The Synergetic Effect of L-PrfTM & The Ossean® Implant Surface
Robert J Miller*1, Marco Del Corso2 and Andrea Mascolo
1Chairman Department of Oral Implantology, Atlantic Coast Dental Research Clinic, US
2Private Practice, Turin, Italy
3D.D.S. MSc. (Oral Surg.) Associate Professor UFP University Fernando Pessoa, Portugal
Submission: October 02, 2017; Published: October 16, 2017
*Corresponding author: Robert J Miller DDS, FACD;DABOI, USA, Email: drrjmiller1@msn.com
How to cite this article: Miller R.J, DelCorso M. , Mascolo A. The Synergetic Effect of L-Prf� & The Ossean® Implant Surface. Open Access J Surg.2017; 6(4): 555691. DOI: 10.19080/OAJS.2017.06.555691
Short Communication
Clinical outcomes in implant dentistry are dependent on a host of factors that are well documented. The evolution of our discipline has involved the physical manipulation of the site and materials used but now also includes the shaping of the biologic response. Speed of healing and the quality of the tissue regenerated are now considered two of the most important attributes as it relates to surgical success. As clinical outcomes are dependent on the types of biomaterials used, one of the strategies that we now employ is using the synergistic effect of a combination of these materials to ensure the best clinical outcomes.Our capacity to shorten treatment time for patients and still achieve successful function and aesthetics requires compression of the wound response.
Starting 20 years ago, the initial use of autologous growth factors, in the form of platelet rich plasma (PRP), enabled us to speed up the regeneration of soft tissue. As it had very little effect on hard tissue, researchers developed recombinant human growth factors in the form of platelet derived growth factor (rhPDGF) and bone morphogenic protein (rhBMP) to address this inadequacy. While there was success in speeding up the process of regenerating hard tissue, the bone was relatively immature unless the clinicianwaitedlonger before placingimplants.A decade ago, a second-generation autologous material in the form of leucocyte containing platelet rich plasma (L-PRF®) became available. The entire range of growth factors necessary for regeneration of both hard and soft tissue, in concentrated form, now enables us to predictably compress treatment time while creating a tissue envelope that normally would be seen following a much longer period of healing1 . But if it is true that a paradigm is only as good as the weakest link, the choice of implant architecture and surface must also be considered [1].
One of the most exciting developments in implant surface design has been the incorporation of calcium phosphate into the titanium oxide layer in a surface treatment called OSSEAN®. It is a fractal nano-roughstructure, and isideal for fibrin attachment and platelet deposition. It has receptor sites that further encourage cell growth, which has tens early osseointegration. It creates a calcium rich environment, potentiating osteoblastic cell activity by altering gene expression and produces a higher percentage of vital bone at an earlier stage of healing. This rapid bone bonding is a requirement for earlyloading of implants i.However, when the OSSEAN® surface implants are placed in the presence of autologous biologics (L-PRF®), something remarkable occurs Synergy.In its simplest form, a synergistic effect is the creation of a whole that is greater than the sum of its parts.The benefit of this effect is that it serves only to add never subtract. The clinical outcomes that we observe far outweigh those that we see when these materials are used independently. This Synergy of these two different materials now enables us to compress the wound response in ways we could not have imagined just a few years ago [1,2].
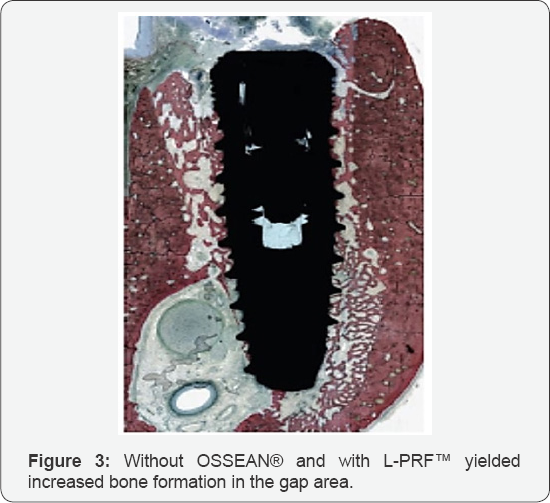
Recently published advances in these two individual biotechnologies lead a group of scientists to investigate justsuch a scenario3. They reasoned that if an implant with an osteoinductive, bioactivesurface (OSSEAN®) was immersed into a super blood clot (L-PRF®) obtained with the Intraspin™ system, bone should naturallygrowin an extraction site defectbetween the bone and implantsurface, even in the absence of initialstability. By simultaneously activating the bonysocket and implant surface, this effect was confirmed in terms of treatment outcomes, what are the immediate and long-term synergistic effects of Intraspinal-PRF® and the OSSEAN® implant surface mean? More predictable results with fewer complications, faster healing and the ability to load the implants sooner. In the long-term, the bond that is formed between the implant and bone is stronger and able to better with stand functional forces. All very positive and profound outcomes for clinicians and their patients [3](Figure 1-4).



References
- Dohan Ehrenfest DM, Piattelli A, Sammartino G, Wang HL (2017) New Biomaterials and Regenerative Medicine Strategies in Periodontology Oral Surgery Esthetic and Implant Dentistry BioMed Research International 2017(2017): 1-3.
- Coelho PG, Takayama T, Yoo D, Jimbo R, Karunagaran S, et al. (2014) Nanometer-scale features on micrometer-scale surfacetexturing: A bone histological gene expression and nanomechanicalstudy. Bone 65: 25-32.
- Neiva RF, Gil LF, Tovar N, Janal MN, Marao HF, et al. (2016) The synergisticeffect of leukocyte platelet-richfibrin and micrometer/ nanometer surface texturing on bone healing around immediately placed implants: An experimental study in dogs. BioMedResearch International 2016(2016): 1-9.






























