The Spurious Swelling - Proliferative Fasciitis
Anubha Bajaj*
Consultant Histopathologist, Panjab University, India
Submission:November 14, 2019;Published:November 24, 2020
*Corresponding author:Anubha Bajaj, Consultant Histopathologist, Panjab University, India
How to cite this article:Anubha B. The Spurious Swelling - Proliferative Fasciitis. Open Access J Surg. 2020; 12(3): 555836. DOI 10.19080/OAJS.2020.12.555836.
Preface
Proliferative fasciitis is a benign, reactive, solid, rapidly progressive, pseudo-sarcomatous cellular proliferation of myofibroblastic soft tissue. Proliferative fasciitis was initially scripted by Chung and Enzinger in 1975 [1]. The mass-forming, subcutaneous cellular proliferation exemplifies a heterogeneous group of benign, myofibroblastic lesions which incorporate nodular fasciitis and proliferative myositis. Proliferative fasciitis is a subcutaneous or fascial, tumour- like fasciitis or an intramuscular myositis exhibiting spindle-shaped cellular proliferation with concurrent emergence of enlarged, basophilic, ganglion-like fibroblasts. Myofibroblasts and fibroblasts configuring proliferative fasciitis recapitulate the cellular component of nodular fasciitis [2]. A disorder of controversial pathogenesis and obscure genetic manifestations, proliferative fasciitis recapitulates sarcomas such as rhabdomyosarcoma or epithelioid sarcoma on account of histological manifestations and rapid tumour progression [2,3].
Keywords:Proliferative fasciitis; Myofibroblasts; fibroblasts; Paediatric neoplasms; Surgical exploration.
Disease Characteristics
Proliferative fasciitis is a rapidly progressive, self limiting neoplasm which typically appears within middle aged adults or elderly population between 40 years to 70 years. Proliferative fasciitis is uncommon in children below< 15 years. Paediatric neoplasms of proliferative fasciitis are unaccompanied by spontaneous involution. A specific ethnic or gender predilection is absent [2,3]. Proliferative fasciitis is commonly discerned within upper extremities, forearm, lower extremities, or trunk, in a descending order. Majority of lesions are confined to the fascia or subcutaneous tissue of upper extremities. The neoplasm is infrequently discerned upon the hand [3].
A clonal aberration (MYH-USP6) with genetic fusion is documented in nodular fasciitis, indicative of a benign, neoplastic origin. Aforesaid chromosomal mutation is absent in proliferative fasciitis which can be contemplated as a reactive, myofibroblastic proliferation with obscure genetic manifestation. It is posited that minor trauma or chronic inflammation can trigger proliferative fasciitis although a history of preceding injury is infrequent [2,3]. Proliferative fasciitis is associated with nonspecific features on imaging; thus a cogent tissue evaluation is necessitated for confirmation.
Clinical Elucidation
Proliferative fasciitis represents as a painful, palpable, oedematous, mobile, pseudo-sarcoma like, firm, tender, non-adherent or non-inflamed soft tissue tumefaction situated within subcutaneous adipose tissue which spares the muscular aponeurotic fascia. Alternatively, the neoplasm can emerge as a painless, reddish, rapidly progressive, papular tumefaction, un-associated with a clinical history of trauma [3,4]. The firm tumefaction can induce flexion of adjacent joints. An estimated two thirds lesions are painful, unlike nodular fasciitis and almost one third neoplasms are misinterpreted as a sarcoma due to rapid tumour progression and bizarre histological features [3,4].
Histological Elucidation
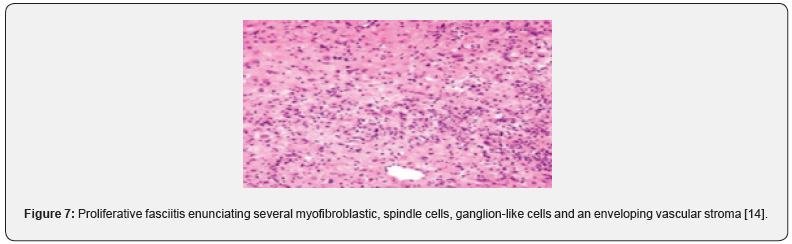
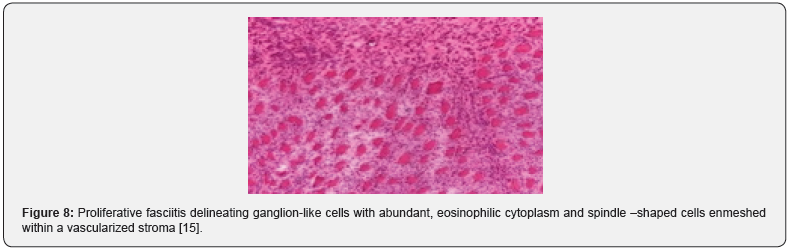
On gross examination, a pale, grey/white elliptical neoplasm of variable magnitude is discerned. Commonly, the tumefaction demonstrates a magnitude of two centimetres to three centimetres. The ill defined, subcutaneous mass may expand along horizontal fascial planes. Paediatric neoplasms demonstrate a well circumscribed, lobulated, firm, grey/ white tumefaction of magnitude varying from 1.5 centimetres to 3 centimetres [3,4]. As proliferative fasciitis is a benign, pseudo-sarcomatous proliferation of myofibroblastic soft tissue, preliminary lesions demonstrate foci of enhanced cellularity and myxoid alterations, akin to nodular fasciitis. On cytological examination, smears are cellular and comprised of spindle-shaped and enlarged cells with abundant cytoplasm, singular or dual eccentric nuclei and macro-nucleoli [3,4]. The neoplasm demonstrates proliferation of spindle –shaped, myofibroblasts intermingled within a variably myxoid and collagenous stroma. Additionally, enlarged, polygonal, ganglion-like myofibroblasts are observed, typically incorporated with abundant basophilic or amphophilic cytoplasm, enlarged, vesicular nuclei and prominent nucleoli [3,4]. The poorly circumscribed neoplasm is composed of plump, spindle-shaped cells configuring abridged fascicles admixed with enlarged, ganglion-like cells enmeshed within a fibro-collagenous stroma. Ganglion-like cells are polygonal or elongated and imbued with abundant, eosinophilic to amphophilic cytoplasm with singular or dual eccentric, vesicular nuclei and prominent nucleoli [5,6]. Characteristically, ganglion-like myofibroblasts intermixed with spindle-shaped myofibroblasts comprise of a biphasic, cellular population. The neoplasm with poorly defined margins is composed of plump, fibroblastic or myofibroblastic, spindle-shaped cells intermixed with enlarged, ganglion-like cells delineating abundant, amphophilic or basophilic cytoplasm, spherical, singular or multiple, vesicular nuclei and prominent nucleoli. Enveloping tumour stroma is collagenous or myxoid and frequently depicts foci of myxoid-cystic degeneration along with arborizing vascular configurations. Mitotic figures are variable and commonly discerned although atypical mitosis is absent [4,5]. Paediatric proliferative fasciitis is a lobular, non infiltrative neoplasm with a solid attern of tumour evolution, enhanced cellularity, enhanced mitotic activity with elevated proportion of myxoid to collagen stroma. The essentially cellular, paediatric neoplasm delineates a dissemination of prominent, ganglionlike cells recapitulating rhabdomyoblasts. Cellular and nuclear pleomorphism, mitotic figures and an intermingling of acute inflammatory cells with focal tumour necrosis is observed. Mature fibroblasts are minimal to absent. Tumour perimeter is pushing, in contrast to an infiltrative perimeter observed in adult neoplasms As paediatric tumefaction manifests foci of acute inflammation, tumour necrosis with exuberant mitosis, a misinterpretation as a sarcoma may be incurred. Proliferative fasciitis and proliferative myositis are identical in histology although appear in diverse locations. Upon ultrastructural examination, tumour cells of proliferative fasciitis demonstrate lack of cross striations or features of skeletal muscle differentiation [4-8]. (Figures 1-8).

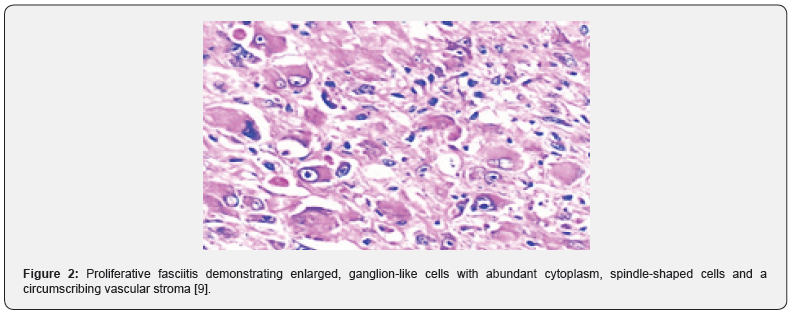
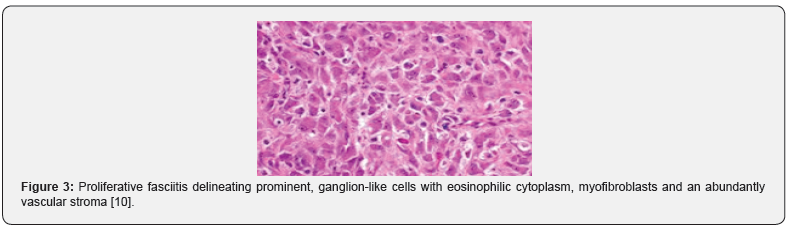

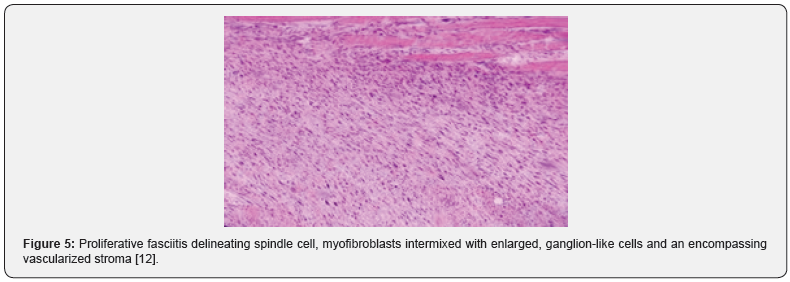
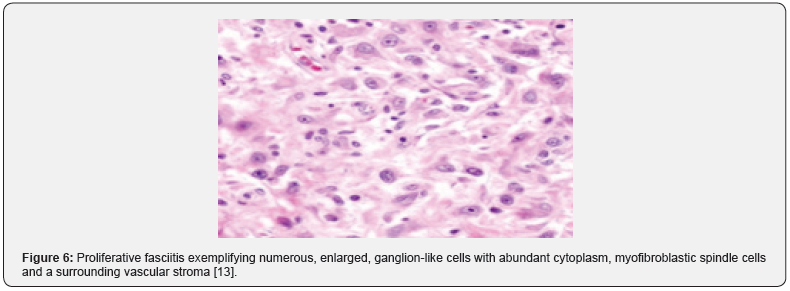
Immune Histochemical Elucidation
Spindle-shaped tumour cells are immune reactive to smooth muscle actin (SMA), muscle specific actin (MSA) and focally immune reactive to CD68. Ganglion-like cells are immune reactive to vimentin and inconsistently immune reactive to actin [2]. The neoplasm is immune-non reactive to cytokeratin, S100 protein, desmin, myogenin, β-catenin, h-caldesmon or CD34 [3,4]. Aforesaid pattern of immune reactivity is identical to immune staining denominated by nodular fasciitis and proliferative myositis.
Differential Diagnosis
Proliferative fasciitis mandates a segregation from
i. Giant cells tumour of tendon sheath (GCTTS) which requires a segregation on account of tumour location and contiguity with flexor tendon. Giant cell tumour of tendon sheath displays a lobular tumour architecture with polymorphous cellular composition of enlarged, histiocytoid cells with abundant eosinophilic cytoplasm, eccentric nucleus and prominent nucleolus. Intracytoplasmic hemosiderin or pigment deposition circumscribing enlarged, histiocytoid cells is discerned. Foamy or hemosiderin laden histiocytes are intermixed with osteoclastlike giant cells. Encompassing stroma is sclerotic and collagenous [6,7].
ii. Ganglioneuroma is composed of proliferation of spindleshaped cells, akin to neurofibroma. Incorporated ganglion cells and spindle-shaped cells are immune reactive to neural markers such as S100 protein.
iii. Nodular fasciitis is composed of myxoid or collagenous matrix, foci of cystic degeneration, spindle-shaped or stellate cells with loose, fascicular or storiform pattern and prominent mitosis. Disseminated lymphocytes, histiocytes and osteoclast-like giant cells are observed although ganglion-like cells are minimal to absent [6,7].
iv. Proliferative myositis is a morphologically similar neoplasm although located within intramuscular sites [6]. Paediatric proliferative fasciitis requires a separation from pertinent sarcomas such as rhabdomyosarcoma and epithelioid sarcoma.
v. Rhabdomyosarcoma demonstrates rhabdomyoblasts which may depict cross striations or simulate ganglion -like cells. Spindle-shaped cellular component may appear malignant and depicts cellular or nuclear atypia. The neoplasm is immune reactive to desmin or myogenin [6,7].
vi. Epithelioid sarcoma classically delineates spindleshaped tumour cells with abundant cytoplasm. Foci of tumour necrosis or inflammation are discerned. Epithelioid sarcoma is immune reactive to cytokeratin and exhibits a SMARCB1 chromosomal deletion or mutation [6,7].
vii. Undifferentiated sarcoma exhibits an enlarged tumour mass composed of spindle-shaped cells with nuclear atypia and cellular or nuclear pleomorphism, atypical mitotic figures and frequently discerned foci of tumour necrosis [6,7].
Additionally, proliferative fasciitis necessitates a demarcation from fibroma of tendon sheath, schwannoma or leiomyoma. Paediatric proliferative fasciitis requires a distinction from reticulohistiocytoma, xanthogranuloma or epithelioid haemangioendothelioma [6,7].
Investigative Assay
Upon plain radiography, bony lesions of proliferative fasciitis demonstrate a periosteal reaction. Adoption of singular plain radiography may be unsatisfactory for appropriate tumour discernment [7]. Ultrasonography exhibits a spherical, well circumscribed, soft tissue mass or a tumefaction of variable magnitude with circumscribing soft tissue oedema. Colour Doppler displays an echogenic, centroidal tumour zone with an absence of visible signal, a hypoechoic rim and mild vascularization of the nodule [7,8]. Magnetic resonance imaging (MRI) delineates a soft tissue mass situated upon extraneous surface of muscular superficial fascia. Upon T1 weighted imaging, tumefaction is homogenously isointense as compared to skeletal muscle. Upon T2 weighted, fat- suppressed imaging, neoplasm demonstrates a heterogeneous, peripherally intense signal [6,8]. Fat-suppressed T1 weighted imaging following contrast administration demonstrates an attenuated rim enhancement along with enhancement of miniature tumour zones concurrent with microvascular zones delineating enhanced signal intensity upon T2 weighted imaging. Peripheral soft tissue oedema and moderate tenosynovitis of abutting joints and flexor tendons is exemplified. Magnetic resonance imaging (MRI) can delineate an elliptical tumour mass situated between flexor tendons. The benign, non infiltrative neoplasm typically depicts an absence of diffusion- restriction upon diffusion- weighted imaging and lack of spontaneous retrogression [6,8].
Therapeutic Options
Surgical exploration demonstrates a tumefaction arising from tendon sheaths of deep-seated flexor muscles. Comprehensive or localized surgical extermination of the neoplasm is an optimal treatment strategy. As adoption of a fine needle or core needle biopsy may be accompanied by tumour involution, conservative management is advocated for lesions confined to the extremities. Postoperative clinical course is usually uneventful with immediate termination of symptoms. Localized tumour reoccurrence is usually absent. However, localized surgical excision is recommended for tumour reoccurrence [7,8].
References
- Chatterjee S, Chaudhuri T, Ghosh G, Ganguly A (2008) Gallstone ileus-an atypical presentation and unusual location. Int J Surg6:e55–e56.
- Chou JW, Hsu CH, Liao KF, Lai HC, Cheng KS et al.(2007) Gallstone ileus: report of two cases and review of the literature. World J Gastroenterol13:1295-
- Kurtz RJ, Heimann TM, Kurtz AB (1983) Gallstone ileus: a diagnostic problem. Am J Surg 146: 314-317.
- Rigler L, Borman C, Noble J(1941) Gallstone obstruction: pathogenesis and roentgen manifestation. JAMA 117: 1753.
- Danzi M, Grimaldi L, Fabozzi M, Reggio S, Danzi R, et al. (2013) Usefulness of CT-scan in the diagnosis and therapeutic approach of gallstone ileus: report of two surgically treated cases. BMC Surg 13(Suppl 2): S6.
- Lassandro F, Gagliardi N, Scuderi M (2004) Gallstone ileus analysis of radiologicalfindings in 27 patients. Eur J Radiol 50: 23-29.
- Balthazar EJ, Schechter LS (1978) Air in gallbladder: a frequent finding in gallstone ileus. AJR Am J Roentgenol 131:219-222.
- Martin F (1912) Intestinal obstruction due to gallstones. Ann Surg 55:725.
- Rodríguez Hermosa JI, CodinaCazador A, GironèsVilà J, Roig García J, FigaFrancesch M, et al. (2001) Gallstone Ileus: results of analysis of a series of 40 patients. Gastroenterol Hepatol 24: 489-494.
- Glenn F, Reed C, Grafe WR (1981) Biliary enteric fistula. SurgGynecolObstet 153: 527-531.
- Reisner RM, Cohen JR (1994) Gallstone ileus: a review of 1001 reported cases. Am Surg 60: 441-446.
- Turner AR, Sharma B, Mukherjee S (2020) Gallstone Ileus. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing, United States.
- Al-Obaid O (2007) Gallstone ileus: a forgotten rare cause of intestinal obstruction. Saudi J Gastroenterol13:39-
- Yu CY, Lin CC, Shyu RY, Hsieh CB, Wu HS, et al. (2005) Value of CT in the diagnosis and management of gallstone ileus. World Gastroenterol 11(14): 2142-2147.
- Lobo DN, Jobling JC, Balfour TW (2000) Gallstone ileus: diagnostic pitfalls and therapeutic successes. J Clin Gastroenterol 30: 72-76.
- Syme RG (1989) Management of gallstone ileus. Can J Surg32: 61-64.
- Giulia Artioli, Margherita Muri, Francesco Emanuele Praticò, Emanuele Angela Marcantoni, Silvia Eleonora Gazzani, et al. (2016) Gallstone ileus: literature review. Acta Biomed 87(3): 40-44.
- L Bouveret (1896) Stenose du pylore adherent a la vesicule. Revue Medicale (Paris) 16: 1-16.
- Bedogni G, Contini S, Meinero M, Pedrazzoli C, Piccinini GC (1985) Pyloroduodenal obstruction due to a biliary stone (Bouveret’s syndrome) managed by endoscopic extraction. Gastrointest Endosc 31:36-
- Dai XZ, Li GQ, Zhang F, Whang XH, Zhang CY (2013) Gallstone ileus: Case report and literature review. World J Gastroenterol 19(33): 5586-5589.
- Halabi WJ, Kang CY, Ketana N, Lafaro KJ, Nguyen VQ, et al. (2013)Surgery for Gallstone Ileus: A Nationwide Comparison of Trends and Outcomes. Ann Surg.






























