Epidemiological Investigation of Gallbladder Stone Disease in Shanghai China
Qihan Wang1,2, Zhongwen Zhang1, Jian Fei1, Jian Wu1, Qiang Cai1, Qu Cai1, Zhaoyan Jiang ab2*, Tianquan Han1* and Shengdao Zhang1
1Department of Surgery, Shanghai Institute of Digestive Surgery, Ruijin Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai 200025, China.
2Center of Gallbladder Disease, Shanghai East Hospital, School of Medicine, Tongji University, Shanghai 200092, China
Submission:June 23, 2021; Published:June 28, 2021
*Corresponding author: Tianquan Han, Zhaoyan Jiang, Department of Surgery, Shanghai Institute of Digestive Surgery, Ruijin Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai 200025, China.
Center of Gallbladder Disease, Shanghai East Hospital, School of Medicine, Tongji University, Shanghai 200092, China
How to cite this article: Qihan W, Zhongwen Z, Jian F, Jian W, Qiang C, et al. Epidemiological Investigation of Gallbladder Stone Disease in Shanghai China. Open Access J Surg. 2021; 13(2): 555860 DOI: 10.19080/OAJS.2021.13.555860.
Abstract
Background: The gallbladder stone disease increased more than thirty years and became popular medical problem in China.
Objective: To investigate the prevalence of gallbladder stone disease (GSD) in Shanghai China and to analyze the risk factors associated with gallbladder stone disease.
Methods: The cross-section survey was done from August 2010 to July 2011. A total of 15 256 habitants in three districts were enrolled including 8 617 males and 6639 females with the age between 20-79 years old. The relevant clinical data of the subjects were collected by questionnaire, physical examination and biochemical analysis. Gallbladder stone disease was diagnosed by type B ultrasonography. T test and Logistic stepwise regression were used to analyze the risk factors associated with gallbladder stone disease.
Results: There were three aspects of study results. First is the prevalence of gallbladder stone disease 7.02% in this survey increased with age (P<0.05) and higher in females than in males (8.10% vs 6.19%, P<0.05). Second is the rate of cholecystectomy was 2.48% in total almost twice in female than in males (3.42% vs 1.75%, P<0.05). The last is that Logistic regression analysis showed the risk factors for gallbladder stone disease including female, older, presence of fatty liver disease, family history of cholelithiasis, hypertension and increased body mass index.
Conclusions: It was shown in this study the prevalence 7.02% of gallbladder stone disease and cholecystectomy rate 2.48% in Shanghai China combined with the risk factors associated with gallbladder stone disease.
Keywords: Epidemiology; Gallbladder stone disease; Cholecystectomy; Prevalence; Risk factors
Introduction
At present, with the continuous improvement of living standards and the aging of the population in China, the incidence of metabolic-related diseases, such as cardiovascular disease, diabetes, obesity, and metabolic syndrome, is increasing. Gallbladder stone disease (GSD) is related to abnormal cholesterol metabolism, and its prevalence is also on the rise. In the 20th century, two national clinical surveys of gallstones were carried out in China, and the surveys covered intrahepatic and extrahepatic bile duct stones. The first survey in 1989 showed that in general surgery patients, patients with GSD accounted for 10.05% [1]. This proportion rose to 11.53% in the second survey in 1995 [2]. There are great differences in the prevalence of GSD among people in different parts of the world. Similarly, due to differences in regions, environments, eating habits and ethnic groups, the prevalence of GSD varies across China. Currently, there is no data on the prevalence of gallstone disease in the Chinese population.
The occurrence of GSD is related to the combination of genetic susceptibility and environmental factors. Epidemiological studies have summarized the risk factors of gallstone disease as “5F”: female, fatty, forty (years old), fertility and family (history of cholelithiasis) [4]. There is no epidemiological evidence for the risk factors related to GSD in the Chinese population.
This study took regional populations in the community as the research object to investigate the epidemiology of GSD in Shanghai China. The objective was to investigate the prevalence of cholelithiasis and its related risk factors, and to provide evidence for the prevention and treatment of cholelithiasis.
Methods
Sample size and distribution
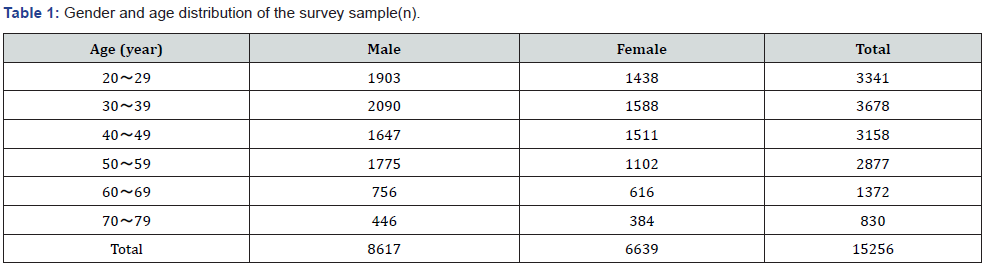
This study is a cross-sectional study. From August 2010 to July 2011, 15256 cases (8617 males and 6639 females) of permanent residents in the communities of Hongkou District, Huangpu District and Minhang District in Shanghai were investigated. All the residents surveyed are in the age range of 20 to 79 years old and have lived in Shanghai for more than 5 years. The gender and age distribution of the samples are shown in Table 1.
Questionnaire survey and physical examination
The investigator used the third national cholelithiasis questionnaire to conduct a questionnaire survey. After uniform training, medical staff adopt uniform standards and procedures to conduct questionnaire surveys and physical examinations of the surveyed population. The contents of the questionnaire include gender, age, surgical history, family history of cholelithiasis, and the others. This manuscript does not show ethnicity, education level, smoking, drinking, medication history, past medical history, etc.
Routine physical examination includes height, weight, waist circumference, and resting blood pressure. Body mass index (BMI) = weight (kg) / height2 (m2). The diagnostic criteria for hypertension are: (1) systolic blood pressure ≥140mmHg; (2) diastolic blood pressure ≥90mmHg; (3) taking hypotensive drugs; (4) normal blood pressure and taking hypotensive drugs. With any of the above, the diagnosis is hypertension.
Diagnosis of gallbladder disease and fatty liver
The result of fasting abdominal B-ultrasound was used for diagnosis. Diagnosis of gallbladder stones: (1) There is a strong echo with sound shadow in the gallbladder cavity. When body position is changed, it can move except for the stones incarcerated in the neck of the gallbladder. (2) When the stone fills the cyst cavity, the gallbladder cavity disappears, and the gallbladder bed is an arc-shaped hyperechoic zone with a clear boundary. (3) When the gallbladder wall is thickened, there is a corresponding weak echo band on the surface and a sound shadow behind it. In the case of muddy stones, the size of the stones is similar, and they are deposited on the back wall of the gallbladder in the form of coarse sand particles, moving with the change of body position, with or without sound shadow behind. Fatty liver diagnosis: (1) The near-field echo of the liver area is diffusely enhanced, and the far-field echo is gradually weakened; (2) The intrahepatic duct structure is unclear.
Biochemical examination
Fasting venous blood is drawn from the anterior cubital vein. Fasting blood glucose, aspartate transaminase, glutamate transaminase, triglyceride and total cholesterol were analyzed by automatic biochemical analyzer.
Data analysis
Microsoft Access program was adopted to compile the questionnaire, input and save the data of the questionnaire. SPSS 17.0 software was used for statistical analysis. The incidence of GSD and cholecystectomy were calculated, respectively. The count data was compared by X2 test. Measurement data are expressed as mean ± standard deviation, and t test is used for comparison between groups. First, t test was performed between the GSD group and the control group, as well as between the cholecystectomy group and the control group, using age, family history of cholelithiasis, BMI, waist circumference, systolic and diastolic blood pressure, fasting blood glucose, total cholesterol and triglyceride, aspartate transaminase and glutamate transaminase. With positive results as independent variables, logistic stepwise regression was performed to analyze the risk factors related to GSD. OR and 95%CI were calculated according to the partial correlation coefficient and its standard deviation. P<0.05 indicates that the difference is statistically significant.
Result
The prevalence of gallstone disease
In the population investigated in this study, the overall prevalence of GSD was 7.02%, and the prevalence of women (8.10%) was higher than that of men (6.19%) (P<0.05). The incidence of GSD increases with age in both males and females. From age 40, the incidence of GSD increased significantly in men and women (Table 2).

Cholecystectomy rate
Overall cholecystectomy rate was 2.48%. Female cholecystectomy rate was about twice that of male (3.42% vs. 1.75%, P< 0.05). Cholecystectomy rates increased with age in both men and women (Table 3).
General clinical features and biochemical indexes
After excluding the samples with incomplete data, this study conducted statistical analysis on the biochemical indexes and other variables of 12,572 samples, including 743 in the GSD group, 258 in the cholecystectomy group, and 11,571 in the control group (Table 4). Age, BMI, waist circumference, systolic blood pressure, diastolic blood pressure, fasting blood glucose, triglyceride, glutamate transaminase, aspartate transaminase and family history of cholelithiasis in GSD group were significantly higher than those in control group (P< 0.05). Compared with the control group, age, BMI, waist circumference, systolic blood pressure, diastolic blood pressure, fasting blood glucose, total cholesterol, triglyceride, glutamate transaminase, aspartate transaminase and family history of cholelithiasis were also significantly increased in the cholecystectomy group (P< 0.05) (Table 4).
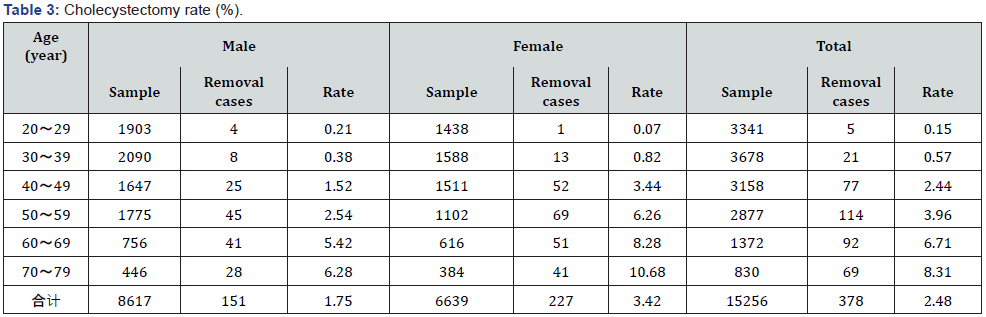
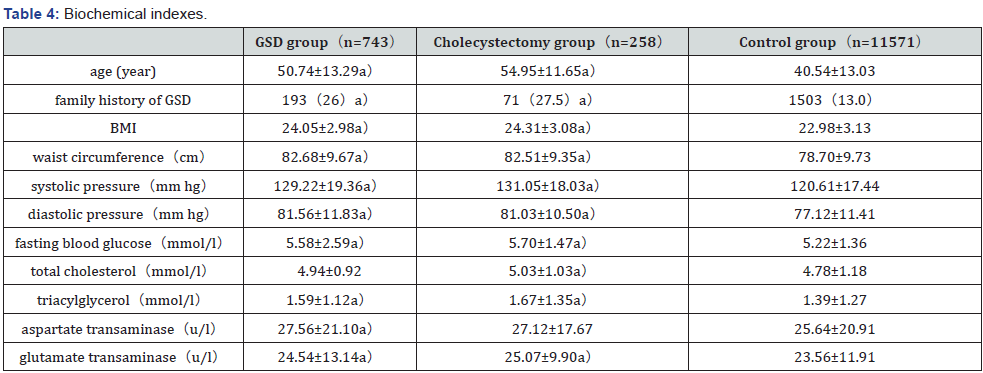
a): Compared with the control group, the difference was statistically significant. P<0.05
Logistic stepwise regression analysis of risk factors
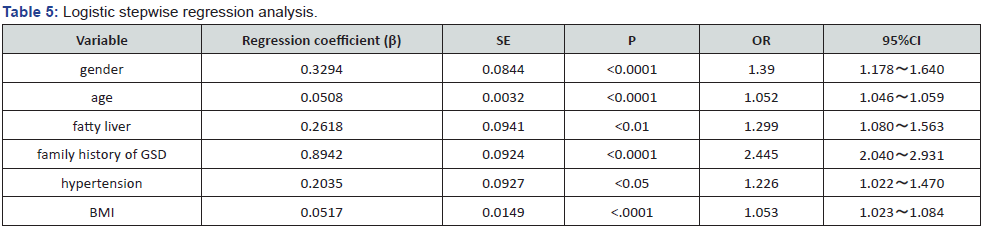
Logistic stepwise regression analysis was used to eliminate 4 variables from the 10 variables of the cholelithiasis group and the control group, and 6 significant variables were obtained. Sex, age, fatty liver disease, family history of cholelithiasis, hypertension and BMI were risk factors for GSD, with OR of 1.390, 1.052, 1.299, 2.445, 1.226, 1.053, respectively (Table 5).
Discussion
In this study, the prevalence of cholelithiasis in the natural population in Shanghai was investigated from 2010 to 2011. A total of 15,256 patients were investigated, ranging in age from 20 to 79 years old. The results showed that the prevalence of cholelithiasis was 7.02%, 6.19% in males and 8.10% in females. The prevalence was significantly higher in females than in males. This study was designed under the guidance of Yutang Gao, Epidemiology Research Group, Shanghai Institute of Cancer, with reference to the age distribution of the population in Shanghai.
GSD, as one of the most common biliary tract diseases in the world, has an incidence of 3.1%-10.7% in Asia and 5.9%-21.9% in Western countries [5]. Two clinical cholelithiasis studies were conducted nationwide in the 20th century, showing that inpatients with GSD accounted for 10.05%~11.53% of general surgery patients [2]. But none of these surveys were based on natural populations. Since then, several regional gallstone disease incidence surveys have been carried out in China. The incidence rate of cholelithiasis in Xinjiang is 13.11% [6], Taipei is 5.3% [7], and Beijing is 4.2% [8]. Our survey shows that the current incidence of GSD is higher than the results of the survey in Shanghai by Ye et al. in 2003 [9], indicating that the current incidence of GSD has an increasing trend. The prevalence of gallstone disease investigated in this study is relatively higher than that in other regions in China, which is mainly related to factors such as the improvement of living standards, the change of dietary habits, and the trend of westernization of living habits in Shanghai.
With the widespread use of laparoscopic cholecystectomy in recent years, patients with cholecystectomy have gradually increased. A German study reported that as age increases, the clinical complications of gallstone disease also increase, resulting in more than 40% of the population undergoing cholecystectomy after the age of 40. Ye et al. [9] conducted a survey on the population in Jiangwan area of Shanghai and showed that cholecystectomy rate was 2.8%, and the cholecystectomy rate showed an increasing trend with the increase of age. In this study, the cholecystectomy rate was 1.75% in males, 3.42% in females, and 2.48% overall. The cholecystectomy rate increased with age in both men and women. Cholecystectomy rate is significantly higher in women than in men, which is associated with a higher incidence of cholelithiasis in women. The surgical indications of cholecystectomy are mostly cholelithiasis. Therefore, the rate of cholecystectomy indirectly reflects the prevalence of gallstone disease. In this study, the prevalence of cholelithiasis was 7.02% and the cholecystectomy rate was 2.48%, indicating that the prevalence of cholelithiasis in Shanghai was close to 9.5%.
This study found that the prevalence of gallstone disease in women is slightly higher than that in men. From the epidemiological data of many countries, the prevalence rate of women is higher than that of men of the same age [10]. This may be related to female-specific physiological factors and the level of estrogen and progesterone. Estrogen can affect the normal activity of the female gallbladder, inhibit gallbladder emptying, and affect the composition of bile. The incidence of gallstone disease in women with oral contraceptives during the childbearing period is higher, and the incidence of gallstone disease in postmenopausal women who use hormone replacement therapy is also increased, further indicating that it is related to elevated estrogen levels [11]. The gender difference in gallstone disease is not obvious after the age of 50, which is related to the decrease of estrogen secretion in women after menopause [12].
Age is a recognized risk factor for cholelithiasis. Our results also confirm that the prevalence of cholelithiasis increases with age in both men and women. Logistic regression analysis showed that age was a risk factor associated with GSD (OR=1.052, 95%CI= 1.046~1.059). Einarsson et al. found that bile cholesterol saturation was positively correlated with age, while cholic acid synthesis and cholic acid pool size were negatively correlated with it, which was conducive to the formation of supersaturated bile and promoted the occurrence of calculi [13]. Secondly, with the increase of age, gallbladder contraction is weakened, which can also promote stone formation [14].In contrast, cholelithiasis rarely occurs in infants and adolescents [15].Many epidemiological studies have shown that the prevalence of cholelithiasis after the age of 40 is many times higher than that in the younger age group, and the dividing line between high and low incidence is 40 years old [16], which is consistent with the results of this survey. The aging of the current urban population may contribute to the increase in prevalence.
Family history of gallstone disease is a risk factor for gallstone disease. The author’s research on 93 Han families in Shanghai shows that gallstone disease has the characteristics of chromosomal dominant inheritance and delayed inheritance [17]. A Swedish study of 43411 pairs of twins confirmed that 25% of the pathogenic factors of gallstone disease are related to genetic factors [18]. This study again confirmed that a family history of cholelithiasis was a high-risk factor for cholelithiasis (OR=2.445, 95%CI= 2.040-2.931). This study again confirmed that a family history of cholelithiasis was a risk factor for cholelithiasis with a high risk (OR=2.445, 95%CI= 2.040-2.931). In this study, DNA samples from the population were not obtained, and the polymorphic loci of the susceptibility genes related to cholelithiasis, such as the ATP binding assette (ABC) G5/G8 gene, could not be determined [19], which requires further study.
Obesity is also a risk factor for gallstone disease, especially among women [20]. Obese people often have cholesterol metabolism disorders, leading to bile cholesterol oversaturation and promoting the formation of stones [21]. Similarly, obese people are often accompanied by hyperinsulinemia, which can promote the synthesis of cholesterol in the liver, leading to oversaturation of cholesterol in the bile, which is easy to precipitate and become stones [3, 22]. In this study, BMI was used as an index to measure the degree of obesity, and it was found that the incidence of cholelithiasis increased 1.053 times for every 1 increase in BMI. This suggests that obesity represented by a high BMI is a risk factor for cholelithiasis.
With the improvement of living standards, the incidence of fatty liver, especially non-alcoholic fatty liver disease (NAFLD), has gradually increased, becoming one of the main causes of chronic liver disease [23]. A meta-analysis based on 12 observational studies showed that gallstone disease is closely related to NAFLD, and NAFLD increases the risk of gallstone disease by 1.5 times [24]. This is consistent with the conclusion that fatty liver increased the incidence of cholelithiasis by 1.299 times in the Logistic analysis of this study. The association between fatty liver and cholelithiasis may stem from common risk factors, such as insulin resistance, obesity, hyperlipidemia, and age. Insulin resistance can lead to the accumulation of free cholesterol in the liver, which in turn leads to NAFLD [25]. On the other hand, insulin resistance can also lead to increased liver cholesterol excretion, causing hypersaturation of bile cholesterol, and at the same time causing gallbladder dyskinesia, promoting the formation of gallstone disease [26]. Obesity can lead to increased liver cholesterol synthesis and promote the formation of fatty liver and gallstones [27].
The relationship between hypertension and gallstone disease is controversial. This study shows that hypertension increases the risk of gallstone disease by 1.226 times. Zamani et al. [28] and Chen et al. [29] have shown that hypertension is a risk factor for gallstone disease. However, some studies have reached the opposite conclusion that there is no significant correlation between gallstone disease and hypertension [30]. These different results may be caused by differences in race, sample size, diagnostic methods for gallstone disease, and interference from other risk factors.
This research shows that the current overall prevalence of gallstone disease in Shanghai is 7.02%, and the cholecystectomy rate is 2.48%, showing an increasing trend. The population over 40 years old has a high incidence of cholelithiasis and should be taken as the main target for prevention and treatment. This study found that gender, age, fatty liver, family history of gallstone disease, hypertension and BMI are risk factors for gallstone disease, suggesting that gallstone disease is closely related to multiple factors. Enhancing health education, exercising moderately, controlling obesity and other metabolic abnormalities, and restricting high-fat diet will help prevent gallstone disease. Regular physical examination after the age of 40, early detection and early intervention are of great significance for preventing the formation and progression of gallstones.
References
- Gu ZY, Huang ZQ (1987) Feature of cholelithiasis in China——National clinical investigation from surgical patients: a study of 11432 gallstone. Chin J Surg 25(6): 321-329.
- Biliary Surgery Group of Surgery Branch, Chinese Medical Association (1995) Changes in Chinese cholelithiasis during ten years. Chin J Surg 33(11): 652-658.
- Portincasa P, Moschetta A, Palasciano G (2006) Cholesterol gallstone disease. Lancet 368(9531): 230-239.
- Nakeeb A, Comuzzie AG, Martin L, Sonnenberg GE, Swartz-Basile D, et al. (2002) Gallstones: genetics versus environment. Ann Surg 235(6): 842-849.
- Chuang SC, Hsi E, Lee KT (2013) Genetics of gallstone disease. Adv Clin Chem 60(2): 143-185.
- Zhu L, Aili A, Zhang C, Saiding A, Abudureyimu K (2014) Prevalence of and risk factors for gallstones in Uighur and Han Chinese. World J Gastroenterol 20(40): 14942-14949.
- Chen JY, Hsu CT, Liu JH, Tung TH (2014) Clinical predictors of incident gallstone disease in a Chinese population in Taipei, Taiwan. BMC Gastroenterol14: 83.
- Xu Q, Tao LY, Wu Q, Gao F, Zhang FL, et al. (2012) Prevalences of and risk factors for biliary stones and gallbladder polyps in a large Chinese population. HPB (Oxford)14(6): 373-381.
- Ye X, Wang BG, Xiang XY, Xiao ZK, Jiang ZY, et al. (2003) Investigation of prevalences for adults with gallstone in Jiangwan district, Shanghai. J Hepatopancreatobiliary Surg 15(1): 28-30.
- Shaffer EA (2006) Gallstone disease: Epidemiology of gallbladder stone disease. Best Pract Res Clin Gastroenterol 20(6): 981-996.
- Cirillo DJ, Wallace RB, Rodabough RJ, Greenland P, LaCroix AZ, et al. (2005) Effect of estrogen therapy on gallbladder disease. JAMA 293(3): 330-339.
- Sanders G, Kingsnorth AN (2007) Gallstones. BMJ 335(7614): 295-299.
- Einarsson K, Nilsell K, Leijd B, Angelin B (1985) Influence of age on secretion of cholesterol and synthesis of bile acids by the liver. New Engl J Med 313(5): 277-282.
- Shaffer EA (2005) Epidemiology and risk factors for gallstone disease: has the paradigm changed in the 21st century?. Curr Gastroenterol Rep 7(2): 132-140.
- Kaechele V, Wabitsch M, Thiere D, Kessler AL, Haenle MM, Mayer H, Kratzer W (2006) Prevalence of gallbladder stone disease in obese children and adolescents: influence of the degree of obesity, sex, and pubertal development. J Pediatr Gastroenterol Nutr 42(1): 66-70.
- Stinton LM, Myers RP, Shaffer EA (2010) Epidemiology of gallstones. Gastroenterol Clin North Am 39(2): 157-169.
- Fei J, Han TQ, Jiang ZY, Jiang ZH, Zhang Y, et al. (2002) Genetics of family with gallbladder stone disease: preliminary study. J Hepatopancreatobiliary Surg 14(1): 4-6.
- Katsika D, Grjibovski A, Lammert F, Lichtenstein P, Marschall HU (2005) Genetic and environmental influences on symptomatic gallstone disease: a Swedish twin study of 43 141 twin pairs. Hepatology 41(5): 1138-1143.
- von Kampen O, Buch S, Nothnagel M, Azocar L, Molina H, Brosch M, Erhart W, von Schönfels W, Egberts J, Seeger M, Arlt A, Balschun T, Franke A, Lerch MM, Mayerle J, Kratzer W, Boehm BO, Huse K, Schniewind B, Tiemann K, Jiang ZY, Han TQ, Mittal B, Srivastava A, Fenger M, Jørgensen T, Schirin-Sokhan R, Tönjes A, Wittenburg H, Stumvoll M, Kalthoff H, Lammert F, Tepel J, Puschel K, Becker T, Schreiber S, Platzer M, Völzke H, Krawczak M, Miquel JF, Schafmayer C, Hampe J (2013) Genetic and functional identification of the likely causative variant for cholesterol gallstone disease at the ABCG5/8 lithogenic locus. Hepatology 57(6): 2407-2417.
- Maclure KM, Hayes KC, Colditz GA, Stampfer MJ, Speizer FE, et al. (1989) Weight, diet, and the risk of symptomatic gallstones in middle-aged women. New Engl J Med 321(9): 563-569.
- Marschall HU, Einarsson C (2007) Gallstone disease. J Intern Med 261(6): 529-542.
- Fornari F, Imberti D, Squillante MM, Squassante L, Civardi G, et al. (1994) Incidence of gallstones in a population of patients with cirrhosis. J Hepatol 20(6): 797-801.
- Younossi ZM, Blissett D, Blissett R, Henry L, Stepanova M, et al. (2016) The economic and clinical burden of non-alcoholic fatty liver disease in the United States and Europe. Hepatology 64(5): 1577-1586.
- Jaruvongvanich V, Sanguankeo A, Upala S (2016) Significant association between gallstone disease and nonalcoholic fatty liver disease: a systematic review and Meta-analysis. Dig Dis Sci 61(8): 2389-2396.
- Utzschneider KM, Kahn SE (2006) Review: The role of insulin resistance in nonalcoholic fatty liver disease. J Clin Endocr Metab 91(12): 4753-4761.
- Tsai CJ, Leitzmann MF, Willett WC, Giovannucci EL (2008) Macronutrients and insulin resistance in cholesterol gallstone disease. Am J Gastroenterol 103(11): 2932-2939.
- Lambou-Gianoukos S, Heller SJ (2008) Lithogenesis and bile metabolism. Surg Clin North Am 88(6): 1175-1194.
- Zamani F, Sohrabi M, Alipour A, Motamed N, Saeedian FS, et al. (2014) Prevalence and risk factors of cholelithiasis in Amol city, northern Iran: a population based study. Arch Iran Med17(11): 750-754.
- Chen LY, Qiao QH, Zhang SC, Chen YH, Chao GQ, et al. (2012) Metabolic syndrome and gallstone disease. World J Gastroenterol 18(31): 4215-4220.
- Froutan Y, Alizadeh A, Mansour-Ghanaei F, Joukar F, Froutan H, et al. (2015) Gallstone disease founded by ultrasonography in functional dyspepsia: prevalence and associated factors. Int J Clin Exp Med 8(7): 11283-11288.






























