Pleomorphic Variant of Primary Breast Liposarcoma: A Case Report of Rare Entity with Radiopathological Review
Anusha Paluri*, Shivakumar Swamy Shivalingappa, Sudhakar Sampangi, Avinash Kesari, Lohith G, Veena Ramaswamy and Kumar Kallur
Fellow in Breast Imaging, HCG Hospitals, Bangalore
Submission:June 03, 2021; Published:June 09, 2021
*Corresponding author: Anusha P, Fellow in Breast Imaging, HCG Hospitals, Bangalore
How to cite this article: Anusha P, Shivakumar Swamy S, Sudhakar S, Avinash K, Lohith G, et al. Pleomorphic Variant of Primary Breast Liposarcoma: A Case Report of Rare Entity with Radiopathological Review. Open Access J Surg. 2021; 13(2): 555856 DOI: 10.19080/OAJS.2021.13.555856.
Abstract
Mammary sarcomas are infrequent and locally aggressive, non-epithelial malignant tumors that arise from the mesenchymal elements of the breast with an incidence of less than 1% among all breast malignancies. They can occur as a pure primary form or arise secondarily from heterologous differentiation and malignant transformation of a phyllodes tumor. Liposarcomas of the breast are a rare subtype of sarcomas, seen in less than 0.3% of all breast malignancies. Pleomorphic variant of breast liposarcoma is the least common variant and extremely rare with only a few reported cases in the literature. We discuss a case of a middle-aged woman who presented with a rapidly growing lump in the left breast. She was evaluated by x-ray mammogram, ultrasound breast examination including ultrasound elastography study and 18F-FDG PET CT scan. She underwent ultrasound guided biopsy of the lump followed by left mastectomy and was subsequently histologically diagnosed as a primary pleomorphic liposarcoma of the left breast. Imaging features and pathological findings of this tumor along with differential diagnosis and novel treatment strategies are discussed in this case report.
Keywords: Pleomorphic, Liposarcoma, Breast, BIRADS, X-ray Mammogram, Ultrasound breast examination, 18F-FDG PET CT
Abbreviations: BIRADS: Breast Imaging and Reporting Data System; E/B ratio: Elastography/B- Mode Ratio; G: Gauge; 18F-FDG: Fluorine-18 Fluorodeoxyglucose; PET CT: Positron Emission Tomography Computed Tomography; CC: Cranio-caudal; MLO: Medio-lateral Oblique; VTI: Virtual Touch Tissue Imaging; H&E: Hematoxylin and Eosin; WHO: World Health Organization; MRI: Magnetic Resonance Imaging; SUV Max: Maximum Standardized Uptake Value; MDM2: Murine Double Minute 2; CDK24: Cyclin Dependent Kinase 24
Introduction
Among the primary soft tissue sarcomas, liposarcomas are the second most common subtype and usually involve the extremities. Sarcomas seldom occur within the breast and have an incidence of less than 1% among all mammary malignant neoplasms. Liposarcomas of breast are very rare tumors with an incidence of less than 0.3% among all breast malignancies, out of which pleomorphic liposarcoma is an extremely uncommon subtype with very few reported cases in the literature.
Case Report
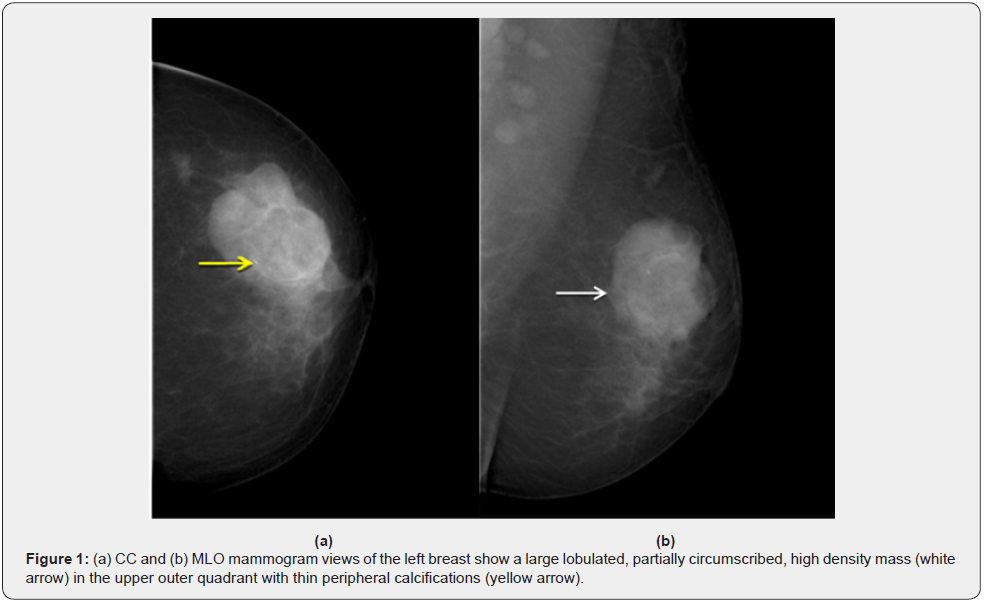
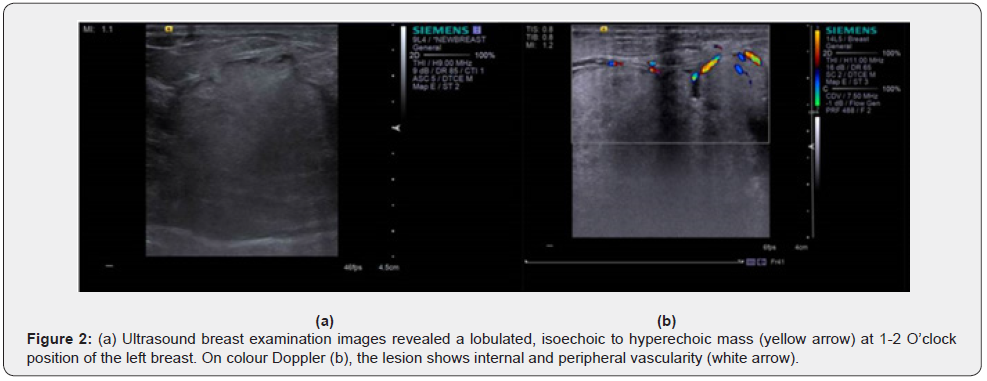
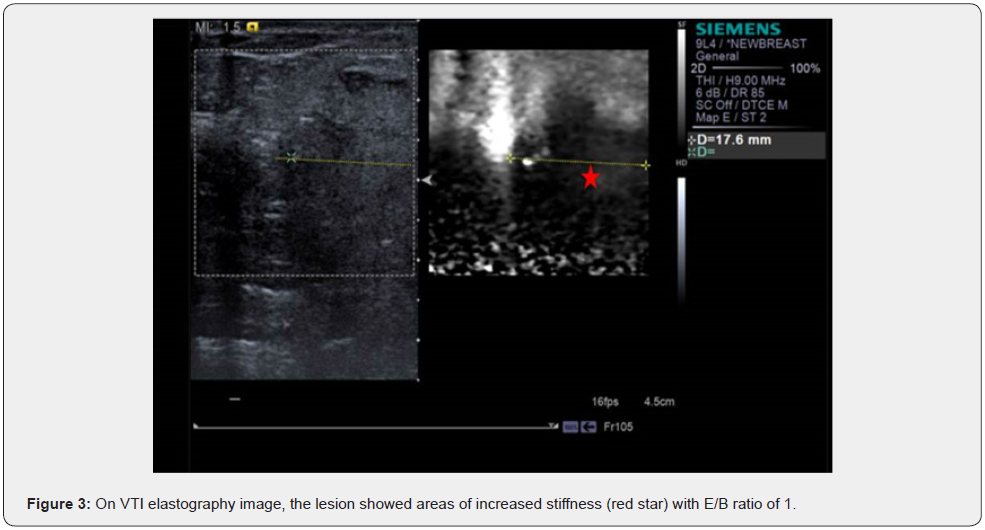
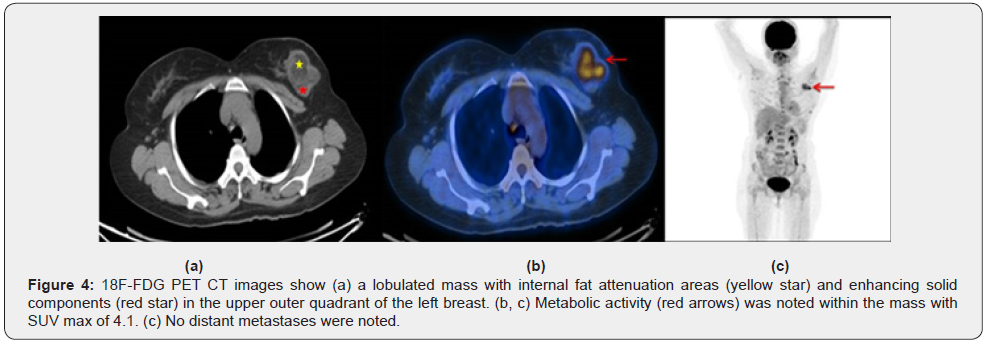
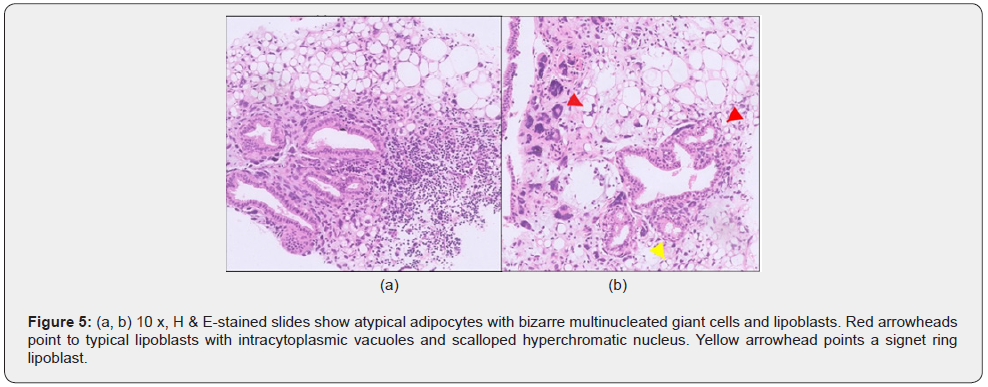
A middle-aged woman presented with a history of lump in the left breast which was rapidly increasing in size over a period of 2 months. Clinical examination revealed a large, non-tender, palpable mass in the upper outer quadrant of the left breast measuring 5 x 5 cm. There was no associated nipple retraction, skin edema, nipple discharge or palpable axillary nodes. There was no familial history of malignancies. She was advised x-ray mammogram and ultrasound breast examination for further evaluation. Mammogram revealed a large, partially circumscribed, high density mass in the upper outer quadrant of the left breast at 1-2 O’clock position with a few thin peripheral calcifications (Figure 1). Ultrasound breast examination revealed a lobulated, isoechoic to hyperechoic mass measuring about 5.5 x 3.5 cm with internal and peripheral vascularity (Figure 2). No pathologically enlarged axillary lymph nodes were noted. On ultrasound elastography study, the mass showed areas of increased stiffness with E/B ratio of 1(Figure 3). It was categorized as BIRADS IV- Suspicious of malignancy requiring histopathological evaluation. Ultrasound guided biopsy from the lesion was performed using 14G automatic biopsy gun with 13G co-axial and the histopathological diagnosis proved to be a liposarcoma. In view of the sarcomatous etiology of the lesion, 18F-FDG PET CT was performed as a part of institutional work up for metastases. 18F-FDG PET CT revealed a metabolically active lobulated mass with areas of internal fat attenuation and enhancing solid soft tissue components, measuring 5.4 x 4.9 x 4.6 cm in the upper outer quadrant of the left breast with SUVmax of 4.1(Figure 4). No distant metastases were detected. Patient later underwent a radical left mastectomy. On gross examination, a poorly circumscribed greyish white tumor measuring 4 x 3.5 x 3.5 cm was observed. Histopathological findings of the tumor comprised sheets of adipocytes, numerous lipoblasts with intracytoplasmic vacuoles and multinucleate giant cells (Figure 5). Most of the cells showed pleomorphic, hyperchromatic and mitotically active nuclei (up to 30 mitotic figures per high power field) with no epithelial elements confirming the final diagnosis of primary pleomorphic liposarcoma. All the 17 resected axillary lymph nodes were negative for tumor involvement.
Discussion
Breast cancers are one of the most common malignancies in women which usually arise from the epithelial components of fibro glandular parenchyma. Sarcomas of the breast are however rare with less than 1% incidence among all breast malignancies and arise from the interlobular mesenchymal components of the breast(1). Different types of sarcomas which arise in the breast include angiosarcoma, liposarcoma, fibrosarcoma, leiomyosarcoma, sarcomas with bone and cartilage and malignant fibrous histiocytomas. Angiosarcoma is the most common and aggressive variant among them [1]. Most breast sarcomas occur from 5th to 6th decades, except angiosarcoma which occurs in the 3rd to 4th decade. However, radiotherapy-associated angiosarcoma (RAAS) can occur at later ages as a rare complication of previous radiotherapy. Breast sarcomas are aggressive tumors which have the potential to grow into large masses ranging from 1 to 30 cm, with a mean size of 3 cm [2]. Tumor size is an important prognostic factor for sarcomas wherein tumors less than 5 cm have a better prognosis. They appear as well or partially circumscribed masses grossly, in spite of histological invasion at the borders [2].
Liposarcoma is an extremely rare type of mammary sarcoma with an incidence of less than 0.3% of all breast malignancies and about 2% to 10% among all breast sarcomas [3]. They can arise from heterologous liposarcomatous differentiation of phyllodes tumor or can occur as a pure primary mammary liposarcoma. Primary liposarcomas of breast have been classified by WHO into five variants. These include well differentiated, myxoid, pleomorphic, dedifferentiated and myxoid pleomorphic [4] with well differentiated variant being the most common subtype and pleomorphic variant being the rarest subtype [5].
Liposarcomas usually present as large masses with a median tumor size of 8 cm and age of incidence ranging from 4th to 5th decades [6]. Because of the rarity of this tumor, knowledge of its imaging appearances is important to diagnose the lesion early, considering the aggressive nature of sarcomas.
Diagnostic evaluation of liposarcoma of breast includes x-ray mammogram, ultrasound breast examination including ultrasound elastography study and MRI for characterization of the primary lesion. PET CT may be performed as part of metastatic work up.
On mammogram, liposarcomas appear as well-defined, lobulated and predominantly radiolucent masses containing soft tissue components [7]. On breast ultrasound examination, they appear as lobulated, typically well circumscribed, and heterogeneous masses with internal vascularity on color Doppler study. The identification of hyperechoic fat within the mass is often difficult and subjective [7]. On ultrasound elastography study, the non-lipomatous components show increased stiffness. Axillary lymph node metastasis, pectoral muscle invasion and skin involvement are rare with liposarcomas [6]. The presence of more than 1 cm large non-lipomatous component within the tumor points towards dedifferentiation which can be identified on cross-sectional imaging. Dedifferentiated liposarcomas are aggressive neoplasms with high metastatic potential [7].
MRI is valuable in the internal characterization of the lesion, determining the extent of lesion and presence of axillary lymphadenopathy. On dynamic gadolinium enhanced MRI study, these lesions appear as enhancing masses with early peak and gradual fading on kinetic analysis [6]. 18F-FDG PET CT determines metabolic activity within the lesion, presence of metastatic axillary lymph nodes, loco regional spread of tumor and distant metastases. They show avid uptake on 18F-FDG PET CT with a SUV of more than 4(6).
Axillary nodal involvement is rare with distant metastases being more common. The most common site of distant metastasis is the lung, followed by liver and bones [2]. Image guided core needle biopsy, preferably with ultrasound imaging is usually performed.
Histologically, liposarcomas are characterized by presence of pleomorphic lipoblasts of varying sizes and shapes. A typical lipoblast is identified by intracytoplasmic vacuoles and with scalloped hyperchromatic nucleus. The presence of these typical lipoblasts is an important feature in diagnosing a liposarcoma. In case of liposarcomas arising from the malignant phyllodes tumor, in addition to the lipogenic components, presence of glandular epithelial component is a key identification feature [8]. On immunohistochemistry, S100 is positive in liposarcomas due to staining of lipoblasts and adipocytes [9]. CDK 4 and MDM 2 markers are used to differentiate pleomorphic variants from dedifferentiated liposarcomas [4].
A comprehensive approach to treatment which includes surgery, radiotherapy and chemotherapy has been recommended. Surgical treatment, with either wide local excision or total mastectomy, is the most crucial step in the treatment plan [10]. Wide local excision is performed with a 2 cm margin. Sarcomas show predilection for hematogenous spread than lymphatic spread, due to which the axillary lymph nodes are rarely involved. Hence, axillary dissection is avoided during surgery unless there is evidence of lymph node metastasis. Adjuvant chemotherapy or radiotherapy is suggested in cases where size of the tumor is over 5 cm, negative tumor margins could not be attained by surgery or in liposarcomas with higher grade [11,12]. In a few studies, the use of neoadjuvant chemotherapy has been advocated to reduce the size of large tumors prior to surgery and to achieve negative tumor margins [10].
A major cause of recurrence is the marginal status of resected tumors, as evidenced by high rate of local recurrence after excision, inferring their locally aggressive nature. The prognosis depends on the histological subtype, size of tumor and resection status. Well differentiated variants mostly have a favorable outcome with a 5-year survival rate of 90% and high grade liposarcomas like dedifferentiated and pleomorphic variants have an aggressive course with a 5-year survival rate of 20%. Post treatment, well differentiated liposarcomas generally present with local recurrence whereas high grade liposarcomas present with distant metastases [6]. Differential diagnosis includes benign fat-containing lesions (lipoma, hamartoma, intramammary node, fat necrosis, pseudo-hamartoma type of galactocele, angiolipoma and spindle cell lipoma), phyllodes tumor, metaplastic carcinoma, invasive ductal carcinomas not otherwise specified and other sarcomas [7,13].
Conclusion
Liposarcomas are extremely rare non-epithelial malignancies of the breast, the pleomorphic variant being one of the rarest subtypes among them. The identification of the internal fat component is key imaging diagnostic feature which helps in limiting the differential diagnosis. Axillary lymph nodal metastases seldom occur due to preferential hematogenous spread of the tumor. Since they are prone for high rate of loco-regional recurrence, continued follow-up is recommended. Overall prognosis largely depends on the tumor size, histological subtype, and post resection marginal status. Owing to the rarity and aggressive nature of these tumors, knowledge of their imaging features and differential diagnosis is essential for prompt diagnosis and treatment.
Key Learning Points
i. Sarcomas of the breast are rare and aggressive tumors. Liposarcomas are extremely uncommon with less than 0.3% incidence among all mammary neoplasms, with pleomorphic variant being the rarest subtype.
ii. Liposarcomas can present as pure primary variant arising from the stromal elements of breast or as a secondary variant due to liposarcomatous differentiation of a phyllodes tumor.
iii. Identification of fat component within the lesion is a crucial feature for imaging diagnosis. The presence of more than 1 cm non-lipomatous component within the tumor points towards dedifferentiation.
iv. Sarcomas have preferential hematogenous route of spread due to which metastasis to the axillary lymph nodes occurs rarely.
v. Presence of typical lipoblasts histologically, is a hallmark feature of liposarcoma. Detailed sampling of the tumor for epithelial elements is essential to differentiate primary from the secondary variant.
vi. Management of liposarcomas is with wide local excision or mastectomy and adjuvant radiotherapy and/or chemotherapy.
vii. According to the current available literature, prognosis of pleomorphic liposarcoma is unfavorable with a five-year survival rate of 20%.
References
- Kluwer W, Reserved AR Rosen PP, Hoda SA, Koerner FC, Borgi E (2009) Rosen’s breast pathology. Philadelphia, PA: Wolters Kluwer Health/Lippincott Williams & Wilkins 26(7): 903-959.
- Smith TB, Gilcrease MZ, Santiago L, Hunt KK, Yang WT (2012) Imaging features of primary breast sarcoma. Am J Roentgenol 198(4).
- Banys Paluchowski M, Burandt E, Quaas A, Wilczak W, Geist S, et al., (2015) Liposarcoma of the breast arising in a malignant phyllodes tumor: A case report and review of the literature. World J Clin Oncol. 6(5): 174-178.
- The WHO Classification of Tumours Editorial Board. WHO Classification of Tumours Soft Tissue and Bone Tumours, 5th ed. Lyon: IARC Press; 2020.
- O Regan KN, Jagannathan J, Krajewski K, Zukotynski K, Souza F, et al., (2011) Imaging of liposarcoma: Classification, patterns of tumor recurrence, and response to treatment. Am J Roentgenol 197(1): W37-43.
- Nagarajan B, Autkar G, Patel K, Sanghvi M (2018) Primary breast liposarcoma. J Radiol Case Rep. 12(12):10–15.
- Ayyappan AP, Crystal P, Torabi A, Foley BJ, Fornage BD (2013) Imaging of fat-containing lesions of the breast: A pictorial essay. J Clin Ultrasound 41(7): 424-433.
- Mardi K, Gupta N (2011) Primary pleomorphic liposarcoma of breast: A rare case report. Indian J Pathol Microbiol 54(1): 124–126.
- Shankar V Pathology Outlines - Pleomorphic liposarcoma.
- Gutman H, Pollock RE, Ross MI, Gutman H, Pollock RE, et al., (1994) Sarcoma of the breast: implications for extent of therapy. The MD Anderson experience. Surgery 116: 505-509.
- Hummer CD, Burkart TJ (1967) Liposarcoma of the breast. Am J Surg 113(4): 558–561.
- McGowan TS, Cummings BJ, O Sullivan B, Catton CN, Miller N, et al., (2000) An analysis of 78 breast sarcoma patients without distant metastases at presentation. Int J Radiat Oncol Biol Phys 46(2): 383-390.
- Matsumoto RAEK, Hsieh SJK, Chala LF, de Mello GGN, de Barros N (2018) Sarcomas of the breast: Findings on mammography, ultrasoundand magnetic resonance imaging. Radiol Bras 51(6): 401-406.






























