Multiple Pathogenic Mechanisms of Alzheimer’s Disease
Jianning Zeng1, Hongyan Pei1, Weijia Chen1, Rui Du2 and Zhongmei He1*
1College of Chinese Medicinal Materials, Jilin Agricultural University, Changchun 130118, Jilin, China
2Engineering Research Center for High Efficiency Breeding and Product Development Technology of Sika Deer, 130118 Jilin, China
Submission: May 18, 2023; Published: June 19, 2023
*Corresponding author: Jianning Zeng, College of Chinese Medicinal Materials, Jilin Agricultural University, Changchun 130118, Jilin, China
How to cite this article: Jianning Z, Hongyan P, Zhongmei H, Weijia C, Rui D. Multiple Pathogenic Mechanisms of Alzheimer’s Disease. Open Access J Neurol Neurosurg 2023; 18(2): 555983. DOI: 10.19080/OAJNN.2023.18.555983.
Abstract
Alzheimer’s disease is a growing global health problem with enormous implications for individuals and societies. In this review, we review the current understanding of the mechanism of sex differences in Alzheimer’s disease, the physiological structure of the brain - the entorhinal cortex, the genetic factors APOE, cytokines, mitochondrial homeostasis, and vascular dementia, and discuss the clinical manifestations of Alzheimer’s disease and the current treatment strategies. Finally, based on our understanding of the pathogenesis of Alzheimer’s, including an understanding of the long-term preclinical phase, informs new treatment strategies with the aim of moving from treatment to prevention.
Keywords: Alzheimer’s disease; Sex differences in AD; Entorhinal cortex; APOE cytokines; Mitochondrial homeostasis; Vascular dementia
Introduction
Alzheimer’s disease (AD) is a neurodegenerative disease characterized by widespread brain atrophy and loss of cognitive function [1]. Alzheimer’s disease is currently one of the major public health problems and is expected to increase exponentially in the coming decades, and has been the focus of research without being able to clarify its pathogenesis. Clinical symptoms of AD include progressive memory loss, impaired executive function, and difficulty performing routine daily activities; Early symptoms of AD include changes in thinking or unconscious behavior, memory impairment of new information, and language and speech dysfunction. In addition, 20% to 30% of patients with early AD show significant depressive symptoms and mood changes [2].
Mechanisms attributing to sex differences in AD
There is compelling yet incomplete evidence that the sex-specific, age-related depletion of estrogens in women and androgens in men are significant factors in the association between age and AD. Sex steroid hormones exert a wide range of neuroprotective actions in adults, termed sex hormone active effects, which diminish with age-related losses in hormones and hormone responsiveness. In addition, emerging evidence suggests that developmental effects of sex steroid hormones that lead to sexual differentiation of the brain, termed organizational effects, yield a female brain that may be inherently more vulnerable to AD pathogenesis [3].
The response to Aβ deposition evolves stereotypically in amphibian brain regions (cortex and hippocampus). Notably, Carlo showed that microglia in female mice progressed faster on the ARM trajectory compared to microglia in male mice, which is consistent with earlier histological analyses. This sex-dependent difference in microglia response is noteworthy because of the higher incidence of AD in females. Women may be more sensitive to these pathological biomarkers of AD than men. This is consistent with a recent study that reported higher levels of total TAU and Aβ42 in the cerebrospinal fluid, faster cognitive decline, and hippocampal atrophy in women, indicating more severe pathological changes than in men. A meta-analysis showed that men (OR, 3.09; 95% CI, 2.79-3.42) and women (OR, 3.31; CI, 3.03-3. 61) No difference in AD risk was shown for APOE ε3/ε4 genotypes at age 55 to 85 years; however, at age 65 to 75 years, women had a higher risk than men (women, OR, 4.37; 95% CI, 3.82-5.00; men, OR, 3.14; 95% CI, 2.68-3.67; P = .002) [4].
It was originally proposed that lifetime depression is a risk factor for Alzheimer’s disease because early meta-analyses showed a significant correlation between the duration of time between depression and the diagnosis of AD and the risk of AD. Stress exposure during adolescence has a stronger proximal effect on girls, including an increased risk of mood-related and stress-related conditions such as depression, anxiety, and posttraumatic stress disorder [4]. Depression is more common and severe in women [5], and moderate/major depressive symptoms are associated with a tripled increased risk of MCI in women but are not associated in men, although mild symptoms are associated with a tripled risk of MCI in men, but are not associated in women [6,7].
Women born in the first half of the 20th century had less education than men, which may explain the elevated risk for women [11], as limited formal education is a risk factor for dementia. This explanation requires more research, but there is evidence that in the United States, women’s educational attainment increased more than men’s over time, which led to a lower risk of dementia. Interestingly, studies in Europe have found that the association between lower educational attainment and dementia outcomes may actually be stronger in women than in men.
A sudden decrease in menopausal sex hormones (estrogen and progesterone) appears to affect the metabolic activity of the brain and the state of decreased / oxidation (redox) because: (i) 17β-estradiol, the main estrogen produced not only by the peripheral glands (ovaries), but also within the nervous system, regulates glycolysis, tricyclic acid circulation and mitochondrial respiration; (ii) decrease in sex hormone levels is paralleled by an increase of oxidative stress in female brains [8,9]. After menopause, ovarian sex hormones such as 17β-estradiol and progesterone decline rapidly. Before menopause, oophorectomy results in significant loss of estrogen, progesterone, and testosterone, disrupting the hypothalamic-pituitary axis. As we age, male sex hormones decrease, but their effects are less severe than those of female sex hormones such as progesterone and estrogen [10]. Estrogen compounds can prevent mitochondrial toxicity β amyloid [11], so estrogen action may be important for protecting cells from β amyloid toxicity, promising to become a possible treatment or prevention strategy for future female patients.
Gender differences in the risk of developing sleep disorders are recognized and women have more sleep problems and peak during menopause. The production of Aβ occurs mainly during waking, while its clearance occurs mainly during sleep. Older people tend to have poor sleep quality, which leads to a decrease in Aβ clearance, an increase in the accumulation of Aβ in the brain, and an increased risk of developing AD [12].
Although ɛ2 is generally believed to be protective against AD, evidence exists to suggest this is true in ɛ2/ɛ3 individuals of both sexes, but only in ɛ2 homozygous females [13].
Finally, there is evidence that although women’s β-amyloid and tau levels are similar, women show faster cognitive decline and neurodegeneration than men, meaning that Alzheimer’s marker protein may have more negative effects on women than men [14,15].
Entorhinal cortex
According to functional magnetic resonance studies, neural network connections appear to break in AD. Connectivity between the entorhinal cortex and the hippocampus decreases with age, and these functional changes are associated with memory deficits [16]. The entorhinal cortex (EC) is one of the first cortical brain regions to exhibit neuronal loss in AD. Neurons in the outer EC layer, especially in layer II, accumulate tau-positive neurofibrillary inclusion bodies and die early in AD. However, these selectively fragile neurons have not yet been characterized at the molecular level [17]. Alexandra proposed that the Alzheimer’s disease risk gene APOE was specifically inhibited in Alzheimer’s disease oligodendrocyte progenitor cells and astrocyte subsets, and upregulated in Alzheimer’s disease-specific microglia subcellular [18]. The study provides insights into the coordinated control of Alzheimer’s disease risk genes and their specific contributions to cell type susceptibility to disease. Glutamatergic systems are widely associated in AD pathophysiology. Glutamatergic dysfunction in AD appears to be mediated by a variety of mechanisms, including Aβ binding to glutamate receptors, tau binding to intrinsic cytoskeletal proteins leading to receptor overactivation, and internalization of glutamate transporters leading to glutamate accumulation in synaptic and extrasynaptic areas [19,20]. The glutamatergic system plays a key role in regulating synaptic activity, so any disruption of glutamatergic receptor composition can have a significant impact on normal neuronal function. Quantification of GluA2 expression in human post-mortem hippocampus revealed a significant increase in the stratum moleculare of the dentate gyrus in AD compared with control. Increased GluN1 receptor expression was found in the str. moleculare and hilus of the DG, str. oriens of the CA2 and CA3, str. pyramidale of the CA2, and str. radiatum of the CA1, CA2, and CA3 subregions and the entorhinal cortex. GluN2A expression was significantly increased in AD compared with control in the str. oriens, str. pyramidale, and str. radiatum of the CA1 subregion [21]. This study provides evidence of specific glutamatergic receptor subunit changes in the AD hippocampus and entorhinal cortex. In AD, the density of GluN1 around the entire hippocampus, hypothalamus, the entorhinal cortex, and neurons around STG decreases, membrane localization markers in the CA1 region are more, and neural staining is increased in all areas examined. Cases of AD showed increased immunoreactivity within the DG chain molecule compared to the control group.
In healthy children with ApoE4 carriers, the effect of ApoE4 on cognition and neurodegeneration has even been observed [22], as it impairs their working memory and causes the entorhinal cortex to thin with age, which is the initial sowing site of tau pathology [18]. Young people with the ɛ4 allele showed greater efficiency in learning memory, which was measured by reducing the blood oxygen level dependence (BOLD) response more rapidly than in learning experiments, and therefore using memory resources more efficiently [23]. They also showed better performance in terms of processing speed, attention, and language fluency. Despite the potential early benefits, older ɛ4 individuals showed evidence of an early decrease in entorhinal cortex volume and an increased rate of hippocampal volume loss [13].
APOE
Apolipoprotein E (ApoE) is a 34-kDa lipid-binding protein that acts in the transport of triglycerides and cholesterol in multiple tissues [24,25], including the brain [26], by interacting with lipoprotein receptors on target cells [27]. ApoE is especially important for the brain because other cholesterol transporters [28], such as ApoA1 [29] and ApoB [30], which are abundant in plasma, are almost non-existent in the brain, making the brain particularly dependent on ApoE for cholesterol transport [31].
A new ApoE-knockout AD mouse model experiment shows that plasma lipid levels can affect cognition and synaptic function independently of ApoE expression in the brain [32].
APOE, a3.6 kb long gene, is located on chromosome 19 and encodes for apolipoprotein E (APOE), a 299 amino acid long lipoprotein. Three APOE isoforms exist in humans: ApoE2, ApoE3, and APOE4, which differ from one another by single amino acid substitutions at positions 112 and 158, APOE2 (Cys-112, Cys- 158) [33], ApoE3 (Cys-112, Arg-158) [34], and ApoE4 (Arg-112, Arg-158) [35]. Many studies have shown that ApoE undergoes fragmentation in the human brain, and that fragmentation patterns vary by subtype. Previously shown that ApoE4 has a neurotoxic function, but recent data have also determined the neuroprotective effect of the ApoE N-terminal 25 kDa fragment, which is more prevalent in ApoE3 individuals. The ability of ApoE 25 kDa fragments to promote neurites has recently been demonstrated, suggesting that in addition to the increased toxic function of the specific ApoE4 fragments described earlier, ApoE4 individuals have a potential neuroprotective loss [36]. Substitution of cysteine at position 158 in ApoE2 results in hypocholesterolemia caused by low levels of low-density lipoprotein (LDL), cholesterol. In contrast, substitution of cysteine with arginine at position 112 in ApoE4 results in elevation of plasma cholesterol and LDL levels and predisposes the carrier to cardiovascular disease and neurodegenerative disorders, including Alzheimer’s disease [13]. Maria’s study showed that astrocytes and microglia differentially express and secrete glycosylated forms of ApoE, and that APOE4 astrocytes and microglia are deficient in immunomodulation compared to APOE2 and APOE3 [37].
Individuals carrying the APOE4 allele are at higher risk of AD than those carrying the ε3 allele, while the risk is reduced for the ε2 allele. APOE isoforms play a central role in the control of brain lipids, neuronal signaling, mitochondrial function, glucose metabolism, and neuroinflammatory transport [38]. Men with APOE ε3/ε4 were at increased risk of developing AD compared to men with APOE ε3/ε3. The APOE ε2/ε3 genotype protects women more than men to reduce the risk of AD. There was no difference in the risk of MCI between women and men aged 55 to 85 years, but there was an increased risk in women between the ages of 55 and 70. Between the ages of 55 and 85, there was no significant difference in the risk of switching from MCI to AD between men and women. Individuals at increased risk of APOE ε4/ε4 compared with individuals with ε3/ε4, but no significant differences were observed between men and women at ε4/ε4 [39].
Apolipoprotein E4 carriers had higher levels of plasma proinflammatory markers TNFα and IL-6 than apolipoprotein E3 carriers, suggesting that apolipoprotein E4 promoted inflammation compared to apolipoprotein E3. Using 18F-fluorodeoxyglucose (FDG) as a radioactive tracer, ApoE4 carriers exhibited low metabolism of local glucose relative to ApoE3 carriers [40]. There also appears to be a link between low metabolism and brain atrophy in apolipoprotein E4 carriers, as they exhibit decreased CMRglc and decreased MRI gray matter volume [41]. Interestingly, the effects of apolipoprotein E4 on amyloid deposition and glucose metabolism appear to be reversed, as ApoE4 is associated with more amyloid deposition in the frontal lobe and a more profound metabolic impairment in the posterior cortex [42].
Cytokines
In AD, the accumulation of Aβ in the brain disrupts the physiological functions of the brain, including synaptic and neuronal dysfunction, microglia activation, and neuronal loss. Interleukin-1 receptor accessory protein (IL-1RAcP) is a member of the immunoglobulin superfamily proteins consisting of soluble and membranous isoforms [43]. Elevated serum soluble ST2 (sST2) levels (a bait receptor for interleukin (IL)-33) in patients with MCI suggest that impaired IL-33/ST2 signaling may contribute to the pathogenesis of AD. IL-33 administration reduces soluble Aβ levels and amyloid plaque deposition by promoting microglia recruitment and Aβ phagocytosis activity, which is mediated by ST2/p38 signal activation. In addition, IL- 33 injection modulates the innate immune response by microglia/ macrophages to the anti-inflammatory phenotype and reduces the expression of pro-inflammatory genes (including IL-1β, IL-6 and NLRP3) in the APP/PS1 mouse cortex [44]. Lau defined a PU.1-dependent transcriptional pathway that drives IL-33- induced microglia functional state transitions that enhance Aβ clearance [45]. IL-33 induces CCL2, TNF-α and nitric oxide release by phosphorylation of ERK in mouse astrocytes. Incubation of mixed cultures containing glial cells and neurons or neuronal culture with IL-33 alone reduces the number of microtubuleassociated protein 2-positive neurons [46]. The elevation of both IL-10 and IL-33 is significantly associated with an improvement of episodic memory of treated patients, as measured by the Delayed Verbal Ray Test [47]. There is evidence that the inflammatory process is associated with AD. On the other hand, in patients with AD, inflammatory products aggregate at a different rate than in a healthy control group [10]. In addition, in patients with AD, IL-6 is present in age plaques, and an increase in immunoreactivity to IL-6 is noted in the ventricles and lumbar cerebrospinal fluid [48].
Female APOɛ4 carriers had higher IL-16 than non-carriers, whereas the opposite was true for IL-8 in males. In addition, women had higher plasma CRP and ICAN1 levels on average, but lower CSF ICAM1, IL-8, IL-16 and IgA levels than men. Potential cytokine biomarkers of the aging process vary by gender. A key molecule of inflammation is the pro-inflammatory cytokine TNF-α. Some evidence using genetic and pharmacological manipulations suggests that TNF-α signaling exacerbates Aβ and tau pathology in vivo. Interestingly, preventive and interventional anti-inflammatory strategies have shown reductions in brain pathology and improvements in cognitive function in rodent models of AD. phase I and phase II clinical trials suggest that TNF-α inhibitors may slow cognitive decline and improve daily activities in patients with AD.
Mitochondria
Decades of research have shown that mitochondria in Alzheimer’s disease (AD) patients differ from those in non-AD patients [54]. Mitochondria in AD patients are altered in number, ultrastructure and enzymatic activity. Effective clearance of aging and dysfunctional mitochondria by mitochondrial phagocytosis is essential for mitochondrial maintenance and neuronal health.
Aβ accumulates in the mitochondria of the AD brain, with altered mitochondria, decreased mitochondrial respiratory function and ATP production, impaired mitochondrial dynamics, and increased mitochondrial-associated oxidative stress [2]. It is clinically manifested by the presence of amyloid plaques (Aβ) and neurofibrillary tangles (NFT) within the brain. Due to intraneuronal processing, Aβ interacts with cellular targets such as mitochondria, ER, and Golgi apparatus and hampers their normal functions [49]. Typical neuropathological features of the disease (β amyloid) and sporadic AD risk genes (APOE) may trigger mitochondrial disorders, but mitochondrial dysfunction may precipitate pathology [50]. Alteration in the mitochondrial function, closely related to the production of reactive oxygen species (ROS), Ca2+ overload, and apoptosis in the brain, is one of the key pathological events studied in AD pathogenesis.
The development of chronic disease usually involves an increase in mitochondrial matrix Ca2+. Thus, by examining the content of endothelial cell mitochondrial Ca2+ to provide the ability to release into the cytoplasmic matrix, someone has altered the development of overall AD pathology by using FCCP mitochondrial uncoupling. FCCP is a proton ionophore (typically -180 mV relative to the cytoplasm), which reduces the affinity of Ca2+ ions within the mitochondrial matrix. Thus, Ca2+ on FCCP was increased after Ca2+ purinoceptor stimulation (except for the Aβ condition where Ca2+ was higher, with any visible sex difference between females (0.48 ± 0.02, n = 5) and males (0.33 ± 0.02, n = 5) on FCCP of approximately 40% (p<0.05) and any visible sex difference in AD and pre-addid disease. ΔF340/F380 is masked when considering only the pathology of AD in combination with data from both sexes. Mitochondrial dysfunction is often associated with altered redox status, and both bioenergetic deficiency and chronic oxidative stress are major causes of cognitive decline associated with brain aging and Alzheimer’s disease. Studies suggest that induction of mitochondrial stress responses is essential for maintaining mitochondrial protein homeostasis and health is critical. Notably, increasing mitochondrial protein homeostasis through pharmacologically and genetically targeted mitochondrial translation and mitochondrial autopsy could increase the fitness and longevity of GMC101 worms and reduce amyloid aggregation in cells, worms, and transgenic mouse models of Alzheimer’s disease.
It has been found that neurons with upregulated OxPhos genes have been observed in animal models of AD to exhibit higher oxidative damage (elevated 8-OHG levels), suggesting that mitochondrial dysfunction causes oxidative stress in the brain of AD [51]. Interestingly, hypermetabolism was detected in several studies in the early stages of patients with mild cognitive impairment (MCI) [52-55].
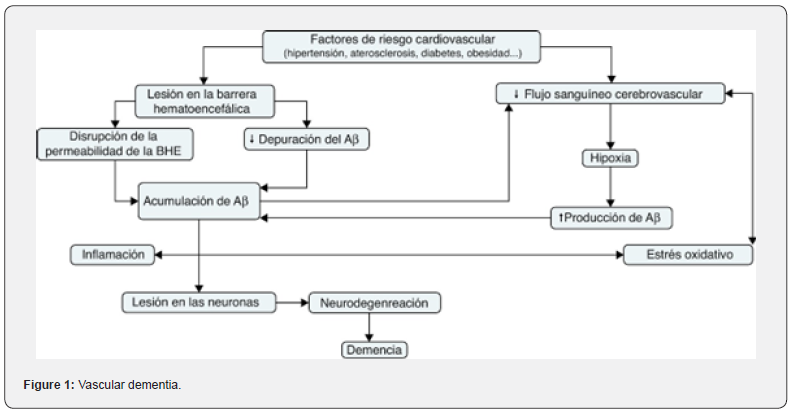
Vascular dementia
Vascular dementia (VaD) is recognised as the second most prevalent type of dementia [56].
Vascular hypothesis explaining the link between vascular dysfunction and AD. (Reprinted/adapted with permission from [57]).
Impaired blood flow response to neural activity due to neurovascular lesions can lead to a mismatch between neural activity and the supply of oxygen and glucose needed to meet adequate metabolic needs. As a result, neural activity is reduced and therefore associated with impaired brain function, which clinically manifests as cognitive decline.
Given the critical importance of cerebral blood supply to the structural and functional integrity of the brain, it is not surprising that changes in cerebrovascular vessels have a profound impact on cognitive function [58-60]. Overlap of AD neuropathology (amyloid plaque and neurofiber tangles) with cerebrovascular lesions is observed in up to 50% of cases of dementia [61]. Vascular lesions are also present in other age-related neurodegenerative diseases, such as synucleosis, hippocampal sclerosis, and frontotemporal lobe degeneration associated with tau or TDP-43, but coexistence with AD is the most common [62-64]. Due to the coexistence of different lesions and the overlap with neurodegenerative pathology, it is difficult to determine the effect of each disease on cognitive dysfunction. Vascular risk factors, including hypertension [65], diabetes mellitus [66], hyperlipidemia [67], smoking [68], atrial fibrillation [69] and hyperhymocytosis [70], increase the risk of dementia, in addition, metabolic syndromes, including insulin resistance, hypertension and dyslipidemia, are associated with lower cognitive abilities [71]. Vascular risk factors and associated oxidative stress and vascular inflammation alter BBB permeability [72], and BBB is destroyed during the course of the disease leading to worsening of the disease [71,73].
Since cerebrovascular disease and AD are common in the elderly, the coexistence of these two conditions may be just a coincidence. According to the additive model, the overall impact on cognition would come from the combined burden of vascular and neurodegenerative pathologies. Alternatively, vascular disease could promote AD and vice versa, causing an interaction between them that amplifies their pathogenic effects. The impact of vascular and AD neuropathology on cognition depends on the severity of AD pathology and the location of the vascular lesion. In advanced cases of AD, vascular pathology does not appear to have a significant impact on the progression of cognitive impairment, suggesting that AD pathology is a major driver of cognitive dysfunction. On the other hand, in older adults with moderate AD pathology, subcortical vascular lesions are a major determinant of dementia expression.
Discussion
In this review, we introduce the pathogenesis of AD through six aspects: sex differences, brain physiological structure- Entorhinal cortex, genetic factors APOE, cytokines, mitochondrial homeostasis, and vascular dementia. The causes of AD are multifactorial, complex and difficult to explain, and conditions involving aging and neuroscience are often difficult to explain.
Although our understanding of AD has increased dramatically in recent years, it is still not possible to fully clarify its mechanisms. Next-generation genetic research has touched on a number of pathways that are important for the pathogenesis of AD: These pathways are currently being explored in cell and animal models and have led to the identification of new drug targets. A more nuanced model of preclinical AD, no longer seeing β amyloid, tau, and inflammation as steps along a sequential pathway, but rather as part of the cellular phase of the pathogenesis of AD, which will also lead to more complex treatment and prevention approaches [74].
Despite the far-reaching chronic effects of AD, current treatments are unable to achieve satisfactory therapeutic outcomes or stop disease progression. Today, the FDA approves only five drugs for AD treatment: donepezil, kabbalatine, galantamine, tacrine, and memantine. The first 4 drugs are acetylcholinesterase inhibitors, while the last drug is N-methyl- D-aspartate receptor (NMDAR) antagonist [75]. The failure of many major Phase 3 clinical trials using monoclonal antibodies that target brain β amyloid has raised doubts about the amyloid hypothesis, but perhaps more worrying is the prospect of AD disease alteration. Regarding the Aβ56 fraud incident some time ago, the credibility of the Aβ hypothesis has been questioned to a certain extent, and people are more willing to believe that amyloid is a product of autoimmune reactions and is not the cause of the disease.
The characteristics of future patients involve designing treatments, including age, sex, genetic factors, environmental factors and lifestyle, etc., and the treatment methods may also be different for AD patients with different conditions.
Acknowledgments
The authors are grateful to the Jilin Province Science and Technology Development Program (Nos. 20210204001YY).
Funding Sources
This work was supported by the Jilin Province Science and Technology Development Program (No.20210204001YY), the Jilin Province Education Department Science and Technology Research Project (JJKH20210373KJ). We declare that there are no financial or other contractual agreements that are likely to cause or be deemed to cause conflicts of interest.
Author Contributions
Jianning Zeng wrote this manuscript. Hongyan Pei and Weijia Chen are responsible for collecting literature. Zhongmei He and Rui Du guided this article.
References
- Koutsodendris N, Nelson MR, Rao A, Huang Y (2022) Apolipoprotein E and Alzheimer’s Disease: Findings, Hypotheses, and Potential Mechanisms. Annu Rev Pathol Mech Dis 17: 73-99.
- Guo T, Zhang D, Zeng Y, Huang TY, Xu H, et al. (2020) Molecular and cellular mechanisms underlying the pathogenesis of Alzheimer’s disease. Mol Neurodegener 15(1): 40.
- Pike CJ (2017) Sex and the development of Alzheimer’s disease. J Neurosci Res 95(1-2): 671-680.
- Serrano-Pozo A, Das S, Hyman BT (2021) APOE and Alzheimer’s disease: advances in genetics, pathophysiology, and therapeutic approaches. Lancet Neurol 20(1): 68-80.
- Hodes GE, Epperson CN (2019) Sex Differences in Vulnerability and Resilience to Stress Across the Life Span. Biol Psychiatry 86(6): 421-432.
- Sassarini DJ (2016) Depression in midlife women. Maturitas 94: 149-154.
- Nebel RA, Aggarwal NT, Barnes LL, Gallagher A, Goldstein JM, et al. (2018) Understanding the impact of sex and gender in Alzheimer’s disease: A call to action. Alzheimers Dement 14(9): 1171-1183.
- Goveas JS, Hogan PE, Kotchen JM, Smoller JW, Denburg NL, et al. (2012) Depressive symptoms, antidepressant use, and future cognitive health in postmenopausal women: the Women’s Health Initiative Memory Study. Int Psychogeriatr 24(8): 1252-1264.
- Grimm A, Mensah-Nyagan AG, Eckert A (2016) Alzheimer, mitochondria and gender. Neurosci Biobehav Rev 67: 89-101.
- Brinton RD (2008) Estrogen regulation of glucose metabolism and mitochondrial function: therapeutic implications for prevention of Alzheimer’s disease. Adv Drug Deliv Rev 60(13-14): 1504-1511.
- Ashraf GM, Ebada MA, Suhail M, Ali A, Uddin MS, et al. (2021) Dissecting Sex-Related Cognition between Alzheimer’s Disease and Diabetes: From Molecular Mechanisms to Potential Therapeutic Strategies. Oxid Med Cell Longev 2021: 4572471.
- Viña J, Lloret A (2010) Why women have more Alzheimer’s disease than men: gender and mitochondrial toxicity of amyloid-beta peptide. J Alzheimers Dis 20 Suppl 2: S527-S533.
- Zhu D, Montagne A, Zhao Z (2021) Alzheimer’s pathogenic mechanisms and underlying sex difference. Cell Mol Life Sci 78(11): 4907-4920.
- Riedel BC, Thompson PM, Brinton RD (2016) Age, APOE and sex: Triad of risk of Alzheimer’s disease. J Steroid Biochem Mol Biol 160: 134-147.
- Koran MEI, Wagener M, Hohman TJ, Alzheimer’s Neuroimaging Initiative (2017) Sex differences in the association between AD biomarkers and cognitive decline. Brain Imaging Behav 11(1): 205-213.
- Buckley RF, Mormino EC, Amariglio RE, Properzi MJ, Rabin JS, et al. (2018) Sex, amyloid, and APOE ε4 and risk of cognitive decline in preclinical Alzheimer’s disease: Findings from three well-characterized cohorts. Alzheimers Dement 14(9): 1193-1203.
- Yassa MA, Mattfeld AT, Stark SM, Stark CEL (2011) Age-related memory deficits linked to circuit-specific disruptions in the hippocampus. Proc Natl Acad Sci U S A 108(21): 8873-8888.
- Leng K, Li E, Eser R, Piergies A, Sit R, et al. (2021) Molecular characterization of selectively vulnerable neurons in Alzheimer’s disease. Nat Neurosci 24(2): 276-287.
- Chang L, Douet V, Bloss C, Lee K, Pritchett A, et al. (2016) Gray matter maturation and cognition in children with different APOE ε genotypes. Neurology 87(6): 585-594.
- Butterfield DA, Pocernich CB (2003) The glutamatergic system and Alzheimer’s disease: therapeutic implications. CNS Drugs 17(9): 641-652.
- Kwakowsky A, Waldvogel HJ, Faull RL (2021) The effects of amyloid-beta on hippocampal glutamatergic receptor and transporter expression. Neural Regen Res 16(7): 1399-1401.
- Yeung JHY, Walby JL, Palpagama TH, Turner C, Waldvogel HJ, et al. (2021) Glutamatergic receptor expression changes in the Alzheimer’s disease hippocampus and entorhinal cortex. Brain Pathol 31(6): e13005.
- Calderón-Garcidueñas L, Gónzalez-Maciel A, Reynoso-Robles R, Delgado-Chávez R, Mukherjee PS, et al. (2018) Hallmarks of Alzheimer disease are evolving relentlessly in Metropolitan Mexico City infants, children and young adults. APOE4 carriers have higher suicide risk and higher odds of reaching NFT stage V at ≤ 40 years of age. Environ Res 164: 475-487.
- Evans S, Dowell NG, Tabet N, Tofts PS, King SL, Rusted JM (2014) Cognitive and neural signatures of the APOE E4 allele in mid-aged adults. Neurobiol Aging 35(7): 1615-1623.
- Tagalakis AD, Dickson JG, Owen JS, Simons JP (2005) Correction of the neuropathogenic human apolipoprotein E4 (APOE4) gene to APOE3 in vitro using synthetic RNA/DNA oligonucleotides (chimeraplasts). J Mol Neurosci 25(1): 95-103.
- Drury J, Narayanaswami V (2005) Examination of lipid-bound conformation of apolipoprotein E4 by pyrene excimer fluorescence. J Biol Chem 280(15): 14605-14610.
- Qi G, Mi Y, Shi X, Gu H, Brinton RD, et al. (2021) ApoE4 Impairs Neuron-Astrocyte Coupling of Fatty Acid Metabolism. Cell Rep 34(1): 108572.
- Wang JM, Irwin RW, Brinton RD (2006) Activation of estrogen receptor alpha increases and estrogen receptor beta decreases apolipoprotein E expression in hippocampus in vitro and in vivo. Proc Natl Acad Sci U S A 103(45): 16983-16988.
- Wang ZH, Xia Y, Liu P, Liu X, Edgington-Mitchell L, et al. (2021) ApoE4 activates C/EBPβ/δ-secretase with 27-hydroxycholesterol, driving the pathogenesis of Alzheimer’s disease. Prog Neurobiol 202: 102032.
- Montañola A, de Retana SF, López-Rueda A, Merino-Zamorano C, Penalba A, et al. (2016) ApoA1, ApoJ and ApoE Plasma Levels and Genotype Frequencies in Cerebral Amyloid Angiopathy. Neuromolecular Med 18(1): 99-108.
- Khalil YA, Rabès JP, Boileau C, Varret M (2021) APOE gene variants in primary dyslipidemia. Atherosclerosis 328: 11-22.
- Leduc V, Jasmin-Bélanger S, Poirier J (2010) APOE and cholesterol homeostasis in Alzheimer’s disease. Trends Mol Med 16(10): 469-477.
- Lane-Donovan C, Wong WM, Durakoglugil MS, Wasser CR, Jiang S, et al. (2016) Genetic Restoration of Plasma ApoE Improves Cognition and Partially Restores Synaptic Defects in ApoE-Deficient Mice. J Neurosci 36(39): 10141-10150.
- Li Z, Shue F, Zhao N, Shinohara M, Bu G (2020) APOE2: protective mechanism and therapeutic implications for Alzheimer’s disease. Mol Neurodegener 15(1): 63.
- Gallego-Villar L, Hannibal L, Häberle J, Thöny B, Ben-Omran T, et al. (2017) Cysteamine revisited: repair of arginine to cysteine mutations. J Inherit Metab Dis 40(4): 555-567.
- Yin Y, Wang Z (2018) ApoE and Neurodegenerative Diseases in Aging. Adv Exp Med Biol 1086: 77-92.
- Muñoz SS, Garner B, Ooi L (2019) Understanding the Role of ApoE Fragments in Alzheimer’s Disease. Neurochem Res 44(6): 1297-1305.
- Lanfranco MF, Sepulveda J, Kopetsky G, Rebeck GW (2021) Expression and secretion of apoE isoforms in astrocytes and microglia during inflammation. Glia 69(6): 1478-1493.
- Uddin MdS, Kabir MdT, Al Mamun A, Abdel-Daim MM, Barreto GE, et al. (2019) APOE and Alzheimer’s Disease: Evidence Mounts that Targeting APOE4 may Combat Alzheimer’s Pathogenesis. Mol Neurobiol 56(4): 2450-2465.
- Neu SC, Pa J, Kukull W, Beekly D, Kuzma A, et al. (2017) Apolipoprotein E Genotype and Sex Risk Factors for Alzheimer Disease: A Meta-analysis. JAMA Neurol 74(10): 1178-1189.
- Carbonell F, Charil A, Zijdenbos AP, Evans AC, Bedell BJ (2014) β-Amyloid is associated with aberrant metabolic connectivity in subjects with mild cognitive impairment. J Cereb Blood Flow Metab 34(7): 1169-1179.
- Chen K, Ayutyanont N, Langbaum JBS, Fleisher AS, Reschke C, et al. (2012) Correlations between FDG PET glucose uptake-MRI gray matter volume scores and apolipoprotein E ε4 gene dose in cognitively normal adults: a cross-validation study using voxel-based multi-modal partial least squares. Neuroimage 60(4): 2316-2322.
- Ossenkoppele R, van der Flier WM, Zwan MD, Adriaanse SF, Boellaard R, et al. (2013) Differential effect of APOE genotype on amyloid load and glucose metabolism in AD dementia. Neurology 80(4): 359-365.
- Zarezadeh Mehrabadi A, Aghamohamadi N, Khoshmirsafa M, Aghamajidi A, Pilehforoshha M, et al. (2022) The roles of interleukin-1 receptor accessory protein in certain inflammatory conditions. Immunology 166(1): 38-46.
- Duan Y, Ye T, Qu Z, Chen Y, Miranda A, et al. (2022) Brain-wide Cas9-mediated cleavage of a gene causing familial Alzheimer’s disease alleviates amyloid-related pathologies in mice. Nat Biomed Eng 6(2): 168-180.
- Lau SF, Chen C, Fu WY, Qu JY, Cheung TH, et al. (2020) IL-33-PU.1 Transcriptome Reprogramming Drives Functional State Transition and Clearance Activity of Microglia in Alzheimer’s Disease. Cell Rep 31(3): 107530.
- Kempuraj D, Khan MM, Thangavel R, Xiong Z, Yang E, et al. (2013) Glia maturation factor induces interleukin-33 release from astrocytes: implications for neurodegenerative diseases. J Neuroimmune Pharmacol 8(3): 643-650.
- Toppi E, Sireno L, Lembo M, Banaj N, Messina B, et al. (2022) IL-33 and IL-10 Serum Levels Increase in MCI Patients Following Homotaurine Treatment. Front Immunol 13: 813951.
- Ormazabal V, Nair S, Elfeky O, Aguayo C, Salomon C, et al. (2018) Association between insulin resistance and the development of cardiovascular disease. Cardiovasc Diabetol 17(1): 122.
- Khatoon R, Pahuja M, Parvez S (2020) Cross Talk between Mitochondria and Other Targets in Alzheimer’s Disease. J Environ Pathol Toxicol Oncol 39(3): 261-279.
- Perez Ortiz JM, Swerdlow RH (2019) Mitochondrial dysfunction in Alzheimer’s disease: Role in pathogenesis and novel therapeutic opportunities. Br J Pharmacol 176(18): 3489-3507.
- Demetrius LA, Eckert A, Grimm A (2021) Sex differences in Alzheimer’s disease: metabolic reprogramming and therapeutic intervention. Trends Endocrinol Metab 32(12): 963-979.
- Ashraf A, Fan Z, Brooks DJ, Edison P (2015) Cortical hypermetabolism in MCI subjects: a compensatory mechanism? Eur J Nucl Med Mol Imaging 42(3): 447-458.
- Blazhenets G, Ma Y, Sörensen A, Rücker G, Schiller F, et al. (2019) Principal Components Analysis of Brain Metabolism Predicts Development of Alzheimer Dementia. J Nucl Med 60(6): 837-843.
- Cohen AD, Price JC, Weissfeld LA, James J, Rosario BL, et al. (2009) Basal cerebral metabolism may modulate the cognitive effects of Abeta in mild cognitive impairment: an example of brain reserve. J Neurosci 29(47): 14770-14778.
- Willette AA, Modanlo N, Kapogiannis D, Alzheimer’s Disease Neuroimaging Initiative (2015) Insulin resistance predicts medial temporal hypermetabolism in mild cognitive impairment conversion to Alzheimer disease. Diabetes 64(6): 1933-1940.
- Kalaria RN, Maestre GE, Arizaga R, Friedland RP, Galasko D, et al. (2008) Alzheimer’s disease and vascular dementia in developing countries: prevalence, management, and risk factors. Lancet Neurol 7(9): 812-826.
- Rius-Pérez S, Tormos AM, Pérez S, Taléns-Visconti R (2018) Vascular pathology: Cause or effect in Alzheimer disease? Neurologia (Engl Ed) 33(2): 112-120.
- Kisler K, Nelson AR, Montagne A, Zlokovic BV (2017) Cerebral blood flow regulation and neurovascular dysfunction in Alzheimer disease. Nat Rev Neurosci 18(7): 419-434.
- Iadecola C (2017) The Neurovascular Unit Coming of Age: A Journey through Neurovascular Coupling in Health and Disease. Neuron 96(1): 17-42.
- Schaeffer S, Iadecola C (2021) Revisiting the neurovascular unit. Nat Neurosci 24(9): 1198-1209.
- Laurent C, Buée L, Blum D (2018) Tau and neuroinflammation: What impact for Alzheimer’s Disease and Tauopathies? Biomed J 41(1): 21-33.
- Attems J, Jellinger KA (2014) The overlap between vascular disease and Alzheimer’s disease--lessons from pathology. BMC Med 12: 206.
- Mehta RI, Schneider JA (2021) What is “Alzheimer’s disease”? The neuropathological heterogeneity of clinically defined Alzheimer’s dementia. Curr Opin Neurol 34(2): 237-245.
- McKee AC (2020) The Neuropathology of Chronic Traumatic Encephalopathy: The Status of the Literature. Semin Neurol 40(4): 359-369.
- Boutouyrie P, Chowienczyk P, Humphrey JD, Mitchell GF (2021) Arterial Stiffness and Cardiovascular Risk in Hypertension. Circ Res 128(7): 864-886.
- Petrie JR, Guzik TJ, Touyz RM (2018) Diabetes, Hypertension, and Cardiovascular Disease: Clinical Insights and Vascular Mechanisms. Can J Cardiol 34(5): 575-584.
- Peng J, Luo F, Ruan G, Peng R, Li X (2017) Hypertriglyceridemia and atherosclerosis. Lipids Health Dis 16(1): 233.
- Messner B, Bernhard D (2014) Smoking and cardiovascular disease: mechanisms of endothelial dysfunction and early atherogenesis. Arterioscler Thromb Vasc Biol 34(3): 509-515.
- Bunch TJ (2020) Atrial Fibrillation and Dementia. Circulation 142(7): 618-620.
- Tarbell J, Mahmoud M, Corti A, Cardoso L, Caro C (2020) The role of oxygen transport in atherosclerosis and vascular disease. J R Soc Interface 17(165): 20190732.
- He JT, Zhao X, Xu L, Mao CY (2020) Vascular Risk Factors and Alzheimer’s Disease: Blood-Brain Barrier Disruption, Metabolic Syndromes, and Molecular Links. J Alzheimers Dis 73(1): 39-58.
- Yamazaki Y, Kanekiyo T (2017) Blood-Brain Barrier Dysfunction and the Pathogenesis of Alzheimer’s Disease. Int J Mol Sci 18(9): E1965.
- Button EB, Robert J, Caffrey TM, Fan J, Zhao W, et al. (2019) HDL from an Alzheimer’s disease perspective. Curr Opin Lipidol 30(3): 224-234.
- Lane CA, Hardy J, Schott JM (2018) Alzheimer’s disease. Eur J Neurol 25(1): 59-70.
- Yu TW, Lane HY, Lin CH (2021) Novel Therapeutic Approaches for Alzheimer’s Disease: An Updated Review. Int J Mol Sci 22(15): 8208.






























