Lung Transplant Due to Pulmonary Chronic Graft Versus Host Disease after Bone Marrow Transplant: Single Tertiary Care Center Experience
Imran Yaqoob Nizam1, Fayyaz Ahmed Baig2, Syed Tufail Khizir3, Saeed Akram4 and Rozeena Huma5,6*
1Consultant and Section Head, KSA
2,3,4Assistant Consultant OTC Lung Transplant, KFSHRC, KSA
5Consultant Researcher, Clinical Research Department, KFSHRC, KSA
6Department of Transplant Medicine, King Faisal Specialist Hospital and Research Center, KSA
Submission:September 02, 2021; Published:September 13, 2021
*Corresponding author: Rozeena Huma, Clinical trial Department, MBC 117, King Faisal Specialist Hospital, P.O Box: 3354, Riyadh 11211, KSA.
How to cite this article: Imran Y N, Fayyaz A B, Syed T K, Saeed A, Rozeena H. Lung Transplant Due to Pulmonary Chronic Graft Versus Host Disease after Bone Marrow Transplant: Single Tertiary Care Center Experience. Open Access J Surg. 2021; 13(4): 555866 DOI: 10.19080/OAJS.2021.13.555866.
Abstract
Lung transplant is a rare treatment option for BMT-related pulmonary chronic GVHD (which includes Bronchiolitis obliterans syndrome BOS, Organizing Pneumonia OP, and Interstitial Pneumonitis or Fibrosis). In our study, we reported eight patients who underwent bilateral LTX due to pulmonary cGVHD after BMT from Jan 2012 to Dec 2019 in our center at King Faisal Specialist Hospital and Research Centre, Riyadh, Saudi Arabia. 4 of these patients had BOS and 4 with other types of cGVHD, i.e., three interstitial fibrosis and one organizing pneumonia. The median time from BMT to the diagnosis of pulmonary cGVHD was 1.86 years. All underwent bilateral Lung transplants with a median interval of time 7.3 years from the diagnosis of pulmonary cGVHD. Two of them had acute cellular rejection (A2) in the 1st year Post LTX for which they received pulse dose steroids. Only one of them developed CLAD and died after seven years of LTX. The notable feature of our study was that no one had a relapse of a malignant hematologic condition.
Introduction
Hematopoietic stem cell transplantation (HSCT), which in simple is called Bone marrow transplant, has been used as a lifesaving procedure for many malignant hematologic, immunologic, and inherited metabolic diseases. There are many pulmonary complications of BMT. The non-infectious pulmonary complications are grouped as chronic pulmonary Graft Versus Host Disease or Pulmonary cGVHD (which includes bronchiolitis obliterans syndrome (BOS), organizing pneumonia (OP), and interstitial pneumonitis or fibrosis). BOS makes 14% of the pulmonary cGVHD post-BMT [1-3]. Different studies suggested that for the therapies given to pulmonary cGVHD patients, only less than 20% improve, and 65% of patients with pulmonary cGVHD (mainly BOS) die within three years of this diagnosis [4-6]. In these studies, standard treatments included immunosuppressive agents such as corticosteroids, cyclosporine, azathioprine, and anti-thymocyte globulin. For those patients who did not respond to the initial course of immunosuppressive therapy, the 2-year and 5-year survival rates were 20% and 13%, respectively [7].
Recommended therapy includes high-dose systemic corticosteroids (1mg/kg/day) for a protracted course with expected improvements in 8% to 20% of patients, of which few are likely curable given the poor overall survival [7-9]. There is no standard treatment for pulmonary cGVHD. Lung transplant (LTX) has become a viable option for selected patients suffering from pulmonary cGVHD.
Case Presentation
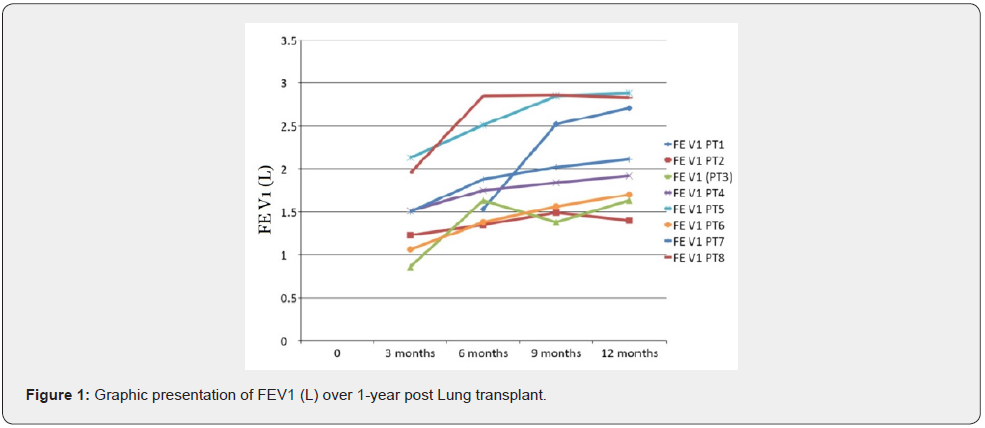
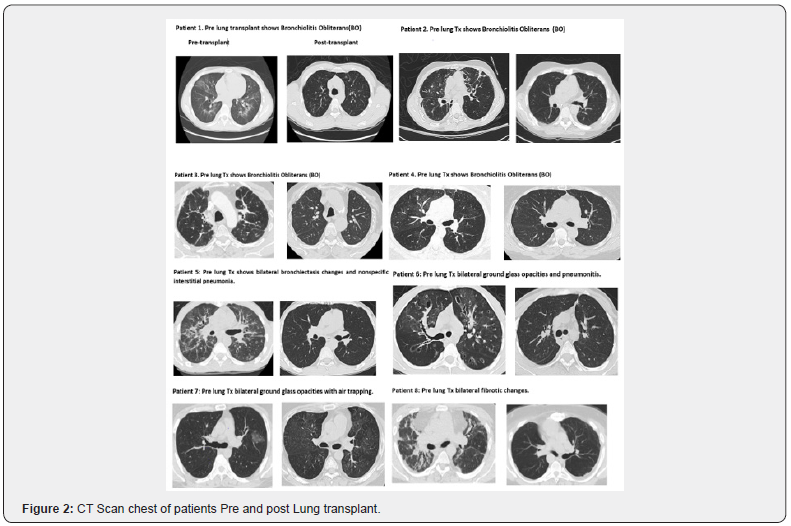
We reviewed patients with Lung transplants who underwent bone marrow transplants between 2012 and 2019. We found eight patients who underwent lung transplants due to pulmonary cGVHD/ BOS, which developed after the BMT. We assessed especially following characteristics, age at LTX, time from BMT to the diagnosis of cGVHD, time from diagnosis of cGVHD to LTX, type of BMT, renal impairment (CrCl less 50%), leukopenia, acute cellular rejection in 1-year post LTX, and chronic allograft Lung dysfunction (CLAD) (Figures 1 & 2).
Patients’ characteristics
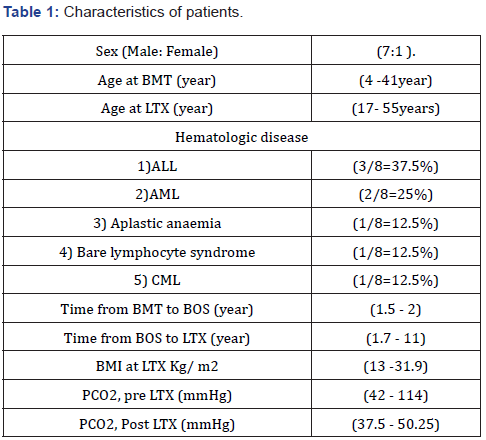
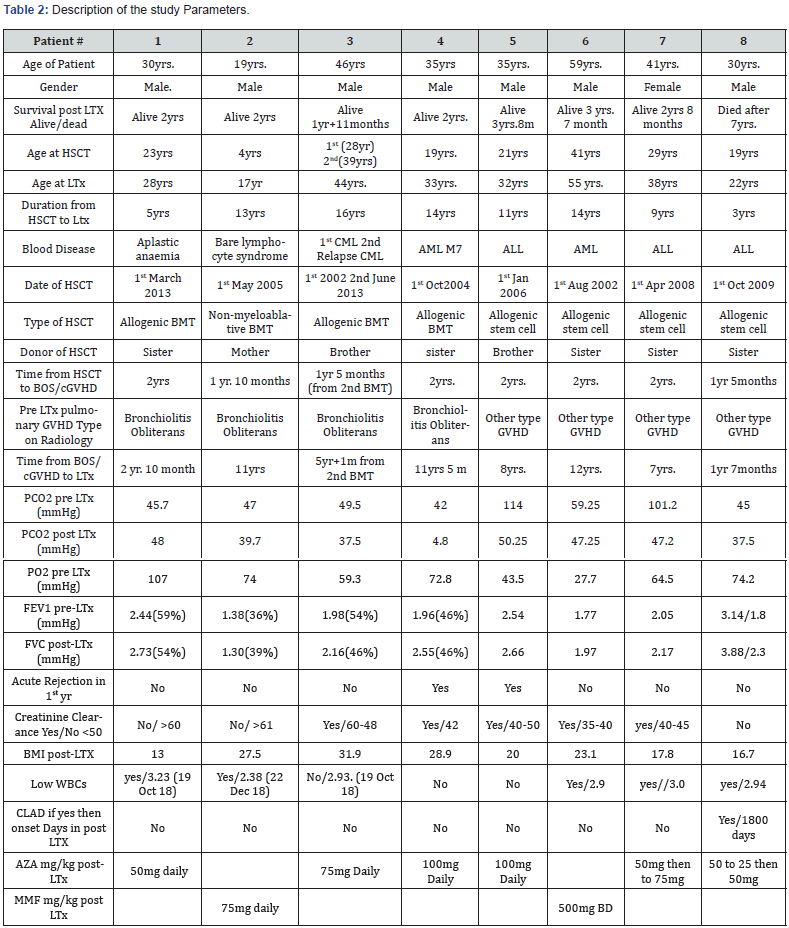
Eight patients (male, n=7, female, n=1) with a median age of 36.87 years underwent allogenic BMT for their hematologic disease. All of them had allogenic BMT from their HLA-matched siblings (brothers or sisters) except one whose donor for BMT was his mother. The median time from BMT to the diagnosis of pulmonary cGVHD was 1.86 yrs. Most of the patients had higher PCO2 (n=6), and all of them had lower SPO2 requiring supplemental oxygen. CT scans showed bronchodilation, mosaic pattern (air trapping), other nonspecific fibrotic changes, and organizing pneumonia. Lung transplantation was carried out at a median time of 7.3 years (ranging from 1 year and 5 months to 12 years) after the diagnosis of pulmonary cGVHD. All of them received bilateral sequential LTX. All of them were with acceptable BMI except one with BMI=13, and they were all size-matched lung transplants. No major thoracic surgical complications were observed. No induction therapy was given in all patients, and the initial immunosuppressive therapy consisted of tacrolimus, MMF, and steroid.
Post lung transplant, only two patients had acute cellular rejection (only once in the 1styear) post LTX, which was treated with a pulse dose of intravenous methylprednisolone for three days and then tapering doses of prednisone orally. Also, one of them had an invasive fungal infection, but none had surgical anastomotic stenosis. Most of them (n=5) had renal impairment (CrCl less than or equal to 50%), which could be related to calcineurin inhibitors such as Tacrolimus or Cyclosporin. Also, most of them (n=6) had episodes of leukopenia which may be associated with Cellcept (MMF). A majority (n=6) were switched to Imuran due to the inability to tolerate MMF on the gastrointestinal system.
Post LTX blood gases showed improvement in PCO2 significantly in six patients compared to pre-LTX PCO2, while the remaining two patients had no significant change because pre- LTX PCO2 was not too high (below 50mmHg). All patients posttransplant became free of oxygen supplementation. Their room air SPO2 was between 96%–98% checked during follow-up in clinics. Pulmonary Function Test performed during follow-ups at 3, 6, 9 and 12 months showed stable lung functions (see Post- LTX FEV1 table over one year). One patient had chronic allograft lung dysfunction (CLAD) and died after seven years of LTX. At the time of writing this manuscript, the remaining seven patients are normal and leading an active life, 24 months to 44 months from their lung transplant surgery. None of them had a relapse of hematological disease or malignancy. From 1 year 5 months minimum to 12 years maximum), and no relapse was observed. This is in accordance with the observations reported previously (Tables 1 & 2).
Glossary: BMT= Blood and Marrow Transplantation; BO= Bronchiolitis obliterans; cGVHD =Chronic Graft Versus Host Disease; PCO2=Concentration of Carbon Dioxide; Po2 =oxygen concentration; FEV1=Forced Expiratory Volume in 1minute; LTx=Bilateral lung transplantation; Meth. Pred =methylprednisolone; MMF=mycophenolate mofetil; Pred=prednisolone (daily dose in mg); Aza=Azathioprine.
Conclusion
We found in our case series of eight patients who underwent a double transplant for underlying pulmonary cGVHD due to BMT, lower incidence of acute cellular rejection, less occurrence of CLAD (chronic Lung allograft dysfunction), and less common fungal infection in comparison with international literature. These could be due to more frequent surveillance bronchoscopy and follow-ups in the clinic. Also, we did not find any recurrence of hematological disease and malignancy.
Declarations
Authors’ contribution: 1,2,3,4 designed & developed the study. All Authors were responsible for content & authenticity. 5 oversaw data collection, data entry. 1,5 carried out a final review of data and analysis. All authors were responsible for direction of the study team, and facilitation of the project plan. Informed consent was obtained from the research.
Ethics approval and consent to participate: The study was approved by the Institutional Review Board of King Faisal Specialist Hospital and Research Center, Kingdom of Saudi Arabia, under approval RAC # 2201155 Dated 22 August 2020.






























