Chronic Abdominal Aortic Dissection with Large Aneurysm Unsuitable for Endovascular Treatment
J Ansell1, U Von Oppell2 and IM Williams1*
1University Hospital of Wales, UK
2Department of Cardiothoracic Surgery, University Hospital of Wales, UK
Submission: May 05, 2016; Published: May 18, 2016
*Corresponding author: IM Williams, Regional Vascular Unit, Ward B2, University Hospital of Wales, Heath Park, Cardiff, Wales, CF14 4XW, UK, Tel: +44(0)2920747747; Fax: +44(0)292074; Email: Ian.Williams5@wales.nhs.uk
How to cite this article: J Ansell, U Von O, IM Williams. Chronic Abdominal Aortic Dissection with Large Aneurysm Unsuitable for Endovascular Treatment. Open Access J Surg. 2016; 1(1): 555551. DOI: 10.19080/OAJS.2016.01.555551
Abstract
Primary aortic dissection originating in the abdominal aorta is rare and accounts for only 1-4% of cases. A case of chronic abdominal aortic dissection (AAD) with aneurysmal dilatation of the abdominal aorta is discussed. An endovascular option was not considered feasible due to the small diameter true lumen, close proximity of visceral vessels and the need for coverage of fenestrations to eliminate false lumen flow. Open repair was successfully performed for a large AAA secondary to a perirenal intimal tear and dissection 8 years previously. Primary aortic dissection localised to the abdominal aorta dissection is rare with an incidence of 1.3-4% [1]. Little is known about the natural history of abdominal aortic dissection (AAD) in contrast to thoracic aortic dissections. This article reports the management of a chronic AAD complicated by aneurysmal degeneration eight years after the initial causation.
Abbreviations: AAD: Abdominal Aortic Dissection; AAA: Abdominal Aortic Aneurysm; CT: Computed Tomography; CA: Coeliac Axis; RAs: Renal Arteries; SMA: Superior Mesenteric Artery; IMA: Inferior Mesenteric Artery
Case Report
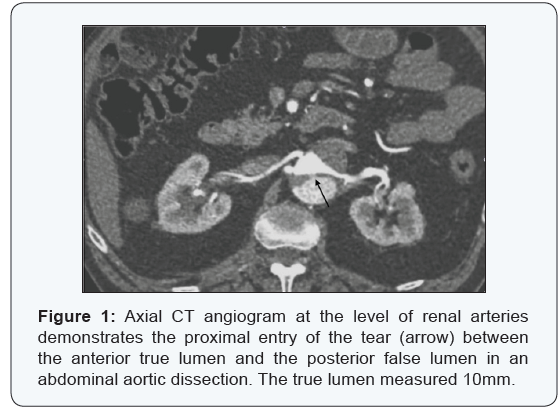
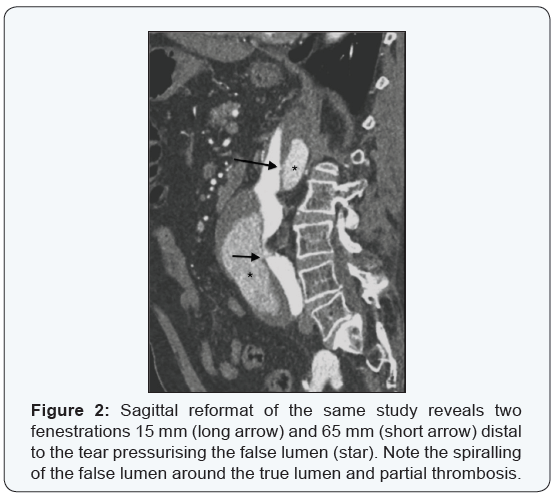
A 70 year-old male sustained an AAD 8 years previously and was being monitored for an enlarging abdominal aortic aneurysm (AAA). A computed tomography (CT) scan confirmed an 8cm diameter AAA extending 9cm above the origin of the coeliac axis (CA). At the level of renal arteries (RAs) there was a dissection flap pressurising the false lumen. Both the RAs arose from the true lumen as did the superior mesenteric artery (SMA) with the dissection flap continuing into the left common iliac artery and then re-entering the true lumen at the origin of the left external iliac artery. The right iliac artery was unaffected. There were two further fenestrations along the length of the dissection flap pressurising the false lumen. As well as the primary tear at the level of both RA (Figure 1), a second tear 15mm distal to this and the last a further 65mm inferiorly (Figure 2). The inferior mesenteric artery (IMA) was occluded at its origin.
Due to the small diameter of the true lumen (10mm) of the abdominal aorta and the need to revascularise all four major visceral arteries an endovascular approach was not considered feasible. Furthermore, any endovascular stent graft would also have to cover all three fenestrations in the dissection flap in order to seal off the false lumen. In view of these considerations an open thoracoabdominal repair was undertaken using partial femoro-femoral veno-arterial partial cardiopulmonary bypass with systemic cooling to 32°C. An end to end Dacron graft was sutured to the thoracic aorta at the hiatus and a Carrel patch anastomosis including the CA, SMA & right RA was done to the side of the graft. The dissection flap was divided longitudinally and a separate side branch was anatomised end to end to the left RA. Distally, the anastamosis was performed to both common femoral arteries, providing retrograde perfusion the hypo gastric arteries after ligation of the aortic bifurcation.
Discussion
This case presented a difficult clinical problem as the entry tear was at the RAs and this rapid expansion of the false lumen had caused the presenting symptoms of back pain. The AAD stabilised but due to continuing false lumen pressurisation aneurysmal dilatation occurred over 8 years and indeed extended proximally for 9cm. Further complications were 2 distal fenestrations pressuring the false lumen with only partial thrombosis. The diameter of the perirenal aortic true lumen was 10mm and distally the dissection flap spiralled through 180 degrees to extend into the left common iliac artery before reentering the true lumen and ending at the external iliac artery. The complexity of the anatomy following aortic remodelling made us consider open aortic surgery. The proximal end to end anastomosis was to normal aortic tissue at the level of the hiatus. Any anastomosis distal to this would have required buttress sutures with felt to close the true to the false lumen.
Clinical presentation of AAD is similar to the more common type B thoracic aortic dissection but the international registry suggests a higher incidence of mesenteric ischaemia, hypotension, abdominal pain and lower limb ischaemia [1]. Over 90% of the abdominal intimal entry tears arise between the RA and the aortic bifurcation with distal extension invariably into the iliac arteries [2]. On diagnosing AAD, up to 50% already have an associated AAA [2]. Pre-existing aneurysmal dilatation of the thoracic aorta is a known predisposing factor to subsequent acute dissections. There is also evidence spontaneous dissections have a higher incidence of chronic AAAs subsequently appearing compared to traumatic or iatrogenic causes. Some have proposed AAA secondary to AAD need treatment when they are as little as 2.7 to 3cm diameter which implies they may behave differently to the much more common atherosclerotic aneurysms [3,4]. As it is rare, the natural history of an aneurysm arising in an AAD is unknown. Dissected thoracic aortas tend to grow faster than aneurysmal aortas but predominantly dilate in the proximal upper descending aorta [5].
Optimum treatment of AAD remains unclear as published series consist of low numbers. Early reports invariably recommended open surgery as many preceded endovascular technology. A series of 10 patients with AAD by Farber described 9/10 of the entry tears at or below the renal arteries [3]. Intervention was performed in 4 patients where 3 underwent open surgery with a prosthetic graft and the other a stent graft after thrombolysis. The remaining 6 were treated conservatively and interestingly pain (if the presenting symptom) settled within 4 weeks. Becquemin published 7 cases of AAD in 1990 where 6 had an anastomosis to the infrarenal aorta with reinforcing sutures to approximate any dissection flap [6]. Mantelas reported a series of 6 patients treated with both open and endovascular surgery over a six year period. In the stent graft group all received an aortouni-iliac device with a femoral femoral cross over graft [7].
The current recommended management of acute type B dissections is a complication specific approach because of the higher mortality associated with acute intervention. The majority of uncomplicated thoracic dissections are treated medically with anti impulse therapy (beta blockade and angiotensin converting enzyme inhibitors). Organ ischaemia from branch vessel occlusion is treated primarily by fenestration and impending rupture manifested by ongoing pain, increasing periaortic haematoma or haemorrhagic pleural effusion by surgical replacement or by an endovascular approach [5]. The criteria for late interventions after Type B thoracic aortic dissections follow the size criteria of chronic non-dissected aneurysmal aortas.
Conclusion
It is likely AAD will be detected more frequently in the future due to increased imaging techniques available. Many already have an AAA when they present and careful planning for the complicated sequelae of the initiating entry tear is necessary. This is especially true with retrograde extension of the dissection as in order to incorporate normal aortic tissue in any surgical repair supracoeliac exposure may be required. Large AAA following AAD are rare indicating they may have a more aggressive natural history compared to the more common atherosclerotic type and warrant closer surveillance monitoring for progressive enlargement.
References
- Trimarchi S, Tsai T, Eagle KA, Isselbacher EM, Froehlich J, et al. (2007) Acute abdominal aortic dissection: Insight from the International Registry of Acute Aortic Dissection(IRAD). J Vasc Surg 46(5): 913-919.
- Jonker FH, Schlösser FJ, Moll FL, Muhs BE (2009) Dissection of the Abdominal Aorta. Current Evidence and Implications for Treatment Strategies: A Review and Meta-Analysis of 92 Patients. J Endovasc Ther 16(1): 71-80.
- Farber A, Wagner WH, Cossman DV, Cohen JL, Walsh DB, et al. (2002) Isolated dissection of the abdominal aorta: Clinical presentation and therapeutic options. J Vasc Surg 36(2): 205-210.
- Borioni R, Garofalo M, De Paulis R, Nardi P, Scaffa R, et al. (2005) Abdominal Aortic Dissections. Tex Heart Inst J 32(1): 70-73.
- Ziganshin BA, Dumfarth J, Elefteriades JA (2014) Natural history of Type B aortic dissection: ten tips. Ann Cardiothorac Surg 3(3): 247- 254.
- Becquemin JP, Deleuze P, Watelet J, Testard J, Melliere D (1990) Acute and chronic dissections of the abdominal aorta: Clinical features and treatment. J Vasc Surg 11(3): 397-402.
- Michail Mantelas, Polychronis Antonitsis, Dimitrios Kaitzis, Ananias Hatzibaloglou, Ioannis Moros (2009) Spontaneous isolated dissection of the abdominal aorta: single-center experience. Interactive CardioVasc Thoracic Surgery 8(4): 398-401.






