in transporter expressions on signal intensity of Gd-EOB-DTPA
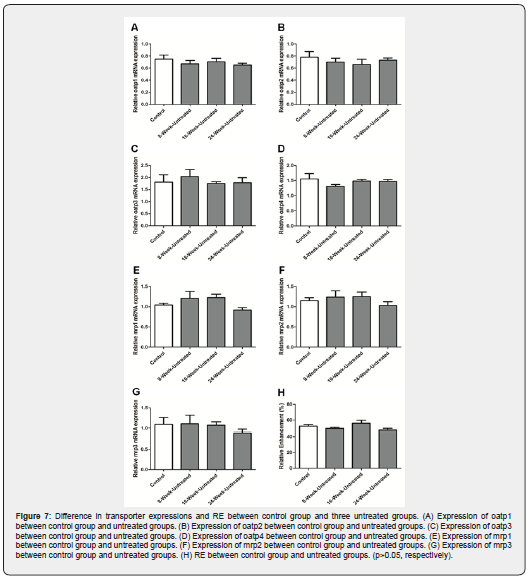
Because of the long-term study, age should be considered as one of the influence factors. Three untreated groups (8-Week- Untreated, 16-Week-Untreated, 24-Week-Untreated) at different time points were compared with control group to investigate the influence of age on parameters. The data suggested there was no difference between untreated groups and control group in transporter expressions and signal intensity of Gd-EOB-DTPA (Figure 7, p<0.05, respectively). These results indicated that age did not affect these parameters significantly.
Discussion
Several preclinical models are used including DEN in rodents to elucidate the molecular and cellular mechanism of HCC development. The carcinogen DEN-induced rat is a wellestablished model of liver carcinogenesis, in which multistep of HCC was chemically induced with chronic inflammation; fibrosis, cirrhosis and HCC that resembles the HBV or HCV infected progress of human HCC [28-32]. Therefore, how the hepatic transporters of Gd-EOB-DTPA changed in the process could help us to have a better understanding of the mechanism of Gd-EOBDTPA, since little is known about the consecutive changes of hepatic transporters in the DEN-induced hepatocarcinogenesis.
Using a rat model of DEN-induced primary HCC, we examined the consecutive changes of transporters (oatps and mrps) located on hepatocytes during hepatocarcinogenesis. We have observed that the expression of oatp1, oatp2 and oatp4 gradually decreased while the expression of mrp1 and mrp2 gradually increased conversely during hepatocarcinogenesis. We had also measured signal intensity of each group in the process; the results indicated that the signal intensity of Gd-EOB-DTPA-enhanced MRI gradually decreased during induction of HCC. Then the Pearson correlation analysis was used to assess the correlation between relative enhancement of intensity and the expression of transporters. The results suggested that the relative enhancement of intensity was correlated with the decreased expression of oatp1 and oatp4 and the increased expression of mrp1 and mrp2. Thus, it could be elucidate the uptake and excretion mechanism of Gd-EOB-DTPA by these transporters. To analyze the role of age during the longterm study, we compared of transporter expressions and signal intensity of Gd-EOB-DTPA between untreated groups and control group. It was concluded that age did not affect these parameters significantly.
Besides, we also investigated the expressions of transporters and relative enhancement between 24-Week-T and 24-Week-NT, which were coincidence with the consecutive changes during hepatocarcinogenesis. It was also found that the expression of oatp1 and oatp4 was decreased in the 24-Week-T group than the 24-Week-NT group significantly, while the expression of mrp1 and mrp2 was increased in the 24-Week-T group than the 24-Week- NT group. Accordingly, this indicated that oatp1 and oatp4 were a decreased expression and the mrp1 and mrp2 were an increased expression during hepatocarcinogenesis especially between the 24-Week-T group and 24-Week-NT group. In addition, the relative enhancement of 24-Week-T group was even lower than the 24-Week-NT group, which were correlated with other studies [33- 35]. The changes of signal intensity in accordance with the gene expression of transporters, which might be, explain the uptake and secretion mechanism of Gd-EOB-DTPA that taken up by oatp1 and oatp4 and secreted by mrp1 and mrp2.
Previous studies showed that transporters of oatp1 and mrp2 played a key role in the uptake and excretion of Gd-EOBDTPA [24,25,36,37], which was coincidence with our results. However, there was no study reported about the expressions of other oatp and mrp family in liver during hepatocarcinogenesis. In our study, we also investigated the consecutive expressions of oatp2, oatp3, oatp4, mrp1 and mrp3 located on hepatocyte during hepatocarcinogenesis. We have found more novel results that oatp4 and mrp1 may also played an essential role in the transportation of Gd-EOB-DTPA. In addition, not only oatp1 and mrp2 but also oatp4 and mrp1 were correlated with the uptake and excretion of Gd-EOB-DTPA. According to these results, we speculated these proteins might have a synergistic effect in the process.
Based on our studies, we suggest that two directions (Figure 8) cause the possible uptake and excretion mechanism of Gd- EOB-DTPA: decreased expression of oatp1 and oatp4 which responsible for the uptake of Gd-EOB-DTPA; on the other hand, increased expression of mrp1 and mrp2 which responsible for the secretion of Gd-EOB-DTPA. Less expression of oatp1 and oatp4 and more expression of mrp1 and mrp2 on tumor cell result in less distribution of Gd-EOB-DTPA in cytoplasm. Accordingly, the consecutive changes of these transporters on hepatocyte could elucidate the mechanism of HCC showed hypo intensity in the hepatobiliary phase in Gd-EOB-DTPA-enhanced MRI.
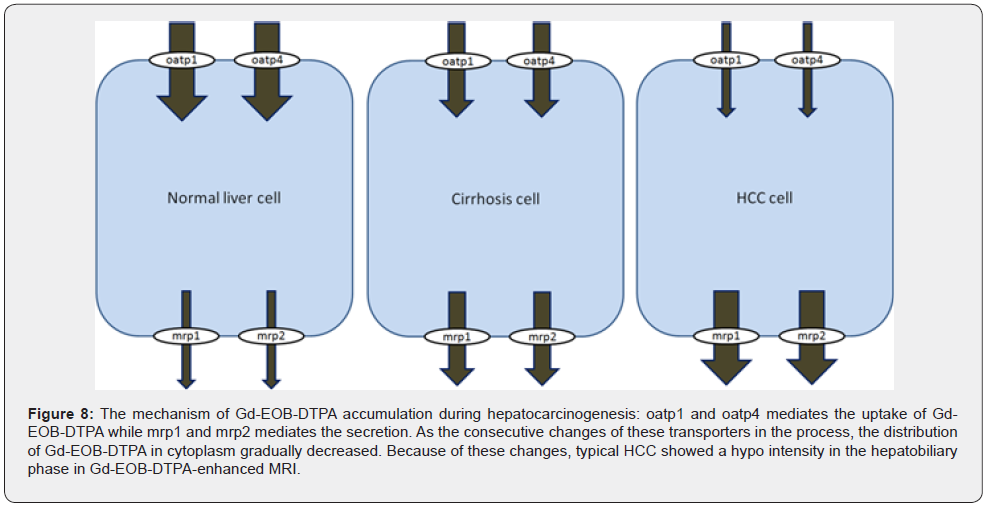
Conclusion
In conclusion, the consecutive changes of oatps and mrps are correlated with the signal intensity of Gd-EOB-DTPA-enhanced MRI, which suggested that the signal intensity was determined by the down-regulation of oatp1 and oatp4 and the up-regulation of mrp1 and mrp2 during the progress of DEN-induced hepatocarcinogenesis.
Acknowledgement
This work was supported by funds from National Natural Sciences Foundation of China (No. 81773140).
References
- Jemal A, Bray F, Center MM, Ferlay J, Ward E, et al. (2011) Global cancer statistics. CA Cancer J Clin 61: 69-90.
- Parkin DM, Bray F, Ferlay J, Pisani P (2005) Global cancer statistics, 2002. CA Cancer J Clin 55: 74-108.
- Jemal A, Siegel R, Ward E, Hao Y, Xu J, et al. (2008) Cancer statistics, 2008.CA Cancer J Clin 58: 71-96.
- Donato F, Boffetta P, Puoti M (1998) A meta-analysis of epidemiological studies on the combined effect of hepatitis B and C virus infections in causing hepatocellular carcinoma. Int J Cancer 75: 347-354.
- Park MJ, Kim YK, Lee MW, Lee WJ, Kim YS, et al. (2012) Small hepatocellular carcinomas: improved sensitivity by combining gadoxetic acid enhanced and diffusion-weighted MR imaging patterns. Radiology 264: 761-770.
- Golfieri R, Renzulli M, Lucidi V, Corcioni B, Trevisani F, et al. (2011) Contribution of the hepatobiliary phase of Gd-EOB-DTPA-enhanced MRI to dynamic MRI in the detection of hypovascular small ((greater-than or equal to)2cm) HCC in cirrhosis. Eur Radiol 21: 1233-1242.
- Blondin D, Erhardt A, Crynen K, Sagir A, Scherer A, et al. (2011) Diagnosis ofFocal Liver Lesions in Cirrhotic Patients: Comparison of Contrast-EnhancedUltrasound using Sulphur Hexafluoride (SF6) Microbubbles and MRI UsingGd-EOB-DTPA. Z Gastroenterol 49: 23-29.
- Bashir MR, Gupta RT, Davenport MS, Allen BC, Jaffe TA, et al. (2013) Hepatocellular carcinoma in a North American population: Does hepatobiliaryMR imaging with Gd-EOB-DTPA improve sensitivity and confidence fordiagnosis? J Magn Reson Imaging 37: 398-406.
- Haradome H, Grazioli L, Tinti R, Morone M, Motosugi U, et al. (2011)Additional value of gadoxetic acid-DTPA-enhanced hepatobiliary phase MRimaging in the diagnosis of early-stage hepatocellular carcinoma: comparisonwith dynamic triple-phase multidetector CT imaging. J MagnReson Imaging 34: 69-78.
- Akai H, Kiryu S, Matsuda I, Satou J, Takao H, et al. (2011) Detection of hepatocellular carcinoma by Gd-EOB-DTPA-enhanced liver MRI: comparison with triple phase 64 detector row helical CT. Eur J Radiol 80: 310-315.
- Ooka Y, Kanai F, Okabe S, Ueda T, Shimofusa R, et al. (2013) Gadoxetic acid enhanced MRI compared with CT during angiography in the diagnosis of hepatocellular carcinoma. Magn Reson Imaging 31: 748-754.
- Baek CK, Choi JY, Kim KA, Park MS, Lim JS, et al. (2012) Hepatocellular carcinoma in patients with chronic liver disease: A comparison of gadoxetic acid enhanced MRI and multiphasic MDCT. Clin Radiol 67: 148-156.
- BluemkeDA, Sahani D, Amendola M, Thomas Balzer, Josy Breuer, et al. (2005) Efficacy and safety of MR imaging with liver specific contrast agent: U.S. multicenter phase III study. Radiology 237(1): 89-98.
- Zech CJ, Herrmann KA, Reiser MF, Schoenberg SO (2007) MR imaging in patients with suspected liver metastases: value of liver-specific contrast agent Gd-EOB-DTPA .Magn Reson Med Sci 6(1): 43-52.
- Reimer P, Schneider G, Schima W (2004) Hepatobiliary contrast agents for contrast-enhancedMRI of the liver: properties, clinical development and applications. Eur Radiol 14 (4): 559-578.
- Kato N, Yokawa T, Tamura A, Heshiki A, Ebert W, et al. (2002) Gadolinium-ethoxybenzyl-diethylenetriamine-pentaacetic acid interaction with clinical drugs in rats. Invest. Radiol 37: 680-684.
- Huppertz A, Haraida S, Kraus A, Christoph J Zech, Juergen Scheidler, et al. (2005) Enhancement of focal liver lesions at gadoxetic acid-enhanced MR imaging: correlation with histopathologic findings and spiral CT—initial observations. Radiology 234(2): 468-478.
- Saito K, Kotake F, Ito N, Taizo Ozuki, Ryuji Mikami, et al. (2005) Gd-EOB-DTPA enhanced MRI for hepatocellular carcinoma:quantitative evaluation of tumor enhancement in hepatobiliary phase. Magn Reson Med Sci 4(1): 1-9.
- Libra A, Fernetti C, Lorusso V, Massimo Visigalli, Pier Lucio Anelli, et al. (2006) Molecular determinants in the transport of a bile acid-derived diagnostic agent in tumoral and nontumoral cell lines of human liver. J Pharmacol Exp Ther 319: 809-817.
- Pastor CM, Planchamp C, Pochon S, Vito Lorusso, Xavier Montet et al. (2003) Kinetic of gadobenatedimeglumine in isolated perfused rat liver: MR imaging evaluation. Radiology 229: 119-125.
- VoglTJ, Kümmel S, Hammerstingl R, M Schellenbeck, G Schumacher, et al. (1996) Liver tumors: comparison of MR imaging with Gd-EOB-DTPA and Gd-DTPA . Radiology 200(1): 59-67.
- Reimer P, Schneider G, Schima W (2004) Hepatobiliary contrast agents for contrast enhanced MRI of the liver: properties, clinical development and applications. Eur Radiol 14: 559-578.
- Zizka J, Klzo L, Ferda J, Milan Mrklovský, Josef Bukac (2007) Dynamic and delayed contrast enhancement in upper abdominal MRI studies: comparison of gadoxetic acid and gadobutrol. Eur J Radiol 62: 186-191.
- Tsuda N, Matsui O (2010) Gd-EOB-DTPA-enhanced MRI in rat cirrhotic liver; with reference to transporter activity and morphological changes of bile canaliculi. Radiology 256: 767-73.
- Natsuko T, Kenichi H, Osamu M (2011) Effect of change in transporter expression on gadolinium-ethoxybenzyl-diethylenetriaminepentaacetic acid-enhanced magnetic resonance imaging during hepatocarcinogenesis in rats. J Gastroenterol Hepatol 26(3): 568-576.
- Mikkaichi T, Suzuki T, Tanemoto M, Ito S, Abe T(2004) The organic anion transporter (OATP) family. Drug Metab Pharmacokinet 19(3): 171-179.
- Tsuda N, Kato N, Murayama C, Narazaki M, Yokawa T (2004) Potential for differential diagnosis with gadolinium-ethoxybenzyldiethylenetriaminepentaacetic acid-enhanced magnetic resonance imaging in experimental hepatic tumors. Invest Radiol 39: 80-88.
- Jia XL, Li SY, Dang SS, Yan-An Cheng, Xin Zhang, et al. (2012) Increased expression of chondroitin sulphate proteoglycans in rat hepatocellular carcinoma tissues. World J Gastroenterol 18(30): 3962-3976.
- Zhang HL, Yu LX, Yang W, Liang Tang, Yan Lin, et al. (2012) Profound impact of gut homeostasis on chemically-induced pro-tumorigenic inflammation and hepatocarcinogenesis in rats. J Hepatol 57(4): 803-812.
- Nakazato K, Takada H, Iha M, et al. (2010) Attenuation of N-nitrosodiethylamine-induced liver fibrosis by high-molecular-weight fucoidan derived from Cladosiphonokamuranus. J GastroenterolHepatol 25(10): 1692-1701.
- Jin N, Deng J, Chadashvili T, Takeaki Nagamine (2010) Carbogen gas-challenge BOLD MR imaging in a rat model of diethylnitrosamine-induced liver fibrosis. Radiology 254(1): 129-137.
- Deng J, Jin N, Yin X, Guang-Yu Yang, Zhuoli Zhang, et al. (2010) Quantitative Multiparametric PROPELLER MRI of Diethylnitrosamine-Induced Hepatocarcinogenesis in Wister Rat Model. J Magn Reson Imaging 31(5): 1242-1251.
- Kitao A, Zen Y, Matsui O, Toshifumi Gabata, Satoshi Kobayashi, et al. (2010) Hepatocellular carcinoma: signal intensity at gadoxetic acid-enhanced MR Imaging--correlation with molecular transporters and histopathologic features. Radiology 256(3): 817-826.
- Kitao A, Matsui O, Yoneda N, Kazuto Kozaka, Satoshi Kobayashi, et al. (2012) Hypervascular hepatocellular carcinoma: correlation between biologic features and signal intensity on gadoxetic acid-enhanced MR images.Radiology 265(3): 780-789.
- Kobayashi S, Matsui O, Gabata T, Kazuto Kozaka, Satoshi Kobayashi, et al. (2012) Relationship between signal intensity on hepatobiliary phase of gadolinium ethoxybenzyldiethylenetriaminepentaacetic acid (Gd-EOB-DTPA)-enhanced MR imaging and prognosis of borderline lesions of hepatocellular carcinoma. Eur J Radiol 81(11): 3002-3009.
- Roth M, Obaidat A, Hagenbuch B (2012) OATPs, OATs and OCTs: the organic anion and cation transporters of the SLCO and SLC22A gene superfamilies. Br J Pharmacol 165: 1260-1287.
- Leonhardt M, Keiser M, Oswald S, Kuhn J, Jia J, et al. (2010) Hepatic uptake of the magnetic resonance imaging contrast agent Gd-EOB-DTPA: role of human organic anion transporters. Drug Metab Dispos 38: 1024-1028.






























