Minimally Invasive Transforaminal Lumbar Interbody Fusion versus Posterior Lumbar I
Christie SD1*, Kiberg M1, Song JK2, Hrubes M3, Abraham E4 and Fessler RG5
1Department of Surgery (Neurosurgery), Dalhousie University, Canada
2Department of Neurological Surgery, Northwestern University, USA
3Department of Orthopedics, University of Illinois at Chicago, USA
4Department of Orthopaedics, Horizon Health Network, Canada
5Department of Neurologic Surgery, Rush University, USA
Submission: March 06, 2017; Published: June 09, 2017
*Corresponding author: Christie SD, Department of Surgery (Neurosurgery) Dalhousie University, Halifax, Nova Scotia, Canada, Email: Sean.Christie@Dal.Ca
How to cite this article: Christie S, Kiberg M, Song J, Hrubes M, et al. Minimally Invasive Transforaminal Lumbar Interbody Fusion versus Posterior Lumbar Interbody Fusion: Surgical and Clinical Results. Open Access J Neurol Neurosurg. 2017; 4(2): 555634. DOI: 10.19080/OAJNN.2017.04.555634
Summary
Objective: Surgical management of degenerative lumbar disorders continues to evolve. Recent publications have demonstrated an advantage for fusion procedures in certain pathologies, such as degenerative spondylolisthesis. The optimal surgical approach still needs to be defined. We sought to compare two approaches, minimally invasive TLIF and open PLIF, for the management of symptomatic spondylolisthesis.
Methods: Two ambispective cohorts were compared with primary outcomes being surgical blood loss and time in hospital and secondary outcomes being VAS back and leg and ODI, at 1 year po stop.
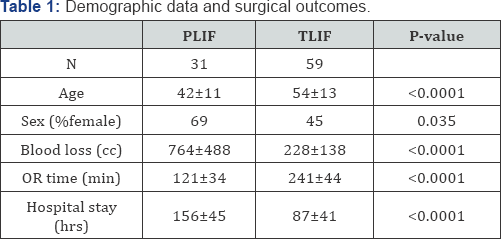
Results: There were 90 patients enrolled (PLIF 31, mTLIF 59). Both groups improved significantly at the 1 year follow-up. Despite significant differences in pre-operative pain scores the TLIF group demonstrated lower blood loss (228±138cc versus 764±488cc, p<0.001) and shorter hospital stay (87±41hrs versus 156±45hrs, p<0.0001). Furthermore, on multivariate analysis the surgical group was a significant predictor in the change in outcomes scores at 1 year, favoring mTLIF (p<0.0001).
Conclusion: Our data suggest that mTLIF is a comparable surgical approach to traditional open PLIF and may have advantages in surgical blood loss, speed of recovery and functional outcomes.
Introduction
Surgical treatment of spondylolisthesis, degenerative disc disease and recurrent disc herniation is becoming increasingly refined. Since the landmark study, by Fritzell et al. [1] showed surgery as a favorable option compared to conservative management of lower back pain, the number of fusion surgeries has increased [1]. These landmark results were further, and more convincingly, confirmed in the recent Spine Patient Outcome Research Trial (SPORT) [2,3].
Historically, lumbar fusion surgeries relied on posterior lateral approaches and laying bone laterally against the facets, pars interarticularis and the transverse process of the vertebrae. This approach had several theoretical biomechanical limitations as it did not closely approximate normal functional mechanics [4,5]. These limitations are improved with the option of 360 degree fusion; currently, there are several widely used techniques that offer 360 degree fusion: posterior lumbar interbody lumbar fusion (PLIF), transforaminal lumbar interbody fusion (TLIF), anterior lumbar interbody fusion (ALIF) and an extreme lateral lumbar interbody fusion (XLIF) [6-8]. The PLIF and TLIF procedure offer an additional advantage in that they only require one approach. The TLIF technique has gained popularity despite the limited evidence supporting the approach. Several theoretical, biomechanical advantages are largely responsible for the gain in popularity, as well as a few unrandomized studies [9,10]. Biomechanically, TLIF offers access to spinal canal and disc via a path that runs through the far lateral portion of the vertebral foramen. This route results in minimal retraction on nerve roots and the dural sac. Hence, there is a decrease in neurological deficit post surgery [11-13]. Also, TLIF is a singlestaged procedure that offers circumferential fusion.
Furthermore, TLIF was shown experimentally to have faster recovery, decreased post-operative pain, decreased time in hospital, decreased amount of post-operative analgesics and decreased blood loss intra-operatively compared to PLIF [10,12-15]. These benefits are balanced against a decreased area of exposure and decreased application. Based on very little evidence from poorly or uncontrolled trials, TLIF gained favor over PLIF.
Advancement of the TLIF technique allows for a minimally invasive approach. This approach is designed to have the same advantages TLIF offers, but to further reduce blood loss and iatrogenic muscle damage. Initial results show that mTLIF is safe and efficacious [16-19]. The limited experimental evidence prompted our group to carry out a prospective cohort study comparing the control group PLIF to the experimental group minimally invasive TLIF.
Our group hypothesized that the mTLIF group would have a faster recovery, less blood loss intraoperatively, and less time in hospital. Our group did not hypothesize any difference in longterm pain outcomes because the procedure should result in the same biomechanical outcome.
Methods
Two prospective databases were retrospectively examined and consecutive patients from each were assigned to PLIF and mTLIF surgical groups. The first group was from a single Canadian medical center (PLIF) and the second was from a single US institution (mTLIF). Pain and functioning survey scores were used to compare outcomes between the PLIF and mTLIF groups. The PLIF group was evaluated using the Visual Analogue Score (VAS) and Oswestry disability index (ODI) surveys [2022]. In addition to these surveys, the mTLIF group was also administered the SF-36® [23]. Patients were asked to complete the surveys at the pre-surgical visit, six weeks post-surgery, and one year post-surgery. The mTLIF group additionally completed the survey at six months post surgery and at the last available follow up.
All surveys have been validated elsewhere in a wide range of patients including spine patients [20-24]. Briefly the VAS score consists of a 10cm line on a paper that is anchored on either end by a descriptor (no pain on the left, highest pain on the right). The patient marks on the continuum where their pain is for them. The mark is then measured and this measurement becomes the VAS score [20,24,25]. The Oswestry disability index is a survey that was designed to access daily level of functioning. Generally, the survey assesses the extent to which the subject can perform the necessary activities for daily life. This method has been validated for spine patients [21,22]. The SF-36 is a commercial survey of 36 question designed to broadly access mental and physical health but more specifically to access six sub-categories: Physical Functioning (PF), Role-Physical (RF), Bodily Pain (BP), General Health (GH), Vitality (VT), Social Functioning (SF), Role- Emotional (RE), Mental Health (MH) [23,26,27].
Procedures
Posterior lumbar interbody fusion (PLIF)
Posterior lumbar interbody fusion is a widely-used procedure. The details of the procedure have been previously described in the literature [28,29].
Minimally invasive transforaminal lumbar interbody fusion (mTLIF)
The minimally invasive transforaminal lumbar interbody fusion has also been previously described.18 Briefly, the technique utilized in this cohort required the patient to be positioned prone on a Wilson frame. Fluoroscopy was used to localize the surgical level. An incision was planned 2.5 to 3.5cm lateral of midline ipsilateral to the predominant leg symptoms. Following the surgical incision, a K-wire is passed through the muscle and docked on the medial facet. Position is confirmed with fluoroscopy. Serial dilators are then passed to dilate the musculature and expand the lumbodorsal fascia. A tubular working channel is then passed over the dilators and fixed to the operative table. Through the retractor, under microscopic magnification, a hemi-laminotomy and facetectomy are performed followed by an aggressive disectomy to achieve root decompression. Interbody grafting included morselized autograft and bone morphogenic protein (rhBMP-2-Infuse, Medtronic). The working channel is then removed and percutaneous pedicle screws placed bilaterally.
Statistics
Statistical analysis was performed with SPSS for windows version 15.1 Data are presented as mean and standard deviation. The PLIF group was assigned as the "control" group and the mTLIF group assigned the "experimental" group. Selected graphs were generated using Microsoft excel for Mac version 11.5. Distribution of the data was examined by generating histograms with visual inspection and then by performing the Kolmogorov-Smirnov test. Data that was not normally distributed was compared using non-parametric testing and multiple comparisons were made using the Mann-Whitney test (MW). Data that was normally distributed was compared using ANOVA with Tukey's post hoc testing. Differences in dichotomous outcomes were tested by Chi-square. Analysis of variables predicting patient outcomes was performed using linear regression models. Because of significant differences between age and sex in the surgical interventions, these variables were included in the models. SF-36 data was examined using the graphing feature in Microsoft excel.
Results
Table 1 shows that there were significant differences between groups for age and sex. The table also shows that TLIF was associated with less operative blood loss and shorter stays in hospital. The baseline VAS for the leg and back as well as the Oswestry Disability Index (ODI) were different preoperatively between the PLIF and mTLIF groups (Table 2). However, both interventions resulted in significantly (P<0.0001) improved pain scores.
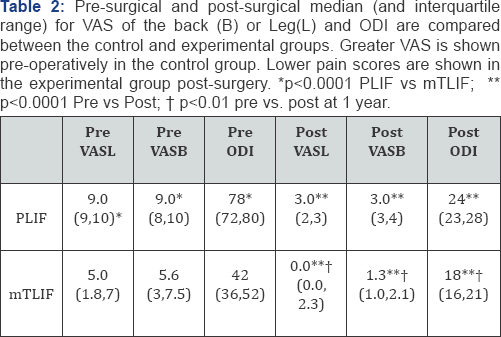
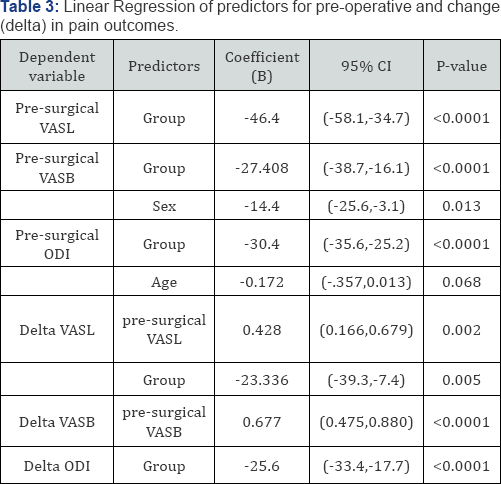
Since there were statistical differences in sex, age and baseline scores, multivariate linear regression was performed to predict variables associated with pre- and pre-post (delta) surgery pain scores. Table 3 shows that pre-surgical scores were largely associated with assigned surgical intervention (group); however, pre-surgical VASB was also associated with sex (more females in PLIF) and pre-surgical ODI was associated with age (older age with mTLIF). When the change (delta) in pain score was examined by linear regression, pre-surgical score was the only significant predictor (P<0.0001) of change at one year for the VAS back. For delta VAS leg, both pre-surgical score and assigned surgical intervention (group) were significant predictors. However, assigned surgical intervention (group) was the only independent predictor of delta ODI score post surgery. In all these analyses age and sex were forced into the model but were not significant. Also, multivariate linear regression of the post-surgical scores was performed. In all cases the surgical group was a significant predictor of final score (p<0.0001). The results for the post-surgical group regression favored the mTLIF group.
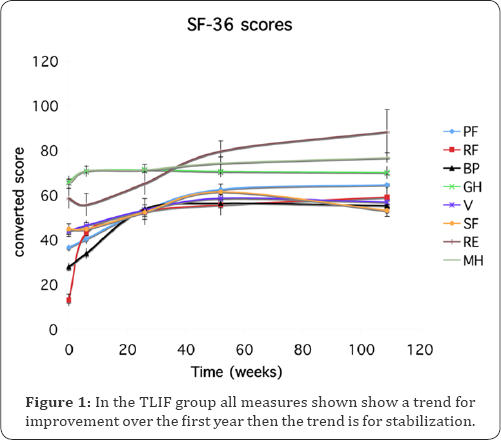
The SF-36 was collected for the mTLIF group only. Scores were converted and plotted. Physical Functioning (PF), Role- Physical (RF), Bodily Pain (BP), General Health (GH), Vitality (VT), Social Functioning (SF), Role-Emotional (RE), Mental Health (MH) all show improvement in the mTLIF group. MH and GH increase slightly over the first 6 weeks and decline to the pre- surgical score in the case of GH. All other measures appear to improve steadily over the first year and then level out after 52 weeks (Figure 1).
Discussion
The mTLIF procedure resulted in several excellent clinical advantages. Although we did not compare the minimally invasive method to the traditional TLIF, our blood loss and hospital stays appear to be lower. Blood loss was 228ml in this report compared to 489-1612ml for the traditional TLIF [6,11,15,19,29]. We report much shorter hospital stay (87hrs) compared to the traditional TLIF (118-168hrs) [11,29]. Moreover, in this study median VAS scores of back pain for the mTLIF group are lower at the last follow up (1.35) compared to the traditional TLIF scores in the literature (3.2-3.7) [10,14,30]. The SF-36 data is further evidence that the mTLIF procedure has good long-term outcomes. In a study by Dhall et al. [19] comparing open and mTLIF a similar reduction in blood loss and hospital stay was seen in the mTLIF group [31]. Furthermore, a recent metaanalysis by Goldstein et al confirmed these trends [19].
The mTLIF procedure results in significantly less blood loss, shorter hospital stays and lower pain-scores at one-year post surgery compared to the PLIF group. With respect to patient pain scores, the best outcome is arguably the post surgical score. However, some would contend that a change in score is more important. We analyzed both separately. Clearly, mTLIF resulted in the lowest pain scores. PLIF had the biggest improvements. We attempted to identify what proportion was the result of the intervention, the demographics and the baseline pain scores. The results were mixed. Delta VAS back appeared to be largely explained by pre-score. ODI was explained by the intervention alone: whereas, both intervention and pre-surgical score predicted delta VAS leg. These findings, slightly favoring mTLIF, are in accord with Goldstein et al. [19].
The baseline preoperative scores between the 2 groups were significantly different. Differences in sex contributed statistically to differences in baseline preoperative pain in the VAS of the back group by linear regression analysis and age approached significance for the ODI score. The assigned surgical intervention (group) was a predictor in all cases. Since the surgical intervention cannot explain baseline pain scores we speculate the patients differed in other ways. The PLIF group was comprised of Canadian only patients. The TLIF group was comprised of patients from the United States. The US patients were, in general, operated on sooner than the Canadian group. In Canada longer wait times may result in the progression of pain. Perhaps increased degeneration over time may partially explain the increased pain in the Canadian group. Poor patient selection may result in a falsely significant decrease in pain. Therefore, the greater drop in pain score may be an artifact of the higher pre- surgical pain level of the PLIF group.
Our group did not hypothesize finding any long-term difference in overall pain reduction. The end result of the procedures should be mechanically the same. The lower one- year VAS in the mTLIF group compared to the PLIF group should be interpreted with caution. Our group did not collect enough demographic data to convincingly determine the effect of demographics on pain scores both pre- and post-operatively. Regardless, both the control and experimental showed both clinically and statistically significant improvement in pain and functioning scores.
Despite the limitations of this non-randomized study, the observations support mTLIF as a procedure to be considered for patients requiring lumbar fusion in the setting of degenerative disease. Our results demonstrate less blood loss, shorter hospital stays and excellent improvement in pain measured by validated instruments.
References
- Fritzell P, Hagg O, Wessberg P, Nordwall A, Swedish Lumbar Spine Study Group (2001) Volvo award winner in clinical studies: Lumbar fusion versus nonsurgical treatment for chronic low back pain: A multicenter randomized controlled trial from the Swedish lumbar spine study group. Spine 26(23): 2521-2532.
- Tosteson AN, Skinner JS, Tosteson TD, Lurie JD, Andersson GB, et al. (2008) The cost effectiveness of surgical versus nonoperative treatment for lumbar disc herniation over two years: Evidence from the spine patient outcomes research trial (SPORT). Spine 33(19): 2108-2115.
- Weinstein JN, Lurie JD, Tosteson TD, Tosteson AN, Blood EA, et al. (2008) Surgical versus nonoperative treatment for lumbar disc herniation: Four-year results for the spine patient outcomes research trial (SPORT). Spine 33(25): 2789-2800.
- Watkins MB (1953) Posterolateral fusion of the lumbar and lumbosacral spine. J Bone Joint Surg Am 35A(4):1014-1018.
- Watkins MB (1959) Posterolateral bone grafting for fusion of the lumbar and lumbosacral spine. J Bone Joint Surg Am 41-A: 388-396.
- Hackenberg L, Halm H, Bullmann V, Vieth V, Schneider M (2005) Transforaminal lumbar interbody fusion: A safe technique with satisfactory three to five year results. Eur Spine J 14(6): 551-558.
- Ozgur BM, Aryan HE, Pimenta L, Taylor WR (2006) Extreme lateral interbody fusion (XLIF): A novel surgical technique for anterior lumbar interbody fusion. Spine J 6: 435-443.
- Weber J, Vieweg U (2006) Anterior lumbar interbody fusion (ALIF) using a cage with stabilization. Z Orthop Ihre Grenzgeb 144: 40-45.
- Chastain CA, Eck JC, Hodges SD, Humphreys SC, Levi P (2007) Transforaminal lumbar interbody fusion: A retrospective study of long-term pain relief and fusion outcomes. Orthopedics 30(5): 389-392.
- Potter BK, Freedman BA, Verwiebe EG, Hall JM, Polly DW, et al. (2005) Transforaminal lumbar interbody fusion: Clinical and radiographic results and complications in 100 consecutive patients. J Spinal Disord Tech 18: 337-346.
- Salehi SA, Tawk R, Ganju A, LaMarca F, Liu JC, Ondra SL (2004) Transforaminal lumbar interbody fusion: Surgical technique and results in 24 patients. Neurosurgery 54(2): 368-74.
- Ozgur BM, Hughes SA, Baird LC, Taylor WR (2006) Minimally disruptive decompression and transforaminal lumbar interbody fusion. Spine J 6: 27-33.
- Ozgur BM, Yoo K, Rodriguez G, Taylor WR (2005) Minimally-invasive technique for transforaminal lumbar interbody fusion (TLIF). Eur Spine J 14: 887-894.
- Lowe TG, Tahernia AD, O'Brien MF, Smith DA (2002) Unilateral transforaminal posterior lumbar interbody fusion (TLIF): Indications, technique, and 2-year results. J Spinal Disord Tech 15: 31-38.
- Whitecloud TS, 3rd, Roesch WW, Ricciardi JE (2001) Transforaminal interbody fusion versus anterior-posterior interbody fusion of the lumbar spine: A financial analysis. J Spinal Disord 14: 100-103.
- Holly LT, Schwender JD, Rouben DP, Foley KT (2006) Minimally invasive transforaminal lumbar interbody fusion: Indications, technique, and complications. Neurosurg Focus 20: E6.?
- Schwender JD, Holly LT, Rouben DP, Foley KT (2005) Minimally invasive transforaminal lumbar interbody fusion (TLIF): Technical feasibility and initial results. J Spinal Disord Tech 18 Suppl: S1-6.
- Khoo LT, Palmer S, Laich DT, Fessler RG (2002) Minimally invasive percutaneous posterior lumbar interbody fusion. Neurosurgery 51(5 Suppl): S166-181.
- Goldstein CL, Macwan K, Sundararajan K, Rampersaud YR (2016) Perioperative outcomes and adverse events of minimally invasive versus open posterior lumbar fusion: meta-analysis and systematic review. J Neurosurg Spine 24(3): 416-427.
- Price DD, McGrath PA, Rafii A, Buckingham B (1983) The validation of visual analogue scales as ratio scale measures for chronic and experimental pain. Pain 17(1): 45-56.
- Fairbank JC (2000) The use of revised oswestry disability questionnaire. Spine 25(21): 2846-2847.
- Fairbank JC, Pynsent PB (2000) The oswestry disability index. Spine 25(22): 2940-2952.
- McHorney CA, Ware JE (1995) Construction and validation of an alternate form general mental health scale for the medical outcomes study short-form 36-item health survey. Med Care 33: 15-28.
- Knop C, Oeser M, Bastian L, Lange U, Zdichavsky M, Blauth M (2001) Development and validation of the visual analogue scale (VAS) spine score. Unfallchirurg 104(6): 488-497.
- Campbell WI, Patterson CC (1998) Quantifying meaningful changes in pain. Anaesthesia 53(2): 121-125.
- Epstein NE (2008) Fusion rates and SF-36 outcomes after multilevel laminectomy and non-instrumented lumbar fusions in a predominantly geriatric population. J Spinal Disord Tech 21: 159-164.
- Ware JE, Kosinski M, Bayliss MS, McHorney CA, Rogers WH, et al. (1995) Comparison of methods for the scoring and statistical analysis of SF-36 health profile and summary measures: Summary of results from the medical outcomes study. Med Care 33(4 Suppl): AS264-AS279.
- Kim DH, Vaccaro AR, Fessler RG (2005) Spinal Instrumentation: Surgical Techniques. Inc: Thieme medical publishers, USA.
- Humphreys SC, Hodges SD, Patwardhan AG, Eck JC, Murphy RB, et al. (2001) Comparison of posterior and transforaminal approaches to lumbar interbody fusion. Spine 26(5): 567-571.
- Hee HT, Majd ME, Holt RT, Myers L (2003) Do autologous growth factors enhance transforaminal lumbar interbody fusion? Eur Spine J 12: 400-407.
- Dhall SS, Wang MY, Mummaneni PV (2008) Clinical and radiographic comparison of mini-open transforaminal lumbar interbody fusion with open transforaminal lumbar interbody fusion in 42 patients with long-term follow-up. J Neurosurg Spine 9(6): 560-565.






























