Epidural Analgesia after Gynecologic Oncology Surgery in the Era of Enhanced Recovery
Lisa Barroilhet1*, Han Cun2, Emmanuel Sampene3, Mayo Fujii4, David Kushner1 and Ryan Spencer1
1Division of Gynecologic Oncology, University of Wisconsin School of Medicine and Public Health, USA
2Department of Obstetrics and Gynecology, University of Wisconsin School of Medicine and Public Health, USA
3Department of Biostatistics and Medical Informatics, University of Wisconsin School of Medicine and Public Health, USA
4Department of Surgery, The University of Vermont Medical Center, USA
Submission: February 02, 2018; Published: March 19, 2018
*Corresponding author: Lisa Barroilhet, Department of Obstetrics and Gynecology, University of Wisconsin, 600 Highland Avenue H4/638 Madison, WI 53792, USA, Tel: 608-265-2319; Fax: 608-265-6572, Email: barroilhet@wisc.edu
How to cite this article: Lisa Barroilhet, Han Cun, Emmanuel Sampene, Mayo Fujii, David Kushner, Ryan Spencer. Epidural Analgesia after Gynecologic Oncology Surgery in the Era of Enhanced Recovery. Open Access J Surg. 2018; 8(4): 555742. DOI: 10.19080/OAJS.2018.08.555742.
Abstract
Objective: To compare pain scores, opioid use, and frequency of complications in gynecologic oncology patients who received epidural analgesia versus those who did not.
Methods: Two hundred fifty-four patients who underwent laparotomy between 2011 and 2013 were included in this retrospective study. We compared demographics, details of surgery, length of hospitalization, complications, pain scores, and total systemic opioids used between the epidural and no epidural groups. Opioid use was reported in intravenous (IV) morphine equivalents.
Results: Demographic data were similar between the two groups. One hundred seventy-eight patients (70%) received an epidural. Reported mean pain scores were slightly lower during the first 24 hours for those with an epidural compared to those without (2.6+1.7 vs. 3.1+2.1, P=.0558). Patients with an epidural used significantly less systemic opioids in the first 24 hours after surgery (47.1+ 22.8 mg vs. 87.1+ 65.6 mg; P<.001). There were no differences in thirty-day complications (epidural group 39.9%, no-epidural group 35.5%; p=0.513), venous thrombo embolism (VTE) (3.0% vs 1.0%; P=.53) or length of hospital stay (5.7+ 4.4 vs. 6.3+ 5.9 days; P=.42).
Conclusion: Patients undergoing laparotomy in gynecologic oncology who received an epidural used half the amount of systemic opioids in the first 24 hours. There was no difference in post-operative complications, thrombo embolic events or hospital stay between the two groups. Pain scores were similar. Decreased systemic opioid use in the first 24 hours after surgery without an increase in complications may be an important step to facilitate goal attainment in enhanced surgical recovery pathways.
Keywords: Surgery; Epidural; Opioids; Enhanced Recovery
Introduction
Multiple peer-reviewed publications address the topic of postoperative pain control in gynecologic oncology surgery [17]. Because many of these surgeries require large laparotomy incisions, patients frequently receive intravenous opioids for pain control. Patient-controlled analgesia, or PCAs, are often used as the mechanism of delivery as they allow patients to deliver pain medications based on their perceived needs [4,5,8]. Intravenous opioids are associated with undesired side effects, including nausea, pruritus, hallucinations, and delay in return of bowel function, which may ultimately lead to prolonged hospitalization and extended recovery times.
Alternatively, epidural analgesia is a commonly used and well-studied method for pain control after major abdominal surgeries [2,4,9]. Epidural analgesia is administered by placing a small gauge flexible catheter into the epidural space to provide continuous dosing of pain medications. The catheters are typically placed pre-operatively but not utilized until the surgery is completed. Occasionally, epidural catheters are placed postoperatively, if the laparotomy was unexpected or when postoperative pain control is sub-optimal. Medications introduced via the catheter into the epidural space may include a local anesthetic, an opioid, or both. Analgesia can then be optimized while balancing adverse effects, allowing for decreased sedation compared to intravenous opioids [3,5].
To date, studies evaluating epidural analgesia for postoperative pain in gynecologic surgery have shown inconsistent results. There are studies demonstrating the efficacy of epidural use during abdominal and pelvic surgery [4,9-11]. While one publication demonstrated that the analgesic effect may be poor for women undergoing gynecologic surgery specifically [5]. In the latter study, those with an epidural were more likely to require supplemental IV opioids to achieve adequate pain control. A separate concern is the potential for reduced mobility when the epidural catheter is in place. Some patients experience a temporary decrease in lower extremity sensation after epidural placement, which could interfere with post-operative ambulation.
Reduced mobility is associated with an increased risk of venous thromboembolism (VTE) and may result in protracted urinary catheterization, with a subsequent increase in urinary tract infections (UTI) [2,4,12,13]. Prolonged catheterization, in particular, has become a quality metric and many hospitals emphasize early removal to reduce infection risk. There are other potential drawbacks to epidural analgesia specific to our patient population. It has known side effects that can be of particular concern in more complicated gynecologic oncology surgeries. For example, hypotension is a common side effect of epidural placement, and can be of serious consequence following bowel resection and reanastamosis [13,14].
"Hypotension can result in hypoperfusion to newly created staple lines..", resulting in breakdown of these vulnerable areas. Although no published data report an increase in bowel leaks in patients who receive epidural analgesia; "...anecdotally, this is a common concern..." among gynecologic oncologists. The use of epidural analgesia varies from institution to institution and surgeon to surgeon. Currently, there are no standardized recommendations for the use of epidural analgesia in gynecologic oncology surgery, and its use is typically determined by surgeon's preferences and hospital culture. In our institution, patients are almost uniformly offered epidurals for gynecologic oncology surgery, but many decline due to fear of additional needle sticks, nerve injury, or previous negative experiences with regional anesthesia.
This retrospective cohort study hopes to shed further light on the use of epidural analgesia for postoperative pain following gynecologic oncology surgery, particularly in the era of enhanced recovery pathways. Enhanced recovery pathways are quickly gaining traction in gynecologic oncology [15,16], but the role of epidural analgesia as part of these pathways is unknown. We hypothesize that patients receiving epidural analgesia used less systemic opioids compared to patients not receiving epidural analgesia. We evaluated retrospectively collected data collected on patients that underwent gynecologic oncology surgeries and had or did not have an epidural placed for postoperative pain control at a single institution between 2011 and 2013.
Materials and Methods
This retrospective cohort study was approved by the University of Wisconsin Institutional Review Board. All patients over the age of 18 who underwent laparotomy performed by a gynecologic oncologist between October 2011 and February 2013 were included in the initial chart review. Pfannensteil and midline vertical skin incisions were included. Vaginal, laparoscopic, robotic or single port surgeries were excluded, unless these surgeries were converted to open laparotomies. Eligible patient were identified using ICD 9 codes for the most common gynecologic oncology procedures performed at our institution (including exploratomy laparotomy, total abdominal hysterectomy, lymphadenectomy, bowel resection, omentectomy and bowel resection). Demographics, details of surgery, body mass index (BMI), length of hospital stay, complication rates, pain scores, and systemic opioid use were compared between the epidural and no-epidural groups.
Three investigators reviewed the data independently to ensure data integrity. Surgeries were labeled "extensive" if they included a bowel resection, splenectomy, or lymph node dissection. All opioid dosages were converted into intravenous (IV) morphine equivalents. Total opioid use included medications administered in the operating room, PACU, and during the patients' post-operative hospital admission. Opioids given via the epidural catheter were included. Pain scores were collected using a Visual Analog Scale (VAS) and Brief Pain Inventory (BPI) pain scores, recorded three times a day and averaged over a 24- hour period. Charts were reviewed for a history of pre-existing pain syndromes, such as fibromyalgia, chronic back pain, endometriosis, or any chronic opioid use, which the investigators felt may be relevant to post-operative opioid use. All covariates were identified a priori. Postoperative complications were recorded, including hospital readmission within the first 30 days after surgery, wound complications, UTIs and postoperative VTE (including both DVT and pulmonary embolism (PE)).
Differences in the frequency distributions of baseline covariates by epidural usage were compared via Chi-square test for categorical variables and a t-test for continuous variables. Covariates considered for all statistical approaches included age, body mass index (BMI), diagnosis, length of hospital stay, complications, pain score, day of discharge and epidural usage. The frequency of complication rates between patients who used epidurals were compared to those patients who declined or were ineligible for epidural placement. The outcome variable was total dosage of systemic opioids used in the first twenty- four hours (reported in IV morphine equivalents) and our main predictor of interest was epidural use.
A multivariable linear regression was performed to determine if any of the covariates were associated with the total opioid equivalents used in the first 24 hours. The analytic strategy for selecting the final model was to investigate each predictor to the outcome through a univariate analysis process. After gaining some inferences from the univariate analysis, interactions among the predictors were checked before proceeding to fit a full model. Since the interaction terms were not significant, our main effect model was used as our final model. In addition, the variables that failed to reach significance at the alpha level of 0.05 were left out of the main effect model. Data analysis was performed using the Stata Statistical Software: Release 14 (College Station, TX: Stata Corp LP).
Results
Three hundred fifty charts were reviewed based on ICD- 9 codes. Two hundred fifty four patients ultimately met our inclusion criteria and had post-operative data available for analysis. Demographic data are presented in Table 1. One hundred seventy-eight patients (70%) received an epidural. The epidural remained in place for an average of 1.6 days. There were no significant differences between patients in the epidural and non-epidural groups with respect to age, BMI, cancer diagnosis and complexity of surgery. The mean age of all subjects included in this study was 58.3 years (range 18-90). The mean BMI was 31.6 mg/kg 2 (range 16.3-61.9). The majority of surgeries (247/254) were performed using a midline vertical incision, the remaining were Pfannensteil incisions. One hundred eighty- nine patients (74.4%) ultimately had a cancer diagnosis on final pathology. One hundred and nineteen patients (46.9%) underwent an extensive surgery, as described in the methods section. No deaths were reported during the study period, and no patients were lost to follow-up in the 30 days following hospital admission.
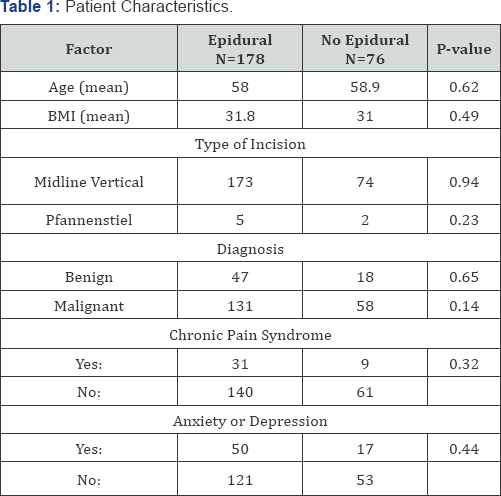
Pain was measured both by patient's subjective experience and total systemic opioid use. In the first 24 hours, reported mean pain scores were slightly lower for those with an epidural compared to no epidural (2.6+1.7 vs. 3.1+2.1, P=.0558) (Figure 1). Mean pain scores were not significantly different in the last 24 hours of hospitalization (2.2+1.53 vs 2.3+2.0, P=.69). The difference in mean pain scores from their first 24 hours and last 24 hours also demonstrated no statistical significance (0.39+2.0x vs 0.77+2.1, P=.17). Patients in the epidural group used nearly half the amount of systemic opioids in the first 24 hours after surgery compared to patients who did not have an epidural (47.1+22.8 mg vs. 87.1+ 65.6 mg; P<.001) (Figure 2).

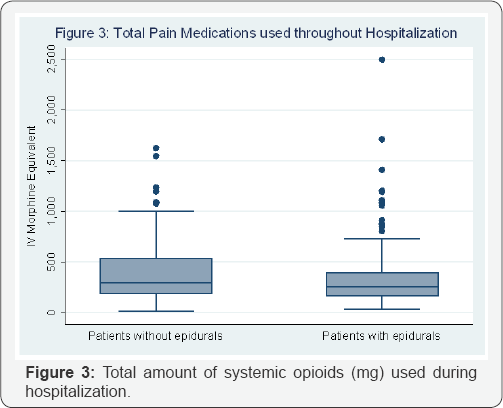

Over the course of their hospitalization, there was no statistical difference in total systemic opioid use in the two groups (343+308.3 mg vs 408+349.9 mg, P=.14) (Figure 3). Epidural catheters were left in place an average of 1.6 days. Twenty-eight patients underwent bowel resection with either reanastomosis or ostomy formation and had an epidural for postoperative pain, compared to 12 patients who underwent bowel surgery without an epidural. Use of epidural analgesia significantly reduced use of systemic opioids used in the first 24 hours in this subgroup (61.6+29.5 mg vs 98.6+75.5 mg, P=.03) (Figure 4). There was no increase in complications in patients who underwent bowel surgery and had an epidural placed. Although this subgroup is too small to make useful comparisons, no bowel leaks or anastamotic breakdowns were reported in the epidural group.
The presence of a pre-existing pain syndrome did not affect systemic opioid use in the first 24 hours in either group. Patients with a pain syndrome used a mean of 57.6 mg of IV morphine equivalents compared to those without a pain syndrome who used a mean of 58.2 mg (P=.94). However, those using an epidural who had a pain syndrome used less systemic opioids throughout the hospitalization (343.4+308.3 mg vs 408.2+349.9 mg, P=.14), although this difference was not statistically significant. There was no difference in mean length of hospital stay between those in the epidural group and no-epidural group (5.7+4.4 vs 6.25+5.9 days, P=.42). There were no differences in postoperative complications (epidural group 39.9%, noepidural group 35.5%; P=.51). Rates of VTE were examined separately and were similar in both groups (epidural group, 3%; no-epidural group, 1%, P=.53).
Discussion
We found that patients who received an epidural used fewer opioids, reported similar pain scores and did not have an increase in post-operative complications. These results would be generalizable to adult populations undergoing laparotomy that includes complicated pelvic and upper abdominal procedures. Patients receiving epidural analgesia did not have delays in ambulation, but were more likely to experience episodes of hypotension, resulting in fluid boluses. "Euvolemia is a critical part of enhanced recovery pathways, and it appears that epidural anesthesia may Post-operative pain control in gynecologic oncology... " has evolved considerably in the last several years. Enhanced Recovery After Surgery (ERAS) pathways have been widely embraced, largely due to excellent data in the colorectal surgery literature [13,17,18].
ERAS focuses on early ambulation, Foley catheter removal, and limited intravenous opioids. The role of epidural analgesia within enhanced recovery pathways is poorly understood. We are attempting to examine the potential benefits of epidural analgesia when used in combination with ERAS with a prospective randomized controlled trial currently accruing at our institution (NCT02423876). In this study, all patients participate in ERAS after gynecologic surgery. Subjects are randomized to either receive an epidural or not, in additional to standard ERAS. Our primary outcome is mean pain score in the first 24 hours after surgery.
Until the role of epidural analgesia as part of ERAS is clarified, one must consider the known benefits of epidurals, particularly in reducing opioid needs in the first 24 hours after surgery. Fewer opioids translate into less nausea and vomiting and earlier return of bowel function, which has been confirmed in multiple publications both in the gynecology and colorectal literature [6,10,11,15]. Improved pain control also leads to early, sustained ambulation. We believe that epidural analgesia may add additional benefit to ERAS. The complications of epidural analgesia most commonly feared based on anecdotal evidence, specifically an increase in infection rates, VTE, and extended hospital stay, were not seen in this retrospective review.
Additional benefits of regional analgesia and anesthesia have been studied in non-gynecologic cancers. The breast cancer literature has numerous publications supporting that neuro-axial blockade (most commonly, para-vertebral blocks) improves cancer outcomes [19,20]. The mechanisms behind this are not fully understood, but may be related to a decrease in inflammation and/or decreased systemic opioid use. Many neoplasms contain opioid receptors [21-23]. Intuitively, decreasing opioids may be beneficial from a cancer standpoint. The benefits of epidural analgesia may ultimately extend far beyond improved pain control.
The strengths of our study are the inclusiveness of a variety of gynecologic surgeries, including extensive oncologic resections, performed primarily through midline vertical skin incisions. A total of six surgeons were involved in the procedures described and the data were abstracted and reviewed by three independent parties. The weaknesses of our study are its retrospective design and relatively homogenous patient population. Also, the medications given to both groups of patients were in no way standardized. The use of non-opioid pain relievers (nonsteroidal anti-inflammatory agents, acetaminophen) was not reported nor standardized between the two groups. Patients who did not receive an epidural either were ineligible due to medical contraindications, declined to have one placed, or the surgeon was opposed to epidural placement. This introduces potential selection bias in the epidural group.
Conclusion
In conclusion, patients undergoing laparotomy for gynecologic oncology indications who received an epidural used less systemic opioids in the first 24 hours after surgery. Patients who received epidurals had similar rates of postoperative complications, thromboembolic events, and length of hospital stay when compared with those who did not. Pain scores were similar in both groups. Decreased systemic opioid use in the first 24 hours after surgery without an increase in complications may be an important step in facilitating goal attainment in enhanced surgical recovery pathways.
References
- Werawatganon T, Charuluxanun S (2005) Patient controlled intravenous opioid analgesia versus continuous epidural analgesia for pain after intra-abdominal surgery. Cochrane Database Syst Rev 1: Cd004088.
- Courtney Brooks M, Kurtz KCT, Pelkofski EB, Nakayama J, Duska LR (2015) Continuous epidural infusion anesthesia and analgesia in gynecologic oncology patients: Less pain, more gain? Gynecol Oncol 136(1): 77-81.
- Moslemi F, Rasooli S, Baybordi A, Golzari SE (2015) A Comparison of Patient Controlled Epidural Analgesia With Intravenous Patient Controlled Analgesia for Postoperative Pain Management After Major Gynecologic Oncologic Surgeries: A Randomized Controlled Clinical Trial. Anesth Pain Med 5(5): e29540.
- Rivard C, Dickson EL, Vogel RI, Argenta PA, Teoh D (2014) The effect of anesthesia choice on post-operative outcomes in women undergoing exploratory laparotomy for a suspected gynecologic malignancy. Gynecol Oncol 133(2): 278-282.
- Chen LM, Weinberg VK, Chen C, Powell CB, Chen LL, et al. (2009) Perioperative outcomes comparing patient controlled epidural versus intravenous analgesia in gynecologic oncology surgery. Gynecol Oncol 115(3): 357-361.
- Oh TK, Lim MC, Lee Y, Yun JY, Yeon S, et al. (2016) Improved Postoperative Pain Control for Cytoreductive Surgery in Women With Ovarian Cancer Using Patient-Controlled Epidural Analgesia. Int J Gynecol Cancer 26(3): 588-593.
- Dickson EL, Stockwell E, Geller MA, Vogel RI, Mullany SA, et al. (2017) Enhanced Recovery Program and Length of Stay after Laparotomy on a Gynecologic Oncology Service: A Randomized Controlled Trial. Obstet Gynecol 129(2): 355-362.
- Abu Rustum NR, Gemignani ML, Moore K, Sonoda Y, Venkatraman E, et al. (2003) Total laparoscopic radical hysterectomy with pelvic lymphadenectomy using the argon-beam coagulator: pilot data and comparison to laparotomy. Gynecologic oncology 91(2): 402-409.
- Block BM, Liu SS, Rowlingson AJ, Cowan AR, Cowan JA, et al. (2003) Efficacy of postoperative epidural analgesia: a meta-analysis. JAMA 290(18): 2455-2463.
- Wu CL, Cohen SR, Richman JM, Rowlingson AJ, Courpas GE, et al. (2005) Efficacy of postoperative patient-controlled and continuous infusion epidural analgesia versus intravenous patient-controlled analgesia with opioids: a meta-analysis. Anesthesiology 103(5): 10791088; quiz 109-110.
- Bauchat JR, Habib AS (2015) Evidence-based anesthesia for major gynecologic surgery. Anesthesiol Clin 33(1): 173-207.
- Elit LM, Thomas H, Trim K, Mazurka J, Moens F (2004) Evaluation of postoperative pain control for women undergoing surgery for gynaecologic malignancies. J Obstet Gynaecol Can 26(12): 1051-1058.
- Marret E, Remy C, Bonnet F, Postoperative Pain Forum G (2007) Metaanalysis of epidural analgesia versus parenteral opioid analgesia after colorectal surgery. Br J Surg 94(6): 665-673.
- Vagts DA, Iber T, Szabo B, Haberstroh J, K Reising, et al. (2003) Effects of epidural anaesthesia on intestinal oxygenation in pigs. British journal of anaesthesia 90(2): 212-220.
- Nelson G, Kalogera E, Dowdy SC (2014) Enhanced recovery pathways in gynecologic oncology. Gynecol Oncol 135 (3): 586-594.
- Barber EL, Van Le L (2015) Enhanced Recovery Pathways in Gynecology and Gynecologic Oncology. Obstet Gynecol Surv 70(12): 780-792.
- Greco M, Capretti G, Beretta L, Gemma M, Pecorelli N, et al. (2014) Enhanced recovery program in colorectal surgery: a meta-analysis of randomized controlled trials. World J Surg 38(6): 1531-1541.
- Zhuang CL, Ye XZ, Zhang XD, Chen BC, Yu Z (2013) Enhanced recovery after surgery programs versus traditional care for colorectal surgery: a meta-analysis of randomized controlled trials. Dis Colon Rectum 56(5): 667-678.
- Snyder GL, Greenberg S (2010) Effect of anaesthetic technique and other perioperative factors on cancer recurrence. Br J Anaesth 105(2): 106-115.
- Fried A (1977) The influence of the anaethetic on survival rates of breast cancer patients after surgery. International journal of cancer Journal 20(2): 213-218.
- Fanning J, Hossler CA, Kesterson JP, Donahue RN, McLaughlin PJ, et al. (2012) Expression of the opioid growth factor-opioid growth factor receptor axis in human ovarian cancer. Gynecol Oncol 124(2): 319324.
- Hatzoglou A, Gravanis A, Margioris AN, Zoumakis E, Castanas E (1995) Identification and characterization of opioid-binding sites present in the Ishikawa human endometrial adenocarcinoma cell line. J Clin Endocrinol Metab 80(2): 418-423.
- Zagon IS, Smith JP, Conter R, McLaughlin PJ (2000) Identification and characterization of opioid growth factor receptor in human pancreatic adenocarcinoma. Int J Mol Med 5(1): 77-84.






























