Towards Improved Treatment Outcomes for Tuberculosis Meningitis - Rethinking the Regimen
Rajeswari Ramachandran1* and Muniyandi M2
1 Consultant Neurologist, Dr Kamakshi Memorial Hospital, India
2Scientist-C, National Institute for Research in Tuberculosis, Indian Council of Medical Research, India
Submission: April 06, 2018; Published: August 16, 2018
*Corresponding author: Rajeswari Ramachandran, Consultant Neurologist, Dr. Kamakshi Memorial Hospital, Tamil Nadu, India; Tel: 9444057486; Email: rajerama@yahoo.com
How to cite this article: Rajeswari R, Muniyandi M. Towards Improved Treatment Outcomes for Tuberculosis Meningitis - Rethinking the Regimen. Open Access J Neurol Neurosurg. 2018; 8(2): 555734. DOI: 10.19080/OAJNN.2018.08.555734.
Abstract
Tuberculosis (TB) ranks as the second leading cause of death from an infectious disease worldwide. TB meningitis (TBM) is the most destructive extra-pulmonary form of TB. The current recommended treatment prevents death or disability in less than half of the patients. TB meningitis still represents an important problem by contributing to a huge number of years of life lost. The management of TBM remains a big challenge. In this context, this manuscript discusses anti TB drugs and treatment regimens that make a difference in the treatment outcomes of TBM considering the pharmacokinetics parameters of anti TB drugs with special reference to the Blood-Brain Barrier (BBB) which is unique to Central Nervous System (CNS). It was observed that generally the treatment outcomes are poor with the recommended regimens (mortality rate ranging from 6.3 to 48.1%). In view of the poor treatment outcomes with standard regimens, different studies were carried out to strengthen the regimen and improve the treatment outcomes by using different interventions. Most of the studies did not show a significant improvement in reducing mortality. There is an urgent need to rethink TBM treatment regimens and to strengthen it considering the following factors: PK parameters of the anti TB drugs including their ability to penetrate the BBB and drugs that achieve good CSF concentrations, drugs that achieve CSF concentrations above the MIC value, considering new drugs, retooling old drugs, using second line drugs for TBM as first line drugs, and modifying the host response. In the management of TBM we need clinical trials to evaluate the efficacy of regimens containing drugs like isoniazid, pyrazinamide, levofloxacin, linezolid, ethionamide, rifampicin and injectable aminoglycoside and trials are also needed to assess the optimum duration of treatment.
Keywords: TB: Tuberculosis; TBM: TB Meningitis; CNS: Central Nervous System; BBB: Blood-Brain Barrier; HIV: Human Immunodeficiency Virus; CSF: CerebroSpinal Fluid
Introduction
Infectious diseases still pose a major problem for public health, mainly in developing countries, where they are the most common reasons contributing to both mortality morbidity and disability. This results in enormous economic burden to the patient, family and the country. Tuberculosis (TB) remains a major global health problem, responsible for ill health among millions of people each year. TB ranks as the second leading cause of death from an infectious disease worldwide, after the Human Immunodeficiency Virus (HIV) [1].
Tuberculosis meningitis (TBM) is the most destructive extra-pulmonary form of TB. It is an inflammation of the meninges, the membranes that envelop the brain and the spinal cord. The reports from India indicate that one tenth of EPTB cases were meningial, spinal and bone TB [2]. Reported long term case fatality due to TBM ranges from 20 to 69% [3], in different settings worldwide with up to half of surviving patients presenting with irreversible sequelae, including paraplegia, blindness, motor and cognitive deficits [4], resulting in enormous economic burden to the family. The current recommended treatment prevents death or disability in less than half of the patients. TB meningitis still represents an important problem by contributing to a huge number of years of life lost [5].
In this manuscript we discuss anti TB drugs and treatment regimens that make a difference in the treatment outcomes of TBM. Specifically, we consider the pharmacokinetics parameters of anti TB drugs with special reference to the Blood-Brain Barrier (BBB) which is unique to Central Nervous System (CNS). We have looked at chemotherapy in terms of choice of drugs, optimum dosage, ability to penetrate the BBB and the possible inclusion of immune modulators such as steroids and the role of shunt surgery.
Current Treatment for TBM
The treatment of EPTB follows standard treatment guidelines depending on the categorization and is consistent with international recommendations by WHO and the International Union Against Tuberculosis and Lung Disease (IUATLD) [6,7]. TBM patients are considered to be seriously ill and treated with two months isoniazid, rifampicin, pyrazinamide and streptomycin followed by 7 months of continuation phase treated with isoniazid and rifampicin. The recommended anti TB drugs and the duration of treatment for TBM is described for TBM in Box-1.
Poor Treatment Outcomes
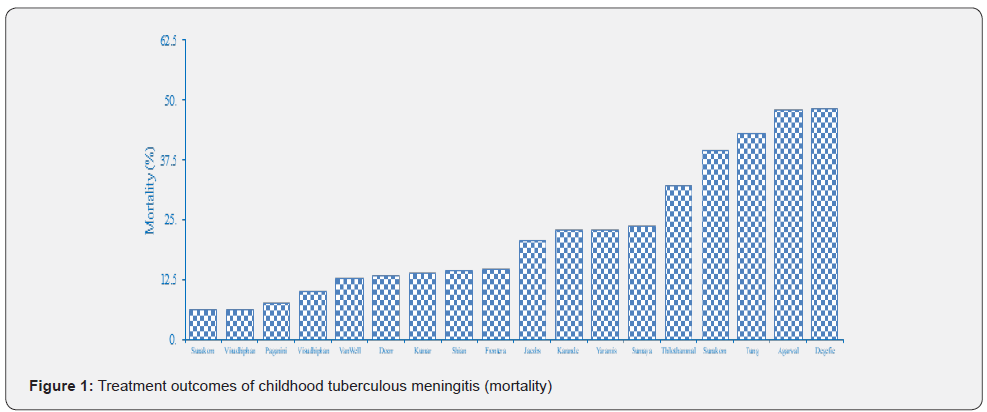
Figure 1 & Figure 2 describes the treatment outcomes such as mortality and morbidity reported by different studies done during 1969 to 2010. It was observed that generally the treatment outcomes are poor with the recommended regimens -mortality rate ranging from 6.3 to 48.1%. The pooled estimate of mortality was 19.1% [8]. A systematic review and meta-analysis of childhood-TBM reported treatment regimens used and their outcomes. The pooled estimates based on 19 studies, with reported treatment outcomes on 1636 children in ten countries between 1952 and 2005 showed a risk of death in 19.3% and probability of survival without neurological sequelae was only 36.7% [9]. In these studies 27 regimens were used and multiple antimicrobial combinations were studied. These studies did not show any associations between outcomes and study-level treatment characteristics such as treatment duration, number of intensive-phase drugs, isoniazid dose, rifampicin dose, and use of rifampicin, streptomycin, pyrazinamide, ethambutol, or corticosteroids.

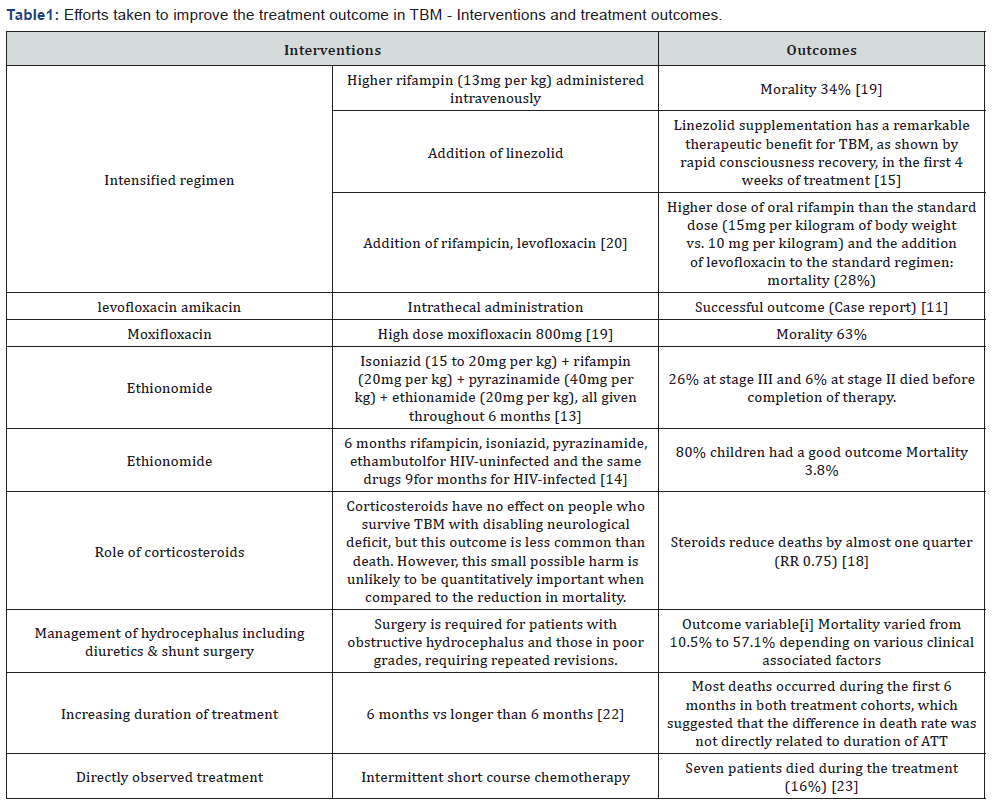
In view of the poor treatment outcomes with standard regimens (Table 1), different studies were carried out to strengthen the regimen and improve the treatment outcomes by using different interventions such as high dose rifampicin, adding newer quinolones, adding ethionomide, adding linezolid, adding both rifampicin and levofloxacin, high dose moxifloxacin, adding steroids, management of hydrocephalus, etc. Majority of the studies did not show a significant improvement in reducing mortality. It is disappointing that there was no advantage associated with the use of these intensified treatment regimens, with regard to overall mortality and most measures of illness.
There is a need to rethink on TBM treatment regimens and to strengthen it based on (6.1) successful regimens tried in pulmonary TB trials, (6.2) on PK parameters of the anti TB drugs including their ability to penetrate the BBB and achieve good CSF concentrations, (6.3) CSF concentration above the MIC value, (6.4) possible use of new drugs, (6.5) retooling of old drugs, (6.6) using second line drugs for TBM as first line drug, and (6.7) modifying host response.
Successful regimens tried in pulmonary TB trials
The current regimens for TBM are designed based on the experience of treating PTB. The standard treatment regimen for PTB consists of REHZ. With the introduction of rifampicin, the treatment outcome of pulmonary TB improved significantly, resulting in shortening of treatment regimens and near 100% cure rates. However, addition of rifampicin to TBM treatment regimens did not significantly improve outcomes. We would like to highlight that TBM is very different from the pulmonary forms of TB and the treatment strategies are to be designed accordingly. In PTB, the response was good even though the number of bacteria is very high (in millions). In TBM, the bacterial population is very low and yet, the same rifampicin, ethambutol, isoniazid, pyrazinamdie regimen does not give good results despite increasing the dose of rifampicin or adding levofloxacin. This may be due the fact that central nervous system is a “unique therapeutic compartment” which requires special consideration in the treatment of TBM [10,11]. We have to think differently while designing treatment regimens for TBM considering the BBB and the ability of the drugs to penetrate the barrier. Time and again, the poor efficacy of the conventional regimen is demonstrated in TBM and redesigning the treatment regimen is an urgent need in this deadly disease.
PK parameters of the anti TB drugs and their ability to penetrate BBB and achieve good CSF concentrations
Pharmacokinetic factors of drugs (such as absorption, metabolism, protein binding, drug clearance) play an important role. It is ideal to select anti TB drugs based on their ability of the drug to penetrate the BBB, and the bactericidal activity. Among the anti TB drugs that are bactericidal with good CSF penetration (more than 80%) are isoniazid, pyrazinamide, levofloxacin, ethionamide and linezolid (Table 2). We feel that, while designing the treatment regimens for TBM, the selection of the drugs should be based on their ability to cross the blood brain barrier and to have a CSF concentration of more than 80%. Bactericidal drugs that cross BBB with the CSF concentration of more than 80% are isoniazid, pyrazinamide, ethionamide, cycloserine and linezolid. These drugs should be considered in the initial intensive phase of the TBM regimen. Pyrazinamide, ethionamide and cycloserine reach levels in CSF identical to blood. Pyrazinamide also shows good penetration into the CerebroSpinal Fluid (CSF); 6 months of pyrazinamide will reduce the rate of relapses [12]. There are few studies and few anecdotal reports showing the efficacy of ethionamide and linezolid [13-16].

In the inflamed meninges, the factors that influence penetration are
(1) Increased drug entry into the CSF,
(2) Delayed removal by a reduction of the CSF bulk flow, and
(3) The inhibition of the activity of efflux pumps.
Among anti TB drugs, isoniazid, pyrazinamide, linezolid, and fluoroquinolones reaches a CSF to serum ratio close to 1.0 and are therefore extremely valuable for the treatment of TBM. In the absence of meningeal inflammation the following factors govern the penetration of anti TB drugs into the CFS and brain tissuemolecular size, lipophilicity, plasma protein binding, active transport and metabolism within the CNS.
In the management of TBM we need clinical trials to evaluate the efficacy of regimens containing drugs like isoniazid, pyrazinamide, levofloxacin, linezolid, ethionamide, rifampicin and injectable aminoglycoside. Trials are also needed to assess the optimum duration of treatment.
CSF concentration above the MIC value
The other pharmacological aspect that has to be considered while designing TBM regimen is to include drugs that have CSF drug levels above the MIC value. Even though rifampicin crosses the barrier partially (20%) due to protein binding, it is above the MIC value in the CSF. In meningitis, rifampicin has been detected in the CSF as early as 2 hours after oral administration and remains above the minimum inhibitory concentration of M. Tuberculosis. Isoniazid diffuses readily into the spinal fluid in the presence or absence of meningeal inflammation. The concentrations obtained in the CSF are above the usual minimum inhibitory concentration of 0.05-0.20μg/ml reported for susceptible strains. It is not known whether lower concentration of drug can lead to failure, relapses and resistance.
Role of new drugs
To optimize antimicrobial therapy, there is an urgent need to try the available newer drugs. It is unfortunate that the three drugs that are closest to wider clinical use bedaquiline (TMC207), delamanid (OPC-67683), and pretomanid (PA-824) are highly protein bound and unlikely to have free penetration into cerebrospinal fluid (CSF) and this will probably limit their usage in TBM [17].
Retooling of old drugs
The current policy is also to retool old drugs and find new purpose. In this light, there is a need to evaluate ethionamide as a first line drug in the treatment of drug sensitive TBM. Not only does ethionamide have good CSF penetration, it is also active in the presence of isoniazid resistance. In a study conducted by Van Torn et al. [14], it was shown that addition of ethionamide in the treatment regimen with a high-end dosing of other drugs showed a good clinical outcome in 80% of children with a mortality of 3.8% [14].
Using second line drugs for TBM as first line drug
Second line drugs (ethionamide, cycloserine and linezolid) should also to be tried as primary chemo therapeutic drugs in this life-threatening form of TB.
Modifying host response
The host response to this bacterial infection like adhesions, exudates, vasculitis and hydrocephalus play a vital role in determining the outcome. So, the treatment regimen should also consider including immune modulatory drugs that may modify the destructive immune host response. In a Cochrane data base of systematic review, it was reported that steroids reduce death by almost one quarter [18]. However, corticosteroids have no effect on people who survive TBM with disabling neurological deficit. While treating TBM, there is a need for closely monitoring for the presence of hydrocephalus and, depending on the type of hydrocephalus and degree of the associated raised intracranial pressure, management is either medical or a shunt procedure.
Cost considerations for TBM
A great economic loss occurs as a result of indirect costs, which involve the cost stemming from losing employees, traveling to health facilities, selling assets to afford TBM treatment, and in particular, losing productivity due to illness and premature death. If all these costs to treat meningitis are added it will be of considerable economic costs to the health system and to the individual households. Further, high morbidity among the survivors may result in increasing dependency ratio, caring cost and productivity loss to the family members.
Conclusion
In the treatment of TBM we need a pragmatic regimen that takes into account the ability of the anti TB drugs to penetrate into the CSF at concentrations that are efficacious, and the doses of the drugs needed to get these concentrations without much of adverse reactions. The WHOs new ‘END TB strategy’ aims to end the global TB epidemic, with targets to reduce TB deaths by 95% and to cut new cases by 90% between 2015 and 2035, and to ensure that no family is burdened with catastrophic expenses due to TB. In order to achieve these targets (to reduce morality due to TBM and also to reduce expenditure both from health system and households) there is an urgent need for investment in conducting controlled clinical trials to develop appropriate efficacious treatment regimens for this devastating disease [19- 23].
References
- World Health Organization (2014) Global tuberculosis report. World Health Organization, France, WHO, Geneva, Switzerland.
- Fraser Wares, Balasubramanian R, Mohan A, Sharma SK (2014) Extrapulmonary Tuberculosis: Management and Control. Central TB Division, Directorate General of Health Services, Ministry of Health and Family Welfare, Government of India.
- Christensen A SH, Roed C, Omland LH, Andersen PH, Obel N, et al. (2011) Long-Term mortality in patients with tuberculous meningitis: A Danish nationwide cohort study. PLoS One 6(11): e27900.
- Ducomble T, Tolksdorf K, Karagiannis I, Hauer B, Brodhun B, et al. (2013) The burden of extrapulmonary and meningitis tuberculosis: an investigation of national surveillance data, Germany, 2002 to 2009. Euro Surveill 18(12): pii: 20436.
- Barbara J McNeil, Mark Thompson, S James Adelstein. Cost effectiveness calculations for the diagnosis and treatment of tuberculous meningitis (1980). Eur J Nucl Med 5(3): 271-276.
- World Health Organization (WHO) Treatment of Tuberculosis (2003). Guidelines for National Programmes, (3rd ed). WHO Geneva, Switzerland.
- International Union Against Tuberculosis and Lung Disease (IUATLD) (2000). Management of tuberculosis: a guide for low-income countries. (5th ed). IUATLD, Paris.
- Silvia S Chiang, Faiz Ahmad Khan, Meredith B Milstein, Arielle W Tolman, Andrea Benedetti, et al. (2014). Treatment outcomes of childhood tuberculous meningitis: a systematic review and metaanalysis. Lancet Infect Dis 4(10): 947-957.
- Faiz Ahmad Khan, Meredith B Milstein, Arielle W Tolman, Andrea Benedetti, Jeffrey R Starke, et al. (2014) Treatment outcomes of childhood tuberculous meningitis: a systematic review and metaanalysis. Lancet Infect Dis 14(10): 947-57.
- Peter R Donald (2016) Chemotherapy for Tuberculous Meningitis. N Engl J Med 374(2): 179-181.
- Berning SE, Cherry TA, Iseman MD (2001) Novel treatment of meningitis caused by multidrug-resistant Mycobacterium tuberculosis with intrathecal levofloxacin and amikacin: case report. Clin Infect Dis 32(4): 643-646.
- (1991) Controlled trial of 2, 4, and 6 months of pyrazinamide in 6-month, three-times-weekly regimens for smear-positive pulmonary tuberculosis, including an assessment of a combined preparation of isoniazid, rifampin, and pyrazinamide. Results at 30 months. Hong Kong Chest Service/British Medical Research Council. Am Rev Respir Dis 143(4 pt 1): 700-706.
- Donald PR, Schoeman JF, Van Zyl LE, De Villiers JN, Pretorius M, et al. (1998) Intensive short course chemotherapy in the management of tuberculous meningitis. Int J Tuberc Lung Dis 2: 704-711.
- vanToorn R, Schaaf HS, Laubscher JA, van Elsland SL, Donald PR, et al. (2014) Short intensified treatment in children with drug-susceptible tuberculous meningitis. Pediatr Infect Dis J 33(3): 248-252.
- Feng Sun, Qiaoling Ruan, Jiali Wang, Shu Chen, Jialin Jin, et al. (2014) Linezolid Manifests a Rapid and Dramatic Therapeutic Effect for Patients with Life-Threatening Tuberculous Meningitis. Antimicrob Agents Chemother 58(10): 6297-6301.
- Roland Nau, Fritz Sorgel, Helmut Eiffert (2010) Penetration of Drugs through the Blood-Cerebrospinal Fluid/Blood-Brain Barrier for Treatment of Central Nervous System Infections. Clin Microbiol Rev 23(4): 858-883.
- Global Alliance for TB Drug Development. Handbook of antituberculosis agents: introduction. Tuberculosis (Edinb) 2008; 88: 85-6.
- Prasad K, Singh MB, Ryan H (2016) Corticosteroids for managing tuberculous meningitis. Cochrane Database Syst Rev 4: CD002244.
- Ruslami R, Ganiem AR, Dian S, Apriani L, Achmad TH, et al. (2013) Intensified regimen containing rifampicin and moxifloxacin for tuberculous meningitis: an open-label,randomised controlled phase 2 trial. Lancet Infect Dis 13(1): 27-35.
- Heemskerk AD, Bang ND, Mai NTH (2016) Intensified antituberculosis therapy in adults with tuberculous meningitis. N Engl J Med 374: 124- 134.
- Rajshekhar V (2009) Management of hydrocephalus in patients with tuberculous meningitis. Neurol India 57(4): 368-374.
- Jullien S, Ryan H, Modi M, Bhatia R (2016) Six months therapy for tuberculous meningitis. Cochrane Database Syst Rev 9: CD012091.
- Sharma SR, Lynrah KG, Sharma N, Lyngdoh M (2013) Directly observed treatment, short course in tuberculous meningitis: Indian perspective. Ann Indian Acad Neurol 16(1): 82-84.






























