Dose Escalation Using Cyberknife Boost in Locally Advanced High Risk Prostatic Carcinoma –A Comparative Study with Historic Cohort Receiving Conventional Fractionated Dose Escalated External Beam Radiotherapy
Dr Krithikaa Sekar, Bindiya Vijayan, Ancy Mathew, Priya sinha, Fareena Taj CK, Radheyshyam Naik, Ajai Kumar BS, Srinivas BJ, Ramesh S Bilimagga, Shekar Patil, Sateesh Chirondoni Thungappa, Veena Ramaswamy, Shivakumar swamy, Sudhakar Sampangi, Kumar Kallur,Ragunath Krishnappa, Somrat Bhattacharjee, Amritanshu ram and G.Lohith*
Consultant Radiation Oncologist, HCG Hospitals-Bangalore, India
Submission:June 03, 2021; Published:June 15, 2021
*Corresponding author: Dr Lohith G, Consultant Radiation Oncologist, HCG Hospitals-Bangalore, India
How to cite this article: Dr Krithikaa S, Bindiya V, Ancy M, Priya s, Fareena Taj C, et al. Dose Escalation Using Cyberknife Boost in Locally Advanced High Risk Prostatic Carcinoma –A Comparative Study with Historic Cohort Receiving Conventional Fractionated Dose Escalated External Beam Radiotherapy. Open Access J Surg. 2021; 13(2): 555858 DOI: 10.19080/OAJS.2021.13.555858.
Introduction
Several randomized trials have compared the outcomes of dose escalated external beam radiation to external radiation followed by a brachytherapy boost in locally advanced prostatic carcinomas and has shown the superiority of the later [1]. However, the biologically equivalent dose (BED) delivered in such schedules must be meticulously assessed, so as the radiobiological difference between the two schedules. Wherein conventionally fractionated external beam radiotherapy provides dose escalation without the radiobiological advantage as that of a naturally higher dose per fraction brachytherapy [2]. SBRT with cyberknife used as a boost circumvents this disadvantage and makes safe dose escalation using external radiation feasible [3]. The role of radio surgical ablation in low-risk prostatic carcinomas is well established [4]. Our study has analyzed the local control rate and toxicity profile of 31 patients with high-risk prostatic adenocarcinoma who have received neoadjuvant hormone therapy followed by external beam radiation using tomotherapy and a cyberknife boost.
Aim
To analyze the biochemical outcomes and toxicity profile of patients receiving cyberknife boost in locally advanced high risk prostatic carcinomas.
Objective
To analyze the effect of cyberknife boost on local control and normal tissue toxicity in localized high risk prostate carcinomas and compare that with the outcomes of historical treatment cohort that received dose escalated external radiation therapy.
Material and Method
We retrospectively identified from our hospital registry,31 patients with histologically proven high risk prostatic adenocarcinoma, treated with neoadjuvant hormonal therapy for 3 months followed by external beam radiation using tomotherapy technique to a total dose of 45-50Gy and 2-3 fractions of cyberknife boost, post fiducial marker placement. Patients continued hormonal therapy for 2 years post treatment.
Inclusion Criteria:
i. Histologically proven adenocarcinoma
ii. Gleason grade >/= 7
iii. Clinical Stage T2b or higher
iv. Baseline PSA >/= 20
v. ECOG Performance status 0-2
Exclusion Criteria:
i. Patients having received previous pelvic radiotherapy.
ii. Patients with distant metastatic disease.
iii. Patients with two or more primary malignancies.
IGRT Treatment Delivery
All patients underwent PSMA PET CT simulation before IGRT planning (Figure 1). Both rectal and bladder protocol as per our institutional policy was followed before simulation and same was followed before each IGRT fraction. GTV, CTV and PTV was contoured according to RTOG guidelines. 95% of isodose coverage was accepted for the contoured volume.

SBRT Treatment Planning & Delivery
All patients had 3 or more gold fiducials placed in the prostate 2 days after the completion of IGRT and prior to treatment planning. To allow for fiducial stabilization, planning images were obtained a minimum of 5-7 days after fiducial placement. Patients underwent thin cut (1.25 mm) contrast enhanced CT scan simulation. The clinical target volume (CTV) included the prostate, areas of radiographic extracapsular extension (ECE), and the proximal seminal vesicles to the point of separation. The prescription dose was 16-24 Gray in 2-3 fractions. The rectum, bladder, penile bulb, and head of femur were contoured and evaluated with dose volume histogram (DVH) analysis during treatment planning.
Follow-up and Toxicity assessment
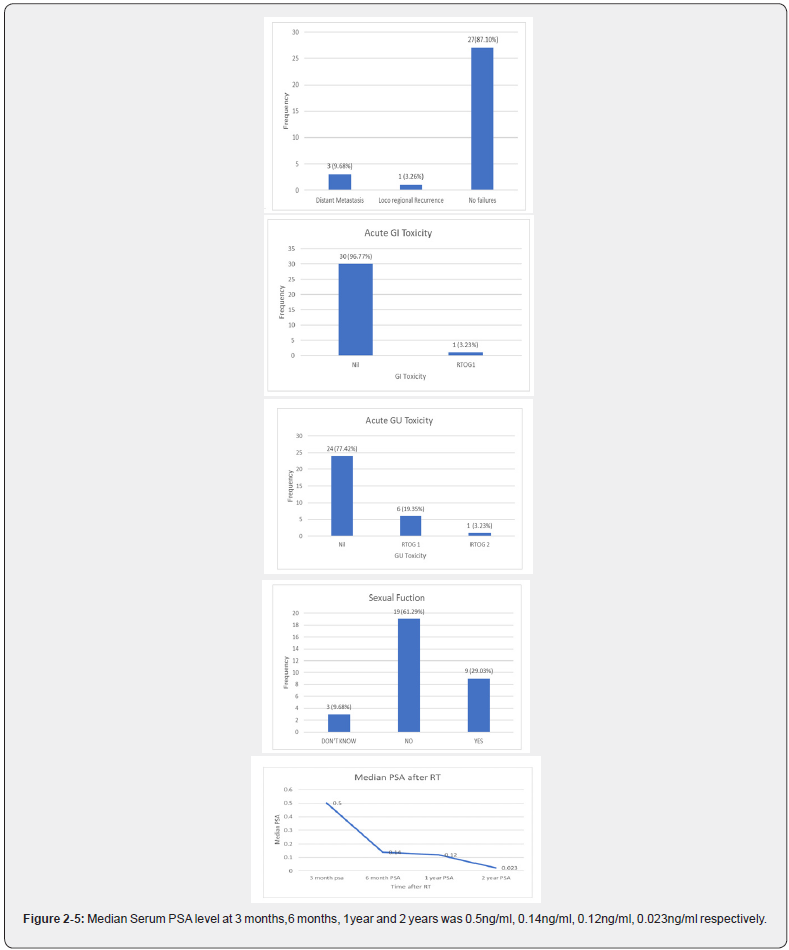
Patients were assessed at 1 month after therapy, every 3 months for the first year, and every 6 months for another year. Chronic GI and GU toxicities were documented at 2 years follow- up visits using RTOG grading score. Any sexual dysfunction was also documented at 2years of follow up period. Serum PSA level was measured at 3months,6 months, 1 year and 2 years of follow up and documented.
Results
From December 2015 to September 2017, 31 prostate cancer patients treated with IMRT plus SBRT boost were considered for the study retrospectively and the descriptive statistics was done on the data collected.
i. The median follow up was 24.4 months.
ii. The median age was 74 years (range 46–89years).
iii. The patients were generally healthy with ECOG PS 0-1.
iv. The median pre-treatment prostate-specific antigen level was 64ng/ml
v. 3 patients i.e., 9.6% were treated with 45Gy in 25 fractions whereas rest of the patients with 50Gy in 25 fractions.
vi. Chronic GI & GU toxicities were documented at 2 years for all patients, where most of the patients did not have any toxicity.
vii. 6 patients i.e., 19.3% had RTOG grade 1 GU toxicity with increase in frequency of micturition and 1 patient had RTOG grade 2 GU toxicity.
viii. Only 1 patient had RTOG grade 1 GI toxicity in terms of diarrhea.
ix. Sexual dysfunction was noted in 9 patients i.e.29% and 3 patients could not comment about the same. The median age for patients having sexual dysfunction was 76 years.
x. One patient had locoregional recurrence: with lymph-nodal recurrence.
xi. Only distant metastasis was in the bone seen in 3 patients. Two patients died among them with one cause of death was not due to disease per se.
Discussion
Increasing incidence of prostatic carcinoma in developed and developing countries warrants newer treatment strategies [5] (Figure 2-5). The most promising treatment strategies developed with respect to radiation therapy being exploration of the radiobiological advantage of hypofractionation and safe dose escalation to a prescription dose of 76–80 Gy [6,7].
Recent studies comparing dose escalated external beam radiation therapy to external beam radiation followed by brachytherapy boost demonstrates higher biochemical control with the later [1,2]. However, radio biologically, conventionally fractionated dose escalation strategies are inferior to the one that combines brachytherapy due to the lower alpha by beta of prostatic carcinoma [8]. External beam radiation followed by cyberknife boost circumvents the disadvantages of an invasive procedure at the same time makes safe dose escalation feasible.
Between dose escalated external beam radiation and external beam radiation followed by brachytherapy boost, there are differences with respect to radiobiology of the overall treatment time, dose per fraction, EQD2 and BED. These differences have translated to a superior biochemical control favoring the use of external beam radiation followed by a brachytherapy boost in high risk localized prostatic carcinoma. However, when external beam radiation is followed by a cyberknife boost, these differences could be minimized and the aim of our study was to analyze the difference in outcomes, if any, among the 31 patients treated with tomotherapy followed by cyber-knife boost compared to the historic cohort receiving dose escalated external beam radiation only (Figure 6).

Comparison of the Dose Schedule
The overall treatment time is less in the cyberknife boost when compared to the dose escalated external radiation. Although the alpha by beta of prostatic carcinoma is similar to or even less than late reacting normal tissues, the impact of overall treatment time in local control of prostatic carcinoma has been inconclusive [9]. However, a shorter treatment duration has its beneficial effects when patient comfort I and resumption to pretreatment life routine is concerned (Figure 7).
Comparison of the Radiobiological differences
BIOLOGICALLY EFFECTIVE DOSE FORMULA- BED=nd(1+d/a/b)
n-Number of fractions
d-dose per fraction
a/b-alpha by beta value
SCHEDULE 1- EXTERNAL BEAM RADIATION 2Gy per fraction, TD 76Gy
TUMOUR –BED-(a/b-1.5)-177 Gy
EARLY RECTING NORMAL TISSUE-BED-(a/b-10)-87.4 Gy
LATE RECTING NORMAL TISSUE-BED-(a/b-3)-126 Gy
SCHEDULE 2-EXTERNAL BEAM RADIATION TD50Gy followed by Cyberknife boost 7Gy x 3 fractions.
TUMOUR –BED-(a/b-1.5)-116.5+118.86 =235.36 Gy
EARLY RECTING NORMAL TISSUE-BED- (a/b-10)-60+35.7=95.7 Gy
LATE RECTING NORMAL TISSUE-BED-(a/b-3)-83.3+70= 153.3 Gy
Cyberknife boost to external beam radiation increases the biologically effective dose delivered to the tumor with a minimal increase in BED to acute and late reacting normal tissue. However, the therapeutic window is widened as the tumor control probability is increased at such high doses and with advanced radiation planning techniques such as cyberknife, dose to the normal tissue can be minimized.
Difference in BED to tumor and normal tissue between external beam radiation and external radiation followed by cyberknife boost. As noted, the proportion of increase in dose to tumor is much higher when compared to increase in proportion of dose to normal tissue thus widening the therapeutic window.
Comparison of mechanism of radiation action
The 4Rs of external beam radiation therapy such as repair, repopulation, redistribution and reoxygenation has very little role in stereotactic radiation. The major mechanism of action of stereotactic radiation is through the direct and indirect vascular damage and the immune stimulating effects of stereotactic radiation [10]. Combing stereotactic radiation as a boost to external radiation provides the combined radiobiological benefits of both. [11].
Comparison of results with historic cohort of dose escalated external beam radiation with conventional fractionation
A Phase II/III Androgen Suppression Combined with Elective Nodal and Dose Escalated Radiation Therapy (ASCENDE-RT) trial demonstrated that a low-dose-rate (LDR) brachytherapy boost resulted in a 50% reduction in biochemical relapse compared to dose-escalated external beam radiation therapy [12].The biochemical failure free survival at 5 years was 89% in the arm that received brachytherapy versus 84% in the External radiation only arm with a trend towards increasing biochemical failure at later years in the only external radiation arm. This trial included intermediate and high risk categories and only 69 percent of the patients belonged to the high risk category. The biochemical recurrence rate in this high-risk group is not known separately our study, although the number of cases is 31, all patients were of high risk and a biochemical failure free survival of 87% was reported at 2 years. Thus, the trend for biochemical failure is superior to dose escalated external radiation and maybe comparable to the brachytherapy boost arm.
While this study used a LDR boost there are similar encouraging outcomes using high-dose-rate (HDR) brachytherapy as a boost [13]. A randomized trial of external beam radiation alone or combined with HDR-brachytherapy boost for localized prostate cancer by Peter J Hoskin et al included 218 patients between the year 1997 to 2005.About 58% of these patients belonged to the high risk category. The relapse free survival at 5 years was 75% in the arm that used brachytherapy as a boost versus 61% in the only external radiation arm. Our results, although are at 2 years is superior to both the reported outcomes.
Comparing the toxicity profile, the rate of acute and late toxicity differs significantly with the technique used [14]. With improving radio therapeutic techniques, we are able to deliver precision radiation with image guidance. A study involving 843 men with localized prostate cancer assessed the toxicity profile in men treated with normal dose and dose escalated conformal radiotherapy. Late bowel toxicity was reported within 5 years of starting treatment by 33% of the escalated and 24% of the standard group. These data demonstrated that escalated dose highly conformed radiation with ADT improves treatment outcomes but is associated with an increased incidence of long-term adverse events [15].
Inverse-planned IMRT has been shown to reduce the dose delivered to the rectum and penile bulb (prostate and seminal vesicle radiotherapy), and the bowel and bladder (pelvic radiotherapy) [16]. data exists to suggest that IMRT can achieve reduced gastrointestinal (GI) toxicity, at least equivalent genitourinary (GU) toxicity, and effects on sexual function may also be improved [17]. The rate of sexual dysfunction reported varies between 20 to 50% with dose escalated external radiation [18].
In our study, external radiation was delivered using tomotherapy and no grade iii acute toxicity was seen not was any chronic toxicity found. The rate of gastrointestinal and genitourinary toxicity is less when compared to other dose escalated external beam radiation technique studies [19,20].
From our observation, external radiation followed by cyberknife boost provides outcomes superior to dose escalated external radiation only with toxicity profile lesser than the dose escalated external radiation group due to high precision radiation sparing normal tissues.
Conclusion
SBRT boost using cyberknife is a promising noninvasive strategy delivering higher tumoricidal dose with outcomes superior to dose escalated external beam radiation only. Further follow-up and outcome analysis including a larger case strength would be recommended.
References
- Bradley R. Pieters, Djuna Z de Back, Caro CE Koning, Aeilko H Zwinderman (2009) Comparison of three radiotherapy modalities on biochemical control and overall survival for the treatment of prostate cancer: A systematic review. Department of Radiation Oncology, Academic Medical Center, University of Amsterdam, The Netherlands Department of Clinical Epidemiology and Biostatistics, Academic Medical Center, University of Amsterdam, The Netherlands Radiotherapy and Oncology 93 (2): 168-173.
- Hoskin PJ, Rojas AM, Bownes PJ, Peter J Ostler, Linda Bryant (2012) Randomised trial of external beam radiotherapy alone or combined with high-dose-rate brachytherapy boost for localized prostate cancer. Radiother Oncol 103(2): 217-222.
- Fuller DB, Naitoh J, Lee C, Hardy S, Jin H (2008) Virtual HDRSM Cyber Knife treatment for localized prostatic carcinoma: dosimetry comparison with HDR brachytherapy and preliminary clinical observations. Int J Radiat Oncol Biol Phys 70(5): 1588-1597.
- Katz AJ, Kang J (2014) Stereotactic body radiotherapy as treatment for organ confined low-and intermediate-risk prostate carcinoma, a 7-year study. Front Oncol 4:240.
- Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68(6): 394-424.
- Dearnaley DP, Gordana Jovic, Isabel Syndikus, Vincent Khoo, Richard A Cowan et al. (2014) Escalated-dose versus control-dose conformal radiotherapy for prostate cancer: long-term results from the MRC RT01 randomised controlled trial. Lancet Oncol 15(4): 464-473.
- Zaorsky NG, Ohri N, Showalter TN, Dicker AP, Den RB (2013) Systematic review of hypo fractionated radiation therapy for prostate cancer. Cancer Treat Rev 39(7): 728-736.
- Fowler J, Chappell R, Ritter M (2001) Is α/β for prostate tumors really low? Int J Radiat Oncol Biol Phys 50(4):1021-1031.
- Vogelius IR, Bentzen SM (2013) Meta-analysis of the alpha/beta ratio for prostate cancer in the presence of an overall time factor: bad news, good news, or no news? Int J Radiat Oncol Biol Phys 85(1): 89-94.
- Hall EJ, Brenner DJ (1993) The radiobiology of radiosurgery: rationale for different treatment regimes for AVMs and malignancies. International Journal of Radiation Oncology Biology Physics 25(2): 381-385.
- Golden EB, Apetoh L (2015) Radiotherapy and immunogenic cell death. Semin Radiat Oncol 25(1): 11-17.
- Morris WJ, Tyldesley S, Rodda S, Halperin R, Pai H, et al. (2017) Androgen Suppression Combined with Elective Nodal and Dose Escalated Radiation Therapy (the ASCENDE-RT Trial): an analysis of survival endpoints for a randomized trial comparing a low-dose-rate brachytherapy boost to a dose-escalated external beam boost for high- and intermediate-risk prostate cancer. Int J Radiat Oncol Biol Phys 98(2): 275-285.
- Randomised trial of external beam radiotherapy alone or combined with high-dose-rate brachytherapy boost for localised prostate cancer. Peter J Hoskina, Ana M. Rojasa,low asterisk,' Peter J Bownesb, Gerry J Lowea, Peter J Ostlera, Linda Bryanta
- Matthew Sean Peach, Timothy N Showalter, and Nitin Ohri (2015) Systematic Review of the Relationship between Acute and Late Gastrointestinal Toxicity after Radiotherapy for Prostate Cancer.
- Dearnaley DP, Jovic G, Syndikus I (2014) Escalated-dose versus control-dose conformal radiotherapy for prostate cancer: Long-term results from the MRC RT01 randomised controlled trial. Lancet Oncol 15(4): 464-473.
- Zelefsky MJ, Chan H, Hunt M (2006) Long-term outcome of high dose intensity modulated radiation therapy for patients with clinically localized prostate cancer. J Urol 176(4 Pt 1): 1415-1419.
- Emily S Weg MD, Xin Pei, PhD, Marisa A Kollmeier MD, Sean M McBride, MD, MPH, Michael J Zelefsky (2019) Dose-Escalated Intensity Modulated Radiation Therapy for Prostate Cancer: 15-Year Outcomes Data. Adv Radiat Oncol 4(3): 492-
- Wiegner EA, King CR (2010) Sexual function after stereotactic body radiotherapy for prostate cancer: results of a prospective clinical trial. Int J Radiat Oncol Biol Phys 78: 442-448.
- Bekelman JE, Mitra N, Efstathiou J, Kaijun Liao, Robert Sunderland, et al. (2011) Outcomes after intensity-modulated versus conformal radiotherapy in older men with nonmetastatic prostate cancer. Int J Radiat Oncol Biol Phys 81(4): e325-e334.
- Patient-reported gastrointestinal and genitourinary toxicity after prostate cancer treatment: A comparison between radiotherapy including pelvic nodes and prostate-only radiotherapy. Per Fransson, Sebastian Olofsson, Anders Widmark, Camilla Thellenberg Karlsson. Journal of clinical Oncology.






























