Effects of Hemin on Ca2+ Permeability in Neurons of C57BL/6 Mouse Brain
Hamid Islampoor*
Department of Biochemistry, Shiraz University of Medical Sciences, Iran
Submission: May 22, 2019;Published: November 04, 2019
*Corresponding author: Hamid Islampoor, Department of Biochemistry, School of Medicine, Shiraz University of Medical Sciences, Shiraz, Iran
How to cite this article: Hamid Islampoor. Effects of Hemin on Ca2+ Permeability in Neurons of C57BL/6 Mouse Brain. Open Access J Neurol Neurosurg. 2019; 12(1): 555826. DOI: 10.19080/OAJNN.2019.12.555826.
Abstract
SExcitotoxicity results in a significant increase in Ca2+ influx; essentially from open N-methyl-D-aspartate receptors (NMDARs) channels that cause a secondary rise in the intracellular Ca2+ concentration. It is correlated with neuronal death induced by Ca2+ overload. Dysfunction of NMDARs is associated with excitotoxic neuronal death in neurodegenerative disorders. In this study, the effects of hemin on Ca2+ permeability in neurons of C57BL/6 mouse brain examined. Isolated from 1-dayold C57BL/6 mice, were cultured in serum-free media. Cells were maintained in growth medium at 37°C in 95% air/5% CO2 for 2 weeks in vitro before treatment. Primary neurons were cultured in serum-free media were treated with hemin (0, 12.5, 25, 50, 75,100 μM) for 18 (h). Intensity of calcium fluorescence was reduced in treated cultures with hemin (100, 86, 78.5, 60, 56, 46%, respective to the concentrations stated previously; P<0.05 for all). Hemin increased Ca2+ influx in cultured neurons. NMDAR stimulation by hemin increased the activating of NMDARs and Ca2+ influx in the cultured neurons. Therefore, hemin is cytotoxic due to increase of intracellular Ca2+ influx.
Keywords: Excitotoxicity Ca2+ overload NMDA receptor Hemin Mammalian cells Neurodegenerative disorders Excitotoxicity Cytoplasmic calcium Neuronal deathMammals Neurotransmission Brain
Introduction
At rest, the cytoplasmic concentration ([Ca2+]i) in mammalian cells is very low (50-100 nM) [1]. Excitotoxicity results in a significant increase in Ca2+ influx essentially from open N-methyl-D- aspartate receptors (NMDARs) channels that cause a secondary rise in the intracellular Ca2+ concentration [2], Glutamate is capable of to killing neurons with excitotoxicity [3]. This secondary overload is correlated with neuronal death induced by Ca2+ overload [4]. Dysfunction of NMDARs is associated with excitotoxic neuronal death in neurodegenerative disorders [5]. Glutamate excitotoxicity is a critical factor in brain damage leading to brain and neurodegenerative disorders, such as; Alzheimer’s disease. The mechanism of excitotoxicity is causally linked to an intense elevation in cytoplasmic calcium concentration ([Ca2+]c) that leads to cell injury and neuronal death [6-8]. Correspondingly, stabilizing low cytosolic Ca2+ by chelation with BAPTA [(1,2-bis(o-aminophenoxy) ethane-N, N, N, N-tetra acetic acid;)] increases the survival rate of neurons exposed to excitotoxic glutamate [9,10].
In the central nervous system of mammals, 𝛼-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPARs) and NMDARs primarily mediate fast excitatory neurotransmission. Ca2+ permeability; allows NMDARs to play a vital role in braindamage, neuropathology, and neuroplasticity [11]. The glutamate receptors of AMPA and NMDA are permeable to Ca2+ and are expressed in the brain regions responsible for cognitive functions, such as neocortex and hippocampus [12]. The receptors are heterothermies comprising a combination of GluN1, GluN2A-D and GluN3A-B subunits [13]. NMDARs receptors activation leads to opening of an ion channels that is permeable for cations, resulting in the influx of Na+ and Ca2+ ions and efflux of K+ ions [14]. During excitatory neurotransmission, presynaptic release of glutamate activates glutamate receptors in the postsynaptic membrane, resulting in the generation of an excitatory postsynaptic potential [15]. Phosphorylation of glutamate receptor regulates the Ca2+ channels and Ca2+ permeability [16].
NMDARs have a high affinity to stimulate L-glutamate [17]. L-glutamate is the strongest NMDA agonist (EC50 2.3 μM) [18]. Hemin is an iron–protoporphyrin molecule consisting of four pyrrole rings [19]. Hemin is cytotoxic due to its ability to contribute to the generation of reactive oxygen species [20]. As respects, hemin play an crucial role in correlated with neuronal death induced by Ca2+, In this study, the effects of hemin on Ca2+ permeability in neurons of C57BL/6 mouse brain examined.
Methods
Cell culture
Twenty-eight 1day-old C57BL/6 male mice were prepared from Animal Center of School of Medicine, Shiraz University of Medical Sciences, Shiraz, Iran. They were kept cages (4 mice per cage) at room temperature and provided with a balanced diet, Light cycle with 12h light, 12h dark. The mice randomly divided into 6 groups; and anesthetized with Ketamine (87 mg/kg) and Xylazine (13 mg/kg). All mice were killed, and their brain and neocortex were removed. Neurons isolated by neutrosphere method and were cultured in Dulbecco’s Modified Eagle Medium (Thermo Fisher Scientific). Cells were maintained in growth medium at 37 °C in 95% air/5% CO2 for 2 weeks in vitro before treatment. Primary postnatal neurons cultured in serum-free media were treated with hemin (Frontier Scientific; 0, 12.5, 25, 50, 75,100 μM) for 18 h [21].
Calcium imaging
The neurons were incubated in medium with (Fluo-8 AM) (ATT Bio quest, Inc) for 1 h at 37°C. The neurons were then washed twice with Hank’s Balanced Salt Solution (Frontier Scientificcontaining CaCl2 (2 mM) and MgCl2 (1 mM). The intensity of Flu-8 AM fluorescence was evaluated after adding NMDA (100 μM) and Nifedepine (10 μM) to neurons, with hemin. The intensity of fluorescence was measured of 482 nm and 505-530 nm (emission reverse microscopy) (Leica HC PL FLUOTAR 20×/0.50 objective, Lambda DG-5 Plus) [22].
Statistical analysis
Statistical analysis was carried out using ANOVA; with SPSS 19. One-Way ANOVA was used to test for differences in mean values from multiple samples (calcium imaging data) with comparison tests. Statistical difference between two means was analyzed by two-tailed unpaired or paired Student’s t-test. Differences were considered significant if p<0.05. All data are expressed as mean±SEM.
Results
Primary postnatal neurons cultured in serum-free media were treated with hemin (0, 12.5, 25, 50, 75,100 μM) for 18 h. fluorescence was reduced in treated cultures with hemin (100, 86, 78.5, 60, 56, 46%, respectively) (P < 0/05 for all) (Figure 1 & Table 1).
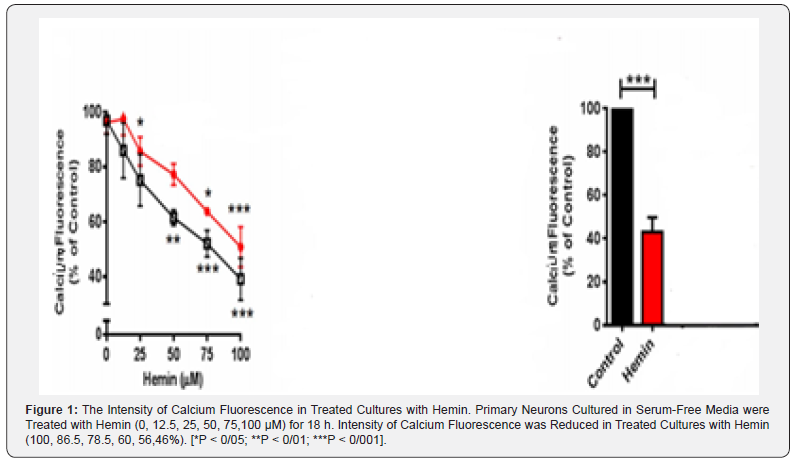
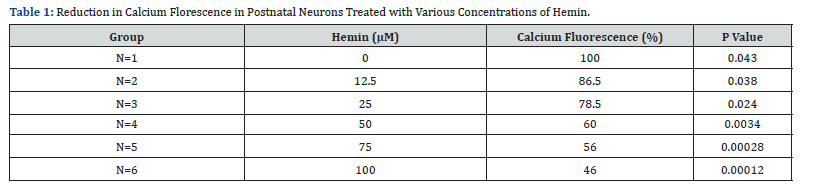
Discussion
Ca2+ influx is regulateds by NMDARs subunits [23]. NMDAR stimulation increases Ca2+ influx [24]. Stimulation of NMDAR activates Ca2+ signaling pathway in neurons [25]. Conversely, inhibition of NMDARs inhibits it [26]. Hemin suppresses NMDAR activity [27], by regulating the GluN1, GluN2A and GluN2B subunits of NMDA [28]. NMDAR stimulation by hemin was increased activating of NMDARs and Ca2+ influx in the cultured neurons.
Data availability
All data relating to the present study are contained in Table.
References
- Berridge MJ, Bootman MD, Roderick HL (2003) Calcium signalling: dynamics, homeostasis and remodelling. Nature reviews. Molecular cell biology 4(7): 517-529.
- Kaku DA, Giffard RG, Choi DW (1993) Neuroprotective effects of glutamate antagonists and extracellular acidity. Science 260(5113): 1516-1518.
- Berliocchi L, Bano D, Nicotera P (2005) Ca2+ signals and death programmes in neurons. Philos Trans R Soc Lond B Biol Sci 360(1464): 2255-2258.
- Lau CG, Zukin RS (2007) NMDA receptor trafficking in synaptic plasticity and neuropsychiatric disorders. Nat Rev Neurosci 8(6): 413-416.
- Tymianski M, Charlton MP, Carlen PL, Tator CH (1993) Secondary Ca2+ overload indicates early neuronal injury which precedes staining with viability indicators. Brain research 607(1-2): 319-323.
- Mattson MP (2003) Excitotoxic and excitoprotective mechanisms. Neuromolecular medicine 3(2): 65-94.
- Lipton SA (2004) Failures and successes of NMDA receptor antagonists: molecular basis for the use of open-channel blockers like memantine in the treatment of acute and chronic neurologic insults. NeuroRx 1(1): 101-110.
- Salińska E, Danysz W, Łazarewicz JW (2005) The role of excitotoxicity in neurodegeneration. Folia neuropathol 43(4): 322-339.
- Tymianski M, Wallace MC, Spigelman I, Uno M, Carlen PL, et al. (1993) Cell-permeant Ca 2+ chelators reduce early excitotoxic and ischemic neuronal injury in vitro and in vivo. Neuron 11(2): 221-235.
- Tymianski MI, Charlton MP, Carlen PL, Tator CH (1994) Properties of neuroprotective cell-permeant Ca2+ chelators: effects on [Ca2+] i and glutamate neurotoxicity in vitro. J neurophysiology 72(4): 1973-1992.
- Kamel IR, Wendling WW, Chen D, Wendling KS, Harakal C, et al. (2008) N-methyl-D-aspartate (NMDA) antagonists-S (+)-ketamine, dextrorphan, and dextromethorphan-act as calcium antagonists on bovine cerebral arteries. J Neurosurg Anesthesiol 20(4): 241-248.
- Cull-Candy S, Brickley S, Farrant M (2001) NMDA receptor subunits: diversity, development and disease. Curr Opin Neurobiol 11(3): 327-335.
- Terry AV, Buccafusco JJ (2003) The cholinergic hypothesis of age and Alzheimer's disease-related cognitive deficits: recent challenges and their implications for novel drug development. J Pharmacol Exp Ther 306(3): 821-827.
- Dzamba D, Honsa P, Anderova M (2013) NMDA receptors in glial cells: pending questions. Curr Neuropharmacol 11(3): 250-262.
- Zito K, Scheuss V NMDA receptor function and physiological modulation.
- Cull-Candy SG, Leszkiewicz DN (2004) Role of distinct NMDA receptor subtypes at central synapses. sci STKE 2004(255): re16.
- Blanke ML, Van Dongen AM (2008) 13 Activation Mechanisms of the NMDA Receptor. Biology of the NMDA Receptor. 29: 283.
- Patneau DK, Mayer ML (1990) Structure-activity relationships for amino acid transmitter candidates acting at N-methyl-D-aspartate and quisqualate receptors. J Neurosci 10(7): 2385-2399.
- Kumar S, Bandyopadhyay U (2005) Free heme toxicity and its detoxification systems in human. Toxicol Lett 157(3):175-188.
- Mohan S, Narumiya S, Doré S (2015) Neuroprotective role of prostaglandin PGE2 EP2 receptor in hemin-mediated toxicity. Neurotoxicology 46: 53-59.
- Mohan S, Glushakov AV (2013) Contribution of PGE2 EP1 receptor in hemin-induced neurotoxicity. Front Mol Neurosci 6: 31.
- Kampa BM, Clements J, Jonas P, Stuart GJ (2013) Kinetics of Mg2+ unblock of NMDA receptors: implications for spike-timing dependent synaptic plasticity. J Physiol 556(2): 337-345.
- Regan RF, Rogers B (2003) Delayed treatment of hemoglobin neurotoxicity. Journal of neurotrauma 20(1):111-20.
- Chen-Roetling J, Cai Y, Lu X, Regan RF (2014) Hemin uptake and release by neurons and glia. Free radical research 48(2): 200-205.
- Lalonde J, Saia G, Gill G (2014) Store-Operated Calcium Entry Regulates Transcription Factor Sp4 in Resting Neurons. Science signaling 7(328): ra51.
- Saia G, Lalonde J, Sun X, Ramos B, Gill G (2014) Phosphorylation of the transcription factor Sp4 is reduced by NMDA receptor signaling. Journal of neurochemistry 129(4): 743-752.
- Gill G, Saia G, Lalonde J (2015) Signal-dependent Regulation of Transcription Factor Activity. The FASEB Journal 29(1 Suppl): 497-501.
- Wang LY, Orser BA, Brautigan DL, MacDonald JF (1994) Regulation of NMDA receptors in cultured hippocampal neurons by protein phosphatases 1 and 2A. Nature 369(6477): 230-232.






























